1. INTRODUCTION
The intensive use of synthetic or inorganic fertilizers is one of the common practices used by potato crop growers for higher marketable yields. This has raised serious concerns over soil health and soil fertility in the long run. Due to the overuse of chemical fertilizers, society is facing the biggest challenge of productivity declining, natural resource depletion, soil fertility reduction, the threat to life on land and in water, adverse effects on climate, and unsecured healthy lives and well-being for human [1-4]. Further, the sole application of fertilizers has caused the depletion of microbial biomass carbon, soil respiration, dehydrogenase, acid phosphatase, and β-glucosidase activities, microbial population, and soil aeration in the rhizosphere [5].
The rhizosphere is attributed with non-metabolic secretions by roots (called root exudates) which are involved in rhizospheric communication between soil, roots, and microbial populations [6-8]. In addition, roots also release some metabolically produced compounds such as mucilage or lysates which act as chemo-attractants for rhizobacteria. The healthy rhizosphere facilitates plant growth promoting rhizobacteria (PGPR) to establish the endophytic association [9,10] or non-symbiotic associations with host plants. However, indiscriminate use of chemicals has resulted a detrimental impact on the health of the rhizosphere ecosystem. The practice of a natural farming system offers some hope; however, the complete package of practices based on natural farming needs to be worked out for proper recommendations to the farmers. The natural farming system, also known as organic or sustainable farming, has received popularity for its emphasis on environmental stewardship, soil health, and reduced dependence on synthetic inputs. Thus, in the current investigation, we have explored the integration of jeevamrut, vermitea, or neem cake with biofertilizers, including phosphate solubilizing bacteria (PSB), azotobacter, and vesicular arbuscular mycorrhiza (VAM).
Jeevamrut is considered one of the important components of Zero Budget Natural Farming [11]. It contains beneficial microorganisms which ensure the mineralization of organic matter or humus to mobilize fixed nutrients [12] and acts as a nutrient reservoir that can improve soil fertility and crop productivity [13]. Vermitea is an aqueous extract from vermicompost that is rich in major nutrients and PGPR [14]. It also contains plant growth promoter’s analog to auxins and cytokinin which could be accountable for improved nutrient and microbial levels of jeevamrut. Adding neem cake will improve the nutritional value as well as the insecticidal potential of jeevamrut. The fortification of jeevamrut with vermitea or neem cake could be a novel approach for the improvement of soil nutrient status for commercial potato cultivation.
Biofertilizers, consortia of living microorganisms, have the ability to dissolve the fixed and unavailable nutrients for making them available to the plants [15,16]. Azotobacter is a free-living bacterium that promotes nitrogen sequestration from atmosphere, synthesizes plant growth promoters such as auxin, cytokinin, and gibberellin, and stimulates microbial activities in rhizosphere to enhance the uptake of nutrients by plants [17,18]. The PSB increase phosphatase activities and release organic acids to lower the pH of the rhizosphere [19,20]. The acid hydrolysis of a bound form of phosphate results in its solubilization and increases the level of available phosphorus in soil [21]. The VAM fungi are involved in the mineralization of organic matter, the production of growth-promoting substances, and the development of tolerance to soil-based stresses in plants. Co-inoculation of VAM with PSB has many fold advantages by decomposition and mineralization of organic matter to release major nutrients in the soil [22].
Although a number of studies have been carried out to explore the potential of various biofertilizers to improve the productivity and fertility of the soil, it is essential to work out the physicochemical and microbial parameters of soil in relation to the application of microbial consortia (PSB and Azotobacter or VAM) in combination with the enriched jeevamrut formulation. Thus, the investigation was carried out with the hypothesis that the combined application of fortified jeevamrut and plant growth-promoting microorganisms would attenuate the negative effects of synthetic fertilizers over microbial and physicochemical properties of soil.
2. MATERIALS AND METHODS
2.1. Experimental Area and Materials
The experiment under the above topic was performed at the CRC farm of agriculture at ITM University, Gwalior, India. The investigation site is located under humid and subtropical climatic conditions and located at an elevation of 412 meters above mean sea level in the gird region of north Madhya Pradesh. The experiment was repeated for 2 years from 2021–2023. Kufri Bahar, a hybrid of Kufri Red × Gineke, which was released by Central Potato Research Institute, Shimla, was used for experimentation due to its commercial acceptability in the region. It has a semi-compact canopy and matures within 100–120 days with an average potential yield of 300–350 q ha-1.
2.2. Experimental Details
2.2.1. Treatment details
The treatments consisted of two factors: Factor-J (Jeevamrut) of 4 levels and Factor B (Biofertilizers) of 5 levels, resulting in 20 treatment combinations. The gross experimental area was 528 m2 while the net experimental area was 345.6 m2 and planting was done at the spacing of 60 cm (row) × 20 cm (plant). The fertilizer application was done as per the recommended dose of N: P:K at the rate of 180:80:120 kg ha-1 (recommended by Central Potato Research Institute–Regional Station, Gwalior). The details of doses and applications of fertilizers and treatments are discussed in Table 1.
Table 1: Details of treatment formulation and application.
| Factor J | Jeevamrut formulation | Details of formulation and application |
|---|---|---|
| J0 | No Jeevamrut | Only inorganic fertilizers were applied* |
| J1 | Jeevamrut | 100 l of water + 10 kg of cow dung + 10 l of cow urine + 2 kg of jaggery + 2 kg of gram flour. The mixture was allowed for fermentation for 10 days |
| J2 | Jeevamrut + Vermitea | 10 l of Vermitea were mixed with J1 at the time of application |
| J3 | Jeevamrut + Neem cake | 10 kg of Neem cake powder was mixed with J1 at the time of application |
| Factor B | Biofertilizer (s) | Details of formulation and application |
| B0 | No biofertilizers | Only inorganic fertilizers were applied* |
| B1 | Azotobacter | 100 ml of Azotobacter in 10 l of water |
| B2 | VAM (Vesicular Arbuscular Mycorrhiza) | 100 g of VAM in 10 l of water |
| B3 | PSB (Phosphate Solubilizing Bacteria) + Azotobacter | 100 ml of Azotobacter 100 mL of PSB in 10 l of water |
| B4 | PSB + VAM (Vesicular Arbuscular Mycorrhiza) | 100 g of VAM and 100 mL of PSB in 10 liters of water |
* Only N: P: K @ 180:80:120 kg ha-1 (recommended by Central Potato Research Institute-Regional Station, Gwalior.
2.2.2. Climatic conditions and soil attributes of the experimental area
The experimental area received an average annual rainfall of 900 mm where monsoon started in June and remained active till September. Although occasional showers were also reported during an investigation (winter season), it was a cool and dry period. The initial properties of the soil are: the sand: silt: clay as 66:22.50:15.10 (sandy loam), pH of 8.04, electrical conductivity (EC) of 0.25 dSm-1, organic carbon (OC) of 0.65 %, available nitrogen of 198.45 kg ha-1, available phosphorus of 12.55 kg ha-1, exchangeable potassium of 155.65 kg ha-1. The details of chemical and microbial attributes of experimental soil and different formulations applied during an investigation are given in Table 2.
Table 2: Details of chemical and microbial properties of soil along with various formulations of Jeevamrut.
| Microbial count | J1: Jeevamrut | J2: Jeevamrut + Vermitea | J3: Jeevamrut + Neem cake | Experimental Soil (dry) |
|---|---|---|---|---|
| Bacterial count (cfu mL-1) | 8.75×106 | 48.42×106 | 35.67×106 | 66×106 cfu g-1 |
| Fungal count (cfu mL-1) | 1.35×104 | 4.06×104 | 3.88×104 | 1.21×106 cfu g-1 |
| Actinomycetes (cfu mL-1) | 3.78×104 | 18.78×104 | 12.46×104 | 0.82×106 cfu g-1 |
| Chemical Properties | J1: Jeevamrut | J2: Jeevamrut + Vermitea | J3: Jeevamrut + Neem cake | Soil |
| pH | 4.65 | 4.43 | 5.57 | 8.04 |
| EC (dSm-1) | 0.56 | 0.67 | 0.66 | 0.25 |
| Carbon (g l-1) | 6.78 | 6.81 | 6.79 | 0.65% |
| Nitrogen (g l-1) | 0.35 | 0.56 | 0.71 | 198.45 kg ha-1 |
| Phosphorus (g l-1) | 0.05 | 0.22 | 0.19 | 12.55 kg ha-1 |
| Potassium (g l-1) | 0.65 | 0.83 | 0.81 | 155.65 kg ha-1 |
2.2.3. Agronomical operations
After harvesting of previous crop (rice), the primary tillage practices were carried out to give a proper tilth. The farm yard manure of 10 tons per hectare was incorporated 30 days before the sowing of potatoes. Seed potato tubers were taken out from cold storage and kept in the shade for 15 days before planting to accelerate the sprouting. The seeds of uniform size (45–50 mm diameter) were planted manually at a uniform distance of ten centimeters between plants. The basal application of fertilizers and other treatments was carried out as per details given in Table 1 at 3 days before planting in the experimental field. Earthing up was done 30 days after planting along with the manual weeding. The exposed tubers were covered to avoid disease and rotting of tubers and to prevent the synthesis of solanin which is responsible for the greening of potato tubers. Five light irrigations (5 cm) were given at 15, 30, 45, 60, and 75 days after the sowing of the potato crop. Harvesting was done in March 2021–2022 and 2022–2023 manually with the help of kudal.
