1. INTRODUCTION
Plant has been known as a host of several endophytic fungi. The endophytic fungi defined as microorganisms that grow inside the plant without causing any harm to the host plant and they are living in a mutualistic symbiosis with their host [1,2]. Although the relationship between endophytes and their host differs each organism, endophytic fungi are an important component of microbial biodiversity [3]. Many endophytic fungi have the same or similar bioactive component because they live together with host plants for long period of time [4]. Some of endophytes may be producing bioactive substances that may have a relationship with host plant such as specific endophytes that produce taxol as an anticancer drug isolated from Taxus brevifolia [5]. They are also known to be producers of bioactive compounds. A number of interesting biological activities of endophytic fungi, i.e., antioxidant, anticancer, immunomodulatory, antiviral, antitubercular, antiparasitic, and insecticidal activities [6], and a number of substances produced by endophytic fungi had been used in medicine, agriculture, and industry [7,8].
Bouea macrophylla Griffith or locally known as Gandaria in Indonesia is a tropical fruit plant from Southeast Asia. In Indonesia, it is distributed in North Sumatra, Java, Kalimantan, and Ambon. This plant belongs to the family of Anacardiaceae [9]. Gandaria fruit has significant amounts of nutrients and can be explored for the commercial purpose [10]. Methanolic extract of unripe fruits has high phenolics content, tannins and ascorbic acid, flavonoids, and flavonols in the ethanolic extract. The unripe fruits of B. macrophylla showed the highest antioxidant activity [11].
In this study, we reported the bioactivity of N. ribis extract isolated from Gandaria as sources of enzyme inhibitor, antimicrobial, and antioxidant and to identify the bioactive compounds of active extract by gas chromatography-mass spectrometry (GC-MS).
2. METHODS
2.1. Isolation and Identification of Endophytic Microbes
Fresh and healthy Gandaria plant was collected from Bogor Botanic Gardens, Indonesian Institute of Sciences in 2009. The healthy twigs showing no disease symptoms were cut 1 cm length and washed under running tap water for 10 min. The surface of twigs was sterilized by soaking in 75% v/v ethanol for 1 min, 5.3% v/v sodium hypochlorite solution for 5 min, and then 75% v/v ethanol for 30 s, after that it was washed with sterilizes water [12]. The sterilized twigs were dried on sterilized tissue towel before cutting longitudinally into two equal parts. Inside part of twigs was placed on sterilized medium of cornmeal malt extract agar supplemented with 50 mg/L chloramphenicol to suppress bacteria growth. The inside part of the twigs contacted with the surface of the agar media. All of the samples were incubated at 27°C for 2–3 weeks, and hyphae growing out from the twigs were cultured on potato dextrose agar (PDA). This was done several times to obtain pure isolate of endophytic fungi. The observation of fungi that growing up was carried out based on the color and surface. One of the pure endophytic fungi that excellent growth has been identified by Indonesian Culture Collection (InaCC) based on 28s rDNA (D1/D2 region) and internal transcribed spacer (ITS) rDNA. Primers used for polymerase chain reaction (PCR) amplification were NL-1 5’-GCA TAT CAA TAA GCG GAG GAAAAG-3’ and NL-4 5’-GGT CCG TGT TTC AAG ACG-3’ for 28S rDNA (D1/D2 region), meanwhile ITS 4 5’-TCC GCT TAT TGA TAT GC-3’ and ITS 5 5’-GGA AGT AAA AGT CGT AAC AAG G-3’ for ITS rDNA. PCR products were purified by polyethylene glycol precipitation method [13]. Sequence reading was done using a built-in software in the DNA sequencer (ABI PRISM 3130 Genetic Analyzer) (Applied Biosystems). Raw sequence trimming and editing were done using Mega 4 and BioEdit. Homology study was done by BLAST, using the Fasta-formatted query sequence. Phylogenetic tree construction and analyses were accomplished using Clustal X and NJ Plot.
