1. INTRODUCTION
Nowadays, rapid industrialization and extensive human activities have a great impact on the environment. Industrial discharge of waste containing harmful compounds in the environment and the wide use of pesticides in agricultural fields reaches nearby soil and water through various means and adversely degrade their quality. These toxicants finally enter the food chain and distress crop quality and health of humans and animals. Pesticides as a whole or in their metabolized form are potent to cause eco-toxicological risks. In aquatic environments, they adversely affect the aquatic organisms and the complete food chain including humans through ecological cycle and biomagnifications. Toxic chemicals even at very low concentration can generate anomalies in various body tissues and organs that hinder the proper growth, development, and physiological processes leading to certain disorders or abnormalities.
Fungicides are a class of pesticides, applied to control the emergence or growth of fungi, responsible for causing fungal diseases in crops [1]. The ethylene bis-dithiocarbamate fungicide, Mancozeb (MZB), a polymeric mixture of zinc- and manganese-ethylene dithiocarbamates, is widely used in agricultural fields as a broad-spectrum contact fungicide acting as a disruptor of cellular metabolism in the target organism, and its consumption has been significantly increased in the recent years [2-4]. Though MZB has restricted persistence in the environment, upon exposure it accounts for high toxicity in non-target species such as aquatic organisms [5]. It is recognized as a potent marine pollutant, harmful for aquatic life [6]. Many studies illustrate MZB-induced toxicity in fish and mammal species, affecting their vital organs and associated physiological processes [7-9].
Pesticides function via imbalanced production of reactive oxygen species (ROS) or free oxygen radicals causing oxidative stress in target species to control their actions. Similarly, they affect nontarget species thus, cause different diseases and anomalies. To manage the generation of free radicals, anti-oxidant system of the body functions naturally, however, it gets disturbed in the state of oxidative stress. Thus, over-produced radicals induce changes in levels of oxidative enzymes [10]. Oxidative stress has been associated with triggering DNA breakage, resulting in genotoxicity. Formation of MN in nucleated erythrocytes provides signal of genotoxicity in fish. MNs are small nuclei formed from the stripped DNA and are present along with the main nucleus in the cells. Both clastogenic and aneugenic events may generate MN during the anaphase stage of the mitotic cycle under the influence of pesticide exposure [11]. Studies have reported that toxicants are potent to cause histological damage, thus reducing their proper growth and development. Damage in tissue serves as a constraint in the normal function of the related organ [10]. The aforementioned impairments have been studied in different species of fish under the exposure of pesticides; however, the information on acute toxic effects of MZB in terms of oxidative stress, genotoxicity, and histopathological changes in vital organs of fish is not sufficient. Therefore, this study has been undertaken to investigate the same in a freshwater teleost, Channa punctatus.
2. MATERIALS AND METHODS
2.1. Test Chemical
The fungicide, MZB (Lumizeb- MZB 75% W.P., Monsoon Agro Chemicals Pvt. Ltd. Azadnagar, New Delhi, India; and Batch No. M0152/031) was purchased from a local retailer for the toxicological study. Other chemicals and reagents of analytical grade were used.
2.2. Test Animal and Acclimatization
Healthy specimens of freshwater edible fish, C. punctatus, (25 ± 5 g in weight and 16 ± 2 cm in length) were procured from the natural water bodies of Lucknow, Uttar Pradesh, India, (Longitude 26° 55′ N, Latitude 80° 59′ E) and were subjected to primary treatment, to disinfect them [11]. Thoroughly sorted, 150 healthy and disinfected fish were acclimatized to the laboratory environment for 20 days in de-chlorinated tap water by following the guidelines of [12,13].