2.3. Observations Recorded
2.3.1. Physical properties of soil
Bulk density and particle density value for soil was calculated using the formula [23]:
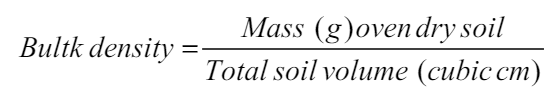
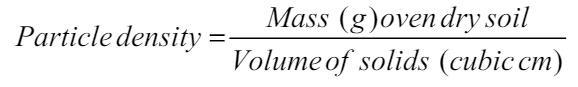
The porosity of soil was estimated by the Piper method (1966) as per the given formula:
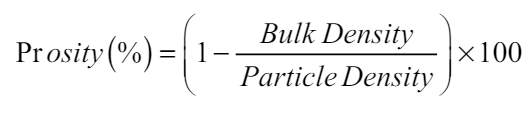
The soil particles were segregated as per the given particle diameter: sand (2–0.05 mm), silt (0.05–0.002 mm), and clay (<0.002 mm). To determine the soil texture, the percentages of sand, silt, and clay were calculated from the laboratory as per the given formula [23], and soil texture was determined using a soil triangle:
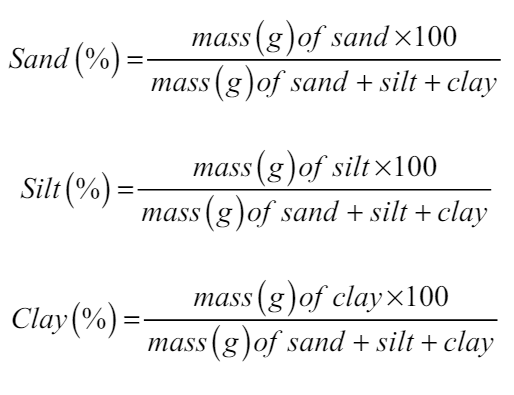
2.3.2. Microbial properties of soil
The microbial populations were estimated by the serial dilution and pour plate techniques using agar media [24]. The media were prepared and sterilized in an autoclave at 121°C and 15 psi for 15 min, while pre-sterilized agar plates were used for plating of diluted samples in triplicates. The agar plates for microbes were incubated at 30+1°C in an inverted position for 5–7 days until countable colonies were developed. The respective colonies were counted on the basis of their morphological characteristics and growth pattern and the microbial population was expressed as colony-forming units (cfu) ml-1.
2.3.3. Chemical properties of soil
The soil samples were randomly collected from four points of each plot and thoroughly mixed to make representative samples for further analysis before planting and after harvesting of crops. Standard protocols were used to determine the various soil parameters. OC, available nitrogen, phosphorus, and potassium were estimated in the process as advocated by Jackson [25]. Soil pH was determined using a pH meter through a suspension of 1: 2.5 soil in water [25] as described by Schwyter and Vaughan [23]. EC was determined using the procedure given by Schwyter and Vaughan [23] with the help of an EC meter. The details of the physico-chemical and microbial properties of soil along with various formulations of jeevamrut is given in Table 2. This value was taken as the initial control for estimating the post-harvest properties of the experimental field.
2.4. Statistical Analysis
The statistical analysis was carried out on the observations recorded on various microbial, physical, and chemical parameters of soil using a two-way analysis of variance at 0.05 probability level in Excel and the SPSS software. The strength of the relationship of initial microbial count in jeevamrut with the various soil attributes was estimated as Pearson’s correlation coefficient. The principal component analysis (PCA) was carried out using OPSTAT software to reveal the existing unexpected associations among variables.
3. RESULTS AND DISCUSSION
3.1. Soil Physical Attributes
The physical attributes of soil including bulk density, particle density, and soil texture (sandy, silt, and clay) were estimated before planting (taken as IC-Initial Control) and after harvesting [Figure 1] in both the years of observation. Bulk density has having strong correlation with a stock of carbon and nutrients in soil [26]. Soil porosity is primarily dependent on soil structure; however, the impact of vegetation and soil use patterns cannot be ignored. As per the available proportion of sand, silt, and clay particles (66: 22.50: 15.10), the soil of the experimental area was reported as silt loam. There was no noticeable variation reported before and after the crop. The bulk density was noticed to range from 1.53 to 1.55 g cm-3 in year 1 and 1.55 to 1.56 g cm-3 in year 2 with initial control of 1.53 and 1.55 g cm-3, respectively. The particle density was varied from 2.63 to 2.65 g cm-3 in year 1 and 2.64 to 2.66 g cm-3 in year 2 with initial control of 2.64 g cm-3. The porosity of the experimental soil was reported to range from 41.36 to 42.13 in year 1 and from 41.25 to 41.77 in year 2.
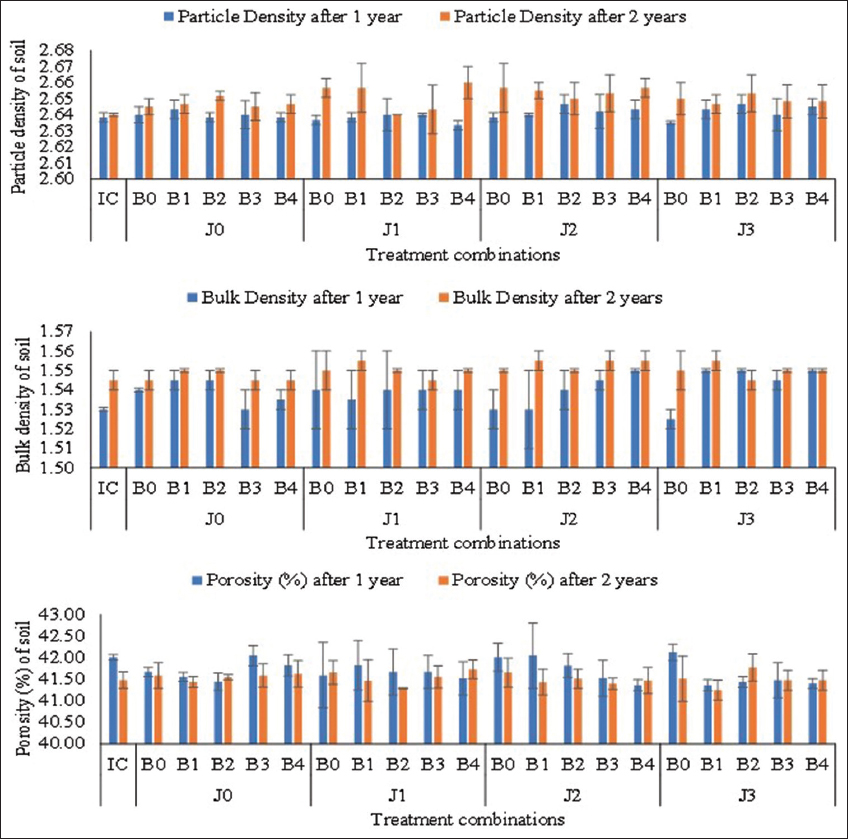 | Figure 1: Physical properties of soil after harvesting of crops under different treatment combinations (J0: No Jeevamrut, J1: Jeevamrut, J2: Jeevamrut + Vermitea, J3: Jeevamrut + Neem cake, B0: No biofertilizers, B1: Azotobacter, B2: Vesicular Arbuscular Mycorrhiza [VAM], B3: Phosphate Solubilizing Bacteria [PSB] + Azotobacter, B4: PSB + VAM, IC: Initial control). [Click here to view] |
The present study observed a non-significant variation in porosity, particle density, and bulk density in the experimental areas which could be due to the static nature of these physical attributes in a short duration. However, the climatic factor and land use pattern are not static in the long term and are closely related to the hydro-ecological and bio-geochemical cycling within that ecosystem [27]. These favors, the ecological succession to bring alteration in the structure and porosity of the soil. The distribution and magnitude of soil porosity regulate the water holding capacity, soil aeration, and microbial diversity which in turn play a significant role in biogeochemical and hydrological cycling so any change in soil structure due to compaction is one of the serious threats to vegetation and biogeochemical cycling [28]. Hence, it is important to maintain proper aeration and drainage in soil for effective improvement in soil nutrient availability. While most ecological models consider porosity as a constant parameter, its dynamic nature is also being recognized in response to changes in climate and land use patterns in the long run [29,30]. Bulk density of soil is used to determine stocks of carbon which is one of the three sub-indicators used to calculate the extent of land degradation under UN SDG Indicator 15.3.1 [26,31].
3.2. Soil Microbial Properties
The soil microbial population was significantly improved due to the application of different formulations of jeevamrut or biofertilizers alone or in combination [Table 3, Supplementary Table 1, Figure 2] in comparison to estimates taken before the experiment [Table 2]. This might be due to the existence of favorable conditions in the rhizosphere region after the addition of jeevamrut or biofertilizers under different treatments [32]. The fermented organic liquid (jeevamrut) contains microbial population and plant-promoting substances which was helpful in buffering the rhizosphere for further co-multiplication of these microbes [33]. Further, the addition of vermitea (J2) or neem cake (J3) during the preparation of jeevamrut-based formulation improved microbial counts. These additives acted as bio-enhancers or catalysts and supplied essential nutrients for the growth and multiplication of beneficial microbes [34,35]. The microbial strength present in jeevamrut or biofertilizers secrete proteins, organic acids, and antioxidants to transform soil organic matter into energy which are supportive of the growth of useful microbes in the rhizosphere [36,37].
