ARTICLE HIGHLIGHTS
Amy LBW 5117 is an alkaline endo-α-1-4-amylase that can desize woven cotton.
- At high pH, implying less contamination from neutrophils.
- At low temperatures, implying low-energy costs.
- In the presence of metal ion impurities, implying savings on the purchase of chelators.
1. INTRODUCTION
The genus Bacillus is one of the major sources of industrial enzymes [1]. The application of such enzymes in the detergent, pulp and paper, and textile industries has prompted the isolation of strains from a variety of alkaline environments as a source of stable enzymes with suitable activities [2,3]. Many alkaliphilic Bacillus species have been identified, and among them is Bacillus halodurans, which was first described by Nielsen [4]. Reports available on this species have only focused on polysaccharide-degrading enzymes (e.g., pectinase, amylase, xylanase, etc.) with potential in different industrial processes [5-7]. We have previously reported the isolation of alkaliphilic Bacillus sp. from samples collected around L. Bogoria, a soda lake found in Kenya’s Rift Valley area [8]. Screening of the microorganisms for starch-degrading enzymes revealed that Bacillus halodurans isolates were among the highest producers of amylases [8].
Amylases catalyze the hydrolysis of the 1,4 -O- and 1,6-O- glycosidic bonds in starch or that of its degradation products [9]. Among these enzymes is endo-α-1-4- amylase (or α-amylase) [EC 3.2.1.1], which randomly cleaves multiple internal 1,4-O-glycosidic bonds in the starch to rapidly produce water-soluble low molecular weight linear α-anomeric maltooligosaccharides and α-dextrins [10]. Endo-α-1-4- amylases are produced mainly by Bacillus species, although production by other bacterial (e.g., Chromohalobacter, Halomonas, and Rhodothermus) and fungal (e.g., Aspergillus, Thermomyces, and Penicillium) species has also been reported [11,12]. Their characterization is vital if they are to find applications in various industrial sectors. This is because enzymes possess unique properties that enable them to operate optimally under different conditions [13,14]. For example, different enzymes require different operational (e.g., pH, temperature, metal ion and surfactant requirements, etc.) conditions to optimally hydrolyze substrates into their respective products. Therefore, an understanding of an enzyme’s characteristics is paramount.
Endo-α-1-4-amylases have widespread applications in several industries, e.g., detergent, brewing, baking, paper, and textiles [15]. In the textile industry, an aqueous solution of starch (sizing material) is applied on warp yarns (sizing) to facilitate a fast and secure weaving process, after which it is removed using crude or partially purified endo-α-1-4-amylases (enzymatic desizing) to facilitate further processing of the fabric, e.g., scouring, bleaching, dyeing, printing, and finishing [16]. Endo-α-1-4- amylases offer numerous advantages as desizing agents, for example, they are: (a) safe and easy to handle; (b) efficient in a wide range of temperatures; and (c) degradable. Furthermore, they require the use of fewer chemicals (eco-friendly) and do not cause damage to cloth (leading to the production of high-quality fabrics that are soft, smooth, flexible, and have good strength retention) [17]. However, most of the desizing amylases are costly, high-temperature enzymes that have to be imported from a few European and Asian countries [9]. The situation is even worse in African countries like Kenya, where conventional chemicals that are unfriendly to both the fabrics and the environment are still being used as desizing agents in a time-consuming (12 h) process (personal communication from Hosea Too, Rivatex (EA) Ltd., Eldoret, Kenya). For these reasons, locally produced endo-α-1-4-amylases are in great demand for the production of high-quality fabrics in an eco-friendly environment.
The objective of our study was to determine the physicochemical properties of crude Amy LBW 5117 from a locally isolated alkaliphilic bacterium, B. halodurans [8], and to demonstrate its potential to desize industrially woven cotton.