2.3. Estimation of 96 h-LC50 of MZB
The 96-h LC50 of MZB was estimated by following the procedures as given by OECD guidelines for acute bioassays of fish (OECD203, 92/69/EC, method C1) and protocols of APHA [12,13]. Primarily, the toxicity range for MZB was determined, by exposing fish to its different concentrations, viz., 9, 10, 11, 12, 13 mg/l. The toxicity on the basis of percentage mortality, ranged in between 11 and 12 mg/l. Thereafter, to evaluate the 96-h LC50 fish were exposed to 11.06, 11.16, 11.26, 11.36, 11.46, 11.56, 11.66, 11.76, 11.86, 11.96 mg/l of MZB. Throughout the experiment, fish were loaded in aquaria at the rate of 4g/l of water [14]. The mortality of fish in percentage was recorded up to 96 h of exposure period. To ensure the reproducibility, experiment was continually repeated thrice. Probit analysis method was used for the determination of 96-h LC50 of MZB [15].
2.4. Experimental Setup
Three different groups were designed in glass aquaria each having duly acclimatized 10 fish, control was designated as group I, group II contained 1.156 mg/L of MZB (1/10th of 96-h LC50) and group III contained 2.312 mg/L of MZB (1/5th of 96-h LC50). No mortality was observed during the experimental period. Aquaria were kept clean throughout the experiment. Physico-chemical parameters namely pH 7.3, temperature 27°C, dissolved oxygen 6.3 mg/l, alkalinity 3.6 mg/l and hardness 58 mg/l were recorded before the start of the exposure period and at its end. At the end of the 96-h exposure period, clove oil (0.40 mL/L) was used to euthanize three fish from each replicate [16]. Thereafter, blood samples were collected for the estimation of ROS and MN. Vital organs (gills, liver, kidney, and muscles) were dissected out, for the assessment of biomarkers of oxidative stress and histopathology.
2.5. Measurement of ROS
In MZB-exposed fish, ROS was measured in blood cells using 2´, 7´-dichloro dichlorofluorescein diacetate fluorescent dye (DCFH-DA, Sigma Aldrich, USA; CAS Number 4091-99-0). Succinctly, for 30 min whole blood was incubated with DCFH-DA dye and slides were prepared and dried in the dark. Afterward, slides were examined under a fluorescent microscope (Nikon Corporation K 12432) to view and capture ROS-generated intracellular fluorescence. Quantification of the fluorescence intensity in cells of every captured picture was done through Image J software (version 1.50, USA) and expressed as corrected total cell fluorescence in the experimental group to the control.
2.6. Estimation of Oxidative Stress-Related Biomarkers
2.6.1. Measurement of reduced glutathione (GSH) activity
In the sample tissues, the activity of reduced GSH was measured by following the methodology of [17]. First, trichloroacetic acid (TCA 5% w/v) was added to the supernatants of gills, liver, kidney, and muscle tissues separately and was centrifuged for 10 min at 6000 rpm. Thereafter, supernatants were separately mixed with Tris-HCl buffer and 0.01 M DNTB (2, 20-dinitro-5, 50-dithiobenzoic acid, Ellman’s reagent) and incubated at room temperature for 10 min. The UV–VIS spectrophotometer (Shimadzu, UV-1800 pharma spec, Kyoto, Japan, EW-83400-20) was used to record the absorbance of the reaction mixtures at 412 nm. The activity of GSH is denoted as mg/mg of protein.
2.6.2. Evaluation of lipid peroxidation (LPO) activity
LPO activity in gills, liver, kidney, and muscles sample tissues was evaluated by the thiobarbituric acid reactive substances (TBARS) method described by Buege and Aust [18]. The homogenized supernatants were mixed with the solution of thiobarbituric acid, trichloroacetic acid, and hydrochloric acid, followed by boiling, cooling, and centrifugation of the mixture at 3000 rpm for 10 min at 4°C. The absorbance of the mixture was taken at 535 nm using UV–VIS spectrophotometer (Shimadzu, UV-1800 pharma spec, Kyoto, Japan; EW-83400-20). The value of LPO is expressed as nMoles of TBARS formed.
