1. INTRODUCTION
The health of human populations is anticipated to be greatly impacted by the widespread issue of environmental pollution. It is becoming evident how important environmental elements are to human population health and well-being. As global industrialization progresses, numerous pollutants are being released, endangering all life forms severely [1]. Global apprehension regarding the impact of environmental pollution on public health has intensified in the past three decades. The amount of pollution that individuals are exposed to today is allegedly at an all-time high [2]. Significant public health issues are caused by the interaction between anthropogenic activities that are not environmentally sustainable [3]. Dangers like climate change are prevalent in the 21st century. It is really concerning how the contamination of the air, water, and soil is escalating over time. The presence of elevated concentrations of heavy metals (HMs) and other toxic compounds often reaches levels that pose a significant risk to downstream ecosystems and life forms in proximity to contaminated sites, while industrial emissions combined with vehicle exhausts significantly worsen the air pollution. Industrial wastes are frequently dumped on unused or public areas, in rivers, or in sewers built to handle solely municipal garbage, which makes the problem worse due to the lack of hazardous waste facilities [4].
Different air pollutants, such as sulfur dioxide, nitrogen oxides, carbon monoxide, ozone, HMs, and respirable particulate matter, volatile organic compounds have different chemical compositions, reaction characteristics, emission rates, times at which they degrade, and capacities for long- or short-distance diffusion [5]. Air pollution has detrimental effects on human health, manifesting in both immediate and prolonged impacts that affect various systems and organs. It can range from minor irritation of the upper respiratory tract to severe consequences such as lung cancer, chronic bronchitis among adults, respiratory infections in children, asthma exacerbations, and worsening of pre-existing cardiovascular and respiratory conditions [6]. Water pollution occurs when undesirable materials enter into water, alters the quality of water and adversely impacts the environment and human health [7]. The quality of water is influenced by various factors, including climatic conditions and the type of soil present, vegetation, precipitation geology, ground water, and human activities [8]. Urbanization and mining have an impact on water quality. The various chemicals, sediment and hazardous substances are additional sources of water contamination [9] [Figure 1]. The presence of low-quality water adversely disrupts the food chain, productivity of crops and contaminates our food, and endangering both human and aquatic life [10,11]. In addition, fishes are adversely affected by heavy metal; particularly iron (Fe), which poses harm to their respiratory system. Salem et al. [12] found that water contaminated with HMs leads to hair loss, liver cirrhosis, and kidney failure in human. The health of people and the environment may be at risk due to the rise in soil contamination over the past few decades. Biological processes related to plant growth and soil fertility are adversely affected by the continual presence of salts, pathogens, persistent hazardous substances, chemical compounds, or radioactive wastes [13]. The increased quantities of harmful substances, primarily HMs, pesticides, and petroleum derivatives, in the soil have adverse effects on the balance of ecosystems and human health [14].
 | Figure 1: Sources that commonly contribute to heavy metal contamination in soil such as mining and smelting, industrial waste, agricultural practices, urban runoff, landfills, and atmospheric deposition. [Click here to view] |
Infections of the respiratory system, conditions of the heart, and several kinds of cancer are all become more common because of increasing pollution levels. Ecologically detrimental pollutants, such as oil hydrocarbons, HMs, and pesticides, have a detrimental impact on the sustainability of ecosystems. da Silva et al. [15] highlighted the prevalence of carcinogenesis, mutagenesis, and other harmful effects, particularly in humans. On entering the soil, pollutants can be absorbed, carried away by wind and runoff, or seep into the deeper layers and contaminate groundwater. Soil pollution primarily arises from agricultural residues, byproducts, air pollutants, irrigation practices, unintentional oil spills, inadequate municipal waste, sewage management, and deposition of hydrocarbons [13]. Due to the problems associated with global population, pressure on soil quality and requirement for sustainable soil fertility are both continuously growing. The confluence of all the aforementioned problems makes soil pollution a prominent concern for microbial community, plant, and agricultural as well as human health [16]. Consequently, the restoration of polluted sites becomes imperative to recover the functionality of the impaired environment, ensuring both environmental preservation and facilitating urban growth. At present, there are three major techniques for the remediation of contaminants from the soil sample including chemical, physical, and sustainable [17] [Figure 2]. Recent years have seen a significant increase in research into bioprocesses including phytoremediation and bioremediation because they are more cost effective than traditional methods, eco-friendly, and capable of removing a wide range of toxins efficiently [18].
 | Figure 2: Different types of soil remediation techniques. [Click here to view] |
2. HEAVY METAL ACCUMULATION
One of the foremost environmental challenges that can have a negative impact on both environmental quality and human health at the moment is the widespread contamination of soil by HMs. According to Ali and Khan [19], a heavy metal is characterized as an element with an atomic number exceeding 20 and an atomic density surpassing 5 g/cm3, exhibiting metal-like properties. Thakare et al. [20] further HMs into two broad categories: Necessary and nonessential HMs. Essential HMs are vital for the fundamental biological processes of growth, metabolism, and organ development in living organisms. Plant species, such as those studied by Gratão et al. [21], require various essential HMs including copper (Cu), Fe, manganese (Mn), cobalt (Co), zinc (Zn), and nickel (Ni). These metals play essential roles in the production of cofactors that maintain the structural and functional integrity of enzymes and proteins. On the other hand, non-essential HMs such as cadmium (Cd), lead (Pb), mercury (Hg), chromium (Cr), or aluminum (Al) are not required by plants or any of their metabolic processes, even in trace amounts [22]. Both anthropogenic and natural factors can cause the discharge of HMs into the soil [23]. Despite the fact that heavy metal environmental contamination dates back to prehistoric times, the issue only became a serious concern following the industrial revolution because of the huge rise in the usage of HMs in contemporary technology.
In soil, commonly encountered HMs include Ni, Pb, Cd, Cr, Cu, Co, Zn, Mn, Al, and Hg [24]. The accumulation of HMs in agricultural soils can Pb to increased uptake of these contaminants by food crops and vegetables, presenting significant health risks to human populations [25]. According to reports, HMs can harm the bones, kidneys, nervous system, brain, and skin as well as cause cardiovascular disease, cancer, cognitive decline, chronic anemia, and damage to the skin, bones, and skin and brain [26-28]. There is a global imperative to maintain heavy metal levels in agricultural soil and crops below regulatory thresholds to mitigate the potential risks associated with heavy metal exposure. Increasing awareness of the adverse effects on human and environmental health stemming from heavy metal-contaminated soils has fostered the development of solutions to remediate such areas [29].
2.1. Chemical Fertilizers
In the past, agriculture was the main human activity that had a significant influence on the soil [30]. In addition to macronutrients nitrogen (N), phosphorus (P), potassium (K), sulfur (S), calcium, and magnesium, plants require essential micronutrients for their growth and life cycle. However, certain soils may exhibit deficiencies of necessary HMs, including Co, Cu, Fe, Mn, Zn, Ni, and Molybdenum, which are crucial for healthy plant development [31], thus crops can be given these nutrients by adding them to the soil or by spraying them on the leaves. On rare instances, Cu is applied to soil as a remedy for Cu-deficient soils to address the nutritional needs of cereal crops. According to Alloway [32], Mn supplementation is also beneficial for cereal and root crops. In intensive agricultural practices, substantial quantities of fertilizers are regularly applied to provide an adequate supply of N, P, and K for crop growth. However, these fertilizer compounds may contain trace amounts of HMs such as Cd and Pb as impurities. Prolonged use of fertilizers can significantly elevate the concentrations of these metals in the soil [33]. Cd and Pb are classified as metals that lack recognized physiological functions in biological systems. The application of certain phosphatic fertilizers can inadvertently introduce Cd and other potentially hazardous metals, including F, Hg, and Pb, into the soil, as highlighted by Moolenaar and Lexmond [34].
2.2. Insecticides
In the field of horticulture and agriculture, numerous conventional pesticides that were extensively employed in the past were found to contain elevated concentrations of metals [35]. Some pesticides, including Bordeaux mixture and Cu oxychloride, are fungicidal sprays that contain Cu [36]. Historically, Pb arsenate was extensively utilized in fruit orchards as a means to combat parasitic insects. Abandoned sites that were treated with Cu, Cr, and arsenic-based formulations chromated copper arsenate to preserve timbers exhibited significantly higher soil concentrations of these elements than background levels, as highlighted by McLaughlin et al. [37]. This form of contamination can pose challenges, particularly if these sites are later repurposed for agricultural or non-agricultural activities. The application of these materials has been more geographically restricted and primarily associated with specific regions or crops, in contrast to the widespread use of fertilizers [38].
2.3. Biosolids
Land application of various biosolids, including composts and municipal sewage sludge, inadvertently results in the contamination of soil with a wide range of HMs [39]. In agriculture, it is customary to utilize animal wastes, including poultry, livestock, and pig manures, by applying them as solids or sludges to crops and pastures [40]. Although manures are generally recognized as valuable fertilizers, the use of Cu and Zn in pig and poultry industries as growth promoters and poultry health products raises concerns regarding potential soil contamination with these metals [41]. Repeated application of manures produced by animals fed diets containing elevated levels of arsenic (As), Cu, and Zn to confined land areas can Pb to substantial accumulation of these metals in the soil [42].
There is growing interest in exploring the potential of composting biosolids together with organic materials such as wood shavings, straw, or yard waste. However, if this trend continues, there is a legitimate concern regarding the potential for soil contamination by HMs. The utilization of biosolids in agricultural practices has raised substantial concerns regarding their potential to introduce toxic substances into the soil [43]. The HMs that are most frequently detected in activated sludge are Pb, Ni, Cd, Cr, Cu, and Zn. The concentrations of metal are affected by the type of industrial activity, its intensity, the procedure used to treat the biosolids, and many other factors [44]. In specific situations, metals that are deposited in soils through the application of biosolids can potentially percolate through the surface soil and contaminate groundwater [45].
2.4. Metal Mining
Metal pollutants in soil have been widely distributed as a result of metal ore mining, ore milling, and related sectors in many different nations. During mining operations, the discharge of tailings, consisting of larger and denser particles that settle at the bottom of flotation cells, directly into natural depressions, including on-site wetlands, leads to elevated concentrations of contaminants [46]. A risk to the health of people and the environment has been created by the extensive extraction and processing of Pb and Zn ore through mining and smelting operations [47]. The soil reclamation techniques utilized at these sites are often time-consuming, costly, and may not effectively enhance soil productivity. The bioavailability of HMs in the soil plays a crucial role in determining the potential environmental risks to human health [48]. The food chain and the availability of contaminants are present in the soil for oral absorption and assimilation [47]. Other materials are produced by numerous industries, including fabric, bronzing, petrochemical products from unintentional oil spills or use of crude oil ingredients, herbicides, and pharmacological amenities, and they have a very varying composition [49]. Despite some being dumped on land, very few are useful for forestry or agriculture. Moreover, some substances are rarely, or never, utilized for land application due to their potential hazard, such as the presence of HMs (such as Cr, Pb, and Zn) or toxic organic compounds. In addition, certain substances exhibit inadequate soil conditioning properties or possess minimal nutritional value for plant growth [40].
2.5. Sewage Water
It has been a regular practice for 400 years in various regions of the world to apply municipal, industrial, and associated wastes and effluents to land [50]. An estimated 20 million acres of cultivable land globally are reported to utilize wastewater for irrigation purposes. Studies indicate that in numerous Asian and African towns, wastewater irrigation-based agriculture provides 50% of total of the city’s supply of vegetables [51]. Farmers are typically more concerned with increasing their yields and earnings than they are with the advantages or risks to the environment. Although the metal concentrations in industrial wastewaters are typically low, prolonged irrigation of land with such waters can eventually result in the accumulation of HMs in the soil.
