1. INTRODUCTION
Pesticides are developed for increased agricultural productivity and yield, forming part of “green revolution” [1]. The application of vast variety of pesticide and chemical fertilizer in modern agriculture practice has impacted negatively on the environment and consumer health [2–4]. Large percentage of the toxic chemicals in pesticides designed to deliberately kill pests end up in air, water, sediments, and food. Health issues, such as diabetes, cancer, Parkinson’s and Alzheimer’s disease, chronic respiratory problems, neurological disorders, spontaneous abortions, and infertility, and increased risk of development of congenital malformations, have being implicated to residues of pesticides in food [5, 6].
Uproot is a glyphosate-based herbicide commonly used at home and commercially in Nigeria. Glyphosate is an important active ingredient of many commercial herbicides, absorbed through the foliage of plants and transported to the roots through xylem and phloem, destroying the root tissue. The quality and activity of this commercial herbicide is improved by preparing it with isopropylamine salt of glyphosate and polyethoxylene amine surfactant [7,8]. The relative low persistence of glyphosate in the environment, results in repeated application [9,10] for the control of weeds in agricultural fields and household lawns, thereby, large quantities find their way into the water bodies [11].
There are records of glyphosate-induced oxidative stress [12] in mammals due to the generation of free radicals resulting in biochemical derangement and organ damage. This condition occurs due to disruption of antioxidant compounds concentration [10], and antioxidant enzyme system [13,14]. Studies have shown that the use of food containing antioxidants can be vital in amelioration of xenobiotic induced toxicity. To maintain good health, dietary supplements with antioxidant capacity play significant role in attenuating xenobiotics. Studies have shown that nutritional intake of ascorbate [15], glutathione [16], and whole or extract of garlic [17] inhibits oxidative-induced cellular damage.
It is on the foregoing that this study was conceived, to determine the toxicity of non-lethal concentration (as may be found as residuals in food and water) of glyphosate-based pesticide on liver of Wistar albino rats and the protective potential of some nutritional supplements.
2. MATERIALS AND METHODS
2.1. Animals
Forty two matured male Wistar albino rats weighing 160±20 g were housed under standard room conditions of temperature 25 ±3°C and photoperiod of 12 hours light and 12 hours dark. The animals were allowed access to Vital growers mesh and clean water. Acclimation was for a week at the animal house of the Department of Biochemistry, Federal University of Technology, Owerri (FUTO), Nigeria. The Ethics Committee of the Department of Biochemistry, FUTO, Nigeria, approved this study with the reference number FUTO/BCH/EC/2017/24b. The animals used in this study were handled according to the guidelines recommended by the National Institute of Health [18]. The herbicide (Uproot) was supplied by the Ministry of Agriculture and Natural Resources, Imo State, Nigeria.
2.2. Hepatotoxicity Studies
At the end of acclimation, the rats were randomly allocated in seven groups with each group having six rats. Normal control group received food and water only. All the groups (less Normal control) received intraperitoneally 50 mg/kg body weight (bw) of uproot herbicide in distilled water at alternate days for the 28 days allowed for the study Finally, all groups (except Normal and Uproot control) were administered orally, nutritional supplements daily in the following order: Garlic group received 20 mg/kg bw Garlic supplement; GSH group received 20 mg/kg bw Glutathione supplement; Vitamin C group received 20 mg/kg bw vitamin C; Garlic plus Vitamin C (GVC) group received 20 mg/kg bw garlic and 20 mg/kg bw vitamin C; Glutathione plus Vitamin C (GHVC) group received 20 mg/kg bw glutathione and 20 mg/kg bw vitamin C. Animals in each group were allowed access to Vital growers mesh and clean water for the 28 days of the study.
2.3. Sample Collection
At the end of four weeks of exposure and treatment, blood samples were collected from the animals via ocular puncture in plain sample bottles and ethylene diamine tetra-acetate (ETDA) bottle. The blood in plain sample bottles were allowed to clot. Serum was obtained by spinning in a centrifuge. Furthermore, the animals were sacrificed, their liver and kidney excised. A portion of the liver and kidney tissues were reserved for homogenate preparation and a portion washed in saline and preserved in 10% normal saline.