2. MATERIALS AND METHODS
2.1. Materials
Starch from Irish potatoes (Solanum tuberosum) was purchased from Merck (Darmstadt, Germany), while those from sweet potatoes (Ipomoea batatas), tapioca (Manihot esculenta), rice (Oryza sativa), corn (Zea mays), and wheat (Triticum aestivum) were bought in the form of flour from a local store (Carrefour Supermarket, Ltd., Nairobi, Kenya). Precoated Kieselgel 60 F254 silica gel sheets were purchased from Macherey-Nagel GmbH & Co. KG (Düren, Germany), while industrially woven plain grey cotton (100%), containing 10% (w/v) corn starch as the sizing material, was kindly donated by Rivatex (EA) Ltd., Eldoret, Kenya. Yeast extract, peptone, Tween 20, 3,5-dinitrosalicylic acid, potassium sodium tartrate tetrahydrate, carboxymethyl cellulose sodium salt, avicel, cellobiose, glucose (G1), and maltose (G2) were purchased from Sigma Aldrich (St. Louis, MO, USA).
2.2. Bacterial Isolate and Culture for Amylase Production
Bacillus halodurans LBW 5117 was isolated in our laboratory from a soil sample collected in Lake Bogoria (00o 15’N and 36o 06’ E), a soda lake found in the Kenyan Rift Valley area, and identified as an amylase producer [8]. A stock of this isolate in 40% (v/v) glycerol was used to inoculate solid Horikoshi II medium containing starch as the sole carbon source [pH adjusted to 10.0 using 20% (w/v) Na2CO3] [18]. The plate was incubated at 37°C for 12 h, after which a bacterial colony was obtained from it and used to inoculate 20 ml of Horikoshi II pre-culture medium (without agar). The bacterium was cultured in a thermoshaker incubator (Gallenkamp, London, UK) at 37°C and 100 rpm for 12 h. This pre-culture was then used to inoculate 80 ml of the same medium in a 500 μl conical flask, and the bacterium was cultured for 48 h under the same conditions. The cell culture was centrifuged at 5,000 × g and 4°C for 30 min to obtain the supernatant, which was then treated as a crude enzyme. The appearance and odor of the enzymatic solution were noted down, after which it was stored at −20°C until use.
2.3. Enzyme Assay
Amylase activity was determined by adding 0.1 ml of crude enzyme solution to 0.4 ml of 0.3% (w/v) Irish potato starch in 50 mM glycine-NaOH buffer, pH 10. The reaction mixture was incubated at 60°C for 10 min, and the amount of reducing sugars formed was monitored as described by Wang et al. [19]. The heat-inactivated enzyme (95°C for 30 min) was used as a control. One unit of enzyme activity was defined as the amount of enzyme that liberated one μmol of glucose per min under the established standard assay conditions.
2.4. Characterization of Amy LBW 5117 Activity
The enzyme was characterized to determine its physicochemical properties, e.g., (a) appearance and (b) optimum storage and operational conditions. This was carried out as described below:
2.4.1. Effect of storage temperature on the stability of the enzyme
The stability of the enzyme during storage was studied by storing it in a refrigerator (4°C) and separate incubators (20 and 30°C, respectively) for 6 weeks. The samples were withdrawn every 2 weeks for the determination of residual enzyme activity under the standard assay conditions.
2.4.2. Effect of pH on the activity of the enzyme
The effect of pH on the activity of the enzyme was investigated by carrying out the assays at pH = 3.0, 4.0, 5.0, 6.0, 7.0, 8.0, 9.0, 10.0, 10.5, 11.0, 12.0, and 13.0 at 60°C under the standard assay conditions. Fifty (50) mM buffer systems consisting of NaOAc/CH3COOH (pH = 3.0, 4.0, and 5.0); NaHPO4/NaH2PO4 (pH = 6.0, 7.0, and 8.0); Tris/HCl (pH = 8.0 and 9.0); Glycine/NaOH (pH = 9.0, 10.0, and 10.5); NaHCO3/NaOH (pH = 10.5 and 11.0); Na2HPO4/NaOH (pH = 11.0 and 12.0) and KCl/NaOH (pH = 12.0 and 13.0) were prepared according to Gomori [20] and information obtained from the website “http://delloyd.50megs.com/moreinfo/buffers2.html”, and used in the assays”.
2.4.3. Effect of temperature on the activity of the enzyme
The effect of temperature on the activity of the enzyme was determined by carrying out the assays at 30, 35, 40, 50, 55, 60, 65, 70, and 90°C using 50 mM glycine-NaOH (pH = 10) under the standard assay conditions.