Table 3: Microbial count in the soil after harvesting of crops under different treatments.
| Factors | Bacterial count (106 cfu g-1) | Fungal count (104 cfu g-1) | Actinomycetes count (104 cfu g-1) |
|---|---|---|---|
| J0 (control) | 76.91±7.91c | 155.61±24.44c | 96.01±8.28d |
| J1 | 79.93±7.99c | 156.22±23.70b | 98.76±7.36c |
| J2 | 95.83±7.83a | 157.40±24.18a | 108.00±7.69a |
| J3 | 90.70±7.80b | 157.54±23.70a | 103.96±6.44b |
| CD (at 0.05) | 0.427 | 0.608 | 0.855 |
| SE (m) ± | 0.149 | 0.211 | 0.298 |
| P-value | 2.65×10-17** | 9.47×10-05** | 4.37×10-09** |
| B0 (control) | 77.29±9.12d | 125.98±1.19e | 93.09±5.81e |
| B1 | 85.63±8.83c | 136.80±1.20d | 101.28±4.87c |
| B2 | 79.35±8.85d | 170.94±0.92b | 96.12±5.69d |
| B3 | 96.33±8.84a | 167.72±0.87c | 111.04±4.57a |
| B4 | 90.61±8.94b | 182.02±0.90a | 106.89±6.11b |
| CD (at 0.05) | 0.478 | 0.679 | 0.956 |
| SE (m) ± | 0.166 | 0.236 | 0.333 |
| P-value | 1.88×10-16** | 6.34×10-21** | 1.57×10-10** |
All values are mean±SD values of three replications, J0: No Jeevamrut (control), J1: Jeevamrut, J2: Jeevamrut + Vermitea, J3: Jeevamrut+Neem cake, B0: No biofertilizers (control), B1: Azotobacter, B2: VAM (Vesicular Arbuscular Mycorrhiza), B3: PSB (Phosphate Solubilizing Bacteria) + Azotobacter, B4: PSB + VAM (Vesicular Arbuscular Mycorrhiza),*0.05 level of significance,
** 0.01 level of significance.
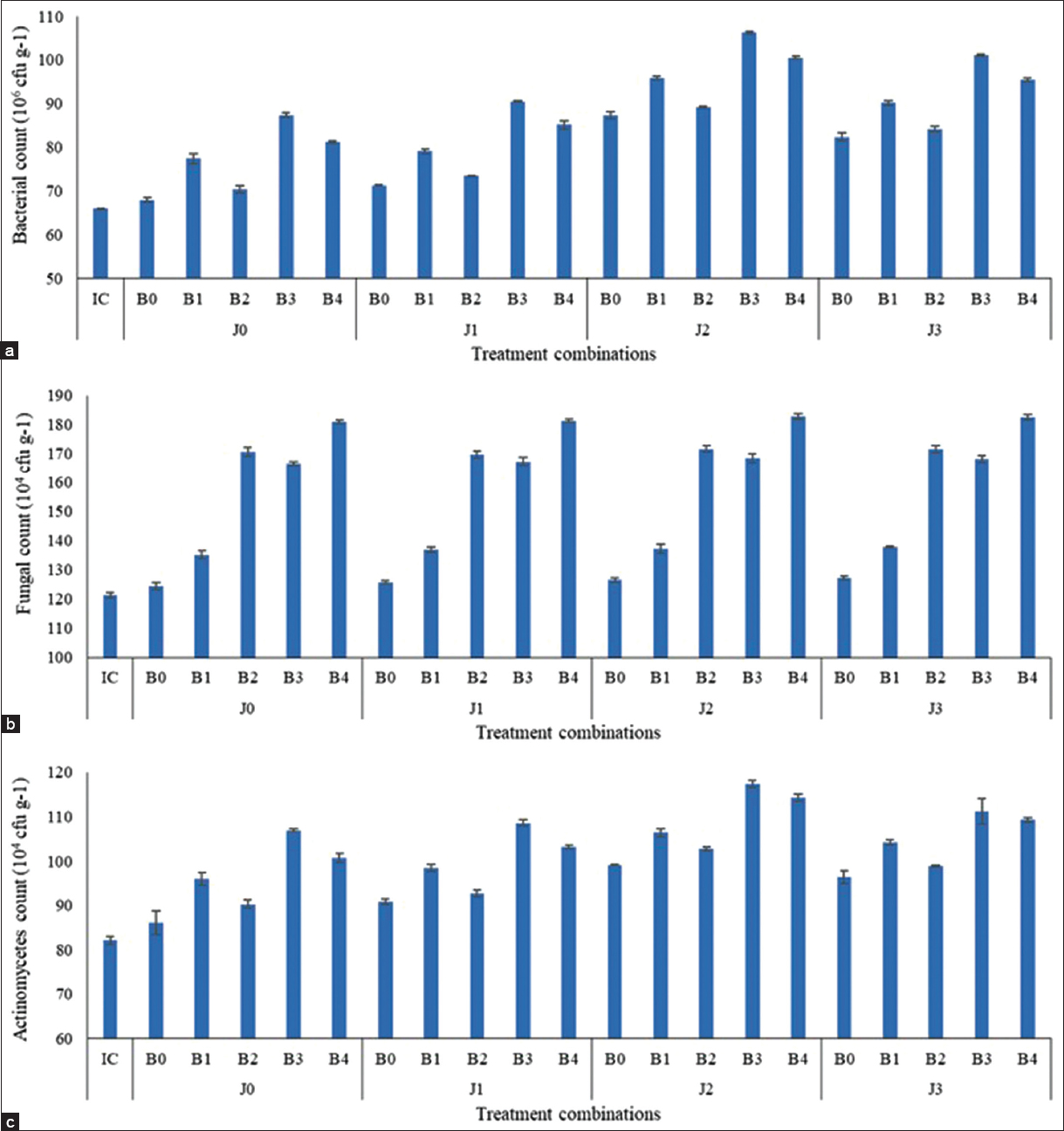 | Figure 2: (a-c) Microbial count in the soil after harvesting of crops under different treatment combinations (J0: No Jeevamrut, J1: Jeevamrut, J2: Jeevamrut + Vermitea, J3: Jeevamrut + Neem cake, B0: No biofertilizers, B1: Azotobacter, B2: Vesicular arbuscular mycorrhiza [VAM], B3: Phosphate solubilizing bacteria [PSB] + Azotobacter, B4: PSB + VAM, IC: Initial control). [Click here to view] |
Bacterial and actinomycetes counts were significantly improved due to the application of Azotobacter and/or PSB (B3 > B4 > B1) in combination with jeevamrut while the fungal count was enhanced due to application of VAM (B4 > B2). VAM develops a synergetic interaction with other beneficial microorganisms of the rhizosphere due to the strong capability of co-inoculation with other biofertilizers [22] and better multiplication of these microbes was observed when applied in combinations [38]. The enrichment of soil microbial population after application of jeevamrut and biofertilizers might be due to favorable rhizospheric micro-environment developed due to root exudates, soil aggregation, decomposition of root cells and organic matter, availability of plant nutrients, and other physical-biochemical processes resulting a higher microbial abundance [39-44].
3.3. Soil Chemical Attributes
The chemical attributes of soil including pH, EC, OC, available N, P, and K were estimated before planting (initial value) and after harvesting of potato crops [Tables 4 and 5, Figures 3 and 4] in both the years of observation.
Table 4: Soil pH, EC (dSm-1), and OC (%) after harvesting of crops in year 1 and year 2 under different treatments.
| Factors | pH (after year 1) | pH 2 (after year 2) | EC (after year 1) | EC (after year 2) | OC (after year 1) | OC (after year 2) |
|---|---|---|---|---|---|---|
| J0 (control) | 8.01±0.05a | 7.96±0.08a | 0.249±0.003a | 0.247±0.005a | 0.615±0.017c | 0.660±0.012b |
| J1 | 7.76±0.08c | 7.53±0.10c | 0.240±0.001a | 0.232±0.003c | 0.638±0.009b | 0.687±0.008a |
| J2 | 7.73±0.09d | 7.45±0.17d | 0.238±0.004c | 0.227±0.007d | 0.645±0.011ab | 0.692±0.011a |
| J3 | 7.88±0.03b | 7.73±0.06b | 0.240±0.001b | 0.240±0.001b | 0.649±0.011a | 0.693±0.008a |
| CD (at 0.05) | 0.013 | 0.036 | 0.001 | 0.002 | 0.011 | 0.012 |
| SE (m) ± | 0.005 | 0.013 | 0.001 | 0.001 | 0.004 | 0.004 |
| P-value | 1.97×10-07** | 2.08×10-08** | 3.75×10-05** | 4.16×10-06** | 6.24×10-05** | 4.84×10-07** |
| B0 (control) | 7.93±0.10a | 7.80±0.19a | 0.243±0.005a | 0.241±0.006a | 0.620±0.022c | 0.670±0.016b |
| B1 | 7.87±0.10b | 7.72±0.18b | 0.243±0.005a | 0.238±0.009b | 0.638±0.020b | 0.680±0.019b |
| B2 | 7.86±0.15b | 7.68±0.25b | 0.243±±0.005a | 0.237±0.010b | 0.638±0.015b | 0.683±0.016ab |
| B3 | 7.77±0.14d | 7.54±0.25d | 0.238±0.006b | 0.233±0.010c | 0.642±0.011ab | 0.687±0.019ab |
| B4 | 7.81±0.15c | 7.59±0.28c | 0.243±0.005a | 0.233±0.010c | 0.650±0.010a | 0.695±0.010a |
| CD (at 0.05) | 0.015 | 0.041 | 0.001 | 0.002 | 0.012 | 0.013 |
| SE (m) ± | 0.005 | 0.014 | 0.001 | 0.001 | 0.004 | 0.005 |
| P-value | 0.00072** | 0.0002** | 0.033* | 0.021* | 0.00092** | 0.0092** |
All values are mean±SD values of three replications, J0: No Jeevamrut (control), J1: Jeevamrut, J2: Jeevamrut+Vermitea, J3: Jeevamrut + Neem cake, B0: No biofertilizers (control), B1: Azotobacter, B2: VAM (Vesicular Arbuscular Mycorrhiza), B3: PSB (Phosphate Solubilizing Bacteria) + Azotobacter, B4: PSB + VAM (Vesicular Arbuscular Mycorrhiza),
* 0.05 level of significance,
** 0.01 level of significance.
Table 5: Soil N, P, and K (kg ha-1) of soil after harvesting of crops under different treatment combinations.
