1. INTRODUCTION
The concept of “sustainability” is becoming increasingly important in almost every aspect of human life, from individual households to the entire planet [1]. Biodiversity is a largely untapped resource in the face of widespread environmental degradation and a rapidly changing climate for supporting and improving the lives on this planet. Fungi form a substantial part of the useful biodiversity available to us and can play an important role in the quest for sustainability.
Arbuscular mycorrhizal fungi (AMF) and most vascular plants have a highly evolved mutually beneficial relationship known as mycorrhizal symbiosis. This symbiosis directly benefits the host plant’s growth and development by acquiring phosphorus (P) and other mineral nutrients from the soil. Furthermore, their functions range from stress mitigation to bioremediation in heavy metal-polluted soils. They may also improve plant resistance to pathogens and increase plant diversity. Proper management of AMF has a great potential to perk up the productivity and sustainability of agricultural systems [2]. In addition to AMF, other fungal communities also play a major role in promoting the growth of the plants under normal environmental conditions by acting as biofertilizers. Under the unfavorable environmental conditions such as drought, salinity, and extremes of temperature, fungal communities act as the stress alleviators and improve the growth of the plants and maintain their health. They also act as the biocontrol agents in presence of the plant pathogens.
Fungi have been widely used in bioremediation. Fungi-based bioremediation offers promising technological avenues that are both efficient and cost-effective. Due to their biomass, hyphal network, and longer life cycle, fungi are more advantageous over bacteria for bioremediation [3]. Antibiotics, antifungal drugs, detergents, heavy metals, herbicides, insecticides, polyaromatic hydrocarbons, and plastics are thus effectively degraded using mycoremediation [4]. Fungi have emerged as a suitable candidate for the pursuit of sustainability, which is actually the major necessity for each aspect of human life in today’s time. The present review deals with the exploitation of the fungi as economical and cost-effective way that could be employed to meet the major challenges for future sustainability.
2. AGRICULTURE
2.1. Biofertilizers
Modern agro-practices such as sustainable agriculture encourage environmental safety while boosting crop production. It is a biotechnological strategy created to satisfy the present and foreseeable food demands of the world. The overuse of agrochemicals for satisfying crop needs has led to an imbalance in the soil quality as well as the composition of its microbial community over time. However, this has made sustainable solutions necessary. In recent years, using biofertilizers in place of chemical fertilizers has grown in popularity. The microbe grouping (fungi, algae, and bacteria) that makes up biofertilizers have the ability to supply nutrients to the soil, fix nitrogen, and solubilize minerals for plant use. They have replaced chemical fertilizers because they are more affordable, more efficient, and a renewable source of plant nutrients [5-7]. These bio-based substances are crucial for preserving soil fertility, nutrient enrichment, and long-term sustenance of healthy soil. As fungal biofertilizers have been used historically, there has been a growing demand in recent years to comprehend their use.
Biofertilizers are made of microbial inoculums, or a mixture of different microorganisms, which promote plant development and increases agricultural output [8]. These microbes aid in nutrient intake from the soil more quickly, solubilize phosphorus, and can fix nitrogen from the atmosphere. One such kind of biofertilizer is AMF, which are prevalent in agricultural soil. It has been suggested that it can boost nutrient intake, stimulate plant growth hormone, and speed up the breakdown of organic wastes, all of which can raise crop output to a higher extent. Mycorrhiza is a term for the type of symbiotic interaction that these fungi have with plants that allows them to survive while also benefiting the plants in so many ways. They interact with these plants, increasing nitrogen, phosphorus, zinc, boron, iron, copper, and sulfur intake [9]. Trees, shrubs, epiphytes, herbs, hydrophytes, and xerophytes are among the plants that interact mutualistically with Mycorrhiza spp. [10]. Mycorrhizae come in seven different varieties, although arbuscular mycorrhizae (AM) and ectomycorrhizae are the two that are the most common mycorrhizae of interest [11].
Endomycorrhizae are fungi that are connected with most agricultural crops and horticulture soils as well as provide natural defense over soil-borne diseases [12]. They are found in many major crop species such as cotton, maize, rice, soybean, and wheat as well as ornamental species such as roses and petunias. AMF are the most common (AMF). The significant rises in crop output following AMF inoculation, which aids in promoting critical impacts such as shaping root architecture, enhanced structural properties of soil, greater nutrient uptake, and ion mobility has been shown [13]. Endomycorrhizae not only improves plant resistance to stressors, but also promotes plant health in general.
Fungal biofertilizers play different roles such as enhance nitrogen uptake, and enhancing phosphate solubilization, can also act as efficient biocontrol agents, and enhance mineral acquisition and transport [7]. The species of Trichoderma are commonly used as biofertilizers [7,14-16]. Aspergillus awamori, and Penicillium citrinum have shown a positive impact on the growth and seed productivity of Cicer arietinum in pot experiments under controlled conditions [17]. The inoculation of C. arietinum with P solubilizing Trichoderma spp. showed growth-promoting effects [18]. The inoculation of Hevea brasiliensis with phosphorus solubilizing Trichoderma harzianum has been studied [19]. The growth promotion of Cajanus cajan with inoculation of Curvularia geniculata has been reported [20].
Some of the commonly used fungal-based biofertilizers are EcoMic®, Micofert®, BuRIZEI, Mycobiol1, Mamezo, and Microrhizae (VAM) [21]. Biofertilizers can be used in the following ways: powder formulations for seed inoculation, in the seed hopper, dry biofertilizers are combined with the seeds, sprinkle method, the slurry approach, pelletizing seeds, slurry biofertilizers, and adhesives are applied to seedlings and covered with ground lime-like substance, water-based peat solution poured into the field while sowing, seed treatment, also known as seed inoculation and root dip for seedlings.
One of the most major disadvantages of biofertilizers is their lower nutritional level when contrasted to inorganic fertilizers, as several studies have found deficient signs in crops cultivated in biofertilizer-treated soils [22]. However, this problem is mitigated by the inclusion of natural ingredients such as bone meal with high content of phosphorus, wood ash with high potassium content, or phosphate rock in the fertilizer. Furthermore, creating biofertilizers from nutrient-rich organic resources such as palm waste and wood ash with high potassium levels is a suitable cure. Notable are the constraints related to biofertilizer storage conditions; this has a significant impact on its performance. Biofertilizers should be kept cool and low-density grade polyethylene with a thickness of 50–75 microns must be used during packing [23]. As beneficial as biofertilizers are, they do not always produce the desired results due to being exposed to high temperatures and other adverse climatic conditions. Other limitation restricting the adoption of biofertilizer technology may be due to a lack of human resources, a lack of acceptable strains, and a lack of suitable carriers, which have been identified as bottlenecks for efficient biofertilizer inoculation [24].