2.4. Liver Tissue Homogenate
Tissue homogenates (5% w/v) were prepared in cold potassium phosphate buffer of pH 7.4. The homogenate was spurned in a cold (4°C) centrifuge to remove cell debris. The supernatant obtained was used to determine oxidative stress parameters: glutathione, total antioxidant capacity, and malondialdehyde (MDA).
2.5. Hepatotoxicity Studies
Serum activity of liver function marker enzymes, such as ALT, ALP, and AST, were assayed using liver enzyme activity assay kits (Bio Merieux France) according to the manufacturer’s instructions. Total bilirubin was determined by Jendrassik and Grof [19] method; total protein by Tietz et al. [20] method, and albumin by Doumas et al. [21] method.
2.6. Oxidative Studies
The concentration of glutathione was determined in tissue homogenates using the method of Raja et al. [22]. Briefly: To remove protein, 1.0 ml sample homogenate was mixed with 4.0 ml of 10% trichloroacetic acid (TCA). This was spurned in a centrifuge for 10 minutes at 3,000 rpm. A pipette was used to deliver 0.01 ml of the supernatant in a test tube holding 2.0 ml of phosphate buffer of pH 8.4, 0.5 ml of 5, 5-dithiobis (2-nitrobenzoic acid), and 0.4 ml doubled distilled water. Finally, the content of the tube was vigorously mixed and absorbance measurement done in spectrophotometer at 412 nm within 15 minutes.
Ferric reducing ability of Plasma [23] was adopted for the determination of total antioxidant activity. Briefly, the following compounds: acetate buffer of pH 3.6, ferric chloride and tripyridyltriazine were constituted as working reagents in 10:1:1 ratio, respectively. With the aid of a pipette, 1.8 ml of this working reagent was delivered to test tubes containing 60 µl of Sample or Standard or Blank. This was thoroughly mixed and allowed to incubate at 37°C for 10 minutes. The coloured (blue) solution that developed was measured in spectrophotometer at 593 nm. The content of blank test tube received similar treatment; however, 60 µl of distilled water was used instead of the plasma. The standard solution is made up of 1,000 µmol/l of ferrous sulphate.
MDA concentration as a product of lipid peroxidation was determined [24]. Four test tubes were prepared and these were added into them; 0.1 ml of sample, 0.9 ml of distilled water, 0.5 ml of 25% TCA, and 0.5 ml of 17% tribarbituric acid (TBA) in 0.3% NaOH. This was allowed to incubate at 95°C for 40 minutes and then allowed to cool. Subsequently, with pipette, 0.1 ml of 20 % sodium dodecyl sulphate was delivered into the tubes and absorbance reading taken at 532 and 600 nm against a blank.
2.7. Histological Studies
Liver samples fixed in formal saline were treated to dehydration, clearing (dealcoholization), and infiltration processes and finally embedded in paraffin [25]. Furthermore, the samples were subjected to serial sectioning at defined thickness and stained using hematoxylin and eosin [26]. With the aid of light microscope set at appropriate magnification (100× and 400×), the tissue sections were examined and photographed.
2.8. Statistical Analysis
The data obtained in this study were subjected to one-way analysis of variance, using computer software statistical package for social sciences, version 18. Significant differences were observed at p ≤ 0.05. The results were expressed as mean and standard deviation.
2.9. Biochemical Results
2.9.1. Liver enzymes
The result of ALP activity presented in Figure 1 shows a significant increase in activity of ALP in uproot control, and supplement treated groups compared to Normal control. However, activity of ALP in uproot control group increased significantly (p < 0.05) when compared to supplement treated groups. The activities of ALT presented in Figure 2 shows significant increase in activities of ALT in uproot control and supplement-treated groups compared to normal control. However, compared to uproot control, all supplement treated groups showed non-significant reduction in ALT activities except Glutathione group which presented significant reduction. Results shown in Figure 3 reveals significant increase in activity of AST in uproot control group compared to normal control group, and supplement-treated groups. The result also showed non-significant difference in activity of AST of normal control compared to all uproot exposed and supplement-treated groups.
2.9.2. Liver proteins and bilirubin
Total protein concentration (Fig. 4) of uproot control showed slight (non-significant) decrease compared to normal control group and supplement-treated groups. Albumin concentration (Fig. 5) presented non-significant variation across the groups with uproot control presenting the lowest concentration. Figure 6 shows significant increases in bilirubin concentration of uproot control and treated groups compared to normal control group. Also, bilirubin reduced significantly in treated groups compared to uproot control group.