2.7. Histopathological Examinations
Tissue samples were sectioned, dehydrated, stained, and fixed for histopathological study by following the methods used by [19]. Briefly, tissue samples were rinsed with 0.9% NaCl saline solution to wash off the blood, mucus, and cellular debris. Thereafter, tissues were fixed in Bouin’s fluid for 48 h and were rinsed twice a day with 70% ethyl alcohol for five continuous days. Thereafter, tissues were dehydrated in graded series of ethyl alcohol. Once dehydrated, tissues were washed with xylol for 30 min. Washed tissue was embedded in paraffin wax and was stored overnight in a refrigerator. The next day, tissues were sectioned using a Yorco Precision Rotary Microtome (YSI062), and flattened over clean glass slides by applying water and heat. Later, sectioned tissues were stained with hematoxylin for 1 min and eosin for 2 min respectively. The stained tissues were mounted with dibutyl phthalate polystyrene xylene (DPX). An oil immersion microscope was used to capture the microphotographs of the tissues and the obtained images were analyzed through ImageJ software (ImageJ bundled with 64-bit Java 1.8.0_172).
2.8. Measurement of Genotoxicity: Micronucleus Test
Genotoxicity was measured in terms of MN frequency, as reported previously [20,21]. A uniform film of blood from each sampled fish was made on pre-cleaned slides and kept for drying at room temperature. Following drying, fixation was done by dipping slides in absolute methanol for 5 min. Subsequently, slides were stained for 3-5 min with May-Grunwald’s dye and for 30 min with 5% Giemsa stain, respectively, and were mounted with DPX. The fixed slides were observed under Nikon Corporation K 12,432 oil immersion microscope for identification and scoring of micronuclei in erythrocytes of the sampled blood.
2.9. Statistical Analysis
Data were analyzed statistically (Significance: P < 0.05) using SPSS software version 20.0. One-way analysis of variance with Tukey’s post hoc test was carried out to compare means between the experimental groups and control groups.
3. RESULTS AND DISCUSSION
Sustainable aquaculture primarily depends upon the health of fish. The biological functions of fish are greatly influenced by the surrounding medium. Fish widely encounter pesticides with varying concentrations and subsequently, their health gets affected. In the present study, health impairments in fish have been studied under the exposure to MZB. The findings demonstrate manifestations leading to oxidative stress, genotoxicity and histological anomalies in the gills, liver, kidney, and muscles of fish. Throughout the study, no change was observed in the values of physiological parameters. The 96-h LC50 value of 11.56 mg/L of MZB to C. punctatus in this study is in correspondence with the earlier findings for Oreochromis mossambicus (11.68 mg/L), and Puntius ticto (12.95 mg/L) [22,23].
Pesticides are found to induce oxidative stress by increasing cellular concentration of ROS, such as hydroxyl ions, superoxide anions, and hydrogen peroxide that causes changes in various biological processes. Along with oxidative damage to the biological membrane, elevated ROS damages DNA and tissues [10,24]. Figure 1 shows a significant increase (P < 0.05) in the formation of ROS in erythrocytes of groups exposed to MZB in comparison to the control after the 96hr of the treatment period in the present study. Group III (1/5th of 96-h LC50 of MZB) recorded the highest generated ROS. These results are in accordance with the finding of earlier studies reporting that zinc and manganese, which are major constituents of MZB, induce ROS production in vital organs of fish such as C. punctatus and Danio rerio [19,24]. Due to the overproduction of ROS, the activity of antioxidant enzymes of the biological system gets altered, which are primarily involved in scavenging ROS and in repairing injured tissues [10,25-28].
 | Figure 1: (a) Graph shows a significant (P < 0.05) increase in ROS represented as corrected total cell fluorescence (CTCF) in exposed groups (II and III) as compared to the group I (control) after 96-h exposure period in erythrocytes of Channa punctatus (The data are expressedas mean ± SD; *denotes the significant (P < 0.05)) (b) Micro photographic images showing ROS induced DCF fluorescence in group II and III after 96-h exposure period. (Scale bar – 100 µm). [Click here to view] |
Fishes contain large amounts of highly unsaturated fatty acids rendering them susceptible to LPO expressed in terms of TBARS, a major contributor to the loss of cell function under oxidative stress. Enzymatic marker of oxidative stress LPO, present in the cell membrane is vulnerable to acute exposure to pesticides [29]. Studies have reported increased LPO activity in different organs of fish species namely, D. rerio exposed to MZB and other pesticides [25,30]. In addition, Astyanax aeneus exposed to diazinon (organophosphate) and buprofezin [31]; whereas Catla catla and C. punctatus were exposed to methyl parathion and atrazine, respectively [32]. In agreement with the aforementioned findings this study shows a significant (P < 0.05) enhancement in the LPO activity in the gills, liver, kidney, and muscles tissue sample in fish of the MZB exposed group compared to the control, with the highest activity in tissues of group III fish (Figure 2a). Among all the tissue samples highest activity for LPO was observed in group III of the liver. Conversely, negative relation of fungicides -metalaxyl and azoxystrobin with LPO has been observed in Astyanax aeneus [31].