2.4.4. Effect of metal ions on the activity of the enzyme
The effect of metal ions on the activity of the enzyme was studied by incubating the enzyme with KCl, CaCl2, MgSO4, Fe2(SO4)3, CuSO4, NaCl, MnCl2, and ZnSO4 (final concentrations = 0–180 mM, respectively) at room temperature (29 ± 3°C) for 30 min and then determining the residual activities under the standard assay conditions.
2.4.5. Effect of surfactants on the activity of the enzyme
The effect of Tween 20, Triton X-100, and SDS on the activity of the enzyme was studied by incubating the latter with each surfactant (final concentration = 0.05, 0.1, 0.5, 1.0, 1,5, 1.75, and 2.0 mM, respectively) at room temperature (29 ± 3°C) for 30 min and then determining the residual activities under the standard assay conditions.
2.4.6. Effect of optimum temperature of activity of the enzyme on its stability
If the enzyme is to be used to degrade starch optimally, then it must be stable at its optimum temperature of activity. Thus, Amy LBW 5117 was incubated at 60°C in the absence and presence of Ca2+ and Tween 20 (final concentrations = 1.0 and 0.05 mM, respectively), individually and together. The samples were withdrawn after 30 min, 1, 2, and 3 h for the determination of the residual activity of the enzyme under the standard assay conditions.
2.4.7. Effect of different substrates on the activity of the enzyme
This was carried out using both semi-quantitative and quantitative assays. For the semi-quantitative assays, 10 μl of enzyme containing Ca2+ and Tween 20 (final concentrations = 1.0 and 0.05 mM, respectively) was aliquoted in small depressions (0.5 cm in diameter) made on the surfaces of agar media (pH = 10.0) containing 0.3% (w/v) Irish potato-, sweet potato-, tapioca-, rice-, corn-, and wheat- starch products, respectively. The plates were incubated at 60°C for 6 h and then stained with an iodine reagent [21]. The presence of halos around the depressions is indicative of enzymatic hydrolysis of the starch. Similarly, 10 μl of enzyme was aliquoted in depressions made on cellulosic plates containing 1.0% (w/v) of carboxymethyl cellulose sodium salt, avicel (microcrystalline cellulose), and cellobiose, respectively. The plates were also incubated at 60°C for 6 h and then stained with Gram’s iodine dye solution [22]. The presence of halos around the critical micellar concentration (CMC), avicel, and cellobiose plates, if any, is indicative of endoglucanase, exoglucanase, and β-glucosidase activities, respectively. For the controls, a heat-inactivated enzyme (95°C for 30 min) was used.
The effect of the different starch products on the activity of the enzyme was also studied quantitatively in the presence of Ca2+ and Tween 20 (final concentrations = 1.0 and 0.05 mM, respectively). This was carried out as described in the assay method (section 2.3) by using 0.3% (w/v) of the substrates. In addition, the activities of endoglucanase, exoglucanase, and β-glucosidase in the crude enzyme were determined using a modified version of the method described by Kiio et al. [23]: 500 μl of enzyme was added to 500 μl of 1% (w/v) carboxymethyl cellulose sodium salt, avicel, and cellobiose, respectively, prepared in 50 mM glycine-NaoH buffer pH = 10. The mixtures were incubated at 60°C for 1 h, and the amount of reducing sugars formed followed using the method described by Wang et al. [19]. One unit of enzyme activity was defined as the amount of enzyme that liberated one μmol of glucose per min under the established standard assay conditions. The heat-inactivated enzyme (95°C for 30 min) was used as a control.
2.4.8. Mode of action of the enzyme
The mode of action of the enzyme was determined according to Wang et al. [24]. 500 μl (0.5 U) of Amy LBW 5117 was added to an equal volume of 1% (w/v) Irish potato prepared in 50 mM glycine NaOH buffer (pH = 10). The reaction mixture was incubated at 60°C, and samples (150 μl) were withdrawn after 0, 2, 4, 8, and 24 h. The samples were heated (95°C, 5 min) and stored at 4°C until use. A defined amount of the sample (5 μL) was spotted on the bottom of a precoated Kieselgel 60 F254 silica gel aluminum sheet, left to dry, and then developed with a mobile phase of 1-butanol: ethanol: water (5:3:2; v/v/v), with two ascents, in a saturated thin layer chromatography (TLC) chamber at room temperature. The sheet was air-dried, immersed in 15% (v/v) H2SO4 (5 s), air-dried again, and finally developed by heating at 110°C until spots appeared. 0.1% (w/v) of the linear sugars glucose (G1) and maltose (G2) were used as standards.