2.9.3. Liver oxidative stress parameters
Glutathione concentrations (Fig. 7) increased significantly in supplement treated groups compared to normal control and uproot control groups. Figure 8 shows that MDA concentration increased significantly in uproot control and supplement treated groups compared to normal control group. However, groups administered selected food supplements presented significantly lower MDA concentration when compared to uproot control group. Figure 9 shows significant decrease in total antioxidant capacity of uproot control group when compared to normal control group and supplement-treated groups.
2.10. Histological Studies Results
The results of the histological studies are presented in Figure 10. Normal control (NC) group shows liver tissue with relatively normal central vein (V) and hepatocytes (H); uproot control (UC) group shows liver parenchyma consisting of piecemeal loss of hepatocytes (H) and edematous sites (ED), section shows liver parenchyma with fatty deposits and inflammatory cells. However, nutritional supplements-treated groups showed mild-to-moderate recovery when compared with the uproot control.
 | Figure 1: Activities of ALP in albino Wistar rats exposed to sub-lethal dose of Uproot-herbicide and treated with nutritional supplements. Bars shows mean ± standard deviation of quadruple determinations and those bearing different alphabets indicate significant difference at p < 0.05. [Click here to view] |
 | Figure 2: Activities of ALT in albino Wistar rats exposed to sub-lethal dose of Uproot-herbicide and treated with nutritional supplements. Bars shows mean ± standard deviation of quadruple determinations and those bearing different alphabets indicate significant difference at p < 0.05. [Click here to view] |
 | Figure 3: Activities of AST in albino Wistar rats exposed to sub-lethal dose of Uproot-herbicide and treated with nutritional supplements. Bars shows mean ± standard deviation of quadruple determinations and those bearing different alphabets indicate significant difference at p < 0.05. [Click here to view] |
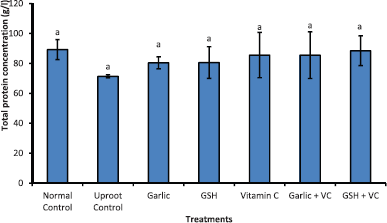 | Figure 4: Concentration of total protein in albino Wistar rats exposed to sub-lethal dose of Uproot-herbicide and treated with nutritional supplements. Bars shows mean ± standard deviation of quadruple determinations and those bearing different alphabets indicate significant difference at p < 0.05. [Click here to view] |
 | Figure 5: Concentrations of albumin in albino Wistar rats exposed to sub-lethal dose of Uproot-herbicide and treated with nutritional supplements. Bars shows mean ± standard deviation of quadruple determinations and those bearing different alphabets indicate significant difference at p < 0.05. [Click here to view] |
 | Figure 6: Concentration of bilirubin in albino Wistar rats exposed to sub-lethal dose of Uproot-herbicide and treated with nutritional supplements. Bars shows mean ± standard deviation of quadruple determinations and those bearing different alphabets indicate significant difference at p < 0.05. [Click here to view] |
.png) | Figure 7: Glutathione concentration in albino Wistar rats exposed to sub-lethal dose of Uproot-herbicide and treated with nutritional supplements. Bars shows mean ± standard deviation of quadruple determinations and those bearing different alphabets indicate significant difference at p < 0.05. [Click here to view] |
3. DISCUSSION
Uproot is a commercially formulated glyphosate mix herbicide and a known terrestrial and aquatic contaminants [27–29]. This makes it very important and urgent to develop a therapy for sub-lethal and lethal exposure to Uproot-herbicide
 | Figure 8: MDA concentration in albino Wistar rats exposed to sub-lethal dose of Uproot-herbicide and treated with nutritional supplements. Bars shows mean ± standard deviation of quadruple determinations and those bearing different alphabets indicate significant difference at p < 0.05. [Click here to view] |
 | Figure 9: Total antioxidant capacity concentration in albino Wistar rats exposed to sub-lethal dose of Uproot-herbicide and treated with nutritional supplements. Bars shows mean ± standard deviation of quadruple determinations and those bearing different alphabets indicate significant difference at p < 0.05. [Click here to view] |
The observed increase in activities of serum AST and ALT indicates hepatotoxic effect of uproot herbicide [30], and this agrees with the earlier observations of the effect of another glyphosate-based herbicide by Cavu?o?lu et al. [31] and dichlorvos pesticide by Celik et al. [32]. The result showed the ameliorative effect of selected food supplement as the activities of liver function enzymes of the supplement treated rats were as recorded in normal control.