2.2. Fermentation and Extraction
The pure isolate of endophytic fungus with excellent growth was cultured on PDA medium for 1 week. After that, approximately three small pieces of endophytic fungus (1 × 1 cm2) were transferred to 200 mL of potato dextrose broth in three of a 1000 mL Erlenmeyer flask. The fermentation process was done using shaking incubator for 12 days with a speed of 120 rpm. The duration of fermentation is reflected the stationary phase of the N. ribis. After fermentation completed, the biomass and media were centrifuged at 8000 rpm for 20 min to get the supernatant. The extraction process was done according to the modified method of Kumala et al. [14]. The supernatant from one Erlenmeyer was extracted with hexane (1:1), collected, and concentrated with a rotary evaporator (a). The supernatant from the second Erlenmeyer was extracted with ethyl acetate (1:1) (b), and the third Erlenmeyer was extracted with methanol (1:1) followed by partitioned with ethyl acetate (1:1) (c). Agitation process was done for 2 h with a speed of 120 rpm, then separated using a separating funnel, and allowed to stand some time until there was a complete separation. This process is performed 3 times. The extract was stored at 4°C until further analysis.
2.3. Acetylcholinesterase Inhibitor (AChE)
Five hundred units of AChE (Sigma) was dissolved in 500 mL Tris-hydrochloric acid buffer (0.05 mol/L, pH 7.8) and then added with 500 mg bovine serum albumin to stabilize the enzyme. Naphthyl acetate (150 mg) was dissolved in 40 mL ethanol and then diluted with 60 mL of distilled water. 50 mg of Fast Blue B salt was dissolved in 100 mL distilled water. 10 μL of sample were transferred onto silica gel thin-layer chromatography (TLC) plate. After the plate was absolutely dried, the plate was sprayed with 1-naphthyl acetate and blown with cold wind. When the plate dried, the plate was sprayed with enzyme solution, dried, and put on closed vessel or Petri dish with wet cotton to keep the humidity and incubate for 20 min at 37°C. After incubation period, the plate was sprayed with Fast Blue B solution. The extract showed AChE inhibitory activity would appear white, while inactive extract appears purple. The active compounds in extract were separated with mobile phase dichloromethane: methanol (10:1), and then, the same procedure was applied to the plate. The white bands indicated the active compounds as AChE inhibitor [15].
2.4. β-Glucosidase Inhibitor
A 1000 units of β-D-glucosidase (sigma) was dissolved in 100 mL of buffer solution. Buffer solution was made by 10.25 g of sodium acetate in 250 mL distilled water; the pH was adjusted to 7.5 by adding acetic acid 0.1 M. The stock solution was kept at 4°C. 10 μL of sample was transferred on to TLC plate and blown with cold wind then sprayed with enzyme solution. Plate was placed in closed Petri dish where the humidity was kept by putting wet cotton. The plate was incubated for 20 min at 37°C. After incubation period was completed, the plate was sprayed with a mixture of naphthyl-glucopyranoside solution and Fast Blue B salt solution (1:4) to give a purple background. The active extract indicated by change color to yellow. The active compounds in the extract were separated with mobile phase dichloromethane: methanol (10:1). The β-D-glucosidase inhibitory activity was done as the same procedure. The white bands indicated the active compounds in the extract [16].
2.5. Antibacterial Assay
The antibacterial test was done against Staphylococcus aureus InaCC B4 and Escherichia coli InaCC B5. Bacteria were culture overnight at 37°C in Mueller Hinton broth (MHB). Antibacterial screening was done by autobiography method at bacterial densities of 108 CFU/mL. 10 μL of extract (100 μg) was transferred onto TLC plate (Silica gel GF254, Merck) and air-dried. The TLC plate then dipped into bacterial suspension, placed in sterile Petri dish, and incubated at 37°C for 18 h. The humidity in Petri dish was kept by putting sterile wet cotton. After incubation completed, the TLC plate was sprayed with iodonitrotetrazolium p-violet (INT). The active extract which suppresses bacterial growth was shown by clear zone formation. The active compounds in extract were separated with mobile phase dichloromethane: methanol (10:1). The antibacterial assay for the developed TLC-bioautography was carried out with the same procedure. The minimum inhibitory concentration (MIC) was determined by serial dilution method in 96-well microplate in triplicate. 100 μL of 2-fold MHB medium was dispensed into each well on the 1st row. In each well, 100 μL of extract (5120 μg/mL) in 10% dimethyl sulfoxide (DMSO) was dispensed and homogenized. Take out 100 μL and put in the second row that has been fill with 100 μL MHB media. This is done up to the last row. After that, 100 μL bacterial suspension (106 CFU/mL) was added in each well. Streptomycin and chloramphenicol were used as positive controls, while broth medium and DMSO were used as negative controls. The microplate was incubated at 37°C for 18 h. After incubation period, 10 μL of INT was added into each well. Growth inhibition was determined by clear solution. MIC value was determined by the lowest concentration that shows no color change [17].