3. HAZARDOUS EFFECT OF HMS ON SOIL
In the industrialized world, heavy metal poisoning of soil is a major concern [52]. In addition to having negative effects on a number of plant quality and yield-related indices, heavy metal pollution also alters the size, make-up and productivity of the microbial community [53]. The detrimental impact of HMs on the biological and chemical properties of soil is widely recognized. The extent of these effects on biological and biochemical characteristics is heavily influenced by specific soil attributes, such as organic matter content, clay concentration, and pH levels [54]. HMs have an indirect effect on soil enzymatic activity by altering the microbial population that generates enzymes [55]. The presence of HMs in soil adversely affects soil biota by disrupting critical microbial functions and diminishing the abundance and activity of microorganisms. On the other hand, long-term effects of HMs can boost bacterial population tolerance as well as fungi like arbuscular mycorrhiza fungi’s tolerance, which can be crucial for the regeneration of damaged ecosystems [56]. Heavy metal contamination resulted in reduction in biomass as well as diversification of the microbial populations in polluted soils, as well as a relative rise in soil actinomycetes. It is said that due to the various biochemical inclinations of the enzymes with in soil system, different metals have varying effects on the enzyme activity [57]. Pb is less harmful to enzymes than Cd because of its weaker affinity for soil colloids and greater mobility. More than cellulose activity, cu inhibits the activity of beta-glucosidase. Urease, catalase, invertase, and acid phosphatase all exhibit significant reductions in activity when exposed to Pb. As (V) inhibits the enzymes phosphatase and sulfatase; however, this did not influence the urease [58]. Protease, urease, alkaline phosphatase, and arylsulfatase are negatively impacted by cd contamination; however, invertase is not significantly affected.
It is crucial to study the behavior of soil microorganisms in ecosystems with prolonged exposure to heavy metal contamination [59]. In soils, Cr exists in two forms: Cr (III) and Cr (VI), each with distinct chemical properties and levels of toxicity. Cr (VI) is a powerful oxidizing agent and highly toxic, while Cr (III) is an essential nutrient and relatively non-hazardous, being 10–100 times less toxic [60]. At high concentrations, Cr (VI) is known to have negative impact on microbial cellular metabolism and has been observed to change the makeup of soil microbial populations [55]. It has also been noted that heavy metal pollution (Cr, Zn, and Cd) has an impact on the metabolism of beneficial microorganisms in all circumstances. This is because HMs have harmful effects on soil microorganisms, changing their variety, population size, and general activity. In general, an increase in metal concentration has a negative impact on the microbiological characteristics of the soil, including the rate of respiration and enzyme activity, both of which seem to be excellent markers of soil pollution [61]. A small alteration in the soil microbial spectrum was seen in lead-contaminated soil [56]. The potential health risks associated with the uptake of significant quantities of HMs by plants from soil warrant careful consideration in relation to their impact on the food chain. A significant food chains route for human exposure is the use of food crops that are polluted with HMs [58]. The ability to collect nutrients from soils is quite strong in food plants whose evaluation system relies on extensive and ongoing culture. Since, the vegetative tissues of such plants have the capacity to absorb HMs growing them in contaminated soil poses a possible concern [62]. When HMs accumulate in the soft tissues without being metabolized, they can become toxic [63]. Chronic exposure to hazardous metals in humans has negative effects that do not become apparent for years after the initial exposure [22].
4. SOIL REMEDIATION TECHNIQUES
4.1. Physical Techniques
4.1.1. Capping
Capping entails covering hazardous material, such as rubbish from landfills or contaminated soil. These coverings are known as “caps”. Caps neither eliminate nor destroy pollutants. To stop the spread of infection, they segregate them and maintain them in situ. Caps shield both humans and animals from harmful substances. Surface capping, sanitary landfills, and encapsulation are a few of the methods used in soil replacement, which entails utilizing a significant amount of uncontaminated soil to mix with or cover the surface of polluted soils [64]. Due to its high cost and labor intensity, this technique is best suited for severely contaminated soil in areas with limited space. It can also effectively reduce the pollutant concentration. To stop additional dispersion from the site, barrier walls might be installed to isolate and contain pollutants. For capping, horizontal and vertical containment, impermeable physical barriers constructed of steel, clay, bentonite, and grout are used. It is clear that the practice of soil isolation or confinement was utilized to minimize the flow of HMs into the groundwater rather than as a direct remedial process [65].
4.1.2. Vitrification
The process of vitrification uses a strong energy source to melt soil at very high temperatures, immobilizing the majority of inorganic impurities and oxidizing or pyrolyzing the majority of organic contaminants. It is possible to use this technology including both situ and ex situ. Ex situ soil heating can be accomplished using a variety of technologies, but the most popular method is the use of electricity. The soil is put in a furnace and heated with electricity to a range of 1100–1400°C. In situ, treatment requires a higher temperature around 1600–2000°C [66]. Graphite electrodes are typically put into the soil to be treated to apply electrical energy [67].
4.1.3. Electrokinetics
The removal of organic, inorganic, and heavy metal particulates from the soil using direct electric current is known as electrokinetics (EK) [68]. When treating subsurface contaminants, this methodology provides an approach that disturbs the surface as minimally as possible [69]. It is a recently created technique that effectively cleans up heavy metal-polluted soils. This technique uses direct electric current to remove HMs from the soil matrix by a variety of methods, comprising electromigration, electroosmosis, electrophoresis, and electrolysis [70]. Chelates are additionally employed to increase the effectiveness of the EK in contaminated soils. By evaluating the impact of several chelators such as Ethylenediamine tetraacetic acid (EDTA), nitrilotriacetic acid, and citric acid in improving EK efficiency, HMs (As, Cd, Cr, Cu, Ni, Pb, and Zn) mobility was examined [71]. In addition, to overcome the drawback of flushing method in fine soil, several researchers have coupled flushing and EK in two different ways: Sequential and integration. The addition of a pump to the EK flushing remediation improved the effectiveness of removing Co2+ and Cs+ (caesium) from the polluted soils.
4.2. Chemical Remediation
It describes a particular technique for removing pollutants that makes use of chemical reagents, processes, and principles. Common methods for remediating contaminated soil include solidification/stabilization, soil washing, and soil flushing [70]. Solidification technology is often employed to reduce the mobility of heavy-metal pollutants by mixing contaminated soils with chemicals or materials. Stabilization involves implementing chemical reactions to reduce the mobility of pollutants, while solidification entails physically encapsulating the impurities within a solid matrix consisting of cement, asphalt, or thermoplastic binders [72]. Chemical bondings as well as encapsulation are the two basic interactions that can immobilize HMs in the glass matrix. Nitrification is a process of solidification and stabilization that requires thermal energy (1400–2000°C). It can be accomplished by combining polluted soil with glass-forming intermediates, heating the compounds until they become liquid, and then cooling the liquid to produce an amorphous relatively homogenous glass [73]. Chemical bondings as well as encapsulation are the two basic interactions that can immobilize HMs in the glass matrix [74]. Soil washing and soil flushing are highly effective techniques for remediation purposes aimed at eliminating pollutants from the soil. Various agents, such as water [75], organic acids [76], chelating agents [77], surfactants [78], saponin [79], and low-molecular-weight organic acids [80], have been identified as successful washing agents, ensuring optimal removal of HMs. Among these, EDTA has proven to be the most efficient chelating agent for extracting HMs from contaminated soils [81]. EDTA offers advantages such as minimal impact on soil microorganisms and enzyme activity, low biodegradability, enhanced metal extraction efficiency, accessibility to appropriate recycling methods, and availability for use in the remediation process [82].
4.3. Biological Techniques
4.3.1. Phytoremediation
Phytoremediation involves the utilization of plants, naturally occurring bacteria, and fungi to biodegrade or detoxify harmful pollutants that pose a threat to the environment or human health. These microorganisms can either originate from the contaminated site itself or be introduced from external sources. Through their metabolic activities, living organisms facilitate reactions that transform contaminant substances, aiding in the remediation process. The utilization of natural processes in bioremediation, which can successfully biodegrade a range of pollutants, even persistent ones, could be useful and effective method for reducing soil contaminations. This technique can make use of a variety of microorganisms, including ligninolytic fungi, methylotrophs, aerobic bacteria, and anaerobic bacteria. In an environmentally responsible manner, revegetating heavy metal-polluted soil with plants is possible through phytoremediation. A deeper comprehension of the mechanisms driving heavy metal concentration and tolerance in plants is essential to increasing the effectiveness of phytoremediation [83]. In this method, hazardous metals are removed from the environment, decomposed, or detoxified using green plants. Five different phytoremediation techniques, including phytovolatilization, phytostabilization, phytodegradation, rhizofiltration, and phytoextraction have been employed for soil decontamination [84]. Phytoremediation involves utilizing plants to eliminate hazardous substances from the surroundings or to make them less accessible in the soil. Ionic compounds with in soil can be taken up by plants via their rhizosphere even in small amounts. Plants strategically extend their root systems into the soil matrix, establishing rhizosphere ecosystems to uptake and control the bioavailability of HMs. This process aids in the restoration of polluted soil and regulation of soil fertility [85].
4.3.2. Mycoremediation
The widespread presence of fungi in the natural environment and their extensive utilization in industrial settings are widely recognized. Given their ability to adapt to their surroundings, fungus is essential for natural processes such as nitrogen cycling and decomposition [86]. Through mycoremediation, pollutants from diverse environmental niches are removed using fungus, either alive or dead [87]. Mycoremediation is a cost-effective approach that avoids the production of harmful waste products, making it a comprehensive solution due to the complete mineralization of pollutants in the environment [88]. The effectiveness of mycoremediation heavily relies on the identification and utilization of a specific fungal species tailored to the targeted HMs or other pollutants. Fungi exhibit efficient accumulation of HMs in their fruit bodies, which renders them inaccessible or lowers their abundance in the environment [89]. Future availability of HMs and other pollutants in the environment is significantly influenced by the longevity of fungus, the chemical characteristics of elements, and the availability of fungi following sequestration [90]. Studies have indicated that Saccharomyces cerevisiae has the potential to sequester a significant amount of Pb and Cd from contaminated soil, ranging from 65% to 79% [91]. Fungal cell walls with functional groups such as carboxyl, amino, hydroxyl, phosphate or sulfate, as well as proteins, glucans, lipids, pigments, and polysaccharides are involved in the biosorption process, which is mediated by interactions including ion-exchange, complexation, and adsorption [92].
4.3.3. Bioremediation
Bioremediation is the process of converting environmental pollutants into less harmful forms by using living organisms, primarily microbes. Methodology used in bioremediation is mostly based on biodegradation. It refers to the complete conversion of harmful or naturally occurring organic contaminants into substances that are safe for human, animal, plant, and marine life [93]. The biodegradation of a wide range of organic compounds has been explained by a variety of mechanisms and pathways; for instance, it can occur both with and without oxygen. Bioremediation is an effective method that aims to mitigate the presence of toxic compounds, including HMs, by converting them into less harmful substances. This technique contributes to the removal of potentially hazardous materials from contaminated environments, as well as the degradation of organic substances, leading to the complete mineralization of organic matter into components such as carbon dioxide, water, and nitrogen gas [94]. The application of bioremediation can be carried out using in situ or ex situ approaches, both of which are suitable for treating soil contamination [Figure 3]. There are several in -situ methods, including bioattenuation, biostimulation, biosparging, and bioventing. To encourage the growth of native or introduced microorganisms, bioventing entails adding oxygen to the soil. Biostimulation entails the provision of particular nutrients and ideal environmental conditions at the site to promote the growth of local microorganisms [95]. A contaminated site can be passively remedied through bioattenuation, which is applicable for both disintegrating and recalcitrant chemicals and necessitates ongoing monitoring. Biosparging is the process of supplying air under compression underneath the water table to raise the oxygen concentration and speed up microbial activity [96]. Over the past two decades, significant advancements have been achieved in bioremediation techniques with the primary goal of restoring polluted areas in an environmentally friendly and cost-effective manner [97].