The presentations of decreased total protein and albumin concentrations in uproot control rats can be a condition predicated by the hepatocytes loss of biosynthetic functions. This finding is in agreement with reports that showed decrease in total protein of albino rats exposed to glyphosate [33–35]. Fonseca et al. [36] implicated reduced serum total protein concentration to increased rate of amino acid degradation to meet the energy demand following exposure to glyphosate-based pesticide. However, groups administered the nutritional supplements (garlic, glutathione, and vitamin C) following Uproot-herbicide exposure maintained total protein and albumin concentration within the range presented by the normal control rat group. Ajith et al. [37] had reported earlier the ameliorative effect of Vitamin C and other supplements on herbicide-intoxicated rats.
 | Figure 10: Histopathological sections of liver in albino Wistar rats exposed to sub-lethal dose of Uproot-herbicide and treated with nutritional supplements. NC. Normal Control (400×); UC, Uproot control group (100×); GAVC, Garlic +Vitamin C group (100×); GSHVC, GSH+Vitamin C group (100×); GA, Garlic treated rats (100×); GSH, Glutathione group (100×); VC. Vitamin C treated rats (100×). V indicates Central vein; H indicates Hepatic parenchyma; F indicates Fatty materials, IF-inflammatory cells. [Click here to view] |
Also, the significant increase in total bilirubin concentration of uproot control group may be due to hepatobiliary damage caused by the herbicide breakdown [38]. Bilirubin is found in bile, and also in the intestines and reticuloendothelial cells of the spleen [39]. Bilirubin is synthesized in bone marrow and in liver as breakdown product of red blood cell. The increase in bilirubin concentration may also be attributed to increased bilirubin production due to red blood cell breakdown or decreased hepatic uptake of bilirubin due to the reduced level of the transport protein-albumin. However, concomitant administration of dietary supplements significantly restored bilirubin concentration to almost normal level. Similar report showed that certain food supplements could stimulate or enhance amino acid biosynthesis and utilization leading to increased level of albumin actively involved in bilirubin transport and clearance [37].
The reduced glutathione and total antioxidant capacity of the hepatocytes of rats in uproot control group is the outcome of exhaustion of reduced glutathione, overwhelmingly used in the redox activities due to increased generation of free radicals and ROS induced by the herbicide [40]. These free radicals when unchecked can induce oxidative stress [41]. Co-administration of nutritional supplement to uproot exposed groups restored cellular glutathione concentration and total antioxidant capacity of hepatocytes as observed in normal control rats. These supplements contain antioxidants that impede and/or attenuate oxidative damage to important cellular biomolecules [42]. Hepatic glutathione have dual actions in that in one state it is directly involved in scavenging free radicals and/or acts as an indispensable cellular substrate in the activities of glutathione peroxidase and transferase. This interplay of substrates and enzymes is an important phase in the detoxification of peroxides, electrophiles and inhibits the oxidation of thiol groups of peptides and proteins [43,44].
Also, the increased MDA concentration in uproot control indicates lipid peroxidation induced by free radicals generated by the herbicide. However, groups treated with dietary supplements showed reduced hepatic MDA, thus connoting effective antioxidant properties by the supplements. Earlier works showed that food supplements can reduce the levels of MDA [45], and phenolic fractions of garlic demonstrated radical scavenging potentials and attenuation of lipid peroxidation [46].
The uproot herbicide-induced cellular alterations, as shown by loss and subsequent death of hepatocytes and presence of edematous liver cells which may eventually rupture. These outcomes predicates increase in liver function enzymes [5] and loss of antioxidative potentials such as reduced concentration of glutathione and increased lipoperoxidation [31] as recorded in this study. corroborating the outcome of current investigation are the reports of Hazarika et al. [47] and Kavitha and Rao [48], which indicated that lipoperoxidation in glyphosate-based herbicide is evidenced by organophosphorus compounds acting directly with cytoplasmic membranes which results to systemic damage.