Biofertilizers are a more environmentally friendly choice than chemical fertilizers, which have gained popularity in agricultural applications in recent years. These products are created by combining various microbial genera and consortium. Fungi are among the most well-known biofertilizers. Fungal biofertilizers reduce plant prevalence of the disease by preventing pathogen growth and reducing their biological processes. They produce hormones and enzymes that stimulate plant development in addition to enhancing soil nutrient absorption and creating bioactive molecules. Biofertilizers, despite being impacted by various environmental circumstances, having a short shelf life, and acting slowly when compared to chemical fertilizers, are nevertheless a desirable alternative to the latter due to their numerous advantages. Among fungal biofertilizers, mycorrhizal biofertilizers have a strong appeal in the agro-business, owing to their adaptability and use of environmentally beneficial technologies. Further study on the subject, aided by sophisticated technology, will increase the usage of biofertilizers globally, as well as their viability for marginal and small-scale farmers [Figure 1].
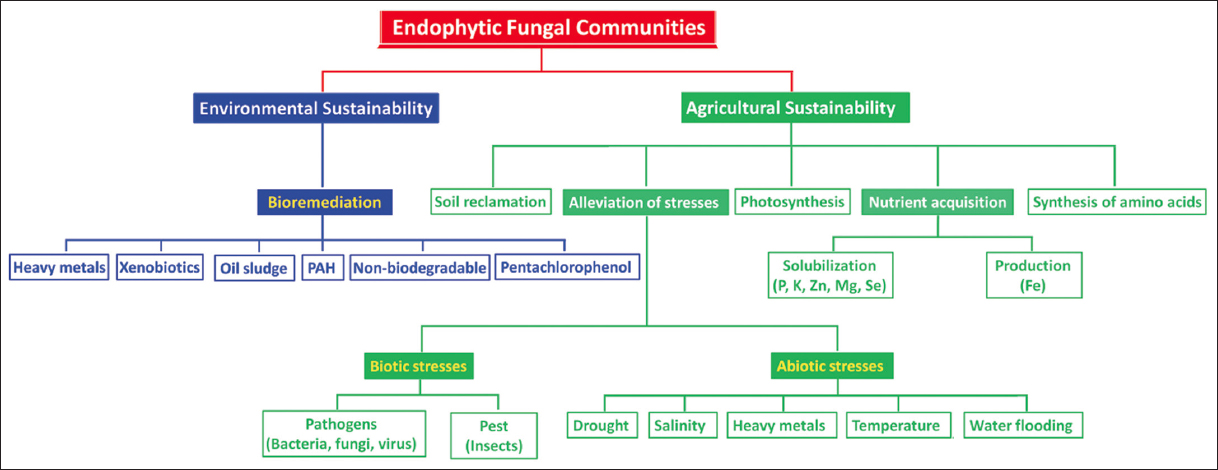 | Figure 1: Biotechnological applications of endophytic fungal communities for agro-environmental sustainability. Adapted with permission from Yadav et al. [98]. [Click here to view] |
2.2. Biocontrol Agents
The misuse of chemical pesticides and their negative impacts on the environment led researchers to focus on safe, environment friendly pest control technology [25]. These have given rise to a brand-new use for biocontrol agents. Controlling plant diseases is urgently needed to preserve global food, feed, and fiber standards. Organisms that inhibit plant diseases are referred to as biological control agents. To put it another way, they are thought of as the use of new or existing biological entities, such as fungi, to lessen the pressure and effects of plant diseases or to limit the reproduction of their species [26]. A significant and crucial function for fungi in acting as biocontrol agents is being played. Fungi are typically genetically stable, exhibit better host destruction, are omnipresent, great dispersal efficiency, and are simple to maintain in cultures. They show effectiveness even at low concentrations and against a variety of pathogenic organisms, possess pesticide resistance, and are non-toxic. These are the elements that qualify them as effective biocontrol agents [27].
Fungi use a variety of strategies, such as direct conflict, antibiosis, or competition, to act as biocontrol agents. Other actions involved in biological disease management include the antagonist’s secretion of extracellular hydrolytic enzymes, competition between organisms for nutrition and space, and detoxification of virulence agents [28,29]. The death of a pathogen by direct antagonistic interaction with other microbes is known as hyperparasitism, and the fungi that use this process are known as mycoparasites [30].
Over the years, both in vivo and in vitro systems have been used to conduct considerable research on the effectiveness of different fungal species in the management of soil-borne fungal diseases. Numerous soil-borne phytopathogenic fungi have been successfully treated using biocontrol agents from Trichoderma spp. Bioformulations based on Trichoderma are frequently used to combat fungal diseases that harm the collar and root systems of agricultural crops. Trichoderma hamatum, Trichoderma viride, Trichoderma atroviride and Trichoderma harzianum, are the major Trichoderma spp. efficient in the treatment of soil-borne fungal diseases [31,32]. Similar to that, there are numerous additional fungi that act as mycofungicides. For instance, Chaetomium cupreum and Chaetomium globosum can be used to combat Phytophthora, Fusarium, and Pythium, Ampelomyces quisqualis can be used to combat powdery mildew, Coniothyrium minitans can be used to combat Sclerotinia, Verticillium albo-atrum can be used to combat Ophyostoma novo-ulmi and Gliocladium virens can be used to combat soilborne pathogens [11,30]. In addition to these, numerous additional fungi are employed as biocontrol agents.
The slow adoption of biological plant disease control in commercial settings is largely attributable to the host-specificity of these methods and their inconsistent field performance under varying environmental conditions. It is crucial to create new biocontrol agent formulations which exhibit higher stability, effectiveness, and survival using novel biotechnological techniques to solve this issue [33]. The cost of commercializing biological control agents is high because there are numerous processes involved, including the isolation of the efficient microbes, their identification, and characterization. It further involves the development of an appropriate formulation, mass production, efficacy testing of the product, examination of storage stability, identification of an industrial partner, consideration of issues relating to human and environmental safety, registration, and marketing [34]. For the management of plant diseases, a variety of biologically based solutions are available in the market all over the world. The use of several microbial antagonists that have been patented and tested for commercial applications is regularly advised for plants [35]. Several commercialized fungal biocontrol agents are Aspire, Sporodex L, ®Bio fungicide, KONI, Primastop, Rotstop®, Rootshield®, Soilgard®, Binab T [36].