2.6. Antioxidant Assay
Antioxidant activity was determined by TLC-bioautography method. 10 μL of extract (10 μg/mL) was transferred onto TLC plate. After air-dried, the plate was sprayed with 0.2% DPPH solution in methanol. The active extract was indicated by yellow spot with purple background after 30 min. To know the active compounds, the TLC plate was developed with dichloromethane:methanol (10:1) as mobile phase and air -dried following 30 min of DPPH sprayed, the yellow bands effected which indicated the active compounds. The active extract was further analyzed for its IC50 in triplicate. 100 μL of sample in a concentration of 1000 ppm and catechin (positive control) was put in the well of 1st row. On the 2nd–8th rows, 50 μL methanol was put in the well. The samples were diluted serially by taking out 50 μL of sample from the 1st row and put in the 2nd–8th rows. After finish diluting the samples, 80 μL of DPPH was added to each well and then incubated in the dark for 30 min, and the absorbance was determined at 517 nm with microplate reader (varioSCAN). The scavenging capacity (SC %) = [(Ao-As)/Ao] × 100%, where Ao is the absorbance of reagent blank, while As is the absorbance of test sample. IC50 is the required concentration of the sample to scavenge 50% DPPH in certain time period [18].
2.7. GC-MS Analysis
Chemical compounds of ethyl acetate extract were carried out using GC-MS instrument (Agilent Technology GC System 6890N), equipped with capillary column HP-5MS (0.25 mm × 60 m × 0.25 μm). The instrument was set to initial temperature 80°C. Injector and detector temperature was 290°C. Helium with the purity of 99.9% was used as carrier gas at a flow rate of 1 mL/min, and the volume of injected sample was 3 μL. The initial oven temperature was programmed at 80°C and increased up to 290°C at the rate of increase 15°C/min. The identification of the compounds was done by comparing mass spectra of the compounds with that of available mass spectral records (NIST library).
3. RESULTS AND DISCUSSION
3.1. Identification and Bioactivity of Strain
A newly isolated endophytic fungi from a twig of Gandaria plant were identified by means of PCR and DNA sequence analysis. Partial 28S rRNA (D1/D2 region) was compared with that of other plant fungi and found to be in the same cluster with N. ribis, Botryosphaeria ribis, and Guignardia cryptomeriae with 99% similarities. Since D1/D2 region could not differentiate N. ribis with G. cryptomeriae, further PCR amplified rDNA ITS analysis which confirms these endophytic fungi to be 99.9% identical with N. ribis.
Bioactivity of N. ribis extract was done by TLC-bioautography method. This assay is the simplest guided bioassay to discover new bioactive compounds from natural product [19]. TLC-bioautography is facilitating the localization and target-directed isolation of active compounds of extract [20]. Extract of endophytic fungi isolated from B. macrophylla was tested for its bioactivities, i.e., AChE and β-glucosidase inhibitor, antibacterial against S. aureus and E. coli, and also antioxidant as DPPH radical scavenger.
3.2. TLC-Bioautography - AChE Inhibitor
The AChE inhibitory activity of endophytic fungi extracts was done by TLC bioautography. The test relies on the cleavage by AChE of 1-naphthyl acetate to form 1-naphthol, which in turn reacts with Fast Blue B salt to give a purple-colored diazonium dye [21,22]. Enzyme inhibitors block the formation of α-naphtol and there is no purple coloration on TLC plates [22]. This method is quick in giving information about the activity and localization of active compounds in the extract [21] and rapid identification of a large number of AChE inhibitors [22]. The active chemical compounds in the extract as acetylcholinesterase inhibitor showed white spots. The extract of endophytic fungi N. ribis showed white spots that indicate that these compounds have AChE inhibitory activity [Figure 1].