However, the systemic damage of hepatocytes membrane structure by Uproot-herbicide was significantly attenuated by the antioxidants of the supplements. Herbicide exposed and nutritional supplements treated groups, recorded mild changes in liver histoarchitecture indicating reduced toxic damage. The result of this study is corroborated by a study that reported the capacity of glyphosate-based herbicide in adverse oxidative changes and prevention of cellular death by co-administration of antioxidants [49].
4. CONCLUSION
The data collected from the present study indicate glyphosate-based pesticide-induced liver dysfunction supported by histopathological lesions. These adverse changes of liver functions could be associated to induction of oxidative stress via ROS generation. However, administration of selected food supplements showed great potentials in reversing the negative consequences of systemic presence of glyphosate-based pesticide.
CONFLICT OF INTEREST
The authors declare that they have no competing interest.
AUTHORS CONTRIBUTION
All the authors worked equally and approved the final manuscript.
FINANCIAL SUPPORT
None.
REFERENCES
1. Yassin MM, Abu Mourad TA, Safi JM. Knowledge, attitude, practice and toxicity symptoms associated with pesticide use among farm workers in the Gaza strip. J. Occup Environ Med 2015;59:387–94. CrossRef
2. Thompson DG. Ecological impacts of major forest-use pesticides. In: Sanchez-Bayo F, van den Brink PJ, Mann RM (eds.). Ecological impact of toxic chemicals, Bentham Science Publishers Ltd., Sharjah, UAE, pp 88–110, 2011. CrossRef
3. Durkin PR. Glyphosate human health and ecological risk assessment. Final report, SERA TR 02_43-09-04a, Syracuse Environmental Research Associates, Inc.: Manlius, NY, p 313, 2011.
4. Naidoo LL, Rother HA. Pesticide safety training and practice in women working in small-scale agriculture in South Africa. J Occup Environ Med 2016;67(12):823–8. CrossRef
5. Benedetti AL, Vituri CL, Trentin AG, Domingues MA, Alvarez-Silva M. The effects of sub-chronic exposure of Wistar rats to the herbicide Glyphosate-Biocarb. Toxicol Lett 2004;153(2):227–32. CrossRef
6. Hasio J. GMOs and pesticides: Helpful or Harmful? Science in the News, 2015. Available via http://situ.hms.havard.edu/flash/2015/gmos-and-pesticides!
7. Woodburn JA. The effect of diuretic, agricultural and industrial effluents on the water quality and Biota of Bindare Stream, Zaria-Nigeria. Unpublished Ph.D Thesis. Ahmadu Bello University Zaria Nigeria, p 256, 2000.
8. Releya HA. Some limnological precautions for fish farmers. Annual report, Kainji Lake Research Institute, New Bussa, Nigeria, pp 54–6, 2005.
9. Dill SM, Shepard KL, Greely MS, Jimenez BD, Ryon MG, Shugart LR, et al. The use of bioindicators for assessing the effects of pollutant stress on fish. Marine Environ Res 2010;28:459–64. CrossRef
10. Pollak DS. Water quality and metal concentration in sediments and fish from Ahmadu Bello University Reservoir Zaria. Ph.D Thesis, Ahmadu Bello University Zaria Nigeria, p 264, 2011.
11. Ayoola AA. Effects of acute and sub-lethal concentrations of Actellic on weight changes and haematology parameters of Clariasgariepinus. World J Biol Res 2008;3:30–9.
12. Larsen KR, Najle A, Lifschitz G, Virkel, Effects of sub-lethal exposure of rats to the herbicide glyphosate in drinking water: glutathione transferase enzyme activities, levels of reduced glutathione and lipid peroxidation in liver, kidneys and small intestine. Environ Toxicol Phar 2012;34:811–8. doi:10.1016/j. etap.2012.09.005
13. Richard S, Moslemi S, Sipahutar H, Benachour N, Seralini GE. Differential effects of glyphosate and Roundup on human placental cells and aromatase. Environ Health Persp 2005;113:716–20. doi:10.1289/ehp.7728
14. Jasper R, Locatelli GO, Pilati C, Locatelli C. Evaluation of biochemical, hematological and oxidative parameters in mice exposed to the herbicide glyphosate-Roundup. Interdiscip Toxicol, 2012;5(3):133–40. CrossRef
15. Institute of Medicine. Vitamin C. Dietary reference intakes for vitamin C, vitamin E, selenium, and carotenoids. The National Academies Press, Washington, D.C, pp 95–185, 2000. ISBN: 0-309-06935-1. Archived from the original on September 2, 2017. Retrieved September 1, 2017.