The major disadvantage of biocontrol agents arises when a broad host range is present in a crop, particularly if beneficial insects (i.e., parasitoids, predators, and pollinators) are existing and non-target death rate in these populations of beneficial insects can negatively impact the overall success of the biological control. The success of biocontrol fungus is also dependent on environmental conditions. For successful control, moist conditions or high relative humidity in the crop canopy are frequently required. Prolonged exposure to sunshine can deactivate spores, hence diminishing crop persistence. Due to these environmental constraints, natural outbursts of fungi in the environment tend to be infrequent, which might reduce their effectiveness in suppressing pests.
If scientists can effectively generate resting spores and competent mycelia, the application of bio-control fungi will grow in the future. Although research into genetic manipulation to increase virulence in fungi is rising, it is not as extensively investigated as research into bacteria and viruses. To identify the best dosage and duration of application, a greater understanding of the biotic and abiotic interactions with fungus is required. Timing of application must take into account the vulnerable host stage, environmental circumstances, and agricultural methods. Fungal biological management could play an important role in sustainable agriculture by reducing the negative effects of pests and weeds , thereby improving crop quality. New fungal strains with strong biocontrol capacity and effectiveness have greatly benefited from modern biotechnological and genetic engineering technologies. Last but not least, fungi and the secondary metabolites they produce will be important components of future plant pathogen bio-control strategies.
2.3. Abiotic Stress Management
Food security for a growing human population is a critical goal in a sustainable ecosystem. The threat of changing climate and unpredictable environmental extremes is influencing plant growth and productivity [37-40]. Climate change is expected to increase the prevalence of abiotic stresses, pests, and pathogens [41,42]. Abiotic stress conditions have been shown to reduce average yields for major crops by more than 50% [43].
Water stress greatly affects the crop growth and productivity which is a major challenge all around the world [44-46]. Salinity stress leads to osmotic stress and creates ionic imbalane [47]. Heat stress is becoming a global problem due to the rising atmospheric concentrations of CO2. In general, heat stress refers to the condition when the temperature exceeds the ambient temperature by 10–15°C [48]. Transient high temperature negatively impacts plant growth and development [49]. Heat stress damages the photosynthetic system [50]. Low temperature impairs metabolism, changes the structure of proteins and inhibits enzymatic activity. The presence of the heavy metals negatively impacts the photosynthetic machinery, uptake of water and minerals and also modifies amino acids, proteins, and leads to cell membrane damage.
Plants must cope with adverse external environmental conditions through intrinsic biological mechanisms, or their growth, development, and productivity will be affected adversely. In fact, all plants have the capability of detecting and responding to stress. Plant responses to the presence of abiotic stress include changes in water transport, the accumulation of various osmolytes, and production of the reactive oxygen species scavengers [51]. Microbes, the most natural plant inhabitants, have enormous metabolic capabilities to alleviate abiotic stresses. Microbial interactions with plants are thought to modulate local and systemic mechanisms in plants to provide defense against adverse external conditions. In this regard, fungi play an important role in imparting stress tolerance to plants. In general, the plant growth-promoting fungi promote plant growth and helps in stress management by producing plant hormones, siderophores, secondary metabolites, improving nutrient access, and enhancing antioxidant system [52].
The inoculation of AMF is widely reported to provide tolerance to host plants against various stressful situations. AMF assists host plants in up-regulating tolerance mechanisms and preventing the down-regulation of key metabolic pathways. AMF provides vital nutrients to their host, ultimately leading to enhanced growth and productivity under stressed as well as unstressed environments. The role of AMF as a bio-fertilizer can potentially strengthen plant’s adaptability to changing environment [53]. Mycorrhizal fungi also aid in the development of soil structure by encasing their micelles and soil aggregates in a ball and reducing soil erosion losses [54]. Bailey et al. [55] revealed that the role of Trichoderma spp. in the tolerance of cacao plant to water stress via gene expression change. The treatment of cucumber plants with Phoma glomerata and Penicillium spp. increased growth and decreased sodium toxicity under sodium chloride and drought stress [56]. Khan et al. [57] reviewed endophytic fungi as a resource of gibberellins production and their role in abiotic stress management. Abd Allah et al. [58] reported the improved growth performance of Sesbania sesban under salinity stress with inoculation of AMF. Bidabadi and Mehralian [59] reported enhanced chilling stress tolerance of watermelon with inoculation of Glomus intraradices. Jumrani et al. [60] reported the alleviation of high-temperature stress in soybean with inoculation of AMF [Table 1].
Table 1: Alleviation of different abiotic stress with inoculation of fungi.
| Fungi | Crop inoculated | Stress alleviated | Effect | References |
|---|---|---|---|---|
| Paecilomyces formosus | Cucumber | Salinity | Growth parameters | Khan et al. [89] |
| Glomus etunicatum | Soybean | Salinity | Proline, carbohydrates and growth parameters | Sharifi et al. [90] |
| Glomus mosseae | Tomato | Salinity | Growth, leaf area, chlorophyll content, fruit fresh weight and fruit yield | Abdel Latef and Chaoxing [91] |
| Rhizophagus irregularis | Peanut | Salinity | higher net photosynthetic rate, leaf relative water content, plant height, osmolyte accumulation, lower leaf relative electrolyte conductivity, lower lipid peroxidation and increased enzymatic activity | Qin et al. [92] |
| Funneliformis mosseae | Peanut | Salinity | Higher net photosynthetic rate, leaf relative water content, plant height, osmolyte accumulation, lower leaf relative electrolyte conductivity, lower lipid peroxidation and increased enzymatic activity | Qin et al. [92] |
| Rhizophagus intradices | Wheat | Salinity | Nutrient content, antioxidant enzyme activity, and decreasing lipid peroxidation. | Fayaz and Zahedi [93] |
| Funneliformis mosseae | Wheat | Salinity | Nutrient content, antioxidant enzyme activity, and decreasing lipid peroxidation | Fayaz and Zahedi [93] |
| Glomus mosseae | Wheat | Salinity | Photosynthetic pigments, proline, protein, spermine and spermidine, and lowered putrescine contents | Abdel-Fattah et al. [94] |
| Septoglomus deserticola | Tomato | Drought | Decreased lipid peroxidation, hydrogen peroxide level and improved leaf and root antioxidant enzyme activities | Duc et al. [95] |
| Septoglomus constrictum | Tomato | Heat | Decreased lipid peroxidation, hydrogen peroxide level and improved leaf and root antioxidant enzyme activities | Duc et al. [95] |
| Funneliformis mosseae | Cucumber | Low temperature | Fresh weight and dry weight, phenols, flavonoids, lignin, DPPH activity and phenolic compounds | Chen et al. [96] |
| Glomus mosseae | Blueberry | Low temperature | Increased leaf activities of superoxide dismutase, ascorbate peroxidase, guaiacol peroxidase, ascorbate, and glutathione, but decreased leaf concentrations of, MDA, and H2O2 | Liu et al. [97] |
| Glomus intraradices | Watermelon | Low temperature | Decreased accumulation of H2O2 and MDA | Shirani Bidabadi and Mehralian [59] |
3. ENVIRONMENT
3.1. Bioremediation