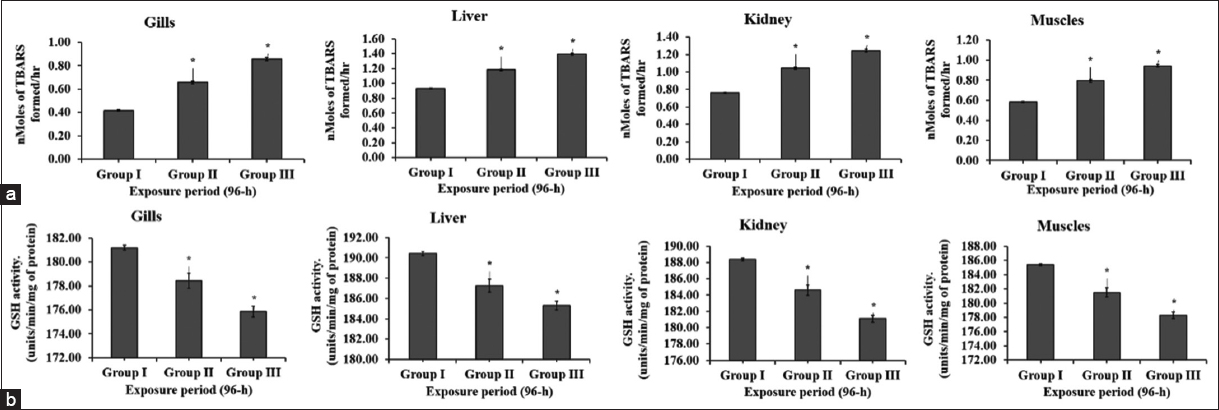 | Figure 2: Activity of LPO and GSH in gill, liver, kidney and muscle tissue of fish in control (group I) and MZB treated groups (II and III) (a) and (b), respectively, for 96hr of exposure period in fish Channa punctatus. (mean ± SD,*denotes the significant (P < 0.05) values of group II and III in comparison to group I). [Click here to view] |
Detoxification of pesticides in fish can be achieved non-enzymatically by a tripeptide GSH, which is observed to scavenge ROS and other substrates of antioxidant enzymes [32-34]. In this study we observed a significant (P < 0.05) decrease in the activity of GSH in MZB-exposed groups in the test samples of the fish as compared to the control, as shown in Figure 2b. Group III of the experiment recorded the lowest GSH content. This decrease in levels of GSH in all the sample tissues demonstrates GSH is involved in neutralizing the generated ROS to mitigate oxidative stress. Results from the study are in accordance with Du et al., [35], who also observed the reduced GSH concentration in fish, Anguilla anguilla, and ethyl carbamate-exposed cuttlefish., exposed to potassium dichromate.
The exceeded level of oxidative stress beyond the activity of the antioxidant system in a body of fish is a probable reason behind the induced genotoxicity. The accumulation of ROS in nucleus triggers DNA damage in association with endonucleases under the state of oxidative stress. Endonucleases cleaves DNA either by breaking single strand or double strand thus, the stripped DNA results in the formation of MN [25,26,36]. A significant (P < 0.05) induction in MN frequency in fish of MZB-treated groups II and III, as compared to control, in the present study (Figure 3), ascertains MZB-induced DNA damage. The highest frequency of MN was observed in group III. Similarly, previous studies done by Srivastava and Singh, 2013 [37] and Marques et al., in 2016 [9] have addressed MZB-induced genotoxicity in fish.