3.3. TLC-Bioautography - β-Glucosidase Inhibitor
Glucosidase inhibitors are responsible for disruption of the activity of glucosidase [23] and potentially useful as antidiabetic, anti-obesity, antiviral, antiadhesive, antibacterial, or antimetastatic agents [24]. The result for TLC-bioautography of endophytic fungi N. ribis for β-glucosidase inhibitor is shown in Figure 2 that the extract contains compounds with β-glucosidase inhibitor activity.
3.4. TLC-Bioautography - Antibacterial Activity
Screening of antibacterial activity of N. ribis extract against S. aureus and E. coli is shown in Figure 3. It was indicated that extract derived from methanol partitioned with ethyl acetate (C) was the most active extract against S. aureus compared to A and B, while extract B and C also active against E. coli.
 | Figure 1: Thin-layer chromatography bioautography for acetylcholinesterase inhibitor (AChE). White bands indicate active compounds as AChE inhibitor [Click here to view] |
 | Figure 2: Thin-layer chromatography bioautography of endophytic fungi Neofusicoccum ribis for β-glucosidase inhibitor. White bands indicate the compounds as β-glucosidase inhibitor [Click here to view] |
The growth inhibition by the extract was indicated by clear zone formation after sprayed with INT. TLC plate as a background changes its color to purple after sprayed with INT. The purple color is caused by the conversion of INT to intensely colored formazan by the dehydrogenases enzyme of living microorganisms [25]. The interaction between viable microorganisms and INT caused a color change of INT from faint yellow to red–purple color [19]. The result of antibacterial activity showed that extracts B and C have several compounds that suppressed the growth of S. aureus and E. coli. The active compound that suppressed both bacteria has similar Rf value. This indicated that the compound might have a broad spectrum in suppressing bacterial growth. The results are shown in Table 1 that all of the extracts had MIC value >512 μg/mL against E. coli and only extract C had the MIC value 512 μg/mL. Extract with MIC value from 500 to 1000 μg/mL was considered as weak antibacterial activity [26].
3.5. TLC-Bioautography - Antioxidant Activity
Antioxidant activity assay used in this study was DPPH radical scavenging activity. DPPH radical scavenging assay is a quick, reliable, and reproducible for screening in vitro antioxidant of crude extract and pure compound. DPPH has the ability to donate hydrogen [19]. The antioxidant compound of extract reduced the capacity of DPPH radical by the decrease in absorbance [27]. The IC50 value of extract A was the lowest compared to extracts B and C. It was indicated that extract A had better antioxidant activity than extracts B and C [Table 2].
3.6. Analysis of Chemical Compounds of N. ribis by GC-MS
There are 12 active compounds identified by GC-MS analysis from N. ribis ethyl acetate extract: Undecane, cyclopentasiloxane, dodecane, 1-tetradecene, phenol, 1-hexadecene, 1-octadecene, N,N-diethyl-N’,N’-diphenyl-6-pyrrol-1-yl-[1,3,5]triazine-2,4-diamine, 1-docosene, N,N-diethyl-N’,N’-diphenyl-6-pyrrol-1-yl-[1,3,5]triazine-2, 4- diamine, and 1-nonadecene [Table 3]. The peak values identified in above extract are shown in Figure 4. Most of the identified compounds have been reported to possess interesting biological activities. Dodecane reported as antifungal/antibacterial [28,29]. 1-Octadecene and 1-hexadecane have been reported as antibacterial/antifungal, antioxidant, and anticancer [30]. Docosene was also reported has the ability as antifungal [31], while phenol has been reported as antioxidant, anticarcinogenic, and anti-inflammatory [32].