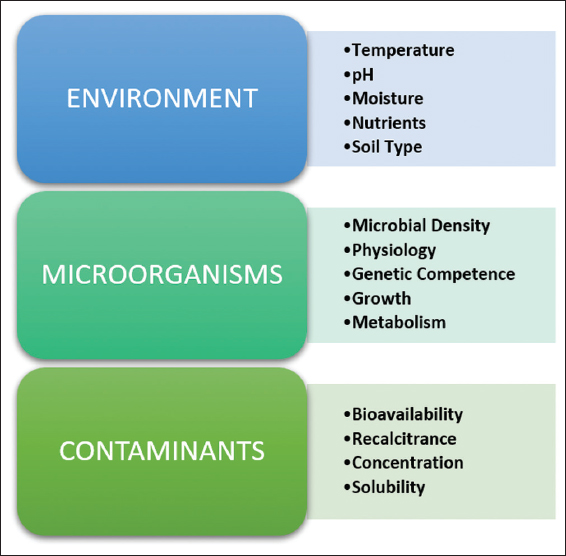 | Figure 3: Factors affecting bioremediation. [Click here to view] |
5. MICROBIAL MANAGEMENT OF HMS IN SOIL
Microorganisms play a crucial role in the essential biogeochemical cycles of metals, facilitating the transformation of metals between insoluble and soluble forms. They are abundant in nature and contribute significantly to these cycles [98]. The interactions between metals and microbes can be advantageous or detrimental. These transformations depend on environmental variables such as pH, humidity, temperature, the occurrence of many other charged particles, in addition to the type of bacteria, and the chemical composition of metal, both of which are essential for microbial colonization and biofilm formation [87]. There are several in situ methods, including bioventing, biostimulation, bioattenuation, and biosparging. To encourage the growth of native or introduced microorganisms, bioventing entails adding oxygen to the soil. Biostimulation entails the provision of particular nutrients and ideal environmental conditions at the site to promote the growth of local microorganisms. A contaminated site can be passively remedied through bio-attenuation, which is applicable for both disintegrating calcitrant chemicals and necessitates ongoing monitoring. Biosparging is the process of supplying air under compression underneath the water table to raise the oxygen concentration and speed up microbial activity [99]. Everywhere you look in the environment, you can find bacteria. A simple and effective method for removing contaminants from wastewater, involving non-biodegradable substances like HMs, is biosorption by bacteria. Cells that make up bacterial biomass might be alive or dead. For the survival, bacterial species have developed mechanisms for resistance to metal ions and remediation. Bacterial species have evolved mechanisms to ensure their survival in the presence of metal ions and to contribute to remediation efforts. Metals including Cu, Zn, Pb, Cd, and Cr can be quickly removed using bacterial biomass [87]. The main physical interface connecting metal ions and bacterial biomass is the bacterial cell wall. Gram-positive bacteria possess cell wall components such as peptidoglycan, teichoic acids, and teichuronic acids, while Gram-negative bacteria have peptidoglycan, lipopolysaccharides, and phospholipids. These components contain anionic functional groups, including amine, hydroxyl, carboxyl, sulfate, and phosphate, which contribute to the metal-binding capability of the cell wall [92]. Extracellular processes are used by decaying biomass cells to remove HMs. These interactions are caused by functional groups on the cell wall, such as carboxyl, phosphonate, amine, and hydroxyl groups [100]. Microorganisms may induce redox reactions that Pb to metal mobilization and immobilization, affecting the bioremediation process. HMs go through cycles of oxidation and reduction, including Fe, As, Cr, and Hg [101]. An element’s transformation from its immobile, insoluble form in sediments to its mobile, soluble state facilitates bioremediation. When hazardous metal ions are reallocated and liberated from their solid phase in sediments into the solution phase, mobilization can potentially have negative effects. As a result, HMs are more bioavailable and can enter microbial metabolic processes. By breaking down Hg (II), the bacteria can produce Hg (0), which is more volatile. By reducing certain ions, such as Fe (III) and As (V), to Fe (II), and As (III), respectively, microbial reduction can also increase the solubility of certain ions and promote leaching from soil. In soil and water, the process of heavy metal biomethylation is crucial and can change the toxicity, instability, and mobility of the metals. Due to the ability to eliminate volatile methylated species from cells, it also functions as a significant detoxification method [102]. Microbial breakdown of organic matter contributes to the indirect process of metal mobilization by accelerating the emission of these ions [Table 1].
Table 1: List of microorganisms associated with heavy metal remediation.
| Microorganism | Genus | Heavy metal removed | References |
|---|---|---|---|
| Bacteria | Bacillus sp. | Pb, Cd, Cr, Cu, As, Zn, Ni, Hg | Alotaibi et al. [161] |
| Vibrio sp. | Cd, Pb, Hg | Parmar et al. [162] | |
| Pseudomonas sp. | Cd, Ni, Pb, Hg, Zn, Cu | Chellaiah [163] | |
| Geobacter sp. | Cr | He et al. [164] | |
| Rhodococcus sp. | Cu | Maslanová et al. [165] | |
| Alkaligenes sp. | Cd, Pb, Hg | Sodhi et al. [166] | |
| Sphingomonas sp. | Ni, Cu, Pb, Cd, | Heidari et al. [167] | |
| Enterobacter sp. | Ni, Cu, Pb, | Heidari et al. [167] | |
| Kocuria sp. | Cu | Kumari et al. [168] | |
| Sporosarcina sp. | Cr | Li et al. [169] | |
| Acinetobacter sp. | Ni | Wu et al. [170] | |
| Staphylococcus sp. | Cr, Cu, Pb | Kumar et al. [171] | |
| Streptomyces sp. | Cd, Pb | Mosbah and Sahmoune [172] | |
| Fungi | Aspergillus sp. | Ni, Pb, Cr, Cd | Mishra and Malik [173] |
| Rhizopus sp. | Cu | Gomes et al. [174] | |
| Trichoderma sp. | Cd | Herliana et al. [175] | |
| Saccharomyces sp. | Pb, Hg, Ni | Infante J et al. [176] | |
| Ganoderma sp. | Cd, Pb | Chang et al. [177] | |
| Fusarium sp. | Pb, Cd, Cu | Al-Hagar et al. [178] | |
| Candida sp. | Cr | Bahafid et al. [179] | |
| Penicillium sp. | Cd, Pb, Hg, As | Sánchez-Castellón et al. [180] | |
| Agaricus sp. | Cd, Pb | Long et al. [181] | |
| Ulva sp. | Pb, Cu, Cd, Zn, Ni | Areco et al. [182] | |
| Algae | Cladophora sp. | Pb, Cu | Deng et al. [183] |
| Spirullina sp. | Pb, Cu, Cd, Zn, Ni | Mane and Bhosle [184] | |
| Sargassam sp. | Pb, Cr, Cd | Tamilselvan et al. [185] | |
| Spirogyra sp. | Pb, Cu, Cd, Zn, Ni | Mane and Bhosle [184] | |
| Codium sp. | Pb, Cu, Cd, Zn, Ni | Laib and Leghouchi [186] | |
| Caulerpa sp. | Pb, Cr, Cd | Tamilselvan et al. [185] | |
| Chaetomorpha sp. | Cd | Kumar et al. [187] |
Pb: Lead, Cd: Cadmium, Cr: Chromium, Cu: Copper, As: Arsenic, Zn: Zinc, Ni: Nickel, Hg: Mercury.
6. UTILIZATION OF Bacillus sp. FOR HEAVY METAL REMEDIATION
Bacillus spp., belonging to the phylum Firmicutes, are Gram-positive bacteria characterized by their rod-shaped morphology and ability to form spores. Categorized primarily as soil microorganisms, Bacillus spp. have the ability to thrive in both aerobic and anaerobic conditions. Furthermore, they are widely distributed across diverse environments such as air, water, food sources, and even the human gut [103]. The Bacillus group is among the most diverse in terms of genetic or economic applications since certain species have gained recognition for their opportunistic nature as infectious agents and their ability to produce toxins, while others have found extensive applications in industrial and pharmaceutical settings [104]. The capacity of Bacillus spp. to produce spores in harsh settings, which is typically driven by a nutrient shortage, is one of their distinctive traits [105]. Spores can withstand extreme environmental stress, including desiccation, high temperatures, humidity, and radiation, thanks to their strong structural makeup. Due to this characteristic, they demonstrate a diverse range of commercial implications and are more ideal for use than vegetative cells [106]. The beneficial applications of Bacillus spp. in numerous domains have drawn attention to their distinctive properties, and research has made it possible to employ these traits to the benefit of humankind. Biocontrol products made from Bacillus are used [107]. The utilization of Gram-positive bacteria, specifically Bacillus spp., presents considerable advantages across medical, industrial, and environmental domains. One notable advantage is their reduced tendency to engage in the transmission of genetic material compared to Gram-negative bacteria. This characteristic enhances their suitability for various applications, making them a valuable resource in these fields.
Numerous Bacillus species, including Bacillus subtilis, Bacillus coagulans, Bacillus pumilus, Bacillus licheniformis, and Bacillus cereus, are used across the globe for a variety of uses [106]. Due to the evidence of its antibacterial and anti-cancerous properties, numerous study findings have suggested that B. subtilis is safe for usage as a probiotic. In the food sector, Bacillus spp. are also employed to produce a number of different enzymes such as amylase, protease, cellulase, and pectinase, as well as a number of additional nutrients such as vitamins and carotenoids [108]. In addition to these applications, extensive research has been conducted on Bacillus spp. due to their active involvement in mitigating heavy metal pollution through diverse mechanisms, including bioaccumulation and biosorption [Table 2]. Their ability to alleviate HMs from contaminated environments has been a subject of intensive study and offers promising prospects for environmental remediation. This is because it has been suggested that Gram-positive bacteria frequently predominate in contaminated locations due to their flexible metabolic capabilities and superior biosorption abilities [109].
Table 2: List of Bacillus sp. associated with heavy metal remediation.
| S. No. | Bacillus species | Heavy metal removed |
|---|---|---|
| 1. | Bacillus cereus, Bacillus aryabhattai, Bacillus megaterium | Arsenic |
| 2. | Bacillus altitudinis, Bacillus subtilis, Bacillus cereus, Bacillus jeotgali, Bacillus licheniformis | Zinc |
| 3. | Bacillus thuringiensis, Bacillus megaterium, Bacillus subtilis | Nickel |
| 4. | Bacillus licheniformis, Bacillus safensis, Bacillus catenulatus | Cadmium |
| 5. | Bacillus cereus | Lead |
| 6. | Bacillus thuringiensis, Bacillus sphaericus, Bacillus licheniformis, Bacillus subtilis | Copper |
| 7. | Bacillus licheniformis, Bacillus cereus | Chromium |
| 8. | Bacillus cereus, Bacillus thuringiensis | Mercury |
6.1. As Remediation
Arsenic is difficult to remove from the environment since it is soluble. Arsenic is remedied physically and chemically by an oxidation process that changes As3+ into As5+. It can happen with air oxygen, which normally happens very slowly, or with chemical oxidants like ozone, chlorine, or hydrogen peroxide. This approach costs a lot of money and results in undesirable externalities [110]. The enzyme As oxidase, primarily located within the protoplasm of bacteria capable of oxidizing As, plays a significant role in As metabolism, converts the poisonous As3+ into its less toxic version, As5+, as a result of microorganisms using Arsenic as a source of energy in their metabolic processes [111]. Numerous researches undertaken over the years have revealed that Bacillus spp. can absorb Arsenic, making it one of the most significant bacteria for minimizing As contamination [Liao et al., 2011]. Arsenic removal conducted by Bacillus spp. such as B. cereus, Bacillus aryabhattai [112], and Bacillus megaterium [113]. Anaerobic respiration of As5+ is also done by B. cereus [114]. Oxidative phosphorylation is essential for all living things, and this is unavoidable. Since As5+ and phosphate are structurally identical, it may act as an inhibitor of oxidative phosphorylation [115]. The pathways known as Pit and Pst, commonly utilized for phosphate uptake, serve as the means through which As5+ enters the biological system of organisms [116]. Adenosine triphosphate production is inhibited and phosphorylation metabolic pathways are hampered [117]. When it internalizes, it attaches to the respiratory enzymes right away using their sulfur residue [118]. In Bacillus species, the Ars operon uses genes such as arsA, arsB, arsC, arsD, and arsR to bioremediate As. ArsA and ArsB are ATPases, ArsC converts As3+ to As5+, ArsD is a metallochaperone, and ArsR is a repressive inhibitor [119,120]. Normally, As3+ is carried out of the cell by ArsB after being converted from As5+ that enters the cell by ArsC [121].