| Factors | N1Y | N2Y | P1Y | P2Y | K1Y | K2Y |
|---|---|---|---|---|---|---|
| J0 | 210.17±2.36d | 216.47±1.61c | 13.61±0.29c | 14.83±0.53c | 161.07±2.58d | 166.07±2.58d |
| J1 | 213.40±4.79c | 220.00±3.76b | 13.88±0.23b | 15.01±0.59b | 164.83±2.32c | 169.20±2.47c |
| J2 | 218.57±5.85a | 226.27±5.71a | 14.45±0.34a | 15.38±0.73a | 171.43±2.24a | 179.67±3.66a |
| J3 | 216.20±5.68b | 224.07±5.72a | 14.52±0.27a | 15.40±0.62a | 167.17±1.20b | 171.40±0.72b |
| CD (at 0.05) | 1.686 | 2.276 | 0.191 | 0.120 | 0.617 | 0.933 |
| SE (m) ± | 0.587 | 0.792 | 0.067 | 0.042 | 0.215 | 0.325 |
| P-value | 0.0002** | 8.23 × 10-05** | 5.39 × 10-11** | 2.36 × 10-06** | 4.94 × 10-09** | 8.51 × 10-09** |
| B0 | 208.08±1.85d | 218.25±2.75c | 13.69±0.39c | 14.18±0.20c | 163.04±5.20d | 167.67±5.36c |
| B1 | 213.75±2.38c | 220.67±4.30bc | 14.03±0.49b | 15.07±0.22b | 165.25±3.49c | 171.33±5.30b |
| B2 | 213.92±4.01c | 219.75±3.75bc | 14.13±0.45b | 15.18±0.30b | 167.17±4.40b | 172.83±6.17a |
| B3 | 220.46±6.31a | 228.75±8.02a | 14.34±0.42ab | 15.61±0.34a | 167.17±4.67b | 172.58±6.59a |
| B4 | 216.71±4.18b | 221.08±3.21b | 14.38±0.45a | 15.74±0.37a | 168.00±4.14a | 173.50±6.12a |
| CD (at 0.05) | 1.885 | 2.545 | 0.216 | 0.134 | 0.690 | 1.044 |
| SE (m) ± | 0.656 | 0.886 | 0.074 | 0.047 | 0.240 | 0.363 |
| P-value | 3.39×10-05** | 0.00024** | 1.84×10-08** | 5.15×10-10** | 4.06×10-05** | 0.00026** |
All values are mean±SD values of three replications, J0: No Jeevamrut (control), J1: Jeevamrut, J2: Jeevamrut + Vermitea, J3: Jeevamrut + Neem cake, B0: No biofertilizers (control), B1: Azotobacter, B2: VAM (Vesicular Arbuscular Mycorrhiza), B3: PSB (Phosphate Solubilizing Bacteria) + Azotobacter, B4: PSB + VAM (Vesicular Arbuscular Mycorrhiza), *0.05 level of significance,
** 0.01 level of significance.
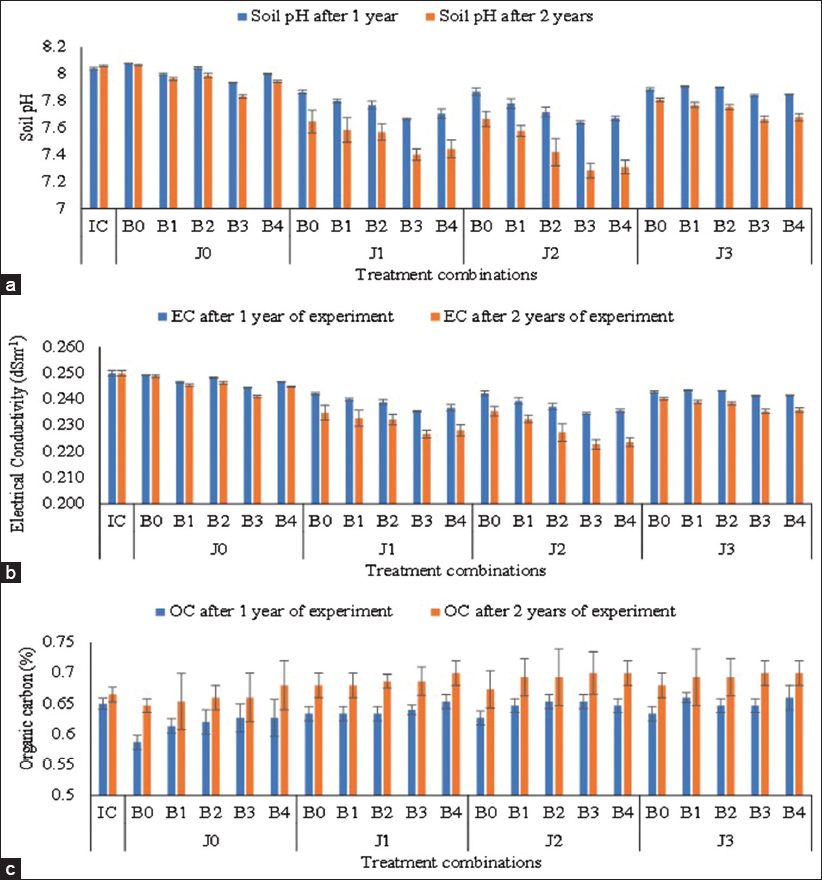 | Figure 3: (a-c) Soil pH, EC, and OC after harvesting of crops under different treatment combinations (J0: No Jeevamrut, J1: Jeevamrut, J2: Jeevamrut + Vermitea, J3: Jeevamrut + Neem cake, B0: No biofertilizers, B1: Azotobacter, B2: Vesicular arbuscular mycorrhiza [VAM], B3: Phosphate solubilizing bacteria [PSB] + Azotobacter, B4: PSB + VAM, IC: Initial control). [Click here to view] |
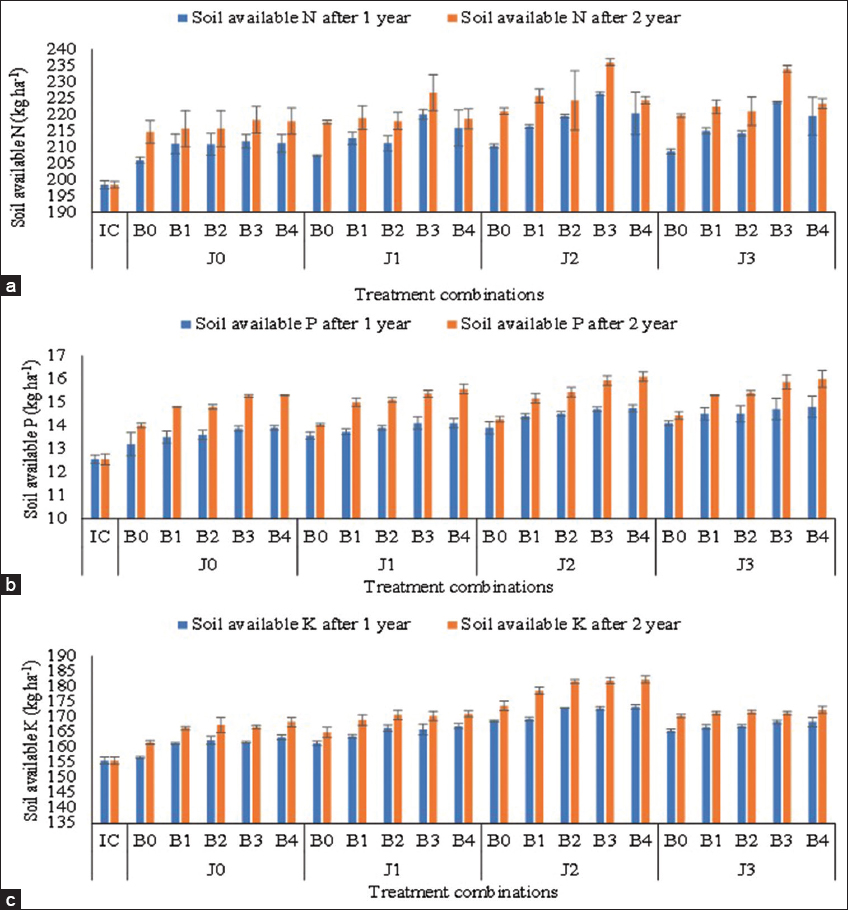 | Figure 4: (a-c) Soil Available N, P and K after harvesting of crops under different treatment combinations (J0: No Jeevamrut, J1: Jeevamrut, J2: Jeevamrut + Vermitea, J3: Jeevamrut + Neem cake, B0: No biofertilizers, B1: Azotobacter, B2: Vesicular arbuscular mycorrhiza [VAM], B3: Phosphate solubilizing bacteria [PSB] + Azotobacter, B4: PSB + VAM, IC: Initial control). [Click here to view] |
3.3.1. Soil pH
The initial soil pH before planting was reported as 8.04 which were significantly reduced to 7.73 and 7.45 after application of jeevamrut + vermitea in year 1 and year 2, respectively [Table 4]. A significant reduction (3.33% and 6.14%) in pH was also reported after the application of PSB with Azotobacter (7.77 and 7.54) in consecutive years. The interaction effect of jeevamrut formulation and biofertilizers was also significant with the lowest pH in J2B3 and J2B4 [Figure 3 and Supplementary Table 2]. The finding confirms that the application of jeevamrut and biofertilizers such as PSB, Azotobacter, or VAM with a reduction of inorganic fertilizers up to 50% provided a constant supply of major nutrients to the plants and made the fixed micronutrients easily available. Further, the increased microbial population is responsible for developing an acidic medium through the decomposition of organic matter which could be responsible for lowering of pH of soil [38].