2.5. Desizing Potential of Amy LBW 5117
A 5 cm × 5 cm piece of plain-woven grey cotton containing corn starch as the sizing material was weighed and subjected to enzymatic desizing using Amy LBW 5117 in a batch process [25]. The fabric was first wetted to approximately 100% in desizing liquor (1.0 mM Ca2+, 0.05 mM Tween 20, and 3.9 U of Amy LBW 5117 in 50 mM glycine NaOH buffer pH = 10) in a thermoshaker incubator (Gallenkamp, London, UK) set at 29 ± 3°C without agitation for 30 min (the material/liquor ratio was maintained at 1:20). Thereafter, the temperature of the incubator was raised to 60°C and 100 rpm for 4 h. The fabric was then removed from the desizing bath and washed in hot water (95°C, 10 min), followed by several cold-water washes. The fabric was then oven-dried (105°C, 1 h), cooled in a desiccator, and re-weighed. As a control, a second piece of woven cotton (5 cm × 5 cm) was weighed, treated similarly but with a heat-inactivated enzyme (95°C for 30 min), and finally re-weighed.
The efficiency of the desizing process was assessed by determining the extent to which the size material was removed from the fabric and then comparing the results with the control fabric. This assessment was carried out as follows:
(a)Surface property determination: The sample was handled or felt freely for softness, firmness, roughness, smoothness and flexibility or stiffness [26].
(b)Weight loss (%) determination: The weight loss (%) that the fabric incurred after the treatment was calculated using the equation below:
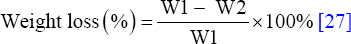 [27]
[27]
Where W1 and W2 are the weights of the fabric before and after treatment, respectively.
(c)Iodine-stain test: This was carried out according to Au and Holme [28]. The fabric was immersed in a 0.005 M iodine solution in a 250 mL beaker. After 1 min, it was removed, washed with cold water, mangled, air dried, and immediately observed for color changes. A deep bluish, purple, or black color indicates the presence of a significant amount of starch, while a brownish or beige color signifies its absence or near absence [29].
(d)TEGEWA rating and residual starch (%) content determination: The color of the stained fabric in (c) above was visually compared with those on the TEGEWA violet scale, and its closest match was identified, along with its numerical value (TEGEWA rating) and corresponding approximate residual starch (%) content [28,29]. The TEGEWA violet scale is shown in Figure 1. It consists of colors ranging from bluish/black/violet to brownish/beige with corresponding numerical values ranging from 1 to 9, where a rating of 1 = no desizing or starch content >1% and a rating of 9 = complete desizing or starch content is approximately 0.04%. In between these two colors are other colors whose shades decrease from dark to light as their ratings and starch contents increase and decrease, respectively.
 | Figure 1: TEGEWA violet scale showing the different colors and their corresponding ratings and starch contents (%). [Click here to view] |
2.6. Data Presentation and Analysis
The data for the characterization of Amy LBW 5117 concerning its catalytic activity and stability are presented in graphical or tabular form and were conducted in duplicate with calculated means. The data for substrate specificity studies of the enzyme are presented in both pictorial and graphical forms, with the latter conducted in duplicate with calculated means. Finally, the data for the enzyme’s mode of action and that for the desized cotton are presented in either tabular or pictorial forms, or both, and were conducted once.
3. RESULTS AND DISCUSSION
The enzyme was characterized to determine its physico-chemical properties, including appearance and optimum storage and operational conditions. This was carried out as described below.
3.1. Physico-chemical Properties of Amy LBW 5117
3.1.1. Appearance
The enzyme Amy LBW 5117, from the culture supernatant of alkaliphilic Bacillus halodurans LBW 5117, was a brown liquid that exhibited a slight fermentation odor. These are some of the typical physical characteristics of crude enzymatic preparations that are used for commercial desizing (http://www.sunsonenzyme.com/Products/Textile/Desizing/), (https://biosolutions.novozymes.com/en/textiles/products/desizing/aquazym). The catalytic activity of the enzyme (0.41 U/ml) is similar to that produced by Bacillus subtilis after utilizing different substrates as carbon sources, e.g., treated wheat straw (0.46 U/ml), maize straw (0.47 U/ml), and rice straw (0.44 U/ml) [30], but much lower than those of known commercial desizing amylases [Table 1]. Thus, if Amy LBW 5117 is to be used as a desizing agent, its catalytic activity will have to be enhanced, e.g., through optimizing the culture conditions that were used to grow the bacterium [31].