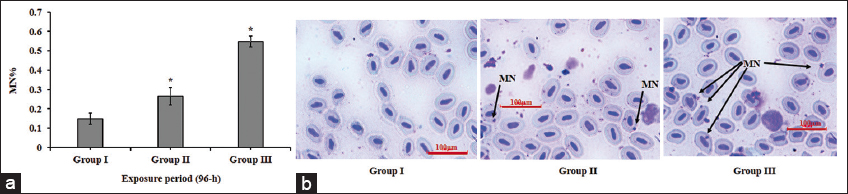 | Figure 3: (a) Increase in MN (%) in MZB-exposered blood cells of groups II and III in relation to group I (control) after 96-h of the exposure period (data are represented as mean ± SD, *denotes the significant (P < 0.05) values of group II and III in comparison to group I).(b) Microphotographic images showing formation of MN in group II and III in comparison to group I. (Scale bar – 100 µm). [Click here to view] |
An increase in ROS can cause tissue damage which can be visualized by histopathological alterations. In the present study, the fish, C. punctatus in the control group were observed to have normal tissue histology, while all the tissue samples of fish in MZB exposed groups showed histopathological abnormalities after 96-h of exposure period, as represented in Figure 4. The gills are the source of the exchange of gases and are constantly exposed to the polluted water. The gills in fish of MZB exposed group III which recorded high level of ROS, were observed to have histological alterations such as destruction in gill arches and necrosis (N) in the present study. Likewise, liver tissues one of the major site of neutralizing toxicant, underwent anatomical manifestations such as vacuolization (V), necrosis (N), and cytoplasmic degeneration in fish of MZB treated group III suggesting oxidative stress leads to tissue destruction. The kidney is also an important organ associated with the biotransformation of toxicants. The results of the present study show anomalies such as cavity reduction in renal tubule, hypertrophy, and vacuolization (V) in kidney tissue of MZB-affected fish of group III that indicates an elevated level of ROS effects the metabolic process of kidney, thereby altering its growth. Motor functions of the body are completely associated with muscles hence, require their proper development. In the present study fish in MZB-treated group III showed muscle degeneration (MD) and vacuolization (V) suggesting oxidative stress hinders the growth of muscle tissue. Similarly, earlier studies have also recorded the histopathological alterations in various organs of fish namely, C. punctatus and D. rerio exposed to toxicants demonstrating change in antioxidant properties of the body under the stress condition damages the tissue, subsequently, leads to damaged body tissue with minimal growth and associated functions [7,11,38].
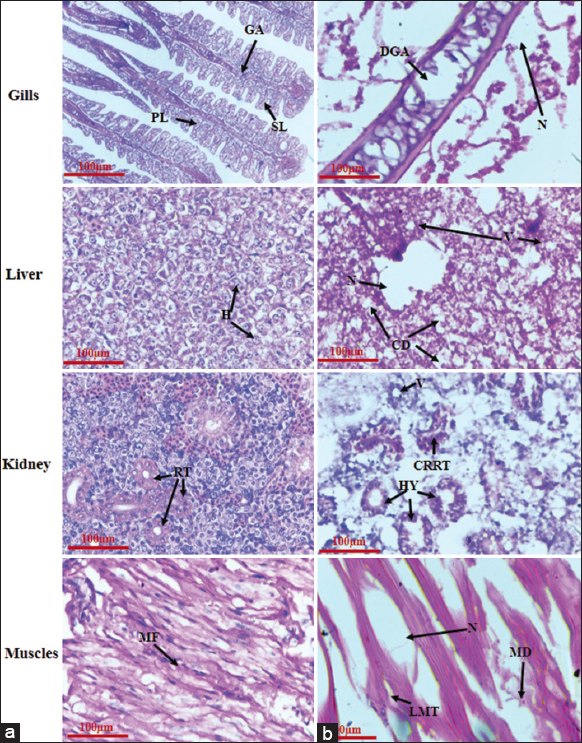 | Figure 4: Microphotographs presenting the histopathology of gills, liver, kidney and muscles of Channa punctatus. (a) Group I (control) represents normal tissue histology, viz., PL-Primary Lamella, SL-Secondary Lamella and GA-Gill Arches in gills; H-Hepatocytes in liver; RT-Renal Tubules in kidney and MF: Muscle fibres in muscles tissues of the fish. (b) Group III (1/5th of 96-h LC50 of MZB) shows histopathological abnormalities, viz., DGA-Destruction of Gill Arches and N-Necrosis in gills; V: Vacuolization, N: Necrosis and CD: Cytoplasmic degeneration in liver, CRRT: Cavity reduced in renal tubule, HY: Hypertrophy and V: Vacuolization in kidney and LMT: Lesion of muscle tissue, MD: Muscle degeneration and N: Necrosis in muscle tissues of C. punctatus. (Scale bar – 100 µm). [Click here to view] |