Jeevamrut formulation is dominated with the fungal members of Ascomycota and Basidiomycota phyla [11,45,46] while vermitea formulation is enriched with bacterial members of Proteobacteria, Bacteroidetes, Firmicutes, Verrucomicrobia, and Chloroflexi and the fungal members of Ascomycota, Basidiomycota, Cryptomycota, Entomophthoromycota, and Glomeromycota phyla which are involved in nutrient recycling in agri-ecosystem through microbial action on organic matter [47]. Jeevamrut contains cow urine which is a source of amino acid and can impart resistance against the pathogen [12] and various formulations of jeevamrut have been reported effective in inhibiting Alternaria alternata in vitro with more than 90% mycelial growth inhibition [48]. Further, vermitea has been reported to enhance the suppression of soil or air borne diseases. Application of the commercial compost tea inhibited the growth of A. solani mycelium (up to 74%), Rhizoctonia solani (isolate 422) (up to 85%), and R. solani (isolate 299) (up to 36%) in potato [47]; and suppressed the growth of Gray Mold (Botrytis cinerea) in geranium [49]. The efficacy has been reported to be influenced by the method of preparation, dilution ratio, application equipment, timing, rates, spray adjuncts, and supplementing specific microbial antagonists [50]. However, the details of mechanism for imparting disease resistance are yet to be explored. In the current study, the application of vermitea with jeevamrut has the additional advantage of developing disease resistance and enhancing the microbial actions on soil organic matters which in turn improves the soil pH, soil EC, and availability of nutrients; however, there was no substantial change in soil OC [51].
3.3.2. Soil EC
The initial soil EC before planting was reported as 0.25 dSm-1 which was significantly reduced to 0.238 dSm-1 and 0.227 dSm-1 after application of jeevamrut + vermitea in consecutive years [Table 4]. A significant reduction in EC was also reported after the application of PSB with Azotobacter (0.238 dSm-1 and 0.233 dSm-1). There was a significant interaction between jeevamrut and biofertilizers with the lowest EC in J2B3 and J2B4 [Figure 3 and Supplementary Table 3]. The finding confirms that the application of jeevamrut and biofertilizers such as PSB, Azotobacter, or VAM has resulted in better uptake of metallic ions by the plants resulting in lowering of EC of soil. The microbial populations present in soil are involved in the secretion of extracellular enzymes which are involved in electrolytic balance in the soil for improved nutrient absorption and assimilation by the plants [52]. The VAM fungi have a strong impact on lowering of EC and maintaining the ionic balance in soil which might be associated with the ability of these fungi to mobilize the mineral ions by direct uptake and translocation of ions to plants through mycorrhizal hyphae [53]. VAM adheres to the plants rhizoids which lead to the development of fungal hyphae. These hyphae further penetrate and form arbuscules within the root cortical which leads to the significant increase in rhizosphere resulting in improvement of nutrient uptake by plants. Further, these fungi developed intracellular vesicles as terminal swellings between the fungal hyphae and the host plant that acts as a storehouse for complex carbon compounds and mineral nutrients [54].
3.3.3. Soil OC
The initial soil OC before planting was reported as 0.65% which was significantly increased to 0.693% and 0.692% after the application of jeevamrut + neem cake and jeevamrut + vermitea, respectively, in year 2 [Table 4]. A significant improvement in OC was also reported after the application of PSB + VAM and PSB + Azotobacter (0.695% and 0.687%, respectively) in year 2. However, the interaction effect of jeevamrut formulation and biofertilizers was also not significant with the highest OC in J2B3 and J2B4 [Figure 3 and Supplementary Table 4]. Sharma [55] and Adekiya et al. [56,57] had also confirmed improvement in the soil physicochemical properties including soil OC after the application of jeevamrut. The strength of microbial population in the soil is largely affected by the soil environment which in turn is influenced by the application of fertilizers and the jeevamrut and/or biofertilizers in various treatments. The investigation by Vieira and Nahas [58] confirmed that the microbial population was significantly influenced by the nature of soil and plants as the bacterial and fungal counts were higher in agricultural soil (sorghum) followed by eucalyptus and forest soil. They have reported that the counts of spore-forming, gram-negative bacteria, and actinomycetes were higher in forest soil followed by eucalyptus and agricultural soil. The genome metagenomic analysis confirms the presence of proteobacteria including Rhizobium, Pseudomonas, and Bacillus, fungal hyphae, and enzymes regulating the protein and carbohydrate metabolisms in jeevamrut which might be contributing factors toward soil OC, soil fertility, and plant growth [11].
3.3.4. Soil available N
The available nitrogen was reported to be 198.45 kg ha-1 before planting and significantly increased to 218.57 and 226.27 kg ha-1 after the application of jeevamrut + vermitea in year 1 and year 2, respectively [Table 5]. A significant increase in available nitrogen was also reported after the application of PSB with Azotobacter (220.46 and 228.75 kg ha-1) in consecutive years. The interaction effect of jeevamrut formulation and biofertilizers was significant in year 1 while non-significant in year 2 with the highest available N in J2B3 and J3B3 [Figure 4 and Supplementary Table 5]. The finding confirms that the application of jeevamrut and biofertilizers like PSB, Azotobacter, or VAM has resulted in the fixation of free atmospheric nitrogen in the microbial bodies which on decomposition were able to release a significant amount of nitrogen in soil [11,59]. Further, the jeevamrut formulation containing vermitea was able to enhance the population of Azotobacter to increase the magnitude of nitrogen fixation [14]. Saharan et al. [11] had reported approximately 98%, 23%, 62%, 55%, 46%, 439%, and 142% increases in zinc, iron, copper, manganese, OC, phosphorus, and potassium, respectively in soil after application of jeevamrut for two consecutive years. Azotobacter utilizes atmospheric nitrogen for the synthesis of cellular protein which on mineralization provides nitrogen to the soil. The sequestration of free atmospheric nitrogen by Azotobacter is regulated by iron-rich nitrogenases which bring reduction of nitrogen during the process. These bacteria are also known to release siderophores which make the metallic nutrients available to the plants through chelation [60]. Laboratory study by Wang et al. [61], aimed to study the dynamic growth of bacteria in incubation soil, reflected the peak of the total bacterial count at the 30th day during inoculation of mixed bacterial addition where the pattern of growth in both, PSB and N2- fixing bacteria, was different. The growth of PSB declined in the past 30 days while the growth of N2-fixing bacteria increased. Although the competition of mixed bacteria retarded the peaking time increased the maximum. Thus, the co-inoculation of these bacteria with PSB can be a more effective tool for soil fertility management [62].
3.3.5. Soil available P
The available phosphorus was reported to be 12.55 kg ha-1 before planting and significantly increased to 15.40 and 15.38 kg ha-1 after application of jeevamrut + neem cake and jeevamrut + vermitea, respectively, in year 2 [Table 5]. A significant increase in available phosphorus in year 2 was also reported after the application of PSB with VAM (15.74 kg ha-1) and PSB with Azotobacter (15.61 kg ha-1). The interaction effect of jeevamrut formulation and biofertilizers was significant in year 1 while non-significant in year 2 with the highest available P in J3B4, J2B4, J2B3, and J3B3 [Figure 4 and Supplementary Table 6]. The finding confirms that the application of jeevamrut and neem cake in combination with biofertilizers such as PSB, Azotobacter, or VAM has resulted in the dissolution of unavailable P to available forms which were able to increase the soil phosphorus. The presence of a greater microbial population in the jeevamrut formulation accelerated the decomposition of soil organic matter to improve soil fertility and nutrient availability to plants [33,38]. Although PSB is a non-symbiotic bacteria, it is beneficial for plants in many ways and can effectively be used under saline soil to increase soil phosphorus availability [63]. Further, its co-inoculation with Azotobacter, Rhizobium, or VAM fungi has a significant impact on soil nutrient status [64,65]. PSB as a biofertilizer has the potential to nullify the effect of induced salinity or alkalinity in calcareous soil and improve P availability through soil acidification in this condition [66]. The PSB releases a proton (H+), phenolics, siderophores, organic acids, and mineral acids which could be involved in the dissolution of precipitated P like Ca3(PO4)2 [67-72]. These exudates from PSB are involved in the chelation of the cations bound to phosphate through their hydroxyl and carboxyl groups, thereby converting them into soluble forms Chen et al. [73]. It is also able to release plant growth-promoting substances which results in increased availability of micronutrients including iron (Fe+2) and zinc (Zn+2). The PSBs are involved in the conversion of the insoluble form of phosphorus to the available form while Azotobacter is both solubilizing and mineralizing P bacteria so in the consortia they work in a complementary manner to improve the availability of P to the plants [72]. Thus, the co-inoculation of these bacteria can be a more effective tool for the improvement of P availability to the plants.
3.3.6. Soil available K
The available potassium was reported to be 155.65 kg ha-1 before planting and significantly increased to 179.67 and 171.40 kg ha-1 after application of jeevamrut + vermitea and jeevamrut + neem cake, respectively, in year 2 [Table 5]. A significant increase in available potassium in year 2 was also reported after the application of PSB with VAM (173.50 kg ha-1), VAM alone (172.83 kg ha-1), and PSB with Azotobacter (172.58 kg ha-1). The interaction between jeevamrut formulation and biofertilizers was significant with the highest available K in J2B4, J2B3, and J2B2 [Figure 4 and Supplementary Table 7]. The finding confirms that the application of jeevamrut and vermitea in combination with biofertilizers such as PSB, Azotobacter, or VAM has resulted in the dissolution of unavailable K to available form which was able to increase the significant amount of potassium in soil. The microbial species available in jeevamrut and biofertilizers were able to improve the quantity of soil microorganisms which are involved in the decomposition (humification and mineralization) of soil organic matters and acted as nutrient reservoirs responsible for soil fertility improvement and stable but dynamic soil ecosystems [74-77]. Jeevamrut enriches the soil with nutrients and improves soil fertility by buffering the soil pH in acidic as well as alkaline soil to make the soil nutrients available to the plants [38]. The release of organic acids and enzymes due to co-inoculation of VAM and PSB might be accountable to the enhanced dissolution of complex minerals to available form and increase in K content in the rhizosphere soil [22]. It has also been observed that the biofertilizer application improved the level of available phosphorus and potassium content in soil when it was supplied in combination with other organic nutrient sources [78].