Table 1: Comparison of some properties of crude Amy LBW 5117 with those of commercially available desizing amylases.
| Enzyme | Catalytic activity (U/ml) | Storage temp (°C) | pH of activity operational | Optimum | Temp* of activity operational | Optimum |
|---|---|---|---|---|---|---|
| LTAA31º | ≥3000 | 5–25, for 12 months | 5–7 | 6 | 20–90 | 80 |
| HTAA25Lº | ≥25000 | 5–25, for 12 months | 5.5–9.0 | 5.5–8.0 | 80–110 | 95 |
| Coenzyme® LTAA3Pº | ≥3000 | 5–35, for 6–9 months | 6.0–7.0 | 6.0 | 35–70 | 60 |
| Coenzyme® DD990Lº | ≥6000 | 5–25, for 12 months | 5.5–7.5 | 6.5 | 40–120 | 80 |
| HTAA190Lº | ≥190000 | 5–25, for 12 months | 5.5–10.0 | 5.8–8.0 | 50–110 | 95 |
| Aquazyme® Prime 12000Lº | - | - | 5.0–10.0 | 5.0–10.0 | 35–90 | 50–80 |
| Aquazyme® Ultra 1200Nº | - | - | 5.0–7.5 | 5.0–7.5 | 40–95 | 55–70 |
| Amy LBW 5117 | 0.41 | 4–30, for 6 weeks | 7.0–10.5 | 10.0 | 50–63 | 60 |
* Temperature. ºCommercial desizing amylases from Sunsonzymes, Sunson Industry Group Co., Ltd, Suite 2302, Zhong’an Shengye Building, Chaoyang District, Beijing, China (100101) (http://www.sunsonenzyme.com/Products/Textile/Desizing/). ºCommercial desizing amylases from Novozymes A/S, Krogshoejvej 36, 2880 Bagsvaerd, Denmark (https://biosolutions.novozymes.com/en/textiles/products/desizing/aquazym).
3.1.2. Storage stability
Temperature is an important limiting factor for the transportation and storage of enzymes following their production. Industrial enzymes are often produced in bulk liquid form, with each container weighing 25 or 30 kg because emphasis is laid on catalysis rather than purity (https://www.sunsonzymes.com/product/9/97/160/detail). These liquid enzymes are therefore not suitable for storage in frozen form and are instead stabilized to enable them to withstand wide ranges of temperature, e.g., 5–25°C, during their transportation and storage (https://www.sunsonzymes.com/product/9/97/160/detail). This is cost-effective in terms of time and energy consumption because freezing is avoided. For these reasons, Amy LBW 5117 was stored at various temperatures in a liquid state, and its catalytic activity was monitored after every 2 weeks for 6 weeks. As seen in Figure 2, the activity of the enzyme declined gradually, reaching 93, 76, and 70% of the activity after 6 weeks of storage at 4, 20, and 30°C, respectively. This shows that the active site and tertiary structure of the enzyme were not adversely affected by the storage temperatures investigated, implying that savings on the cost of its purchase can potentially be made due to its good stability property in the absence of stabilizers. In the future, it would be interesting to store the enzyme for up to 12 months under these conditions and then determine its catalytic activity, as is the case with most commercial desizing amylases [Table 1].