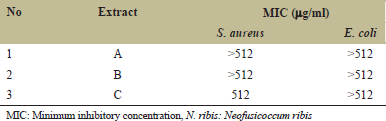 | Table 1: MIC of N. ribis extract [Click here to view] |
 | Table 2: IC50 value of N. ribis extract [Click here to view] |
 | Figure 3: Thin-layer chromatography bioautography of Neofusicoccum ribis extract for antibacterial activity against Staphylococcus aureus and Escherichia coli [Click here to view] |
 | Figure 4: Gas chromatography-mass spectrometry analysis of chemical compounds [Click here to view] |
 | Table 3: GC-MS Identification [Click here to view] |
4. CONCLUSION
This study revealed that endophytic fungi of N. ribis ethyl acetate extract isolated from Gandaria plant contain 12 chemical compounds, some of them have biological activities. The extract contains chemical compounds with antibacterial, antioxidant, and enzyme inhibitors, especially acetylcholine esterase inhibitor and β-glucosidase inhibitor. The N. ribis extract against S. aureus and E. coli was in the range MIC values 512–>512 μg/mL, while hexane extract has better antioxidant activity (IC50 = 479.66 μg/mL) than ethyl acetate extract (IC50 = 928.09–1102.60 μg/mL). The activity as enzyme inhibitors is present only in ethyl acetate extracts. Further, isolation and purification of bioactive compounds need to be done.
5. REFERENCES
1. Petrini O, Sieber TN, Toti L, Viret O. Ecology, metabolite production, and substrate utilization in endophytic fungi. Nat Toxin 1992;1:185-96.
2. Nair DN, Padmavathy S. Impact of endophytic microorganisms on plants, environment and humans. Sci World J 2014. Article ID: 250693.
3. Khaswar RN, Misra A, Gond SK, Stierle A, Stierle D. Anticancer compounds derived from fungal endophytes: Their importance and future challenges. Nat Prod Rep 2011;28:1208-28.
4. Zhou X, Zhu H, Liu L, Lin J, Tang K. A review: Recent advances and future prospects of taxol-producing endophytic fungi. Appl Microbiol Biotechnol 2010;86:1707-17.
5. Strobel GA. Endophytes as sources of bioactive products. Microb Infect 2003a;5:535-44.
6. Hussain H, Kliche-Spory C, Al-Harrasi A, Al-Rawahi A, Abbas G, Green IR, et al. Antimicrobial constituents from three endophytic fungi. Asian Pac J Trop Med 2014;7:S224-7.
7. Strobel GA, Daisy B. Bioprospecting for microbial endophytes and their natural products. Microbiol Mol Biol Rev 2003b;67:491-502.
8. Bhagobaty RK, Joshi SR. Metabolite profiling of endophytic fungal isolates of five ethno-pharmacologically important plants of Meghalaya, India. J Metabol Syst Biol 2011;2:20-31.
9. Kaewpongumpai S, Poeaim S, Vanijajiva O. Sequence-related amplified polymorphism (SRAP) analysis for studying genetic characterization of Bouea macrophylla. Biodiversitas 2016;17:539-43.
10. Rajan NS, Bhat R, Karim AA. Preliminary studies on the evaluation of nutritional composition of unripe and ripe ‘Kundang’ fruit (Bouea macrophylla Griffith). Intl Food Res J 2014;21:985-90.
11. Rajan NS, Bhat R. Antioxidant compounds and antioxidant activities in unripe and ripe kundang fruits (Bouea macrophylla Griffith). Fruits 2016;71:41-7.
12. Petrini O. Taxonomy of endophytic fungi of aerial plant tissues. In: Fokkena NJ, Van Den HJ, editors. Microbiology of the Phyllosphere. Cambridge: Cambridge University Press; 1986. p. 175-87.
13. Hiraishi A, Kamagata Y, Nakamura K. Polymerase chain reaction amplification and restriction fragment length polymorphism analysis of 16S rRNA genes from methanogenes. J Ferment Bioeng 1995;79:523-9.
14. Kumala S, Yuliani KD, Simanjuntak P. Antimicrobial activity of secondary metabolites produced by endophytic fungi isolated from stem of Jati tree (Tectona grandis L.F). Intl J Pharm Sci Res 2015;6:2349-53.
15. Yang Z, Zhang X, Duan D, Song Z, Yang M, Li S. Modified TLC bioautographic method for screening acetylcholinesterase inhibitors from plant extracts. J Sep Sci 2009;32:3257-9.