4. CONCLUSION
The present study reveals that acute exposure of MZB induces severe morphological, anatomical, and biochemical alterations in C. punctatus. They include a significant increase in ROS causing oxidative stress and genotoxicity, leading to irreversible histopathological disorders in the vital organs (gills, liver, kidney, and muscles) of the fish. The results demonstrate detrimental effects caused in aquatic organisms by the use of fungicides like MZB and recommend limitations in the application of fungicides like MZB near aquatic habitats.
5. AUTHORS CONTRIBUTION
Manoj Kumar contributed to the study’s conception and design. Experimental setup and sampling were done by Anjali Mishra and Akash Verma; Adeel Ahmad Khan and Shikha Dwivedi collected data and performed statistical analysis; Sunil P. Trivedi analyzed and interpreted the whole data. The first draft of the manuscript was written by Anamika Jain and Manoj Kumar. All authors read and approved the final manuscript.
6. ACKNOWLEDGMENT
We record our sincere thanks to the Head, Department of Zoology, University of Lucknow, Lucknow (226007) for facilitating in smooth conduct of laboratory work.
7. FUNDING
There is no funding to declare.
8. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
9. ETHICAL APPROVALS
An Institutional Animal Ethics Committee vide registration no. 1861/GO/Re/S/16/CCSEA already exists in the University of Lucknow, Lucknow. All the protocols and methods mentioned therein for the purpose of control and supervision of experiments on animals CPCSEA for maintenance and experiment has been followed religiously.
10. DATA AVAILABILITY
All the data have been provided in the manuscript.
11. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. National Pesticide Information Center. Fungicides. Available from:https://npic.orst.edu/ingred/ptype/fungicide.html [Last accessed on 2022 Jan 20].
2. United States Environment Protection Agency. Reregistration Eligibility Decision for Mancozeb;2005. Available from:https://archive.epa.gov/pesticides/reregistration/web/pdf/mancozeb_red.pdf
3. Tzanova M, Atanasov V, Zaharinov B, Beev G, Dinev T, Valkova E. Reproduction impact of Mancozeb on rainbow trout (Oncorhynchus mykiss W.) and accumulation of its carcinogen metabolite, ethylene thiourea in fish products. J Cent Eur Agric 2017;18:369-87. [CrossRef]
4. Huang Z, Wang P, Pu Z, Lu L, Chen G, Hu X, et al. Effects of mancozeb on Citrus rhizosphere bacterial community. Microb Pathog 2021;154:104845. [CrossRef]
5. Geissen V, Ramos FQ, Bastidas-Bastidas PJ, Díaz-González G, Bello-Mendoza R, Huerta-Lwanga E. Soil and water pollution in a banana production region in tropical Mexico. Bull Environ Contam Toxicol 2010;85:407-13. [CrossRef]
6. Lewis KA, Tzilivakis J, Warner D, Green A. An international database for pesticide risk assessments and management. Hum Ecol Risk Assess 2016;22:1050-64. [CrossRef]
7. Choudhury R, Das P. Histopathological studies on liver and kidney of Channa punctatus (Bloch) exposed to Mancozeb containing pesticide dithane M-45. Pollut Res 2010;39:87-9.