6.2. Zn Remediation
The capacity of Bacillus spp. effectively eliminate Zn from polluted environments has been emphasized in various bioremediation trials. Bacillus species can either evolve resistance mechanisms or acquire resistance using plasmids to control this ability [Christofi and Ivshina, 2002]. The Zur family controls Zn absorption in B. subtilis, allowing Zn ions to be transported using two transporter proteins. When there is oxidative stress, the Zn ion absorption pathway in B. subtilis termed ZosA (P-type ATPase) is expressed [122]. In the bacterium B. subtilis, a CPx-type ATPase extrusion pump called CadA plays a crucial role in facilitating the excretion of Zn from highly concentrated solutions. Furthermore, it has also been revealed in latest studies that certain Bacillus species, including Bacillus altitudinis, B. subtilis, B. cereus, Bacillus jeotgali, and B. licheniformis remove Zn from the environment [123-125].
6.3. Ni and Cd Remediation
Although there are various ways to remove Ni from solid matrices, the best ones are those that can do it before the Ni enters the environment. Throughout the years, various physicochemical techniques have been employed to eliminate Ni from aqueous media [126]. Regardless of the techniques previously employed, recent advancements have introduced more cost-effective and efficient methods for Ni removal. Among these methods, adsorption techniques, particularly utilizing biomass such as sugarcane, corn cobs, citrus peels, and barks, have gained prominence [127]. The more effective way to remove Ni ions from media that has been contaminated with Ni is through bioremediation employing gram-positive bacteria like Bacillus spp. Extensive studies [128,129] have shown the consumption and removal of nano particles from polluted sites like soils, sewage, and streams. It has also often been reported that Bacillus thuringiensis may absorb and eliminate Ni from polluted surroundings [130]. In a latest study, B. megaterium was obtained from soils that had been contaminated with Ni, and it was able to absorb more than 500 mg of Ni there, despite the presence of more than 3000 mg/L of Ni salt there earlier [131]. Bacterial resistance mechanisms, which ultimately make it easier for metal ions to enter and exit the cell, are necessary for the elimination of Ni by bacteria. It has long been known that members of the CDF family, which includes Ni, facilitate the efflux of several metal ions [132]. The cation diffusing transporter CzcD in B. subtilis is considered to prevent the cell from excessive Ni2+, Cu+, Zn2+, and Co2+ concentrations. Under favorable conditions, metal ions in association with citrate can be absorbed by B. subtilis through the metaldicitrate absorption system known as CitM, thereby potentially increasing their toxicity [133]. Since B. licheniformis eliminated more than 98% of Cd2+, it is a highly efficient adsorbent for the effective removal of Cd2+ ions [134]. There are other reports that Bacillus safensis and Bacillus catenulatus are capable of eradicating Cd2+ [135,136].
6.4. Pb Remediation
There have been reports of Pb- resistant capability of Bacillus spp. in the past. Bacillus speciesexploit the pbr operon and active transport as possible defenses against the harmful effects of Pb [137,138]. It is immobilized by microorganisms through biosorption, adsorption, chelation, extrinsic sedimentation, complexation, and adsorption. Several methods incorporate phosphate, carboxyl, carbonyl, sulfhydryl, and hydroxyl groups in bacterial cell walls, which give the cell wall a negative charge. When Pb binds to any of these, an insoluble material is produced. The capacity of organic macromolecules such as proteins, polypeptides, and polysaccharides to sequester Pb using electrostatic forces including covalent bonds, ionic bonds, and Van der Waal’s forces make adsorption through the cell wall a viable alternative route [139]. By altering the DNA, proteins, and lipids and even substituting the necessary ions inside the enzymes, Pb interacts with bacterial metabolism, structure, and biological activities [140]. To protect themselves from Pb toxicity, microbes exhibit interfacial deposition, ostracization, volatilization, biomethylation, cellular binding, intrinsic sequestration, and increased siderophore synthesis. Bacillus spp. especially B. cereus use one or more of these techniques, similar to other Gram-positive bacteria, to remove Pb from polluted sites [141].
6.5. Cu Remediation
Similar to several other HMs, copper is considered to play a key role in the physiology of B. subtilis, although quantities over typical ranges can be hazardous to the cells. B. cereus, B. thuringiensis, Bacillus sphaericus, and B. licheniformis species are known to participate in the removal of Cu from environments when its concentrations exceed the permissible levels [142-145]. The transport of Cu in the cytosol of B. licheniformis is facilitated by CueR, a protein that is similar to CueR found in other bacterial species [146]. B. subtilis CueR controls the copZA operon, which encodes a P-type ATPase for Cu extrusion, a constituent of the vital category that exports metal from the cell, along with a Cu facilitator. CopB is in charge of Cu extrusion and decontamination in the latter, whereas CopZ is essential as a Cu chaperone in the former, transporting Cu to CopA, which helps to Cu absorption [147]. B. subtilis has shown that YcnJ is associated with the Cu absorption function of the copZA operon. According to their model, this protein works in collaboration with specific Cop proteins to facilitate the transport of Cu both within and outside of the cell [148].
6.6. Cr Remediation
The innate resistance of microbes in Cr-contaminated habitats allows them to avoid metal stress by metal absorption, efflux, or detoxifying by lowering metal ion immobilization [149]. The enzyme-mediated biotransformation of Cr6+ by bacteria out of its hazardous to non-hazardous state (Cr3+) is thought to be one of the most effective and affordable ways to remove Cr from polluted site and wastewater. The enzymatic reduction of chromate in both Gram-positive and Gram-negative bacteria is regulated by an enzyme known as ChrR, also known as chromate reductase. This enzyme is predominantly found in Cr-resistant bacteria and is not commonly associated with plasmids [150]. The membrane-bound component and the cytosolic component, respectively, can mediate this reduction both aerobically and anaerobically. Furthermore, the reduction of chromate ions in Bacillus spp. is mediated by an aerobic mechanism, in which electrons are moved from the hexavalent to the trivalent state of Cr. An unstable intermediate is produced during this transition (Cr5+), which is controlled by NADH/NADPH [151]. Numerous studies Nguema et al. [152] and Joshi et al. [153] indicated that B. licheniformis removed 95% and 69.4% of Cr. Over 81% of the Cr was eliminated by B. cereus. The effectiveness of other Bacillus species in reducing Cr contamination has also been reported [154].
6.7. Hg Remediation
According to several studies conducted over the years, the several species of Bacillus contribute to the bioremediation of Hg in a positive way. Hg has a hazardous impact on biological systems due to its high toxicity. Bacteria are armed with a variety of methods to protect their survival when exposed to high levels of harmful Hg. Hg resistance genes and operons, such as the Mer operon, have been identified in both gram-positive and gram-negative bacteria [155]. A limited range Mer operon and a broad range operon are the two different forms of Hg resistance genes found in bacteria. Inside the cell, Hg is forced to undergo reversible detoxification from its hazardous to non-hazardous form, yet diffusion aids in Hg’s exit from the body. Once the metal ions have left the cell, different bacterial species may once more subject them to oxidation [156]. Numerous bacteria have been used to study the phenomena, where the activation of this phenomenon by the activator protein MerR results in the target identification of Hg. The mercuric reductase and lyase enzymes, which are inherited by the MerA and MerB genes, assist the elimination of inorganic Hg2+ and organic Hg [157]. In addition, the various other forms of Hg might regulate its transit and bioaccumulation, which might lead to Hg toxicity in environments and ecosystems close to Hg mines or emission sites [158]. Metallothioneins play a significant role in the bioremediation of Hg by accumulating Hg ions in an inactive state and facilitating their transformation through enzymatic processes. Gram-positive and Gram-negative bacteria both share the Mer operon. Although the pathway of Hg resistance in Gram-negative bacteria is better understood compared to Gram-positive bacteria, significant progress has been made in unraveling the mechanisms of Hg resistance in both bacterial groups [159]. Moreover, it was reported that soil-isolated Bacillus spp. possessed Hg-resistant genes, which aided in the removal and subsequent bioremediation of Hg [160]. To enhance the biosorption and exudation of Hg from contaminated sites, genetic modification was performed on B. cereus by introducing a mer operon derived from another Bacillus species, B. thuringiensis. This modification aimed to confer Hg resistance to B. cereus and improve its capacity for Hg removal [161].
7. CONCLUSIONS
The promising approach of bioremediation, which involves the use of living organisms to detoxify polluted areas, has shown great potential in remedying heavy metal contamination in the environment. Among different types of bacteria, the potential of Bacillus spp. in the bioremediation of HMs has been well-established and validated. Bacillus spp. has been shown to have the capability to remove various HMs, including Pb, Cu, and Cd, from contaminated soils, groundwater, and industrial effluents. Bacillus spp. has been studied extensively for its molecular mechanisms in heavy metal bioremediation, including metal resistance, efflux, haulage, and detoxification. These processes enable Bacillus spp. to maintain metal homeostasis even in environments with elevated concentrations of HMs. The Bacillus spp. can be used to bioremediate settings that have been contaminated with HMs, although there are still unexplained molecular resistance mechanisms that need to be investigated and characterized. The bioremediation process driven by bacteria is not only efficient but also practical from an economic and environmental standpoint. Sewage sludge, effluents, polluted groundwater, and soil can all be treated using this method. Bacillus spp. can considerably lower the levels of HMs and other environmental pollutants when used in bioremediation. In conclusion, using bacteria-driven bioremediation, in particular using Bacillus spp., is a promising and long-lasting alternative for the treatment of environmental heavy metal contamination. To create more efficient methods for bioremediation, additional study is required to completely comprehend the molecular mechanisms underlying heavy metal resistance and detoxification by Bacillus spp. Bacillus spp. play a significant role in bioremediation, which will become clearer with the identification and characterization of hitherto unknown molecular resistance mechanisms. This will also open the door to their prospective application in the effective removal of heavy metal pollutants.
8. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors requirements/guidelines.
9. FUNDING
There is no funding to report.
10. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
11. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
12. DATA AVAILABILITY
All the data is available with the authors and shall be provided upon request.
13. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Kumar V, Shahi SK, Singh S. Bioremediation:An eco-sustainable approach for restoration of contaminated sites. In:Singh J, Sharma D, Kumar G, Sharma N, editors. Microbial Bioprospecting for Sustainable Development. Singapore:Springer;2018. 115-36. [https://doi.org/10.1007/978-981-13-0053-0_6]
2. Schell LM, Gallo MV, Denham M, Ravenscroft J. Effects of pollution on human growth and development:An introduction. J Physiol Anthropol 2006;25:103-12. [https://doi.org/10.2114/jpa2.25.103]
3. Khan FI, Husain T, Hejazi R. An overview and analysis of site remediation technologies. J Environ Manage 2004;71:95-122. [https://doi.org/10.1016/j.jenvman.2004.02.003]
4. Pichtel J. Waste Management Practice. Boca Raton:Taylor and Francis Group;2005. [https://doi.org/10.1201/9781420037517]
5. Blande JD, Holopainen JK, Niinemets Ü. Plant volatiles in polluted atmospheres:Stress responses and signal degradation. Plant Cell Environ 2014;37:1892-904. [https://doi.org/10.1111/pce.12352]
6. Kampa M, Castanas E. Human health effects of air pollution. Environ Pollut 2008;151:362-7. [https://doi.org/10.1016/j.envpol.2007.06.012]
7. Chowdhary P, Bharagava RN, Mishra S, Khan N. Role of industries in water scarcity and its adverse effects on environment and human health. In:Shukla V, Kumar N, editors. Environmental Concerns and Sustainable Development. Singapore:Springer;2020. 235-56. [https://doi.org/10.1007/978-981-13-5889-0_12]
8. Green TR, Taniguchi M, Kooi H, Gurdak JJ, Allen DM, Hiscock KM, et al. Beneath the surface of global change:Impacts of climate change on groundwater. J Hydrol 2011;405:532-60. [https://doi.org/10.1016/j.jhydrol.2011.05.002]
9. Chaudhry FN, Malik MF. Factors affecting water pollution:A review. J Ecosyst Ecography 2017;7:225-31. [https://doi.org/10.4172/2157-7625.1000225]
10. Khan MA, Ghouri AM. Environmental pollution:Its effects on life and its remedies. J Arts Sci Commer 2011;2:276-85.
11. Halder JN, Islam MN. Water pollution and its impact on the human health. J Environ Hum 2015;2:36-46. [https://doi.org/10.15764/EH.2015.01005]
12. Salem HM, Eweida EA, Farag A. Heavy Metals in Drinking Water and their Environmental Impact on Human Health. In:Proceedings of the International Conference for Environmental Hazards Mitigation. Egypt:Cairo University;2000. 542-56.
13. Havugimana E, Bhople BS, Kumar A, Byiringiro E, Mugabo JP, Kumar A. Soil pollution-major sources and types of soil pollutants. Environ Sci Eng 2017;11:53-86.
14. Chen M, Xu P, Zeng G, Yang C, Huang D, Zhang J. Bioremediation of soils contaminated with polycyclic aromatic hydrocarbons, petroleum, pesticides, chlorophenols and heavy metals by composting:Applications, microbes and future research needs. Biotechnol Adv 2015;33:745-55. [https://doi.org/10.1016/j.biotechadv.2015.05.003]
15. Da Silva IG, de Almeida FC, e Silva NM, Casazza AA, Converti A, Sarubbo LA. Soil bioremediation:Overview of technologies and trends. Energies 2020;13:4664. [https://doi.org/10.3390/en13184664]
16. Li AM. Ecological determinants of health:Food and environment on human health. Environ Sci Pollut Res Int 2017;24:9002-15. [https://doi.org/10.1007/s11356-015-5707-9]
17. Agarwal A, Pandey RS, Sharma B. Water pollution with special reference to pesticide contamination in India. J Water Resour Prot 2010;2:432-48. [https://doi.org/10.4236/jwarp.2010.25050]
18. Soleimani M, Jaberi N. Comparison of biological and thermal remediation methods in decontamination of oil polluted soils. J Bioremediat Biodegrad 2014;5:e145. [https://doi.org/10.4172/2155-6199.1000e145]
19. Ali H, Khan E. What are heavy metals?Long-standing controversy over the scientific use of the term 'heavy metals'-proposal of a comprehensive definition. Toxicol Environ Chem 2018;100:6-19. [https://doi.org/10.1080/02772248.2017.1413652]
20. Thakare M, Sarma H, Datar S, Roy A, Pawar P, Gupta K, et al. Understanding the holistic approach to plant-microbe remediation technologies for removing heavy metals and radionuclides from soil. Curr Res Biotechnol 2021;3:84-98. [https://doi.org/10.1016/j.crbiot.2021.02.004]
21. Gratão PL, Polle A, Lea PJ, Azevedo RA. Making the life of heavy metal-stressed plants a little easier. Funct Plant Biol 2005;32:481-94. [https://doi.org/10.1071/FP05016]
22. Sandeep G, Vijayalatha KR, Anitha T. Heavy metals and its impact in vegetable crops. Int J Chem Stud 2019;7:1612-21.
23. Bradl H. Sources and origins of heavy metals. In:Interface Science and Technology. Amsterdam:Elsevier;2005. 1-27. [https://doi.org/10.1016/S1573-4285(05)80020-1]
24. White PJ, Pongrac P. Heavy-metal toxicity in plants. In:Plant Stress Physiology. Wallingford UK:Cabi;2017. 300-31. [https://doi.org/10.1079/9781780647296.0300]
25. Pierart A, Shahid M, Séjalon-Delmas N, Dumat C. Antimony bioavailability:Knowledge and research perspectives for sustainable agricultures. J Hazard Mater 2015;289:219-34. [https://doi.org/10.1016/j.jhazmat.2015.02.011]
26. Vaishaly AG, Mathew BB, Krishnamurthy NB. Health effects caused by metal contaminated ground water. Int J Adv Sci Res 2015;1:60-4. [https://doi.org/10.7439/ijasr.v1i2.1798]
27. Akan JC, Mohmoud S, Yikala BS, Ogugbuaja VO. Bioaccumulation of some heavy metals in fish samples from River Benue in Vinikilang, Adamawa state, Nigeria. Am J Anal Chem 2012;3:727-36. [https://doi.org/10.4236/ajac.2012.311097]
28. Plante C, Blanchet C, Rochette L, O'Brien HT. Prevalence of anemia among Inuit women in Nunavik, Canada. Int J Circumpolar Health 2011;70:154-65. [https://doi.org/10.3402/ijch.v70i2.17811]
29. Khalid S, Shahid M, Niazi NK, Murtaza B, Bibi I, Dumat C. A comparison of technologies for remediation of heavy metal contaminated soils. J Geochem Explor 2017;182:247-68. [https://doi.org/10.1016/j.gexplo.2016.11.021]
30. Hao X, Chen Y, Xu C, Li W. Impacts of climate change and human activities on the surface runoff in the Tarim river basin over the last fifty years. Water Resour Manage 2008;22:1159-71. [https://doi.org/10.1007/s11269-007-9218-4]
31. Lasat MM. Phytoextraction of metals from contaminated soil:A review of plant/soil/metal interaction and assessment of pertinent agronomic issues. J Hazard Subst Res 1999;2:5-25. [https://doi.org/10.4148/1090-7025.1015]
32. Alloway BJ. Micronutrients and crop production:An introduction. In:Micronutrient Deficiencies in Global Crop Production. Dordrecht:Springer;2008. 1-39. [https://doi.org/10.1007/978-1-4020-6860-7_1]
33. Wuana RA, Okieimen FE. Heavy metals in contaminated soils:A review of sources, chemistry, risks and best available strategies for remediation. Int Sch Res Notices 2011;2011:402647. [https://doi.org/10.5402/2011/402647]
34. Moolenaar SW, Lexmond TM. Heavy metal balances, Part I:General aspects of cadmium, copper, zinc, and lead balance studies in agro-ecosystems. J Ind Ecol 1998;2:45-60. [https://doi.org/10.1162/jiec.1998.2.4.45]
35. Ibitayo O. Agricultural pesticides poisoning in Sub-Sahara Africa:past research and future needs. In:Edward C (ed). Trends in Hazardous Material Research, Nova Science Publisher, 2007;pp 201-17.
36. Pscheidt JW, Ocamb C. Copper-based bactericides and fungicides. In:Pacific Northwest Pest Management Handbooks. Corvallis:Oregon State University;2022.
37. McLaughlin MJ, Hamon RE, McLaren RG, Speir TW, Rogers SL. Review:A bioavailability-based rationale for controlling metal and metalloid contamination of agricultural land in Australia and New Zealand. Aust J Soil Res 2000;38:1037-86. [https://doi.org/10.1071/SR99128]
38. Sisay A. Contamination of heavy metals, source, effects on leaving things and different remediation techniques in soil:A review. Am J Nanosci 2019;5:67-75. [https://doi.org/10.11648/j.ajn.20190504.17]
39. Basta NT, Gradwohl R. Remediation of heavy metal-contaminated soil using rock phosphate. Better Crops Plant Food 1998;82:29-31.
40. Sumner ME. Beneficial use of effluents, wastes, and biosolids. Commun Soil Sci Plant Anal 2000;31:1701-15. [https://doi.org/10.1080/00103620009370532]
41. Bahiru DB, Yegrem L. Levels of heavy metal in vegetable, fruits and cereals crops in Ethiopia:A review. Int J Environ Monit Anal 2021;9:96-103. [https://doi.org/10.11648/j.ijema.20210904.11]
42. Petersen SO, Sommer SG, Béline F, Burton C, Dach J, Dourmad JY, et al. Recycling of livestock manure in a whole-farm perspective. Livest Sci 2007;112:180-91. [https://doi.org/10.1016/j.livsci.2007.09.001]
43. Rigueiro-Rodríguez A, Cuiña-Cotarelo R, Mosquera-Losada M. Liming and sewage sludge effects on cd soil and on ni understory development in reforested Pinus radiata d. don plantations. In:Cagas B, Machac R, Nedelnik J (eds). Grassland Science in Europe. UK, pp 223-25.
44. Jayakumar M, Surendran U, Raja P, Kumar A, Senapathi V. A review of heavy metals accumulation pathways, sources and management in soils. Arab J Geosci 2021;14:2156. [https://doi.org/10.1007/s12517-021-08543-9]
45. Silveira ML, Alleoni LR, Guilherme LR. Biosolids and heavy metals in soils. Sci Agric 2003;60:793-806. [https://doi.org/10.1590/S0103-90162003000400029]
46. FindorákováL, ŠestinováO, Ková?ováM. Assessment of potential sediment contamination using screening methods (XRF, TGA/MS) taking into account principles of green chemistry, Eastern Slovakia. Environ Earth Sci 2017;76:119. [https://doi.org/10.1007/s12665-017-6433-z]
47. Hosseinniaee S, Jafari M, Tavili A, Zare S, Cappai G. Investigating metal pollution in the food chain surrounding a lead-zinc mine (Northwestern Iran);an evaluation of health risks to humans and animals. Environ Monit Assess 2023;195:946. [https://doi.org/10.1007/s10661-023-11551-9]
48. Hosseinniaee S, Jafari M, Tavili A, Zare S, Cappai G, De Giudici G. Perspectives for phytoremediation capability of native plants growing on Angouran Pb-Zn mining complex in northwest of Iran. J Environ Manage 2022;315:115184. [https://doi.org/10.1016/j.jenvman.2022.115184]
49. Sankhla MS, Kumari M, Nandan M, Kumar R, Agrawal P. Heavy metal contamination in soil and their toxic effect on human health:A review study. Int J All Res Educ Sci Methods 2016;4:13-9. [https://doi.org/10.2139/ssrn.3428216]
50. Reed SC, Crites RW, Middlebrooks EJ. Natural Systems for Waste Management and Treatment. 2nd ed. New York:McGraw-Hill Inc.;1995.
51. Bjuhr J. Trace Metals in Soils Irrigated with Waste Water in a Periurban Area Downstream Hanoi City, Vietnam. Uppsala, Sweden:Institutionen for Markvetenskap, Sveriges Lantbruksuniversitet (SLU);2007.