Many industrial processes, agricultural practices, and chemical uses in our daily lives result in the intentional or unintentional release of potentially toxic chemicals into the environment. When toxic chemicals are released, they travel through the atmosphere and water, and in many cases, they end up in sediments and soils. Environmental chemicals which are of chief concern include petroleum hydrocarbons, halogenated solvents, endocrine-disrupting agents and drugs, explosives, agricultural chemicals, heavy metals, metalloids, and radionuclides. Bioremediation is an efficient technique for converting toxic and recalcitrant compounds harmful to the environment into environmentally benign products. Fungi play an important role in bioremediation due to their robust morphology and commendable metabolic capacities. Fungi have the biochemical and ecological ability to degrade environmental organic chemicals [61]. Mycoremediation is an important sustainable and green approach for cleaning of contaminated sites [62]. White-rot fungi are chiefly involved in the biodegradation of lignin material in nature which contribute in the global carbon recycling. Many white rot fungi including Bjerkandera adjusta, Phanerochaete chysosporium, Pleurotus spp. and Trametes versicolor have been reported for their bioremediation potential by the production of the enzymes including laccases and peroxidases [63]. Laccases, tyrosinases, lignin peroxidases, and manganese peroxidases are major fungal enzymes involved in the process of bioremediation [61]. The presence of these enzymes enables the fungi to grow on recalcitrant substrates. The well-known laccases are majorly produced in Ascomycetes and Basidiomycetes. Laccases are used for decolorization and detoxification in textile industries, degradation, and detoxification of recalcitrant compounds in wastewater [64,65]. Transferases convert the hazardous pollutants into non-hazardous products. The transferase enzymes degrade the pollutants containing hydroxyl groups [66,67]. Thus, fungi appear to be a potential candidate for bioremediation and can be exploited for environmental protection.
3.2. Waste Water Management
Water is an essential resource for all living things and many human endeavors, including home consumption, agriculture, and industry [68]. Therefore, a paradigm change in thinking about wastewater solutions has occurred within the scientific community as a result of the need for sustainable development, which includes a more cyclic usage of water sources, and the resource inadequacy of current wastewater treatment procedures [69]. Furthermore, as wastewater can contain a variety of dangerous compounds, treatment of wastewater has always been among the main challenges of environmental protection [70].
Numerous contaminants and their derivatives are currently dumped into water bodies as a result of urbanization and industrialization, and it is estimated that 1.8 million people are dying annually from waterborne diseases. A significant proportion of these fatalities can be indirectly related to poor sanitation. To prevent the development of such diseases, a wastewater treatment facility is modified to provide clean drinking water and improved living circumstances for the world’s ever-growing population. The environment and human health might be significantly impacted if wastewater is not handled appropriately. These effects may include damage to fish and wildlife populations, depletion of oxygen, beachside bans and other limits on leisure usage of water, limitations on the harvesting of fish and shellfish, and contaminating drinkable water. For the improvement of society, wastewater treatment is an endeavor that must be addressed more severely.
Wastewater treatment refers to the process of removing impurities from wastewater using a variety of procedures. Physical, chemical, and biological procedures are generally the three categories into which advanced removal techniques for wastewater treatment fall [71]. The biological method used by fungi has garnered the most attention from scientists among these categories. In addition, fungi have been found to improve the sludge management approach and the settleability, degradability, and dewaterability of wastewater sludge. Extracellular enzymes are typically used in fungal-mediated bioremediation to break down pollutants and decrease or remove harmful waste from the environment [72]. In buffer zones around streams, fungal mycelium can also function as a filter network to trap runoff from farms, highways, and suburban areas [73]. In other terms, it is a type of bioremediation, which use fungi to remove or degrade environmental pollutants.
Fungus is a unicellular organism with a promising potential in the treatment of wastewater. A diverse range filamentous fungi may metabolize organic compounds and can be easily isolated from wastewater treatment processes. Fungi have been shown to be the most effective in the removal of azo-dye compounds during the wastewater treatment process. Fungal biomass can absorb pigments from wastewater. Fungi have an abundance of enzymes with degrading abilities and can withstand harsh conditions of low pH, variable pollution load, and low nutrient concentrations. Fungi are also more resistant to inhibitory chemicals as compared to the bacterial strains. Fungal cell walls a layer of extra polysaccharide matrix, shield them from inhibiting substances by adsorption. Furthermore, fungi have far more genes than bacteria, making them more adaptable to inhibiting chemicals [74]. The higher the number of genes in fungus, the greater the reproductive selectivity, which may result in superior environmental adaptations [75]. Clumps, pellets, and suspended mycelia are the three types of fungal morphologies that can be employed for wastewater treatment.
Some commonly used fungi for the wastewater treatment are Aspergillus parasitica, Botryosphaeria rhodina, Cephalosporium aphidicola, Hymenoscyphus ericae, Phlebia spp., Pleurotus pulmonarius, Mucor circinelloides, Neocosmospora vasinfecta, Rhizopus spp., Trametes polyzona, Trichoderma longibrachiatum, and Verticillum terrestre [76-79]. Fungi belonging to genera Aspergillus, Penicillium, and Rhizopus, have also proven efficient in waste water treatment [80-82]. The fungal wastewater treatment process has several significant advantages, such as faster degradation of complex organic compounds, efficient solid separation of the fungal biomass from the mixed liquor, and the ability to recover valuable fungal byproducts.
Fungi are frequently grown in the industry to provide a broad range of useful biochemicals. Integrating wastewater treatment with resource recovery could result in an economically feasible option for long-term waste management. From these viewpoints, the fungal wastewater treatment process may be an appealing alternative since it uses a low-cost organic substrate as a feed to generate high-value fungal byproducts while also remedying the wastewater. To arrive at a viable solution for developing a full-scale fungal wastewater treatment process for resource recovery, an inventory based on economics and regulatory criteria must be constructed.
4. FUNGI IN FOOD INDUSTRY
Color is the first and most appealing characteristic that individual notices when approaching food, and it has been linked to the flavor, safety, and nutritional value of the food item [83]. The investigation of filamentous fungi as readily available bioresources of chemically diverse colourants is an emerging area as research trends are changing, a shift towards healthier lifestyles, and a growing market for natural food colorants in economically developing countries worldwide with globalization. Among non-conventional sources, filamentous fungi are known to produce an amazing range of carotenoids, flavins, phenazines, and more specifically monascins, violacein, or indigo [84]. The red and the yellow polyketide pigments from the Monascus spp. have been commercially produced and legally used in traditionally used dried fermented red rice powder for more than 1000 years [85]. Diverse species of Talaromyces have been known to produce meniscus-like polyketide azaphilone pigments [86,87].