16. Simoes-Pires CA, Hmicha B, Marston A, Hostettman K. A TLC bioautographic method for the detection of α- and β-glucosidase inhibitors in plant extracts. Phytochem Anal 2009;20:511-5.
17. Perumal S, Pillai S, Cai LW, Mahmud R, Ramanathan S. Determination of minimum inhibitory concentration of Euphorbia hirta (L.) extracts by tetrazolium microplate Assay. J Nat Prod 2012;5:68-76.
18. Lee SY, Mediani A, Ashikin NA, Azliana AB, Abas F. Antioxidant and α-glucosidase inhibitory activities of the leaf and stem of selected traditional medicinal plants. Intl Food Res J 2014;21:165-72.
19. Shahverdi AR, Abdolpour F, Monsef-Esfahani HR, Farsam H. A TLC bioautographic assay for the detection of nitrofurantoin resistance reversal compound. J Chromatogr B Analyt Technol Biomed Life Sci 2007;850:528-30.
20. Bag A, Chattopadhyay RR. Evaluation of synergistic antibacterial and antioxidant efficacy of essential oils of spices and herbs in combination. PLoS One 2015. Available from: https://www.doi.org/10.1371/journal.pone.0131321. [Last accessed on 2017 Oct 16].
21. Marston A, Kissling J, Hostettman KA. A rapid TLC bioautographic method for the detection of acetylcholinesterase and butyrylcholinesterase inhibitors in plants. Phytochem Anal 2002;13:51-4.
22. Dewanjee S, Gangopadhyay M, Bhattacharyaa N, Khanra R, Dua TK. Bioautography and its scope in the field of natural product chemistry. J Pharm Anal 2015;5:75-84.
23. Pandey S, Sree A, Dash SS, Sethi DP. A novel method for screening beta-glucosidase inhibitors. BMC Microbiol 2013;13:55. Available from: http://www.biomedcentral.com/1471-2180/13/55. [Last accessed on 2017 Oct 16].
24. Mehta A, Zitzmann N, Rudd PM, Block TM, Dwek RA. α-Glucosidase inhibitors as potential broad-based anti-viral agents. FEBS Lett 1998;430:17-22.
25. Silva MT, Simas SM, Batista TG, Cardarelli P, Tomassini TC. Studies on antimicrobial activity, in vitro, of Physalis angulata L. (Solanaceae) fraction and physalin B bringing out the importance of assay determination. Mem Inst Oswaldo Cruz 2005;100:779-82.
26. Pessini GL, Filho BP, Nakamura CV, Cortez DA. Antibacterial activity of extracts and neolignans from Piper regnellii (Miq.) C. DC. var. Pallescens (C. DC.) Yunck. Mem Inst Oswaldo Cruz 2003;98:1115-20.
27. Kumar S, Pandey AK. Medicinal attributes of Solanum xanthocarpum fruit consumed by several tribal communities as food: An in vitro antioxidant, anticancer and anti HIV perspective. BMC Comp Alt Med 2014;14:112-9.
28. Akpuaka A, Ekwenchi MM, Dashak DA, Dildar A. Biological activities of characterized isolates of n-hexane extract of Azadirachta indica A. Juss (Neem) leaves. N Y Sci J 2013;6:119-24.
29. Sharma D, Pramanik A, Agrawal PK. Evaluation of bioactive secondary metabolites from endophytic fungus Pestalotiopsis neglecta BAB-5510 isolated from leaves of Cupressus torulosa D. Don. 3 Biotech 2016;6:210-4.
30. Belakhdar G, Benjouad A, Abdennebi EH. Determination of some bioactive chemical constituents from Thesium humile Vahl. J Mater Environ Sci 2015;6:2778-83.
31. Nandhini US, Sangareshwari S, Lata K. Gas chromatography-mass spectrometry analysis of bioactive constituents from the marine streptomyces. Asian J Pharm Clin Res 2015;8:244-6.
32. Muthusamy K, Gopal V, Sivanandham V. GC-MS analysis of bioactive components of karisalaikarpa chooranam-a siddha poly herbal formulation. Intl J Phytopharm 2015;6:131-6.