52. Hinojosa MB, Carreira JA, García-Ruíz R, Dick RP. Soil moisture pre-treatment effects on enzyme activities as indicators of heavy metal-contaminated and reclaimed soils. Soil Biol Biochem 2004;36:1559-68. [https://doi.org/10.1016/j.soilbio.2004.07.003]
53. Yao H, Xu J, Huang C. Substrate utilization pattern, biomass and activity of microbial communities in a sequence of heavy metal-polluted paddy soils. Geoderma 2003;115:139-48. [https://doi.org/10.1016/S0016-7061(03)00083-1]
54. Speir TW, Kettles HA, Percival HJ, Parshotam A. Is soil acidification the cause of biochemical responses when soils are amended with heavy metal salts?Soil Biol Biochem 1999;31:1953-61. [https://doi.org/10.1016/S0038-0717(99)00115-7]
55. Huang SH, Peng B, Yang ZH, Chai LY, Zhou LC. Chromium accumulation, microorganism population and enzyme activities in soils around chromium-containing slag heap of steel alloy factory. Trans Nonferrous Met Soc China 2009;19:241-8. [https://doi.org/10.1016/S1003-6326(08)60259-9]
56. De Mora AP, Ortega-Calvo JJ, Cabrera F, Madejón E. Changes in enzyme activities and microbial biomass after “in situ”remediation of a heavy metal-contaminated soil. Appl Soil Ecol 2005;28:125-37. [https://doi.org/10.1016/j.apsoil.2004.07.006]
57. Karaca A, Cetin SC, Turgay OC, Kizilkaya R. Effects of heavy metals on soil enzyme activities. In:Soil Heavy Metals, Soil Biology. Vol. 19. Berlin, Heidelberg:Springer;2010. 237-62. [https://doi.org/10.1007/978-3-642-02436-8_11]
58. Jiwan S, Ajay SK. Effects of heavy metals on soil, plants, human health and aquatic life. Int J Res Chem Environ 2011;1:15-21.
59. Wang Y, Shi J, Wang H, Lin Q, Chen X, Chen Y. The influence of soil heavy metals pollution on soil microbial biomass, enzyme activity, and community composition near a copper smelter. Ecotoxicol Environ Saf 2007;67:75-81. [https://doi.org/10.1016/j.ecoenv.2006.03.007]
60. Garnier J, Quantin C, Martins ES, Becquer T. Solid speciation and availability of chromium in ultramafic soils from Niquelândia, Brazil. J Geochem Explor 2006;88:206-9. [https://doi.org/10.1016/j.gexplo.2005.08.040]
61. Paz-Ferreiro J, Fu S. Biological indices for soil quality evaluation:Perspectives and limitations. Land Degrad Dev 2016;27:14-25. [https://doi.org/10.1002/ldr.2262]
62. McIntyre T. Phytoremediation of heavy metals from soils. Adv Biochem Eng Biotechnol 2003;78:97-123. [https://doi.org/10.1007/3-540-45991-X_4]
63. Sobha K, Poornima A, Harini P, Veeraiah K. A study on biochemical changes in the fresh water fish, Catla catla (Hamilton) exposed to the heavy metal toxicant cadmium chloride. Kathmandu Univ J Sci Eng Technol 2007;1:1-11. [https://doi.org/10.3126/kuset.v3i2.2890]
64. Lee GF, Jones-Lee A. Hazardous chemical site remediation through capping:Problems with long?term protection. Remediat J 1997;7:51-7. [https://doi.org/10.1002/rem.3440070406]
65. Li C, Zhou K, Qin W, Tian C, Qi M, Yan X, et al. A review on heavy metals contamination in soil:Effects, sources, and remediation techniques. Soil Sediment Contam 2019;28:380-94. [https://doi.org/10.1080/15320383.2019.1592108]
66. Rahman RO, Hussain CM. Handbook of Advanced Approaches Towards Pollution Prevention and Control:Volume 1:Conventional and Innovative Technology, and Assessment Techniques for Pollution Prevention and Control. Amsterdam:Elsevier;2021.
67. Pavel LV, Gavrilescu M. Overview of ex situ decontamination techniques for soil cleanup. Environ Eng Manage J 2008;7:815-34. [https://doi.org/10.30638/eemj.2008.109]
68. Wang Y, Li A, Cui C. Remediation of heavy metal-contaminated soils by electrokinetic technology:Mechanisms and applicability. Chemosphere 2021;265:129071. [https://doi.org/10.1016/j.chemosphere.2020.129071]
69. Mosavat N, Oh E, Chai G. A review of electrokinetic treatment technique for improving the engineering characteristics of low permeable problematic soils. Geomat J 2012;2:266-72. [https://doi.org/10.21660/2012.4.3i]
70. Jankaite A, Vasarevi?ius S. Remediation technologies for soils contaminated with heavy metals. J Environ Eng Landsc Manage 2005;13:109-13. [https://doi.org/10.3846/16486897.2005.9636854]
71. Song Y, Ammami MT, Benamar A, Mezazigh S, Wang H. Effect of EDTA, EDDS, NTA and citric acid on electrokinetic remediation of As, Cd, Cr, Cu, Ni, Pb and Zn contaminated dredged marine sediment. Environ Sci Pollut Res Int 2016;23:10577-86. [https://doi.org/10.1007/s11356-015-5966-5]
72. Weitzman L. Factors for selecting appropriate solidification/stabilization methods. J Hazard Mater 1990;24:157-68. [https://doi.org/10.1016/0304-3894(90)87007-5]
73. Yao Z, Li J, Xie H, Yu C. Review on remediation technologies of soil contaminated by heavy metals. Procedia Environ Sci 2012;16:722-9. [https://doi.org/10.1016/j.proenv.2012.10.099]
74. Navarro A. Effect of sludge amendment on remediation of metal contaminated soils. Minerals 2012;2:473-92. [https://doi.org/10.3390/min2040473]
75. Dermont G, Bergeron M, Mercier G, Richer-Laflèche M. Soil washing for metal removal:A review of physical/chemical technologies and field applications. J Hazard Mater 2008;152:1-31. [https://doi.org/10.1016/j.jhazmat.2007.10.043]
76. Kim JO, Lee YW, Chung J. The role of organic acids in the mobilization of heavy metals from soil. KSCE J Civ Eng 2013;17:1596-602. [https://doi.org/10.1007/s12205-013-0323-z]
77. Jiang W, Tao T, Liao Z. Removal of heavy metal from contaminated soil with chelating agents. Open J Soil Sci 2011;1:70-6. [https://doi.org/10.4236/ojss.2011.12010]
78. Sun H, Wang H, Qi J, Shen L, Lian X. Study on Surfactants Remediation in Heavy Metals Contaminated Soils. In:2011 International Symposium on Water Resource and Environmental Protection. New Jersey:IEEE;2011. 1862-5.
79. Maity JP, Huang YM, Fan CW, Chen CC, Li CY, Hsu CM, et al. Evaluation of remediation process with soapberry derived saponin for removal of heavy metals from contaminated soils in Hai-Pu, Taiwan. J Environ Sci (China) 2013;25:1180-5. [https://doi.org/10.1016/S1001-0742(12)60162-4]
80. Almaroai YA, Usman AR, Ahmad M, Kim KR, Moon DH, Lee SS, et al. Effects of synthetic chelators and low-molecular-weight organic acids on chromium, copper, and arsenic uptake and translocation in maize (Zea mays L.). Soil Sci 2012;177:655-63. [https://doi.org/10.1097/SS.0b013e31827ba23f]
81. Leštan D, Luo CL, Li XD. The use of chelating agents in the remediation of metal-contaminated soils:A review. Environ Pollut 2008;153:3-13. [https://doi.org/10.1016/j.envpol.2007.11.015]
82. Olaniran AO, Balgobind A, Pillay B. Bioavailability of heavy metals in soil:Impact on microbial biodegradation of organic compounds and possible improvement strategies. Int J Mol Sci 2013;14:10197-228. [https://doi.org/10.3390/ijms140510197]
83. Yan A, Wang Y, Tan SN, Yusof ML, Ghosh S, Chen Z. Phytoremediation:A promising approach for revegetation of heavy metal-polluted land. Front Plant Sci 2020;11:359. [https://doi.org/10.3389/fpls.2020.00359]
84. Akpor OB, Muchie M. Remediation of heavy metals in drinking water and wastewater treatment systems:Processes and applications. Int J Phys Sci 2010;5:1807-17.
85. Hooda V. Phytoremediation of toxic metals from soil and waste water. J Environ Biol 2007;28 2 suppl:367-76.
86. Rhodes CJ. Mycoremediation (bioremediation with fungi)-growing mushrooms to clean the earth. Chem Speciation Bioavailability 2014;26:196-8. [https://doi.org/10.3184/095422914X14047407349335]
87. Kapahi M, Sachdeva S. Bioremediation options for heavy metal pollution. J Health Pollut 2019;9:191203. [https://doi.org/10.5696/2156-9614-9.24.191203]
88. Hamba Y, Tamiru M. Mycoremediation of heavy metals and hydrocarbons contaminated environment. Asian J Nat Appl Sci 2016;5:48-58.
89. Mani D, Kumar C. Biotechnological advances in bioremediation of heavy metals contaminated ecosystems:An overview with special reference to phytoremediation. Int J Environ Sci Technol 2014;11:843-72. [https://doi.org/10.1007/s13762-013-0299-8]
90. Sarwar N, Imran M, Shaheen MR, Ishaque W, Kamran MA, Matloob A, et al. Phytoremediation strategies for soils contaminated with heavy metals:Modifications and future perspectives. Chemosphere 2017;171:710-21. [https://doi.org/10.1016/j.chemosphere.2016.12.116]
91. Krämer U. Phytoremediation:Novel approaches to cleaning up polluted soils. Curr Opin Biotechnol 2005;16:133-41. [https://doi.org/10.1016/j.copbio.2005.02.006]
92. Gadd GM. Biosorption:Critical review of scientific rationale, environmental importance and significance for pollution treatment. J Chem Technol Biotechnol 2009;84:13-28. [https://doi.org/10.1002/jctb.1999]
93. Jain PK, Bajpai V. Biotechnology of bioremediation-a review. Int J Environ Sci 2012;3:535-49.
94. Srivastava S, Kumar S. Bacterial remediation to control pollution. In:Biological Approaches to Controlling Pollutants. Amsterdam:Elsevier;2022. 285-305. [https://doi.org/10.1016/B978-0-12-824316-9.00017-3]
95. Adams GO, Fufeyin PT, Okoro SE, Ehinomen I. Bioremediation, biostimulation and bioaugmention:A review. Int J Environ Bioremediat Biodegrad 2015;3:28-39. [https://doi.org/10.12691/ijebb-3-1-5]