16. Filomeni GI, Rotilio G, Ciriolo MI. Cell signaling and glutathione redox system. Biochem Pharmacol 2002;64(5–6):1057–64. CrossRef
17. Kweon S, Park KA, Choi H. Chemopreventive effect of garlic powder diet in diethylnitrosamine-induced rat hepatocarcinogenesis. Life Sci 2003;73(19):2515–26. CrossRef
18. National Institute of Health (NIH). Guide for the care and use of laboratory animals. DHEW Publication, Office of Science and Health Reports, Bethesda, MD, 2011.
19. Jendrassik L, Gro PV. Photometrische methoden zur Bestimmung des blutbilirubins. Biochem Z 1938;297:81–9.
20. Tietz NW. Clinical guide to laboratory test. 3rd edition, W.B. Sunders Company, Philadelphia, pp 518–19, 1995.
21. Doumas BT, Watson WA, Biggs HG. Albumin standards’ and the measurement of serum albumin with bromocresol green. Clin Chimica Acta 1971;31:87–97. CrossRef
22. Raja S, Ahmed K, Kumar V, Mukherjee K, Bandyopadhyay A, Mukherjee P. Antioxidant effect of Cytisus scopararius against carbon tetrachloride treated liver injury in rats. J Ehnopharm 2007;109:41–7. CrossRef
23. Benzie IF, Strain JJ. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Anal Biochem 1996;239(1):70–6. CrossRef
24. Wallin B, Rosengren B, Shertzer HG, Cameyo G. Lipoprotein oxidation and measurement of TBARS formation in a single microlitrepeate; its use for evaluation of antioxidants. Annu Rev Med 1993;208:10–5. CrossRef
25. Okoro I. Manual of practical histology, 2nd edition, Peace Publishers, Owerri, 2002.
26. Conn HJ, Darrow MA, Emmels VM. Staining procedures used by the biological stain commission. 2nd edition, The Williams & Wilkins Co, Baltimore, MD, pp 93–4, 1960.
27. Ruiz-Toledo J, Castro R, Rivero-Pérez N, Bello-Mendoza R, Sánchez D. Occurrence of glyphosate in water bodies derived from intensive agriculture in a tropical region of Southern Mexico. Bull Environ Contam Toxicol 2014;93:289–93. doi:10.1007/s00128-014-1328-0
28. Ujowundu CO, Koloseke DM, Uba NO, Achilike JJ, Ogbuagu HD, Ubuoh EA. Ecotoxicological impact of sub-lethal concentrations of glyphosate-based herbicide on Juvenile Clarias gariepinus. J Adv Med Med Res 2017;22(11):1–14. doi:10.9734/JAMMR/2017/34374
29. Székács A, Darvas B. Re-registration Challenges of Glyphosate in the European Union. Front Environ Sci 2018;6:78. doi:10.3389/fenvs.2018.00078
30. Gholami-Seyedkolaei SJ, Mirvaghefi A, Farahmand H, Kosari AA. Effect of a glyphosate-based herbicide in Cyprinus carpio: assessment of acetylcholinesterase activity, hematological responses and serum biochemical parameters. Ecotoxicol Environ Saf 2013;98:135–41. doi:10.1016/j.ecoenv.2013.09.011
31. Cavu?o?lu K, Yapar K, Oruc E, Yalc?n E. Protective effect of Ginkgo biloba L. leaf extract against glyphosate toxicity in Swiss albino mice. J Med Food 2011;14(10):1263–72. CrossRef
32. Celik I, Yilmaz Z, Turkoglu V, Hematotoxic and hepatotoxic effects of dichlorvos at sublethal dosages in rats. Environ Toxicol 2009;24:128–32. CrossRef
33. Borges A, Scotti LV, Siqueira DR, Zanini R, Amaral F, Jurinitz DF. Changes in hematological and serum biochemical values in jundia´ Rhamdiaquelen due to sub-lethal toxicity of cypermethrin. Chemosphere 2007;69:920–6. doi:10.1016/j.chemosphere.05.068