8. Mohammadi-Sardoo M, Mandegary A, Nabiuni M, Nematollahi-Mahani SN, Amirheidari B. Mancozeb induces testicular dysfunction through oxidative stress and apoptosis:Protective role of N-acetylcysteine antioxidant. Toxicol Ind Health 2018;34:798-811. [CrossRef]
9. Marques A, Rego A, Guilherme S, Gaivão I, Santos MA, Pacheco M. Evidences of DNA and chromosomal damage induced by the mancozeb-based fungicide Mancozan® in fish (Anguilla anguilla L.). Pestic Biochem Physiol 2016;133:52-8. [CrossRef]
10. Trivedi SP, Ratn A, Yashika A, Kumar M, Trivedi A. In vivo assessment of dichlorvos induced histological and biochemical impairments coupled with expression of p53 responsive apoptotic genes in the liver and kidney of fish, Channa punctatus (Bloch, 1793). Comp Biochem Physiol C Toxicol Pharmacol 2021;245:109032. [CrossRef]
11. Awasthi Y, Ratn A, Prasad R, Kumar M, Trivedi A, Shukla JP, et al. A protective study of curcumin associated with Cr6+induced oxidative stress, genetic damage, transcription of genes related to apoptosis and histopathology of fish, Channa punctata (Bloch, 1793). Environ Toxicol Pharmacol 2019;71:103209. [CrossRef]
12. APHA, AWWA, WEF. In:Baird RB, Eaton AD, Rice EW, editors. Standard Methods for the Examination of Water and Wastewater. 23rd ed. Washington, DC:APHA;2017.
13. OECD. Test No. 203:Fish, Acute Toxicity Testing, Section 2:Effects on Biotic Systems. In:OECD Guidelines for the Testing of Chemicals. Vol. 10. France:OECD;2019.
14. Palermo FF, Risso WE, Simonato JD, Martinez CB. Bioaccumulation of nickel and its biochemical and genotoxic effects on juveniles of the Neotropical fish Prochilodus lineatus. Ecotoxicol Environ Saf 2015;116:19-28. [CrossRef]
15. Ratn A, Prasad R, Awasthi Y, Kumar M, Misra A, Trivedi SP. Zn 2+induced molecular responses associated with oxidative stress, DNA damage and histopathological lesions in liver and kidney of the fish, Channa punctatus (Bloch, 1793). Ecotoxicol Environ Saf 2018;151:10-20. [CrossRef]
16. Pronina GI, Mannapov AG, Petrushin AB, Rozumnaya LA, Koryagina NY. Technological methods of breeding and rearing European catfish Silurus glanis in carp fish farms. Aquac Aquarium Conserv Legislation 2022;15:520-31.
17. Ellman GL. Tissue sulfhydryl groups. Arch Biochem Biophys 1959;82:70-7. [CrossRef]
18. Buege JA, Aust SD. Microsomal lipid peroxidation. Methods Enzymol 1978;52:302-10. [CrossRef]
19. Finney DJ. Statisical logic in the monitoring of reactions to therapeutic drugs. Methods Inf Med 1971;10:237-45. [CrossRef]
20. Schmid W. The micronucleus test. Mutat Res 1975;31:9-15. [CrossRef]
21. Fenech M, Kirsch-Volders M, Natarajan AT, Surralles J, Crott JW, Parry J, et al. Molecular mechanisms of micronucleus, nucleoplasmic bridge and nuclear bud formation in mammalian and human cells. Mutagenesis 2011;26:12-32. [CrossRef]
22. Saha NC, Giri SK, Chatterjee N, Biswas SJ, Bej S. Acute toxic effects of mancozeb to fish Oreochromis mossambicus (W. K. H. Peters, 1852) and their behaviour. Int J Adv Res Biol Sci 2016;3:40-4.
23. Sharma MR, Mushtaq R, Allayie SA, Vardhan H. Assessment of lethal toxicity of mancozeb and its consequences on the behavior of fresh water fish, Puntius ticto. J Int Acad Res Multidiscipl 2016;4:132-8.