 | Figure 2: Effect of storage temperature on the activity of the enzyme at pH = 10.0. Enzyme activity corresponding to 100% was 0.41 U/ml. [Click here to view] |
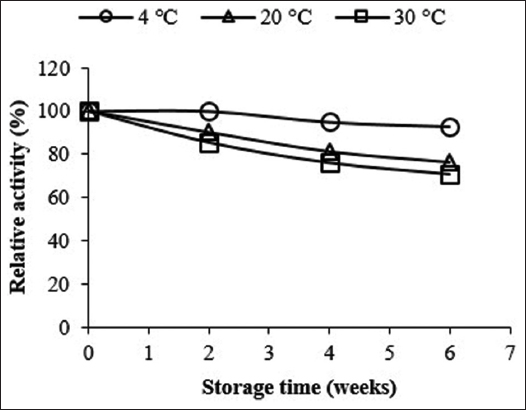
3.1.3. Optimum pH of enzyme activity
Another important limiting factor that affects the activity of enzymes is pH. For this reason, the activity of Amy LBW 5117 was measured using different 50 mM buffer systems at 60°C. As seen in Figure 3, the activity of the enzyme increased rapidly with an increase in pH from 3.0 to 4.0. This was followed by a gradual increase in activity as the pH increased to 10. The activity declined sharply thereafter at higher pH values, reaching 0 at pH 13. The enzyme exhibited an optimum pH of 10 in 50 mM glycine NaOH buffer, with over 80% of its original activity displayed in the pH range of 7.0–10.5 (operational range). Most commercially available desizing amylases are, however, optimally active in the near-neutral pH range [Table 1]. This implies that Amy LBW 5117 has an added advantage over them since it can be used to hydrolyze starch optimally under alkaline conditions and thus help minimize contamination from neutrophilic microorganisms.
 | Figure 3: Effect of pH on the activity of the enzyme at 60°C. The enzyme activity corresponding to 100% was 0.41 U/ml at pH = 10.0. [Click here to view] |
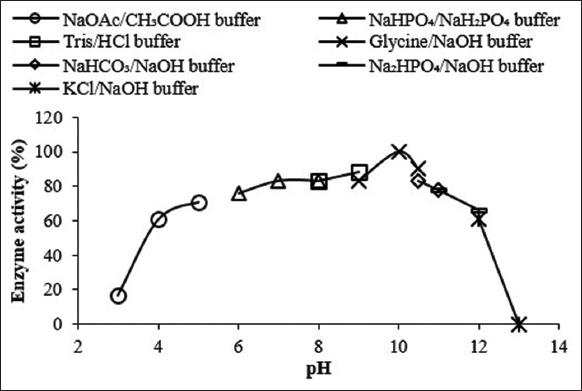
3.1.4. Optimum temperature of enzyme activity
Temperature is yet another important limiting factor that affects the activity of enzymes. The activity of Amy LBW 5117 was thus investigated at different temperatures in 50 mM glycine-NaOH buffer (pH = 10). As shown in Figure 4, the activity of the enzyme increased rapidly with an increase in temperature, reaching optimum at 60°C, and then declining rapidly thereafter to reach 0 at 90°C. The enzyme exhibited optimum temperature at 60°C with over 80% of the activity falling between 50 and 63°C (the operational range). The optimum temperature of activity of the enzyme falls within those listed as low-temperature commercial desizing amylases, e.g., COENZYME® LTAA3P and NOVOZYME AQUAZYME® ULTRA 1200 N [Table 1]. Amylase LBW 5117 can, therefore, also be considered a low-temperature amylase. This comes with numerous advantages, for example, low energy costs and savings on the purchase of specialized equipment that is resistant to heat [9].
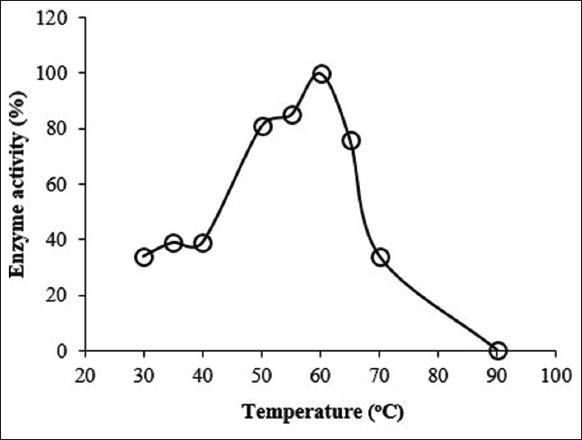 | Figure 4: Effect of temperature on the activity of the enzyme at pH = 10.0. The enzyme activity corresponding to 100% at optimum pH was 0.41 U/ml. [Click here to view] |
3.1.5. Effect of metal ions on the activity of the enzyme
Metal ions play an important role in the functioning of enzymes, with the most notable ones being the alteration of their structures to improve their stability and/or by taking part in their catalytic functions [32]. The type and concentration of the metal ion must, however, be selected carefully, or else the enzyme can become inactivated. In this regard, the effect of different concentrations of metal ions on the activity of Amy LBW 5117 was studied. The activity of the enzyme was: a) either stimulated or insignificantly affected in the presence of up to 10 mM concentrations of most metal ions tested; and b) insignificantly affected by Cu2+ and partially inhibited by Mn2+ at low concentrations [Table 2]. This shows that Amy LBW 5117 requires some metal ions, particularly Ca2+ at 1 mM. Ca2+ ions play the important roles of: (a) linking starch to the active site of amylases to help maintain the enzyme’s catalytic