96. Maitra S. In situ bioremediation-an overview. Res J Life Sci Bioinform Pharm Chem Sci 2018;4:422-37.
97. Azubuike CC, Chikere CB, Okpokwasili GC. Bioremediation techniques-classification based on site of application:Principles, advantages, limitations and prospects. World J Microbiol Biotechnol 2016;32:180. [https://doi.org/10.1007/s11274-016-2137-x]
98. Gadd GM. Metals, minerals and microbes:Geomicrobiology and bioremediation. Microbiology (Reading) 2010;156:609-43. [https://doi.org/10.1099/mic.0.037143-0]
99. Jin Y, Luan Y, Ning Y, Wang L. Effects and mechanisms of microbial remediation of heavy metals in soil:A critical review. Appl Sci 2018;8:1336. [https://doi.org/10.3390/app8081336]
100. Schiewer S, Volesky B. Biosorption processes for heavy metal removal. In:Environmental Microbe-metal Interactions. United States:American Society for Microbiology;2000. 329-62. [https://doi.org/10.1128/9781555818098.ch14]
101. Ma Y, Oliveira RS, Freitas H, Zhang C. Biochemical and molecular mechanisms of plant-microbe-metal interactions:Relevance for phytoremediation. Front Plant Sci 2016;7:918. [https://doi.org/10.3389/fpls.2016.00918]
102. Mishra B, O'Loughlin EJ, Boyanov MI, Kemner KM. Binding of HgII to high-affinity sites on bacteria inhibits reduction to Hg0 by mixed FeII/III phases. Environ Sci Technol 2011;45:9597-603. [https://doi.org/10.1021/es201820c]
103. Ren G, Jin Y, Zhang C, Gu H, Qu J. Characteristics of Bacillus sp. PZ-1 and its biosorption to Pb(II). Ecotoxicol Environ Saf 2015;117:141-8. [https://doi.org/10.1016/j.ecoenv.2015.03.033]
104. Økstad OA, KolstøAB. Genomics of Bacillus species. In:Wiedmann M, Zhang W, editors. Genomics of Foodborne Bacterial Pathogens. Food Microbiology and Food Safety. New York:Springer;2010. p. 29-53. [https://doi.org/10.1007/978-1-4419-7686-4_2]
105. Setlow P, Wang S, Li YQ. Germination of spores of the orders Bacillales and Clostridiales. Annu Rev Microbiol 2017;71:459-77. [https://doi.org/10.1146/annurev-micro-090816-093558]
106. Sanders ME, Morelli L, Tompkins TA. Sporeformers as human probiotics:Bacillus, Sporolactobacillus, and Brevibacillus. Compr Rev Food Sci Food Saf 2003;2:101-10. [https://doi.org/10.1111/j.1541-4337.2003.tb00017.x]
107. Chen K, Tian Z, He H, Long CA, Jiang F. Bacillus species as potential biocontrol agents against citrus diseases. Biol Control 2020;151:104419. [https://doi.org/10.1016/j.biocontrol.2020.104419]
108. Ouattara HG, Reverchon S, Niamke SL, Nasser W. Regulation of the synthesis of pulp degrading enzymes in Bacillus isolated from cocoa fermentation. Food Microbiol 2017;63:255-62. [https://doi.org/10.1016/j.fm.2016.12.004]
109. Chikere CB, Okpokwasili GC, Chikere BO. Bacterial diversity in a tropical crude oil-polluted soil undergoing bioremediation. Afr J Biotechnol 2009;8:2535-40.
110. Alka S, Shahir S, Ibrahim N, Ndejiko MJ, Vo DV, Abd Manan F. Arsenic removal technologies and future trends:A mini review. J Clean Prod 2021;278:123805. [https://doi.org/10.1016/j.jclepro.2020.123805]
111. Paul D, Poddar S, Sar P. Characterization of arsenite-oxidizing bacteria isolated from arsenic-contaminated groundwater of West Bengal. J Environ Sci Health A Tox Hazard Subst Environ Eng 2014;49:1481-92. [https://doi.org/10.1080/10934529.2014.937162]
112. Singh N, Gupta S, Marwa N, Pandey V, Verma PC, Rathaur S, et al. Arsenic mediated modifications in Bacillus aryabhattai and their biotechnological applications for arsenic bioremediation. Chemosphere 2016;164:524-34. [https://doi.org/10.1016/j.chemosphere.2016.08.119]
113. Miyatake M, Hayashi S. Characteristics of arsenic removal from aqueous solution by Bacillus megaterium strain UM-123. J Environ Biotechnol 2009;9:123-9.
114. Ghosh S, Mohapatra B, Satyanarayana T, Sar P. Molecular and taxonomic characterization of arsenic (As) transforming Bacillus sp. strain IIIJ3-1 isolated from As-contaminated groundwater of Brahmaputra river basin, India. BMC Microbiol 2020;20:256. [https://doi.org/10.1186/s12866-020-01893-6]
115. Miller WH Jr., Schipper HM, Lee JS, Singer J, Waxman S. Mechanisms of action of arsenic trioxide. Cancer Res 2002;62:3893-903.
116. Dey U, Chatterjee S, Mondal NK. Isolation and characterization of arsenic-resistant bacteria and possible application in bioremediation. Biotechnol Rep (Amst) 2016;10:1-7. [https://doi.org/10.1016/j.btre.2016.02.002]
117. Santini JM, Stolz JF, Macy JM. Isolation of a new arsenate-respiring bacterium--physiological and phylogenetic studies. Geomicrobiol J 2002;19:41-52. [https://doi.org/10.1080/014904502317246156]
118. Oremland RS, Stolz JF. Arsenic, microbes and contaminated aquifers. Trends Microbiol 2005;13:45-9. [https://doi.org/10.1016/j.tim.2004.12.002]
119. Yang HC, Fu HL, Lin YF, Rosen BP. Pathways of arsenic uptake and efflux. Curr Top Membr 2012;69:325-58. [https://doi.org/10.1016/B978-0-12-394390-3.00012-4]
120. Mukhopadhyay R, Rosen BP, Phung LT, Silver S. Microbial arsenic:From geocycles to genes and enzymes. FEMS Microbiol Rev 2002;26:311-25. [https://doi.org/10.1111/j.1574-6976.2002.tb00617.x]
121. Sharma P, Jha AB, Dubey RS. Arsenic toxicity and tolerance mechanisms in crop plants. In:Pessarakli M, editor. Handbook of Plant and Crop Physiology. Boca Raton:CRC Press;2021. 762-811. [https://doi.org/10.1201/9781003093640-46]
122. Gaballa A, Wang T, Ye RW, Helmann JD. Functional analysis of the Bacillus subtilis Zur regulon. J Bacteriol 2002;184:6508-14. [https://doi.org/10.1128/JB.184.23.6508-6514.2002]
123. Singh PP, Chopra AK. Removal of Zn2+and Pb2+using new isolates of Bacillus spp. PPS03 and Bacillus subtilis PPS04 from paper mill effluents using indigenously designed bench-top bioreactor. J Appl Nat Sci 2014;6:47-56. [https://doi.org/10.31018/jans.v6i1.374]
124. Joo JH, Hassan SH, Oh SE. Comparative study of biosorption of Zn2+by Pseudomonas aeruginosa and Bacillus cereus. Int Biodeterior Biodegrad 2010;64:734-41. [https://doi.org/10.1016/j.ibiod.2010.08.007]
125. Green-Ruiz C, Rodriguez-Tirado V, Gomez-Gil B. Cadmium and zinc removal from aqueous solutions by Bacillus jeotgali:pH, salinity and temperature effects. Bioresour Technol 2008;99:3864-70. [https://doi.org/10.1016/j.biortech.2007.06.047]
126. Moghbeli MR, Khajeh A, Alikhani M. Nanosilica reinforced ion-exchange polyHIPE type membrane for removal of Nickel ions:Preparation, characterization and adsorption studies. Chem Eng J 2017;309:552-62. [https://doi.org/10.1016/j.cej.2016.10.048]
127. Sireesha S, Upadhyay U, Sreedhar I. Comparative studies of heavy metal removal from aqueous solution using novel biomass and biochar-based adsorbents:characterization, process optimization, and regeneration. Biomass Convers Bioref. 2022;[https://doi.org/10.1007/s13399-021-02186-2. https://doi.org/10.1007/s13399-021-02186-2]
128. ?Mardiyono M, Sajidan S, Masykuri M, Setyono P. Bioremediation of nickel heavy metals in electroplating industrial liquid waste with Bacillus subtilis. International Conference on Science and Applied Science (ICSAS), 2019;pp. 200841-5. [https://doi.org/10.1063/1.5141697]
129. Rajendran P, Muthukrishnan J, Gunasekaran P. Current perspectives in nickel bioremediation strategies. Indian J Microbiol 2002;42:1-9.
130. Das P, Sinha S, Mukherjee SK. Nickel bioremediation potential of Bacillus thuringiensis KUNi1 and some environmental factors in Nickel removal. Bioremediat J 2014;18:169-77. [https://doi.org/10.1080/10889868.2014.889071]
131. Njoku K, Akinyede O, Obidi O. Microbial remediation of heavy metals contaminated media by Bacillus megaterium and Rhizopus stolonifer. Sci Afr 2020;10:e00545. [https://doi.org/10.1016/j.sciaf.2020.e00545]
132. Nies DH. Efflux-mediated heavy metal resistance in prokaryotes. FEMS Microbiol Rev 2003;27:313-39. [https://doi.org/10.1016/S0168-6445(03)00048-2]
133. Moore CM, Gaballa A, Hui M, Ye RW, Helmann JD. Genetic and physiological responses of Bacillus subtilis to metal ion stress. Mol Microbiol 2005;57:27-40. [https://doi.org/10.1111/j.1365-2958.2005.04642.x]
134. Basha SA, Rajaganesh K. Microbial bioremediation of heavy metals from textile industry dye effluents using isolated bacterial strains. Int J Curr Microbiol Appl Sci 2014;3:785-94.
135. Priyalaxmi R, Murugan A, Raja P, Raj KD. Bioremediation of cadmium by Bacillus safensis (JX126862), a marine bacterium isolated from mangrove sediments. Int J Curr Microbiol Appl Sci 2014;3:326-35.
136. Kim SY, Jin MR, Chung CH, Yun YS, Jahng KY, Yu KY. Biosorption of cationic basic dye and cadmium by the novel biosorbent Bacillus catenulatus JB-022 strain. J Biosci Bioeng 2015;119:433-9. [https://doi.org/10.1016/j.jbiosc.2014.09.022]
137. Hynninen A, TouzéT, Pitkänen L, Mengin-Lecreulx D, Virta M. An efflux transporter PbrA and a phosphatase PbrB cooperate in a lead-resistance mechanism in bacteria. Mol Microbiol 2009;74:384-94. [https://doi.org/10.1111/j.1365-2958.2009.06868.x]
138. Silver S, Ji G. Newer systems for bacterial resistances to toxic heavy metals. Environ Health Perspect 1994;102 Suppl 3:107-13. [https://doi.org/10.1289/ehp.94102s3107]
139. Qiao W, Zhang Y, Xia H, Luo Y, Liu S, Wang S, et al. Bioimmobilization of lead by Bacillus subtilis X3 biomass isolated from lead mine soil under promotion of multiple adsorption mechanisms. R Soc Open Sci 2019;6:181701. [https://doi.org/10.1098/rsos.181701]
140. Fashola MO, Ngole-Jeme VM, Babalola OO. Heavy metal pollution from gold mines:Environmental effects and bacterial strategies for resistance. Int J Environ Res Public Health 2016;13:1047. [https://doi.org/10.3390/ijerph13111047]
141. Li Q, Zhang W, Liao S, Xing D, Xiao Y, Zhou D, et al. Mechanism of lead adsorption by a Bacillus cereus strain with indole-3-acetic acid secretion and inorganic phosphorus dissolution functions. BMC Microbiol 2023;23:57. [https://doi.org/10.1186/s12866-023-02795-z]
142. Fang L, Cai P, Li P, Wu H, Liang W, Rong X, et al. Microcalorimetric and potentiometric titration studies on the adsorption of copper by P. putida and B. thuringiensis and their composites with minerals. J Hazard Mater 2010;181:1031-8. [https://doi.org/10.1016/j.jhazmat.2010.05.118]
143. Rohini B, Jayalakshmi S. Bioremediation potential of Bacillus cereus against copper and other heavy metals. Int J Adv Res Biol Sci 2015;2:200-9.