3.4. Correlation (r) of various soil parameters with microbial count
The correlation study of initial microbial count in jeevamrut with the various soil attributes estimated after harvesting of potato crops in year 1 and year 2 was estimated and it was reported that after harvesting of the crop, the bacterial count (BC2), fungal count (FC2) and actinomycetes count (AC2), (OC1 and OC2), available nitrogen (N1Y, N2Y), available phosphorus (P1Y, P2Y), and available K (K1Y and K2Y) in soil were positively affected by bacterial, fungal and actinomycetes population of jeevamrut formulation applied [Figure 5a, Supplementary Table 8]. Similarly, the microbial population in the soil after harvest was also having a positive correlation with the chemical parameters of the soil after harvesting [Figure 5b, Supplementary Table 8]. The pH and EC of soil were reported to be negatively affected by the microbial population of jeevamrut and soil. The experimental findings confirm that the application of jeevamrut and biofertilizers have increase the humification and mineralization of organic matter present in the soil to release the nutrients [12,16]. Further, the acidic medium developed during the decomposition of organic matter might be responsible for reducing soil pH and inducing the dissolution of fixed nutrients in available forms.
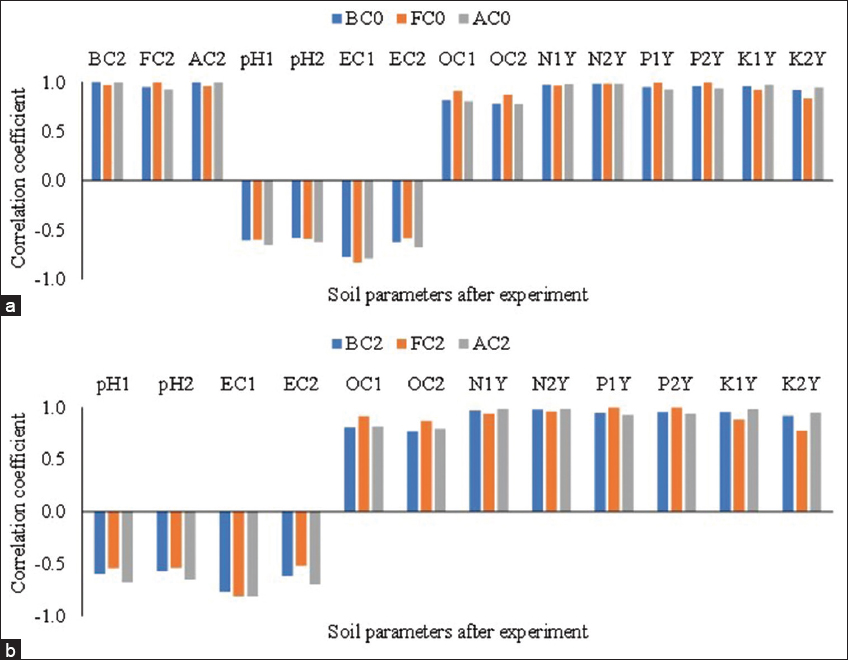 | Figure 5: Correlation coefficient (a) initial microbial count with various soil properties after crop harvest and (b) the microbial count with other soil properties after crop harvest (BC0: Initial bacterial counts in jeevamrut, FC0: Initial fungal counts in jeevamrut, AC0: Initial actinomycetes counts in jeevamrut, BC2: Bacterial count in the soil after 2 years of experiment, FC2: Fungal count in the soil after 2 years of experiment, AC2: Actinomycetes count in the soil after 2 years of experiment, pH1: Soil pH after 1 year of the experiment, pH2: Soil pH after 2 years of experiment, EC1: Soil electrical conductivity after 1 year of the experiment, EC2: Soil electrical conductivity after 2 years of experiment, OC1: Soil organic carbon after 1 year of the experiment, OC2: Soil organic carbon after 2 years of experiment, N1Y: Soil available nitrogen after 1 year of the experiment, N2Y: Soil available nitrogen after 2 years of experiment, P1Y: Soil available phosphorus after 1 year of the experiment, P2Y: Soil available phosphorus after 2 years of experiment, K1Y: Soil available potassium after 1 year of experiment, K2Y: Soil available potassium after 2 years of experiment). [Click here to view] |
3.5. PCA
The inter-relationships between various microbial and chemical attributes were studied using PCA [Table 6 and Supplementary Table 9]. The scree plot of all principal components (PCs) [Figure 6] confirms that the first four PCs are explaining 93 % of the total variable so these components are retained and detailed in the loading of the correlation matrix as shown in Table 6. The PCs-1, explaining 72.90% of the total variances, was significantly contributed to all attributes under study. This could be due to a strong association between the parameters under study and bacterial activity. The negative loading reflected by soil pH and EC in PC-1 suggested a stressed activity of the bacterial community during the utilization of substrate in controls (J0 and B0). However, the positive loading in the other parameters indicated significant improvement in bacterial community with the application of jeevamrut and biofertilizers. The PC-2, explaining 9.10 % of the total variance, was contributed by FC2, pH1, pH2, EC1, EC2, N1Y, P1Y, and P2Y with positive loading which suggests that application of jeevamrut and biofertilizers has enhanced the synergistic influence of the fungal communities over these parameters [5] and plays significant role in improving nutrient use efficiency in the integrated plant nutrient approach [79].
Table 6: Principal component loadings after Varimax rotation.
| Eigenvalues and parameters | Principal components# | |||
|---|---|---|---|---|
| PC1 | PC2 | PC3 | PC4 | |
| Eigenvalues | 10.937 | 1.363 | 0.915 | 0.729 |
| Explained variance (%) | 72.90 | 9.10 | 6.10 | 4.90 |
| Loadings (Eigenvectors) of correlation matrix on three retained components | ||||
| BC2: Bacterial count in soil after 2 years of experiment | 0.275 | NS | NS | 0.34 |
| FC2: Fungal count in soil after 2 years of experiment | 0.185 | 0.393 | 0.384 | -0.588 |
| AC2: Actinomycetes count in soil after 2 years of experiment | 0.276 | NS | NS | NS |
| pH 1: Soil pH after 1 year of experiment | -0.263 | 0.341 | NS | NS |
| pH 2: Soil pH after 2 years of experiment | -0.268 | 0.327 | NS | NS |
| EC1: Soil electrical conductivity after 1 year of experiment | -0.248 | 0.314 | NS | NS |
| EC2: Soil electrical conductivity after 2 years of experiment | -0.243 | 0.400 | NS | 0.319 |
| OC1: Soil organic carbon after 1 year of experiment | 0.201 | NS | 0.665 | 0.383 |
| OC2: Soil organic carbon after 2 years of experiment | 0.255 | NS | 0.431 | NS |
| N1Y: Soil available nitrogen after 1 year of experiment | 0.282 | 0.219 | NS | NS |
| N2Y: Soil available nitrogen after 2 years of experiment | 0.263 | NS | -0.354 | NS |
| P1Y: Soil available phosphorus after 1 year of experiment | 0.279 | 0.213 | NS | NS |
| P2Y: Soil available phosphorus after 2 years of experiment | 0.260 | 0.369 | NS | NS |
| K1Y: Soil available potassium after 1 year of experiment | 0.283 | NS | NS | NS |
| K2Y: Soil available potassium after 2 years of experiment | 0.271 | NS | NS | NS |
The soil parameters are grouped according to the maximum fittings to principal components (correlation coefficients ≥0.25; n=150) NS Loadings where the correlation coefficient is lower than 0.25, # Only principal components with Eigen values >0.5 and those explaining >5% of the total variance were retained.
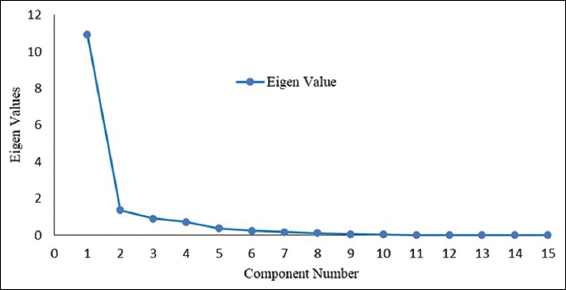 | Figure 6: The Scree plot reflecting Eigen value of different principal components (PC-1 to PC-15). [Click here to view] |
4. CONCLUSIONS
On account of the present experimental findings, it can be concluded that the application of jeevamrut fortified with vermitea or neem cake is essential for improving the soil microbial activity and the available nutrient content. The highest bacterial count (95.83 ± 7.83 × 106 cfu g-1), actinomycetes count (108.00 ± 7.69 × 104 cfu g-1), soil nitrogen (218.57 ± 5.85 and 226.27 ± 5.71 kg ha-1), and soil potassium (171.43 ± 2.24 and 179.67 ± 3.66 kg ha-1) were reported due to application of jeevamrut fortified with vermitea. The highest fungal count (157.54 ± 23.70 × 104 cfu g-1) and soil phosphorus (14.52 ± 0.27 and 15.40 ± 0.62 kg ha-1) was recorded after application of jeevamrut fortified with neem cake which was at par to jeevamrut fortified with vermitea.
The application of microbial consortium (co-inoculation of different types of microbes) consisting of PSB and Azotobacter or PSB and VAM has enhanced the mobilization of primary nutrients and soil microbial population. The highest bacterial count (96.33 ± 8.84 × 106 cfu g-1), actinomycetes count (111.04 ± 4.57 × 104 cfu g-1), and soil nitrogen (220.46 ± 6.31 and 228.75 ± 8.02 kg ha-1) were reported due to application of consortia of PSB and Azotobacter. The highest fungal count (182.02 ± 0.90 × 104 cfu g-1), soil phosphorus (14.38 ± 0.45 and 15.74 ± 0.37 kg ha-1), and soil potassium (168.00 ± 4.14 and 173.50 ± 6.12 kg ha-1) was recorded after application of consortia of PSB and VAM which was at par to PSB and Azotobacter.
Thus, application of jeevamrut fortified with vermitea or neem cake in combination with consortia of PSB and Azotobacter or PSB and VAM in crop nutrient management is necessary to increase soil bacterial, actinomycetes and fungal count; improve soil available nitrogen, phosphorus, and potassium; and balance soil structure, pH, EC and OC.