Hydroxyanthraquinoid pigments have also been found abundantly particularly in the genera Aspergillus and Penicillium. Drechslera spp. are known to produce hydroxyanthraquinones such as helminthosporin which is maroon, brown pigment, catenarin a red pigment, cynodontin, tritisporin a red brown. A similar red compound, erytroglaucin, is produced by Aspergillus glaucus [88]. Emodin pigment has been isolated from P. citrinum and P. islandicum. The natural food colorant Arpink red™ is manufactured by a Czech company and has been claimed to be produced by fermentation and bioprocess engineering of Penicillium oxalicum var. Armeniaca CCM 8242 as the first commercial product within this chemical family. Marine fungi are also known for the production of bright colors, from yellow to red, chiefly polyketides. Pigments not only serve as coloring agents in the food industry, but they also improve the nutritional and pharmacological potential of the food product by acting primarily as antioxidants. Thus, the major focus for the pigment industry is to look for a sustainable and potential source of pigments which is relatively safe for human health and the environment.
5. CONCLUSION
The advancement of science and technology, as well as rapid industrialization and urbanization has a negative impact on the environment, resulting in the extinction of many species. Beneficial plant-microbe interactions are of paramount importance for the present scenario of climate change, stressful environment, and removal of toxic contaminants from the environment. The unique traits and the biochemical characteristics of fungi have emerged as the potential tool to meet the emerging environmental challenges. Fungi can adapt and survive in diverse climatic conditions and are competent for metabolizing several chemicals. Thus, plant-fungal associations are the precious mines for maintaining the health and sustainability of the terrestrial ecosystem.
6. AUTHOR CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work. All the authors are eligible to be an author as per the international committee of medical journal editors (ICMJE) requirements/guidelines.
7. FUNDING
There is no funding to report.
8. CONFICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
9. ETHICAL APPROVAL
This study does not involve experiments on animals or human subjects.
10. DATA AVAILABILITY
All the data is available with the authors and shall be provided upon request.
11. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Spinosa R, Stamets P, Running M. Fungi and sustainability. Fungi. 2008;1(1):38-43.
2. Aggarwal A, Kadian N, Tanwar A, Yadav A, Gupta K. Role of arbuscular mycorrhizal fungi (AMF) in global sustainable development. J Appl Nat Sci 2011;3(2):340-51. [CrossRef]
3. Singh M, Srivastava P, Verma P, Kharwar R, Singh N, Tripathi R. Soil fungi for mycoremediation of arsenic pollution in agriculture soils. J Appl Microbiol 2015;119(5):1278-90. [CrossRef]
4. Akhtar N, Mannan MA-u. Mycoremediation:Expunging environmental pollutants. Biotechnol Rep 2020;26:e00452. doi:[CrossRef]. [CrossRef]
5. Kour D, Rana KL, Yadav AN, Yadav N, Kumar M, Kumar V, et al. Microbial biofertilizers:Bioresources and eco-friendly technologies for agricultural and environmental sustainability. Biocatal Agric Biotechnol 2020;23:101487. [CrossRef]
6. Yadav AN. Plant microbiomes for sustainable agriculture:current research and future challenges. Plant microbiomes for sustainable agriculture. 2020:475-82. [CrossRef]
7. Odoh CK, Eze CN, Obi CJ, Anyah F, Egbe K, Unah UV, Akpi UK, Adobu US. Fungal Biofertilizers for Sustainable Agricultural Productivity. In:Yadav AN, Mishra S, Kour D, Yadav N, Kumar A, editors. Agriculturally Important Fungi for Sustainable Agriculture:Volume 1:Perspective for Diversity and Crop Productivity. Cham:Springer International Publishing;2020. 199-225 [CrossRef
8. Fuentes-Ramirez LE, Caballero-Mellado J. Bacterial Biofertilizers. In:Siddiqui ZA, editor. PGPR:Biocontrol and Biofertilization. Dordrecht:Springer Netherlands;2006. 143-72. [CrossRef]
9. Mukesh Kumar D, Rejitha R, Devika S, Balakumaran M, Immaculate A, Kalaichelvan P. Production, optimization and purification of lipase from Bacillus sp. MPTK 912 isolated from oil mill effluent. Adv Appl Sci Res 2012;3(2):930-8.
10. Rai A, Rai S, Rakshit A. Mycorrhiza-mediated phosphorus use efficiency in plants. Environ Exp Biol 2013;11:107-17.
11. Mishra T, Mukherjee G. Incredible Role of Fungi in Various Fields for Sustainable Development. In:Gehlot P, Singh J, editors. Fungi and their Role in Sustainable Development:Current Perspectives. Singapore:Springer Singapore;2018. 35-49. [CrossRef]
12. Fr?c M, Hannula SE, Be?ka M, J?dryczka M. Fungal biodiversity and their role in soil health. Front Microbiol 2018;9:707. [CrossRef]
13. Bagyaraj D, Ashwin R. Soil biodiversity:role in sustainable horticulture. Biodivers Hortic Crops 2017;5:1-18.
14. Khalid M, Hassani D, Bilal M, Asad F, Huang D. Influence of bio-fertilizer containing beneficial fungi and rhizospheric bacteria on health promoting compounds and antioxidant activity of Spinacia oleracea L. Bot Stud 2017;58(1):1-9. [CrossRef]
15. Gehlot P, Singh J. Fungi and their role in sustainable development:current perspectives. Springer;2018. [CrossRef]
16. Jehangir I, Mir M, Bhat M, Ahangar M. Biofertilizers an approach to sustainability in agriculture:a review. Int J Pure Appl Biosci 2017;5:327-34. [CrossRef]
17. Mittal V, Singh O, Nayyar H, Kaur J, Tewari R. Stimulatory effect of phosphate-solubilizing fungal strains (Aspergillus awamori and Penicillium citrinum) on the yield of chickpea (Cicer arietinum L. cv. GPF2). Soil Biol Biochem 2008;40(3):718-27. [CrossRef]
18. Kapri A, Tewari L. Phosphate solubilization potential and phosphatase activity of rhizospheric Trichoderma spp. Brazilian J Microbiol. 2010;41(3):787-95. [CrossRef]
19. Promwee A, Issarakraisila M, Intana W, Chamswarng C, Yenjit P. Phosphate solubilization and growth promotion of rubber tree (Hevea brasiliensis Muell. Arg.) by Trichoderma strains. J Agric Sci 2014;6(9):8. [CrossRef]
20. Priyadharsini P, Muthukumar T. The root endophytic fungus Curvularia geniculata from Parthenium hysterophorus roots improves plant growth through phosphate solubilization and phytohormone production. Fungal Ecol 2017;27:69-77. [CrossRef]
21. Mishra J, Arora NK. Bioformulations for Plant Growth Promotion and Combating Phytopathogens:A Sustainable Approach. In:Arora NK, Mehnaz S, Balestrini R, editors. Bioformulations:for Sustainable Agriculture. New Delhi:Springer India;2016 pp 3-33. [CrossRef]
22. Itelima J, Bang W, Onyimba I, Sila M, Egbere O. Bio-fertilizers as key player in enhancing soil fertility and crop productivity:a review. 2018;6:73-83