144. Da Costa AC, Duta FP. Bioaccumulation of copper, zinc, cadmium and lead by Bacillussp., Bacillus cereus, Bacillus sphaericus and Bacillus subtilis. Braz J Microbiol 2001;32:1-5. [https://doi.org/10.1590/S1517-83822001000100001]
145. Clausen CA. CCA removal from treated wood using a dual remediation process. Waste Manage Res 2000;18:485-8. [https://doi.org/10.1177/0734242X0001800510 https://doi.org/10.1034/j.1399-3070.2000.00151.x]
146. Changela A, Chen K, Xue Y, Holschen J, Outten CE, O'Halloran TV, et al. Molecular basis of metal-ion selectivity and zeptomolar sensitivity by CueR. Science 2003;301:1383-7. [https://doi.org/10.1126/science.1085950]
147. Magnani D, Solioz M. How bacteria handle copper. In:Nies DH, Silver S, editors. Molecular Microbiology of Heavy Metals. Microbiology Monographs. Berlin, Heidelberg:Springer;2007. 259-85. [https://doi.org/10.1007/7171_2006_081]
148. Chillappagari S, Miethke M, Trip H, Kuipers OP, Marahiel MA. Copper acquisition is mediated by YcnJ and regulated by YcnK and CsoR in Bacillus subtilis. J Bacteriol 2009;191:2362-70. [https://doi.org/10.1128/JB.01616-08]
149. Dhal B, Thatoi H, Das N, Pandey BD. Reduction of hexavalent chromium by Bacillus sp. isolated from chromite mine soils and characterization of reduced product. J Chem Technol Biotechnol 2010;85:1471-9. [https://doi.org/10.1002/jctb.2451]
150. Nag A, Sharma D, Arora S. Molecular advances in bioremediation of hexavalent chromium from soil and wastewater. In:Removal of Refractory Pollutants from Wastewater Treatment Plants. Boca Raton:CRC Press;2021. 113-26. [https://doi.org/10.1201/9781003204442-7]
151. Hossan S, Hossain S, Islam MR, Kabir MH, Ali S, Islam MS, et al. Bioremediation of hexavalent chromium by chromium resistant bacteria reduces phytotoxicity. Int J Environ Res Public Health 2020;17:6013. [https://doi.org/10.3390/ijerph17176013]
152. Nguema PF, Luo Z, Lian J. The biosorption of Cr(VI) ions by dried biomass obtained from a chromium-resistant bacterium. Front Chem Sci Eng 2014;8:454-64. [https://doi.org/10.1007/s11705-014-1456-4]
153. Joshi N, Rathod M, Vyas D, Kumar R, Mody K. Multiple pollutants removal from industrial wastewaters using a novel bioflocculant produced by Bacillus licheniformis NJ3. Environ Prog Sustain Energy 2019;38:S306-14. [https://doi.org/10.1002/ep.13027]
154. Nayak AK, Panda SS, Basu A, Dhal NK. Enhancement of toxic Cr (VI), Fe, and other heavy metals phytoremediation by the synergistic combination of native Bacillus cereus strain and Vetiveria zizanioides L. Int J Phytoremediation 2018;20:682-91. [https://doi.org/10.1080/15226514.2017.1413332]
155. Helmann JD, Wang Y, Mahler I, Walsh CT. Homologous metalloregulatory proteins from both gram-positive and gram-negative bacteria control transcription of mercury resistance operons. J Bacteriol 1989;171:222-9. [https://doi.org/10.1128/jb.171.1.222-229.1989]
156. Dash HR, Das S. Bioremediation of mercury and the importance of bacterial mer genes. Int Biodeterior Biodegrad 2012;75:207-13. [https://doi.org/10.1016/j.ibiod.2012.07.023]
157. Silver S, Phung LT. Bacterial heavy metal resistance:New surprises. Annu Rev Microbiol 1996;50:753-89. [https://doi.org/10.1146/annurev.micro.50.1.753]
158. Jing YD, He ZL, Yang XE. Role of soil rhizobacteria in phytoremediation of heavy metal contaminated soils. J Zhejiang Univ Sci B 2007;8:192-207. [https://doi.org/10.1631/jzus.2007.B0192]
159. Das S, Dash HR, Chakraborty J. Genetic basis and importance of metal resistant genes in bacteria for bioremediation of contaminated environments with toxic metal pollutants. Appl Microbiol Biotechnol 2016;100:2967-84. [https://doi.org/10.1007/s00253-016-7364-4]
160. Chen J, Dong J, Shen S, Mei J, Chang J. Isolation of the Hg(II)?volatilizing Bacillus sp. strain DC?B2 and its potential to remediate Hg(II)?contaminated soils. J Chem Technol Biotechnol 2019;94:1433-40. [https://doi.org/10.1002/jctb.5905]
161. Alotaibi BS, Khan M, Shamim S. Unraveling the underlying heavy metal detoxification mechanisms of Bacillus species. Microorganisms 2021;9:1628. [https://doi.org/10.3390/microorganisms9081628]
162. Parmar P, Shukla A, Goswami D, Gaur S, Patel B, Saraf M. Comprehensive depiction of novel heavy metal tolerant and EPS producing bioluminescent Vibrio alginolyticus PBR1 and V. rotiferianusPBL1 confined from marine organisms. Microbiol Res 2020;238:126526. [https://doi.org/10.1016/j.micres.2020.126526]
163. Chellaiah ER. Cadmium (heavy metals) bioremediation by Pseudomonas aeruginosa:A minireview. Appl Water Sci 2018;8:154. [https://doi.org/10.1007/s13201-018-0796-5]
164. He Y, Gong Y, Su Y, Zhang Y, Zhou X. Bioremediation of Cr (VI) contaminated groundwater by Geobacter sulfurreducens:Environmental factors and electron transfer flow studies. Chemosphere 2019;221:793-801. [https://doi.org/10.1016/j.chemosphere.2019.01.039]
165. VojtkováH, MaslanováI, Sedlácek I, SvanováP, JanulkováR. Removal of heavy metals from wastewater by a Rhodococcus sp. bacterial strain. In:International Multidisciplinary Scientific GeoConference, Surveying Geology and Mining Ecology Management. 2012;685-91.
166. Sodhi KK, Kumar M, Singh DK. Multi-metal resistance and potential of Alcaligenes sp. MMA for the removal of heavy metals. SN Appl Sci 2020;2:1885. [https://doi.org/10.1007/s42452-020-03583-4]
167. Heidari P, Sanaeizade S, Mazloomi F. Removal of nickel, copper, lead and cadmium by new strains of Sphingomonas melonis e8 and Enterobacter hormaechei WW28. J Appl Biotechnol Rep 2020;7:208-14.
168. Kumari D, Pan X, Achal V, Zhang D, Al-Misned FA, Mortuza MG. Multiple metal-resistant bacteria and fungi from acidic copper mine tailings of Xinjiang, China. Environ Earth Sci 2015;74:3113-21. [https://doi.org/10.1007/s12665-015-4349-z]
169. Li J, Tang C, Zhang M, Fan C, Guo D, An Q, et al. Exploring the Cr(VI) removal mechanism of Sporosarcina saromensis M52 from a genomic perspective. Ecotoxicol Environ Saf 2021;225:112767. [https://doi.org/10.1016/j.ecoenv.2021.112767]
170. Wu Z, Su J, Ali A, Hu X, Wang Z. Study on the simultaneous removal of fluoride, heavy metals and nitrate by calcium precipitating strain Acinetobacter sp. H12. J Hazard Mater 2021;405:124255. [https://doi.org/10.1016/j.jhazmat.2020.124255]
171. Kumar A, Bisht B, Joshi V. Bioremediation potential of three acclimated bacteria with reference to heavy metal removal from waste. Int J Environ Sci 2011;2:896-908.
172. Mosbah R, Sahmoune M. Biosorption of heavy metals by Streptomyces species-an overview. Open Chem 2013;11:1412-22. [https://doi.org/10.2478/s11532-013-0268-6]
173. Mishra A, Malik A. Simultaneous bioaccumulation of multiple metals from electroplating effluent using Aspergillus lentulus. Water Res 2012;46:4991-8. [https://doi.org/10.1016/j.watres.2012.06.035]
174. Gomes PF, Lennartsson PR, Persson NK, Taherzadeh MJ. Heavy metal biosorption by Rhizopus sp. biomass immobilized on textiles. Water Air Soil Pollut 2014;225:1834. [https://doi.org/10.1007/s11270-013-1834-4]
175. Herliana O, Soesanto L, Mawadah E. Phytobioremediation of cadmium-contaminated soil using combination of Ipomoea reptans poir and Trichodermasp. and its effect on spinach growth and yield. J Degrad Min Land Manage 2018;6:1519-26. [https://doi.org/10.15243/jdmlm.2018.061.1519]
176. Infante JC, De Arco RD, Angulo ME. Removal of lead, mercury and nickel using the yeast Saccharomyces cerevisiae. Rev MVZ Cordoba 2014;19:4141-9. [https://doi.org/10.21897/rmvz.107]
177. Chang J, Zhang H, Cheng H, Yan Y, Chang M, Cao Y, et al. Spent Ganoderma lucidum substrate derived biochar as a new bio-adsorbent for Pb2+/Cd2+removal in water. Chemosphere 2020;241:125121. [https://doi.org/10.1016/j.chemosphere.2019.125121]
178. Al-Hagar OE, Bayoumi RA, Aziz OA, Mousa AM. Biosorption and adsorption of some heavy metals by Fusarium sp. F6c isolate as affected by gamma irradiation and agricultural wastes. ScienceAsia 2020;46:37-45. [https://doi.org/10.2306/scienceasia1513-1874.2020.008]
179. Bahafid W, Joutey NT, Sayel H, Boularab I, El Ghachtouli N. Bioaugmentation of chromium?polluted soil microcosms with Candida tropicalis diminishes phytoavailable chromium. J Appl Microbiol 2013;115:727-34. [https://doi.org/10.1111/jam.12282]
180. Sánchez-Castellón J, Urango-Cárdenas I, Enamorado-Montes G, Burgos-Nuñez S, Marrugo-Negrete J, Díez S. Removal of mercury, cadmium, and lead ions by Penicillium sp. Front Environ Chem 2022;2:795632. [https://doi.org/10.3389/fenvc.2021.795632]
181. Long Y, Lei D, Ni J, Ren Z, Chen C, Xu H. Packed bed column studies on lead(II) removal from industrial wastewater by modified Agaricus bisporus. Bioresour Technol 2014;152:457-63. [https://doi.org/10.1016/j.biortech.2013.11.039]
182. Areco MM, Salomone VN, Dos Santos Afonso M. Ulva lactuca:A bioindicator for anthropogenic contamination and its environmental remediation capacity. Mar Environ Res 2021;171:105468. [https://doi.org/10.1016/j.marenvres.2021.105468]
183. Deng L, Su Y, Su H, Wang X, Zhu X. Biosorption of copper (II) and lead (II) from aqueous solutions by nonliving green algae Cladophora fascicularis:Equilibrium, kinetics and environmental effects. Adsorption 2006;12:267-77. [https://doi.org/10.1007/s10450-006-0503-y]
184. Mane P, Bhosle AS. Bioremoval of some metals by living algae Spirogyra sp. and Spirullina sp. from aqueous solution. Int J Environ Res 2012;6:571-6.
185. Tamilselvan N, Saurav K, Kannabiran K. Biosorption of Cr (VI), Cr (III), Pb (II) and Cd (II) from aqueous solutions by Sargassum wightii and Caulerpa racemosa algal biomass. J Ocean Univ China 2012;11:52-8. [https://doi.org/10.1007/s11802-012-1843-8]
186. Laib E, Leghouchi E. Cd, Cr, Cu, Pb, and Zn concentrations in Ulva lactuca, Codium fragile, Jania rubens, and Dictyota dichotoma from Rabta Bay, Jijel (Algeria). Environ Monit Assess 2012;184:1711-8. [https://doi.org/10.1007/s10661-011-2072-0]
187. Kumar JI, Oommen C, Kumar RN. Biosorption of heavy metals from aqueous solution by green marine macroalgae from Okha Port, Gulf of Kutch, India. Am Eurasian J Agric Environ Sci 2009;6:317-23.