5. ACKNOWLEDGMENTS
The authors would like to acknowledge the contribution of ITM University, Gwalior to providing facilities and resources during an investigation.
6. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to the conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be authors as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
7. FUNDING
There is no funding to report.
8. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
9. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
10. DATA AVAILABILITY
The data are available with the first and corresponding author as it is from the dissertation work of the first author. It will be made available on request.
11. USE OF ARTIFICIAL INTELLIGENCE (AI)-ASSISTED TECHNOLOGY
The authors declares that they have not used artificial intelligence (AI)-tools for writing and editing of the manuscript, and no images were manipulated using AI.
12. PUBLISHER’S NOTE
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors and the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Bharucha ZP, Mitjans SB, Pretty J. Towards redesign at scale through zero budget natural farming in Andhra Pradesh, India. Int J Agric Sustain 2020;18:1-20. [CrossRef]
2. Bhattacharyya C, Banerjee S, Acharya U, Mitra A, Mallick I, Haldar A, et al. Evaluation of plant growth promotion properties and induction of antioxidative defense mechanism by tea rhizobacteria of Darjeeling, India. Sci Rep 2020;10:15536. [CrossRef]
3. Li R, Pang Z, Zhou Y, Fallah N, Hu C, Lin W, et al. Metagenomic analysis exploring taxonomic and functional diversity of soil microbial communities in sugarcane fields applied with organic fertilizer. Biomed Res Int 2020;2020:9381506. [CrossRef]
4. Kitamura R, Sugiyama C, Yasuda K, Nagatake A, Yuan Y, Du J, et al. Effects of three types of organic fertilizers on greenhouse gas emissions in a grassland on andosol in southern Hokkaido, Japan. Front Sustain Food Syst 2021;5:649613. [CrossRef]
5. Dinesh R, Anandaraj M, Kumar A, Srinivasan V, Bini YK, Subila KP, et al. Effects of plant growth-promoting rhizobacteria and NPK fertilizers on biochemical and microbial properties of soils under ginger (Zingiber officinale) cultivation. Agric Res 2013;2:346-53. [CrossRef]
6. Ahmed T, Shahid M, Noman M, Hussain S, Khan MA, Zubair M, et al. Plant growth-promoting rhizobacteria as biological tools for nutrient management and soil sustainability. In:Plant Growth Promoting Rhizobacteria for Agricultural Sustainability:From Theory to Practices. Berlin:Springer;2019. 95-110. [CrossRef]
7. Lucini L, Colla G, Miras Moreno MB, Bernardo L, Cardarelli M, Terzi V, et al. Inoculation of Rhizoglomus irregulare or Trichoderma atroviride differentially modulates metabolite profiling of wheat root exudates. Phytochemistry 2019;157:158-67. [CrossRef]
8. Mohanty P, Singh PK, Chakraborty D, Mishra S, Pattnaik R. Insight into the role of PGPR in sustainable agriculture and environment. Front Sustain Food Syst 2021;5:667150. [CrossRef]
9. Papik J, Folkmanova M, Polivkova-Majorova M, Suman J, Uhlik O. The invisible life inside plants:Deciphering the riddles of endophytic bacterial diversity. Biotechnol Adv 2020;44:107614. [CrossRef]
10. Wozniak M, Galazka A, Tyskiewicz R, Jaroszuk-SciselJ. Endophytic bacteria potentially promote plant growth by synthesizing different metabolites and their phenotypic/physiological profiles in the biolog GEN III MicroPlate(TM) test. Int J Mol Sci 2019;20:5283. [CrossRef]
11. Saharan BS, Tyagi S, Kumar R, Vijay, Om H, Mandal BS, et al. Application of Jeevamrit improves soil properties in zero budget natural farming fields. Agric 2023;13:196. [CrossRef]
12. Reddy AV, Menon S. A study on role of Jeevamruth in natural farming:A replacement for synthetic fertilizers. J Emerg Technol Innov Res 2021;8:89-93.
13. Ray P, Lakshmanan V, LabbéJL, Craven KD. Microbe to microbiome:A paradigm shift in the application of microorganisms for sustainable agriculture. Front Microbiol 2020;11:622926. [CrossRef]
14. Musa SI, Njoku LK, Ndiribe CC. The effect of vermitea on the growth parameters of Spinacia oleracea L. (Spinach). J Environ Sci Pollut Res 2018;3:236-8.
15. Sharma S, Gupta R, Dugar G, Srivastava AK. Impact of application of biofertilizers on soil structure and resident microbial community structure and function. In:Bacteria in Agrobiology:Plant Probiotics. Berlin:Springer;2012. 65-77. [CrossRef]
16. Fitriatin BN, Amanda AP, Kamaluddin NN, Khumairah FH, Sofyan ET, Yuniarti A, et al. Some soil biological and chemical properties as affected by biofertilizers and organic ameliorants application on paddy rice. Euras J Soil Sci 2021;10:105-10. [CrossRef]
17. Jnawali AD, Ojha RB, Marahatta S. Role of Azotobacter in soil fertility and sustainability-a review. Adv Plants Agric Res 2015;2:1-5.
18. Nongthombam J, Kumar A, Sharma S, Ahmed S. Azotobacter:A complete review. Bull Environ Pharmacol Life Sci 2021;10:72-9.
19. Fitriatin BN, Khumairah FH, Setiawati MR, Suryatmana P, Hindersah R, Nurbaity A, et al. Evaluation of biofertilizer consortium on rice at different salinity levels. Asian J Microbiol Biotechnol Environ Sci 2018;201:1108-12.
20. Kalayu G. Phosphate solubilizing microorganisms:Promising approach as biofertilizers. Int J Agron 2019;2019:4917256. [CrossRef]
21. Fitriatin BN, Fauziah D, Fitriani FN, Ningtyas DN, Suryatmana P, Hindersah R, et al. Biochemical activity and bioassay on maize seedling of selected indigenous phosphate-solubilizing bacteria isolated from the acid soil ecosystem. Open Agric 2020;5:300-4. [CrossRef]
22. Sandhya A, Vijaya T, Narasimha G. Effect of microbial inoculants (VAM and PSB) on soil physico-chemical properties. Bio Technol Indian J 2013;7:320-4.
23. Schwyter AR, Vaughan KL. SOIL 2010-Lab Manual and Materials (Doctoral Dissertation, University of Wyoming. Libraries);2020.
24. Tankeshwar A. Serial Dilution Method for Estimating Viable Count of Bacteria. General Microbiology;2022. Available from:? https://microbeonline.com/serial-dilution-method [Last accessed on 2023 Nov 04].
25. Jackson ML. Soil Chemical Analysis. New Delhi:Prentice Hall Pvt. Ltd.;1973.
26. Walter K, Don A, Tiemeyer B, Freibauer A. Determining soil bulk density for carbon stock calculations:A systematic method comparison. Soil Sci Soci Am J 2016;80:579-91. [CrossRef]
27. Robinson DA, Thomas A, Reinsch S, Lebron I, Feeney CJ, Maskell LC, et al. Analytical modelling of soil porosity and bulk density across the soil organic matter and land-use continuum. Sci Rep 2022;12:7085. [CrossRef]
28. Nawaz MF, Bourrie G, Trolard F. Soil compaction impact and modelling. A review. Agron Sustain Dev 2013;33:291-309. [CrossRef]
29. Fatichi S, Or D, Walko R, Vereecken H, Young MH, Ghezzehei TA, et al. Soil structure is an important omission in earth system models. Nat Commun 2020;11:522. [CrossRef]
30. Hirmas DR, Giménez D, Nemes A, Kerry R, Brunsell NA, Wilson CJ. Climate-induced changes in continental-scale soil macroporosity may intensify water cycle. Nature 2018;561:100-3. [CrossRef]
31. Sims NC, Barger NN, Metternicht GI, England JR. A land degradation interpretation matrix for reporting on UN SDG indicator 15.3. 1 and land degradation neutrality. Environ Sci Policy 2020;114:1-6. [CrossRef]
32. Aulakh CS, Singh H, Walia SS, Phutela RP, Singh G. Evaluation of microbial culture (Jeevamrit) preparation and its effect on productivity of field crops. Indian J Agron 2013;58:182-6. [CrossRef]
33. Nitin, Purohit HS. Effect of different Jeevamrut based liquid organic formulations on biochemical properties of soil and on plant growth of blackgram [Vigna mungo (L.) Hepper] under pot culture conditions. Indian J Chem Stud 2021;9:2280-3. [CrossRef]
34. Sreenivasa MN, Naik N, Bhat SN. Beejamrutha:A source for beneficial bacteria. Karnataka J Agric Sci 2010;22:1038-40.
35. Gore NS, Sreenivasa MN. Influence of liquid organic manures on growth, nutrient content and yield of tomato (Lycopersicon esculentum Mill.) in the sterilized soil. Karnataka J Agric Sci 2011;24:153-7.
36. Devakumar N, Shubha S, Gowder SB, Rao GG. Microbial analytical studies of traditional organic preparations Beejamrutha and Jeevamrutha. Build Org Bridges 2014;2:639-42.
37. Somasundaram E, Sankaranan N, Meena S, Thiyagarajan TM, Chandaragiri K, Pannerselvam S. Response of green gram to varied levels of Panchagavya (organic nutrition) foliar spray. Madras Agric J 2003;90:169-72.
38. Kulkarni SS, Gargelwar AP. Production and microbial analysis of Jeevamrutham for nitrogen fixers and phosphate solubilizers in the rural area from Maharashtra. IOSR J Agric Vet Sci 2019;12:85-92.