23. Mishra B, Dadhich S. Methodology of nitrogen biofertilizer production. J Adv Dev Res 2010;1(1):3-6.
24. Ritika B, Utpal D. Biofertilizer, a way towards organic agriculture:A review. Afr J Microbiol Res 2014;8(24):2332-43. [CrossRef]
25. Haggag WM, Mohamed H. Biotechnological aspects of microorganisms used in plant biological control. Am.-Eurasian J Sustain Agric 2007;1(1):7-12.
26. Yadav AN, Rastegari AA, Yadav N, Kour D. Advances in plant microbiome and sustainable agriculture. Springer;2020. [CrossRef]
27. Irtwange S. Application of biological control agents in pre-and postharvest operations. Agricultural Engineering International:CIGR Journal. 2006.
28. Zheng Y, Xue Q-Y, Xu L-L, Xu Q, Lu S, Gu C, et al. A screening strategy of fungal biocontrol agents towards Verticillium wilt of cotton. Biol Control 2011;56(3):209-16. [CrossRef]
29. Deketelaere S, Tyvaert L, França SC, Höfte M. Desirable traits of a good biocontrol agent against Verticillium wilt. Front Microbiol 2017;8:1186. [CrossRef]
30. Palmieri D, Ianiri G, Del Grosso C, Barone G, De Curtis F, Castoria R, et al. Advances and Perspectives in the Use of Biocontrol Agents against Fungal Plant Diseases. Horticulturae. 2022;8(7):577. [CrossRef]
31. Alfiky A, Weisskopf L. Deciphering Trichoderma–plant–pathogen interactions for better development of biocontrol applications. J Fungi 2021;7(1):61. [CrossRef]
32. Zin NA, Badaluddin NA. Biological functions of Trichoderma spp. for agriculture applications. Ann Agric Sci 2020;65(2):168-78. [CrossRef]
33. Heydari A, Pessarakli M. A review on biological control of fungal plant pathogens using microbial antagonists. J Biol Sci 2010;10(4):273-90. [CrossRef]
34. Montesinos E. Development, registration and commercialization of microbial pesticides for plant protection. Int Microbiol 2003;6(4):245-52. [CrossRef]
35. Nabi S, Raja W, Kumawat K, Mir J, Sharma O, Singh D, et al. Post harvest diseases of temperate fruits and their management strategies-a review. Int J Pure App Biosci 2017;5 (3):885-898 [CrossRef]
36. Thambugala KM, Daranagama DA, Phillips AJ, Kannangara SD, Promputtha I. Fungi vs. fungi in biocontrol:An overview of fungal antagonists applied against fungal plant pathogens. Front Cell Infect Microbiol 2020;10:604923. [CrossRef]
37. Abberton M, Batley J, Bentley A, Bryant J, Cai H, Cockram J, et al. Global agricultural intensification during climate change:a role for genomics. Plant Biotechnol J 2016;14(4):1095-8. [CrossRef]
38. Suprasanna P, Ghag SB. Plant Tolerance to Environmental Stress:Translating Research from Lab to Land. Molecular Plant Abiotic Stress. 2019 pp 1-28. [CrossRef]
39. Gray SB, Brady SM. Plant developmental responses to climate change. Dev Biol 2016;419(1):64-77. [CrossRef]
40. Lobell DB, Gourdji SM. The influence of climate change on global crop productivity. Plant Physiol 2012;160(4):1686-97. [CrossRef]
41. Chakraborty S, Newton AC. Climate change, plant diseases and food security:an overview. Plant Pathol 2011;60(1):2-14. [CrossRef] [CrossRef]
42. Batley J, Edwards D. The application of genomics and bioinformatics to accelerate crop improvement in a changing climate. Curr Opin Plant Biol 2016;30:78-81. [CrossRef]
43. Tripathi DK, Singh S, Singh S, Chauhan DK, Dubey NK, Prasad R. Silicon as a beneficial element to combat the adverse effect of drought in agricultural crops. Water Stress and Crop Plants. 2016;pp 682-694. [CrossRef]
44. Kour D, Rana KL, Yadav N, Yadav AN, Singh J, Rastegari AA, et al. Agriculturally and Industrially Important Fungi:Current Developments and Potential Biotechnological Applications. In:Yadav AN, Singh S, Mishra S, Gupta A, editors. Recent Advancement in White Biotechnology Through Fungi:Volume 2:Perspective for Value-Added Products and Environments. Cham:Springer International Publishing;2019;pp 1-64. [CrossRef]
45. Kour D, Rana KL, Kaur T, Sheikh I, Yadav AN, Kumar V, et al. Microbe-mediated alleviation of drought stress and acquisition of phosphorus in great millet (Sorghum bicolour L.) by drought-adaptive and phosphorus-solubilizing microbes. Biocatal Agric Biotechnol 2020;23:101501. [CrossRef]
46. Tiwari P, Bajpai M, Singh LK, Mishra S, Yadav AN. Phytohormones Producing Fungal Communities:Metabolic Engineering for Abiotic Stress Tolerance in Crops. In:Yadav AN, Mishra S, Kour D, Yadav N, Kumar A, editors. Agriculturally Important Fungi for Sustainable Agriculture:Volume 1:Perspective for Diversity and Crop Productivity. Cham:Springer International Publishing;2020;pp 171-97. [CrossRef]
47. Hossain MS, Dietz K-J. Tuning of redox regulatory mechanisms, reactive oxygen species and redox homeostasis under salinity stress. Front Plant Sci 2016;7:548 [CrossRef] [CrossRef]
48. Wahid A, Gelani S, Ashraf M, Foolad MR. Heat tolerance in plants:an overview. Environ Exp Bot 2007;61(3):199-223. [CrossRef]
49. Song Y, Chen Q, Ci D, Shao X, Zhang D. Effects of high temperature on photosynthesis and related gene expression in poplar. BMC Plant Biol 2014;14(1):1-20. [CrossRef]