34. O¨ner M, Atli G, Canli M. Changes in serum biochemical parameters of freshwater fish Oreochromis niloticus following prolonged metal (Ag, Cd, Cr, Cu, Zn) exposures. Environ Toxicol Chem 2008;27:360–6. doi:10.1897/07-281R.1
35. F?rat O, Cogun HY, Yüzerero?lu TA, Gök G, F?rat O, Kargin F, Kötemen Y. A comparative study on the effects of a pesticide (cypermethrin) and two metals (copper, lead) to serum biochemistry of Nile tilapia, Oreochromis niloticus. Fish Physiol Biochem 2011;37:657–66. doi:10.1007/s10695-011-9466-3
36. Fonseca MB, Glusczak L, Moraes BS, Menezes CC, Pretto A, Tierno MA, et al. The 2,4-D herbicide effects on acetylcholinesterase activity and metabolic parameters of piava freshwater fish (Leporinus obtusidens). Ecotoxicol Environ Saf 2008;69:416–20. CrossRef
37. Ajith TA, Nivitha, V, Usha, S. Zingiber officinale Roscoe alone and in combination with alpha-tocopherol protects the kidney against cisplatin induced acute renal failure. Food Chem Toxicol 2007;45:921–7. CrossRef
38. Ramaiah SK. A toxicologist guide to the diagnostic interpretation of hepatic biochemical parameters. Food Chem Toxicol 2007;45:1551–7. CrossRef
39. Cullen JM. Mechanistic classification of liver injury. Toxicol Pathol 2005;33:6–8. CrossRef
40. Singh M, Sandhir R, Kiran R. Oxidative stress induced by atrazine in rat erythrocytes: Mitigating effect of vitamin E. Toxicol Mech Methods 2010;20(3):119–26. CrossRef
41. Banudevi S, Arunkumar A, Sharmila M, Senthilkumar J, Balasubramanian K. Diallyl disulfide-induced modulation of a few phase I and II drug metabolizing enzymes on Aroclor 1254 toxicity in Rattusnorvegicus liver and ventral prostate. J Clin Biochem Nutr 2005;36:59–65. CrossRef
42. Halliwell B. Antioxidant in human health and disease. Annu Rev Nut 1996;16:33–8. CrossRef
43. Mulder GJ, Adang AEP, Brussee J, Ketterer B, Meyer D. The glutathione binding site of glutathione S-transferaseisoenzymes from the rat: selectively towards tripeptide analogues of glutathione. Glutathione S-transferase and Drug Resistance Taylor & Francis, UK, p 75, 1990.
44. Masella R, Di Benedetto R, Vari R, Filesi C, Giovannini C. Novel mechanisms of natural antioxidant compounds in biological systems: involvement of glutathione and glutathione - related enzymes. J Nutr Biochem 2005;16(10):577–86. CrossRef
45. Amin A, Hamza AA. Effects of roselle and ginger on cisplatin induced reproductive toxicity in rats. Asian J Androl 2006;8:607–12. CrossRef
46. Siddaraju MN, Dharmesh SM. Inhibition of gastric H(+), K(+)- ATPase and Helicobacter pylori growth by phenolic antioxidants of Zingiberofficinale. Mol Nutr Food Res 2007;51:324–32. CrossRef
47. Hazarika A, Sarkar SN, Hajare S, Kataria M, Malik JK. Influence of malathion pretreatment on the toxicity of anilofos in male rats: a biochemical interaction study. Toxicology 2003; 185:1–8. CrossRef
48. Kavitha P, Rao V. Oxidative stress and locomotor behavior response as biomarkers for assessing recovery status of mosquito fish Gambusia affi nis after lethal effect of an organophosphate pesticide, monocrotophos. Pest Biochem Physiol 2007;87:182–8. CrossRef
49. Gehin A, Guyon C, Nicod L. Glyphosate-induced antioxidant imbalance in HaCaT: the protective effect of Vitamins C and E. Environ Toxicol Pharmacol 2006;22:27–34. CrossRef