24. Kayhan FE, Kaymak G, Duruel HE, K?z?lkaya S. Some oxidative stress parameters in heart tissue of zebrafish (Danio rerio) caused by mancozeb. Ege J Fish Aquat Sci 2019;36:325-8. [CrossRef]
25. Kumar M, Singh S, Dwivedi S, Trivedi A, Dubey I, Trivedi A. Copper-induced genotoxicity, oxidative stress, and alteration in transcriptional level of autophagy-associated Genes in Snakehead Fish. Channa punctatus. Biol Trace Elem Res 2022;201:2022-35. [CrossRef]
26. Kumar M, Singh S, Dwivedi S, Dubey IY, Trivedi A. Altered transcriptional levels of autophagy-related genes, induced by oxidative stress in fish Channa punctatus exposed to chromium. Fish Physiol Biochem 2022;48:1299-13. [CrossRef]
27. Qu R, Feng M, Wang X, Qin L, Wang C, Wang Z, et al. Metal accumulation and oxidative stress biomarkers in liver of freshwater fish Carassius auratus following in vivo exposure to waterborne zinc under different pH values. Aquat Toxicol 2014;150:9-16. [CrossRef]
28. Birben E, Sahiner UM, Sackesen C, Erzurum S, Kalayci O. Oxidative stress and antioxidant defense. World Allergy Organ J 2012;5:9-19. [CrossRef]
29. Tabassum H, Dawood AQ, Sharma P, Khan J, Raisuddin S, Parvez S. Multi-organ toxicological impact of fungicide propiconazole on biochemical and histological profile of freshwater fish Channa punctata Bloch. Ecol Indic 2016;63:359-65. [CrossRef]
30. Severo ES, Marins AT, Cerezer C, Costa D, Nunes M, Prestes OD, et al. Ecological risk of pesticide contamination in a Brazilian river located near a rural area:A study of biomarkers using zebrafish embryos. Ecotoxicol Environ Saf 2020;190:110071. [CrossRef]
31. Mena F, Vargas S, Guevara-Mora M, Vargas-Hernández JM, Ruepert C. Biotransformation and oxidative stress responses in fish (Astyanax aeneus) inhabiting a Caribbean estuary contaminated with pesticide residues from agricultural runoff. Environ Sci Pollut Res Int 2022;30:21481-93. [CrossRef]
32. Khatib I, Rychter P, Falfushynska H. Pesticide pollution:Detrimental outcomes and possible mechanisms of fish exposure to common organophosphates and Triazines. J Xenobiot 2022;12:236-65. [CrossRef]
33. Sukhovskaya IV, Borvinskaya EV, Smirnov LP, Kochneva AA. Role of glutathione in functioning of the system of antioxidant protection in fish (review). Inland Water Biol 2017;10:97-102. [CrossRef]
34. Oruc EO, Sevgiler Y, Uner N. Tissue-specific oxidative stress responses in fish exposed to 2,4-D and azinphos methyl. Comparative biochemistry and physiology. Toxicol Pharmacol 2004;137:43-51. [CrossRef]
35. Du B, Deng G, Zaman F, Ma H, Li X, Chen J, et al. Antioxidant cuttlefish collagen hydrolysate against ethyl carbamate-induced oxidative damage. RSC Adv 2021;11:2337-45. [CrossRef]
36. Trivedi SP, Ratn A, Awasthi Y, Gupta N, Kumar M, Trivedi A. Micronuclei and other nuclear abnormalities in phorate exposed fish, Channa punctatus. J Environ Biol 2021;42:1221-31. [CrossRef]
37. Srivastava P, Singh A. In vivo study of effects of dithiocarbamates fungicide (Mancozeb) and its metabolite ethylene thiourea (ETU) on fresh water fish Clarius batrachus. JBiol Earth Sci 2013;3:228-35.
38. Güro MV, Arman S, Yön ND. Effects of mancozeb on the testicular histology of the zebrafish (Danio rerio). Int J Lim 2021;56:10. [CrossRef]