50. Feng B, Liu P, Li G, Dong S, Wang F, Kong L, et al. Effect of heat stress on the photosynthetic characteristics in flag leaves at the grain-filling stage of different heat-resistant winter wheat varieties. J Agron Crop Sci 2014;200(2):143-55. [CrossRef]
51. Sushma, Verma RK, Thakur S, Singh H, Kapur D. The role of fungi in abiotic stress tolerance of plants. In:Sharma VK, Shah MP, Parmar S, Kumar A (ed) Fungi Bio-Prospects in Sustainable Agriculture, Environment and Nano-Technology. Academic Press;2021, pp 117-54. [CrossRef]
52. Khan N, Asadullah, Bano A. Rhizobacteria and Abiotic Stress Management. In:Sayyed RZ, Arora NK, Reddy MS, editors. Plant Growth Promoting Rhizobacteria for Sustainable Stress Management:Volume 1:Rhizobacteria in Abiotic Stress Management. Singapore:Springer Singapore;2019, pp 65-80. [CrossRef]
53. Begum N, Qin C, Ahanger MA, Raza S, Khan MI, Ashraf M, et al. Role of arbuscular mycorrhizal fungi in plant growth regulation:implications in abiotic stress tolerance. Front Plant Sci. 2019;10:1068 [CrossRef] [CrossRef
54. Demir S, Danesh YR, Boyno G, Najafi S. Arbuscular mycorrhizal fungi in biotic and abiotic stress conditions:Function and management in horticulture. Sustain Hort 2022:157-183. [CrossRef]
55. Bailey B, Bae H, Strem M, Roberts D, Thomas S, Crozier J, et al. Fungal and plant gene expression during the colonization of cacao seedlings by endophytic isolates of four Trichoderma species. Planta 2006;224(6):1449-64. [CrossRef]
56. Waqas M, Khan AL, Kamran M, Hamayun M, Kang S-M, Kim Y-H, et al. Endophytic fungi produce gibberellins and indoleacetic acid and promotes host-plant growth during stress. Molecules 2012;17(9):10754-73. [CrossRef]
57. Khan AL, Hussain J, Al-Harrasi A, Al-Rawahi A, Lee I-J. Endophytic fungi:resource for gibberellins and crop abiotic stress resistance. Crit Rev Biotechnol 2015;35(1):62-74. [CrossRef]
58. Abd_Allah EF, Hashem A, Alqarawi AA, Bahkali AH, Alwhibi MS. Enhancing growth performance and systemic acquired resistance of medicinal plant Sesbania sesban (L.) Merr using arbuscular mycorrhizal fungi under salt stress. Saudi J Biol Sci2015;22(3):274-83. doi:[CrossRef]. [CrossRef]
59. Shirani Bidabadi S, Mehralian M. Arbuscular mycorrhizal fungi inoculation to enhance chilling stress tolerance of watermelon. Gesunde Pflanz 2020;72(2):171-9. doi:10.1007/s10343-020-00499-2. [CrossRef]
60. Jumrani K, Bhatia VS, Kataria S, Alamri SA, Siddiqui MH, Rastogi A. Inoculation with Arbuscular Mycorrhizal Fungi Alleviates the Adverse Effects of High Temperature in Soybean. Plants 2022;11(17):2210. [CrossRef]
61. Harms H, Schlosser D, Wick LY. Untapped potential:exploiting fungi in bioremediation of hazardous chemicals. Nat Rev Microbiol 2011;9(3):177. [CrossRef]
62. Deshmukh R, Khardenavis AA, Purohit HJ. Diverse metabolic capacities of fungi for bioremediation. Indian J Microbiol 2016;56(3):247-64. [CrossRef]
63. dos Santos Bazanella GC, Araujo A, Castoldi R, Maciel G, Inacio F, De Souza C, et al. Ligninolytic enzymes from white-rot fungi and application in the removal of synthetic dyes. Fungal enzymes CRC Press, Boca Raton. 2013, pp 258-79. [CrossRef]
64. Alneyadi AH, Rauf MA, Ashraf SS. Oxidoreductases for the remediation of organic pollutants in water–a critical review. Crit Rev Biotechnol 2018;38(7):971-88. [CrossRef]
65. Spina F, Tigini V, Prigione V, Varese GC. Fungal biocatalysts in the textile industry:Whole-cell systems in real textile wastewater treatment. In:Gupta VK, Mach RL, Srinivasaprasad S (ed) Fungal Biomolecules:Sources, Applications and Recent Developments. 2015, pp 39-50. [CrossRef]
66. Morel M, Meux E, Mathieu Y, Thuillier A, Chibani K, Harvengt L, et al. Xenomic networks variability and adaptation traits in wood decaying fungi. Microb Biotechnol 2013;6(3):248-63. [CrossRef]
67. Singh RK, Tripathi R, Ranjan A, Srivastava AK. Chapter 9 - Fungi as potential candidates for bioremediation. In:Singh P, Kumar A, Borthakur A, editors. Abatement of Environmental Pollutants. Elsevier;2020. pp 177-91. [CrossRef]
68. Besha AT, Gebreyohannes AY, Tufa RA, Bekele DN, Curcio E, Giorno L. Removal of emerging micropollutants by activated sludge process and membrane bioreactors and the effects of micropollutants on membrane fouling:A review. J Environ Chem Eng 2017;5(3):2395-414. [CrossRef]
69. Kehrein P, Van Loosdrecht M, Osseweijer P, GarfíM, Dewulf J, Posada J. A critical review of resource recovery from municipal wastewater treatment plants–market supply potentials, technologies and bottlenecks. Environ Sci Water Res Technol 2020;6(4):877-910. [CrossRef]
70. Lu T, Zhang Q-L, Yao S-J. Application of Biosorption and Biodegradation Functions of Fungi in Wastewater and Sludge Treatment. In:Purchase D (ed). Fungal Applications in Sustainable Environmental Biotechnology. Cham:Springer International Publishing;2016. pp 65-90. [CrossRef]
71. Wang J, Wang S. Removal of pharmaceuticals and personal care products (PPCPs) from wastewater:a review. J Environl Manag 2016;182:620-40. [CrossRef]
72. Kulshreshtha S, Mathur N, Bhatnagar P, Jain B. Bioremediation of industrial waste through mushroom cultivation. J Environ Biol 2010;31(4):441-4.