39. Vivanco L, Austin AT. Intrinsic effects of species on leaf litter and root decomposition:A comparison of temperate grasses from North and South America. Oecologia 2006;150:97-107. [CrossRef]
40. Wardle DA, Yeates GW, Barker GM, Bonner KI. The influence of plant litter diversity on decomposer abundance and diversity. Soil Biol Biochem 2006;38:1052-62. [CrossRef]
41. Ayres E, Steltzer H, Berg S, Wall DH. Soil biota accelerate decomposition in high?elevation forests by specializing in the breakdown of litter produced by the plant species above them. J Ecol 2009;97:901-12. [CrossRef]
42. Bray SR, Kitajima K, Mack MC. Temporal dynamics of microbial communities on decomposing leaf litter of 10 plant species in relation to decomposition rate. Soil Biol Biochem 2012;49:30-7. [CrossRef]
43. Hobbie SE. Plant species effects on nutrient cycling:Revisiting litter feedbacks. Trends Ecol Evol 2015;30:357-63. [CrossRef]
44. Sofo A, Elshafie HS, Camele I. Structural and functional organization of the root system:A comparative study on five plant species. Plants (Basel) 2020;9:1338. [CrossRef]
45. Ma M, Zhou J, Ongena M, Liu W, Wei D, Zhao B, et al. Effect of long-term fertilization strategies on bacterial community composition in a 35-year field experiment of Chinese Mollisols. AMB Express 2018;8:20. [CrossRef]
46. Ding JL, Jiang X, Guan DW, Zhao BS, Ma MC, Zhou BK, et al. Influence of inorganic fertilizer and organic manure application on fungal communities in a long-term field experiment of Chinese Mollisols. Appl Soil Ecol 2017;111:114-22. [CrossRef]
47. Mengesha WK, Gill WM, Powell SM, Evans KJ, Barry KM. A study of selected factors affecting efficacy of compost tea against several fungal pathogens of potato. J Appl Microbiol 2017;123:732-47. [CrossRef]
48. Pandia S, Trivedi A, Sharma SK, Yadav S. Evaluation of Jeevamrut and its constituents against alternaria leaf spot of mungbean in vitro and under cage house condition in Rajasthan. Int J Curr Microbiol Appl Sci 2019;8:2240-51. [CrossRef]
49. Scheuerell SJ, Mahaffee WF. Variability associated with suppression of Gray Mold (Botrytis cinerea) on geranium by foliar applications of nonaerated and aerated compost teas. Plant Dis 2006;90:1201-8. [CrossRef]
50. Scheuerell SJ, Mahaffee WF. Compost tea:Principles and prospects for plant disease control. Compost Sci Util 2002;10:313-38. [CrossRef]
51. Pant A, Radovich TJ, Hue NV, Arancon NQ. Effects of vermicompost tea (aqueous extract) on pak choi yield, quality, and on soil biological properties. Compost Sci Util 2011;19:279-92. [CrossRef]
52. Sapkota A, Thapa A, Budhathoki A, Sainju M, Shrestha P, Aryal S. Isolation, characterization, and screening of antimicrobial-producing actinomycetes from soil samples. Int J Microbiol 2020;2020:2716584. [CrossRef]
53. Shukla AK, Shahi S, Patel N, Patel S. Effect of phosphorus, VAM and FYM on soil fertility status under rice cultivation. Int J Curr Microbiol App Sci 2020;9:3766-75. [CrossRef]
54. Maiti SK, Ghosh D. Plant-soil Interactions as a Restoration Tool. Climate Change and Soil Interactions. Netherlands:Elsevier;2020. 689-730. [CrossRef]
55. Sharma SB. Trend setting impacts of organic matter on soil physico-chemical properties in traditional vis -a- vis chemical-based amendment practices. PLoS Sustain Transform 2022;1:e0000007. [CrossRef]
56. Adekiya AO, Ejue WS, Olayanju A, Dunsin O, Aboyeji CM, Aremu C, et al. Different organic manure sources and NPK fertilizer on soil chemical properties, growth, yield and quality of okra. Sci Rep 2020a;10:16083. [CrossRef]
57. Adekiya AO, Ogunboye OI, Ewulo BS, Olayanju A. Effects of different rates of poultry manure and split applications of urea fertilizer on soil chemical properties, growth, and yield of maize. ScientificWorldJournal 2020b;2020:4610515. [CrossRef]
58. Vieira FC, Nahas E. Comparison of microbial numbers in soils by using various culture media and temperatures. Microbiol Res 2005;160:197-202. [CrossRef]
59. Devakumar N, Rao GG, Shubha S. Evaluation of Locally Available Media for the Growth and Development of Nitrogen Fixing Micro-organisms. In Organic is Life-Knowledge for Tomorrow. Volume 1-Organic Crop Production. In:Proceedings of the Third Scientific Conference of the International Society of Organic Agriculture Research (ISOFAR), held at the 17th IFOAM Organic World Congress in cooperation with the International Federation of Organic Agriculture Movements (IFOAM) and the Korean Organizing Committee (KOC), 28. September-1. October 2011 in Namyangju, Korea Republic. International Society of Organic Agricultural Research (ISOFAR);2011. 504-9.
60. Baars O, Zhang X, Morel FM, Seyedsayamdost MR. The siderophore metabolome of Azotobacter vinelandii. Appl Environ Microbiol 2016;82:27-39. [CrossRef]
61. Wang Z, Chen Z, Fu X. Integrated effects of co-inoculation with phosphate-solubilizing bacteria and N2-fixing bacteria on microbial population and soil amendment under C deficiency. Int J Environ Res Public Health 2019;16:2442. [CrossRef]
62. Sumbul A, Ansari RA, Rizvi R, Mahmood I. Azotobacter:A potential bio-fertilizer for soil and plant health management. Saudi J Biol Sci 2020;27:3634-40. [CrossRef]
63. Soni A, Rokad S, Sharma P. Screening of efficient halotolerant phosphate solubilizing bacteria and their effect on seed germination under saline conditions. J Sci Innov Res 2013;2:932-7.
64. Heisnam P, Sah D, Moirangthem A, Singh MC, Pandey PK, Mahato NK, et al. Effects of Rhizobium, PSB inoculation and phosphorus management on soil nutrient status and performance of cowpea in acid soil of Arunachal Pradesh, India. Int J Curr Microbiol Appl Sci 2017;6:937-42. [CrossRef]
65. Samar S, Kumar A. Co-inoculation potential impact of PSB and Rhizobium on physico-chemical properties of soil and legume crop growth. Res J Agric Sci 2020;11:1-9.
66. Adnan M, Fahad S, Saleem MH, Ali B, Mussart M, Ullah R, et al. Comparative efficacy of phosphorous supplements with phosphate solubilizing bacteria for optimizing wheat yield in calcareous soils. Sci Rep 2022;12:11997. [CrossRef]
67. Saikia J, Sarma RK, Dhandia R, Yadav A, Bharali R, Gupta VK, et al. Alleviation of drought stress in pulse crops with ACC deaminase producing rhizobacteria isolated from acidic soil of Northeast India. Sci Rep 2018;8:3560. [CrossRef]
68. Wei Y, Zhao Y, Shi M, Cao Z, Lu Q, Yang T, et al. Effect of organic acids production and bacterial community on the possible mechanism of phosphorus solubilization during composting with enriched phosphate-solubilizing bacteria inoculation. Bioresour Technol 2018;247:190-9. [CrossRef]
69. Adnan M, Fahad S, Khan IA, Saeed M, Ihsan MZ, Saud S, et al. Integration of poultry manure and phosphate solubilizing bacteria improved availability of Ca bound P in calcareous soils. 3 Biotech 2019;9:368. [CrossRef]
70. Yu LY, Huang HB, Wang XH, Li S, Feng NX, Zhao HM, et al. Novel phosphate-solubilising bacteria isolated from sewage sludge and the mechanism of phosphate solubilisation. Sci Total Environ 2019;658:474-84. [CrossRef]
71. Zheng BX, Ding K, Yang XR, Wadaan MA, Hozzein WN, Peñuelas J, et al. Straw biochar increases the abundance of inorganic phosphate solubilizing bacterial community for better rape (Brassica napus) growth and phosphate uptake. Sci Total Environ 2019;647:1113-20. [CrossRef]
72. Elhaissoufi W, Ghoulam C, Barakat A, Zeroual Y, Bargaz A. Phosphate bacterial solubilization:A key rhizosphere driving force enabling higher P use efficiency and crop productivity. J Adv Res 2022;38:13-28. [CrossRef]
73. Chen YP, Rekha PD, Arun AB, Shen FT, Lai WA, Young CC. Phosphate solubilizing bacteria from subtropical soil and their tricalcium phosphate solubilizing abilities. Appl Soil Ecol 2006;34:33-41. [CrossRef]
74. Mohite B. Isolation and characterization of indole acetic acid (IAA) producing bacteria from rhizospheric soil and its effect on plant growth. J Soil Sci Plant Nutr 2013;13:638-49. [CrossRef]
75. Deluz C, Nussbaum M, Sauzet O, Gondret K, Boivin P. Evaluation of the potential for soil organic carbon content monitoring with farmers. Front Environ Sci 2020;8:113. [CrossRef]
76. Dhawi F. Plant growth promoting Rhizobacteria (PGPR) regulated Phyto and microbial beneficial protein interactions. Open Life Sci 2020;15:68-78. [CrossRef]
77. Han SH, An JY, Hwang J, Kim SB, Park BB. The effects of organic manure and chemical fertilizer on the growth and nutrient concentrations of yellow poplar (Liriodendron tulipifera Lin.) in a nursery system. Forest Sci Technol 2016;12:137-43. [CrossRef]
78. Ye L, Zhao X, Bao E, Li J, Zou Z, Cao K. Bio-organic fertilizer with reduced rates of chemical fertilization improves soil fertility and enhances tomato yield and quality. Sci Rep 2020;10:177. [CrossRef]
79. Bargaz A, Lyamlouli K, Chtouki M, Zeroual Y, Dhiba D. Soil microbial resources for improving fertilizers efficiency in an integrated plant nutrient management system. Front Microbiol 2018;9:1606. [CrossRef]