73. Chiu S-W, Law S-C, Ching M-L, Cheung K-W, Chen M-J. Themes for mushroom exploitation in the 21st century:Sustainability, waste management, and conservation. J Gen Appl Microbiol 2000;46(6):269-82. [CrossRef]
74. Guest R, Smith DW. A potential new role for fungi in a wastewater MBR biological nitrogen reduction system. J Environ Eng Sci 2002;1(6):433-7. [CrossRef]
75. Alic M, Bennett J, Lasure L. More gene manipulations in fungi. Academic Press;1991.
76. Kasonga TK, Coetzee MA, Kamika I, Momba MNB. Data on the degradation of pharmaceuticals and their metabolites by a fungal consortium in a non-sterile stirred fluidized bioreactor. Data Br 2020;28:105057 [CrossRef] [CrossRef]
77. D'Annibale A, Leonardi V, Federici E, Baldi F, Zecchini F, Petruccioli M. Leaching and microbial treatment of a soil contaminated by sulphide ore ashes and aromatic hydrocarbons. Appl Microbiol Biotechnol 2007;74(5):1135-44. [CrossRef]
78. Tunali S, Akar T, Özcan AS, Kiran I, Özcan A. Equilibrium and kinetics of biosorption of lead (II) from aqueous solutions by Cephalosporium aphidicola. Sep Purif Technol 2006;47(3):105-12. [CrossRef]
79. Akar T, Tunali S, Çabuk A. Study on the characterization of lead (II) biosorption by fungus Aspergillus parasiticus. Appl Biochem Biotechnol. 2007;136(3):389-405. [CrossRef]
80. Ahalya N, Ramachandra T, Kanamadi R. Biosorption of heavy metals. Res J Chem Environ 2003;7(4):71-9.
81. Shahid MJ, AL-surhanee AA, Kouadri F, Ali S, Nawaz N, Afzal M, et al. Role of microorganisms in the remediation of wastewater in floating treatment wetlands:a review. Sustainability. 2020;12(14):5559. [CrossRef]
82. M. de Wet MM, G. Brink H. Chapter 18 - Fungi in the bioremediation of toxic effluents. In:Sharma VK, Shah MP, Parmar S, Kumar A (ed) Fungi Bio-Prospects in Sustainable Agriculture, Environment and Nano-technology. Academic Press;2021. pp 407-31. [CrossRef]
83. Kalra R, Conlan XA, Goel M. Fungi as a potential source of pigments:harnessing filamentous fungi. Front Chem 2020;8:369 [CrossRef] [CrossRef]
84. Dufosse L, Fouillaud M, Caro Y, Mapari SA, Sutthiwong N. Filamentous fungi are large-scale producers of pigments and colorants for the food industry. Curr Opin Biotechnol 2014;26:56-61. [CrossRef]
85. Feng Y, Shao Y, Zhou Y, Chen W, Chen F. Monascus pigments. In Vandamme EJ and Revuelta JL (ed) Industrial Biotechnology of Vitamins, Biopigments, and Antioxidants. 2016:497-535. [CrossRef]
86. Mapari SA, Hansen ME, Meyer AS, Thrane U. Computerized screening for novel producers of Monascus-like food pigments in Penicillium species. J Agric Food Chem 2008;56(21):9981-9. [CrossRef]
87. Mapari SA, Meyer AS, Thrane U, Frisvad JC. Identification of potentially safe promising fungal cell factories for the production of polyketide natural food colorants using chemotaxonomic rationale. Microb cell Fact 2009;8(1):1-15. [CrossRef]
88. Durán N, Teixeira MF, De Conti R, Esposito E. Ecological-friendly pigments from fungi. Crit Rev Food Sci Nutr 2002;42(1):53-66. [CrossRef]
89. Khan AL, Hamayun M, Kang S-M, Kim Y-H, Jung H-Y, Lee J-H, et al. Endophytic fungal association via gibberellins and indole acetic acid can improve plant growth under abiotic stress:an example of Paecilomyces formosus LHL10. BMC Microbiol 2012;12(1):3. doi:10.1186/1471-2180-12-3. [CrossRef]
90. Sharifi M, Ghorbanli M, Ebrahimzadeh H. Improved growth of salinity-stressed soybean after inoculation with salt pre-treated mycorrhizal fungi. J Plant Physiol 2007;164(9):1144-51. [CrossRef]
91. Abdel Latef AAH, Chaoxing H. Effect of arbuscular mycorrhizal fungi on growth, mineral nutrition, antioxidant enzymes activity and fruit yield of tomato grown under salinity stress. Sci Hort 2011;127(3):228-33. doi:[CrossRef]. [CrossRef]
92. Qin W, Yan H, Zou B, Guo R, Ci D, Tang Z, et al. Arbuscular mycorrhizal fungi alleviate salinity stress in peanut:Evidence from pot-grown and field experiments. Food Energy Secur 2021;10(4):e314. doi:[CrossRef]. [CrossRef]
93. Fayaz F, Zahedi M. Beneficial effects of arbuscular mycorrhizal fungi on wheat (Triticum aestivum L.) nutritional status and tolerance indices under soil salinity stress. J Plant Nutr 2022;45(2):185-201. doi:10.1080/01904167.2021.1952228. [CrossRef]
94. Abdel-Fattah GM, Ibrahim AH, Al-Amri SM, Shoker AE. Synergistic effect of arbuscular mycorrhizal fungi and spermine on amelioration of salinity stress of wheat ('Triticum aestivum'L. cv. gimiza 9). Aust J Crop Sci 2013;7(10):1525-32.
95. Duc NH, Csintalan Z, Posta K. Arbuscular mycorrhizal fungi mitigate negative effects of combined drought and heat stress on tomato plants. Plant Physiol Biochem 2018;132:297-307. doi:[CrossRef]. [CrossRef]
96. Chen S, Jin W, Liu A, Zhang S, Liu D, Wang F, et al. Arbuscular mycorrhizal fungi (AMF) increase growth and secondary metabolism in cucumber subjected to low temperature stress. Sci Hort 2013;160:222-9. doi:[CrossRef]. [CrossRef]
97. Liu XM, Xu QL, Li QQ, Zhang H, Xiao JX. Physiological responses of the two blueberry cultivars to inoculation with an arbuscular mycorrhizal fungus under low-temperature stress. J Plant Nutr 2017;40(18):2562-70. doi:10.1080/01904167.2017.1380823. [CrossRef]
98. Yadav AN, Kour D, Kaur T, Devi R, Yadav A. Endophytic fungal communities and their biotechnological implications for agro-environmental sustainability. Folia Microbiol 2022;67(2):203-32. doi:10.1007/s12223-021-00939-0. [CrossRef]