activity; (b) interacting with the negatively charged amino acid residues (glutamic- and aspartic-acid) and resulting in the stabilization and maintenance of the enzyme conformation; and (c) salting out the hydrophobic amino acid residues to force the enzyme to acquire a compact structure that can resist extreme pH and temperature [32]. On the other hand, the complete inhibitory effect on the activity of the enzyme by ≥5 mM Cu2+ is ascribed to the fact that this metal ion binds to His-amino acid residues at the active site of enzymes, and this perturbs the proton shuttling effect in which the amino acid participates during catalysis [33]. The inhibitory effect exerted by ≥1 Mn2+ could be due to its ability to outcompete or displace the more essential metal ions that bind to the enzyme’s metal binding sites, thus leading to a modification of the enzymatic activity [34]. The overall result, however, shows that the activity of Amy LBW 5117 can either be stimulated or may not be significantly affected by the metal ions present in cotton fibers and tap water, both of which the textile industry heavily depends on for desizing woven cotton [35], (Chrome-extension://efaidnbmnnnibpcajpcglclefindmkaj/https://wasreb.go.ke/downloads/Water_Quality_&_Effluent_Monitoring_Guidelines.pdf, 2008). This means that if the enzyme is to be used for such an application: a) a high turnover rate of desized fabrics (implies more profits); and b) savings on the cost of purchasing metal ion chelators can be realized. No information is available on the effect of metal ions on commercially available desizing amylases (http://www.sunsonenzyme.com/Products/Textile/Desizing/), (https://biosolutions.novozymes.com/en/textiles/products/desizing/aquazym).
Table 2: Effect of metal ions on the activity of Amy LBW 5117. The activity of the enzyme is expressed as a percentage of the untreated enzyme (enzyme activity corresponding to 100% activity was 0.41 U/ml).
| Metal ion | Enzyme activity (%) | ||||
|---|---|---|---|---|---|
| 0.5 mM | 5 mM | 5 mM | 10 mM | 180 mM | |
| None | 100 | 100 | 100 | 100 | 100 |
| K+ | ND | 146 | 153 | 147 | 78 |
| Ca2+ | ND | 200 | 176 | 106 | ND |
| Mg2+ | ND | 131 | 145 | 96 | ND |
| Fe3+ | 125 | 159 | 162 | 102 | ND |
| Cu2+ | 99 | 96 | 0 | 0 | ND |
| Na+ | ND | 101 | 153 | 116 | ND |
| Mn2+ | 74 | 0 | 0 | 0 | ND |
| Zn2+ | 103 | 96 | 95 | 96 | ND |
ND: Not determined
3.1.6. Effect of surfactants on the activity of the enzyme
The penetration of enzymes into untreated fabric is often difficult and slow during desizing [17]. Therefore, surfactants are added to desizing baths to accelerate the wetting of the fabric and penetration of the enzyme. The type and concentration of the surfactant must, however, be selected carefully, otherwise the enzyme can become inactivated [9,17,36]. In this context, the effect of different Tween 20, Triton X-100, and SDS surfactants on the activity of Amy LBW 5117 was investigated under optimized pH and temperature. As seen in Figure 5, the activity of the enzyme increased to 115 and 104% in the presence of 0.05 mM Tween 20 and 0.1 mM Triton X-100, respectively, and declined gradually thereafter to just below the 95% activity mark at a 2 mM concentration of both surfactants. Both surfactants are classified as nonionic or mild detergents and do not, therefore, interact strongly with enzyme surfaces when used at concentrations that are lower than their CMCs (the CMC for Tween 20 is 0.06 mM and that for Triton X-100 is 0.24). Instead, they lower the surface tension between aqueous solutions, which in turn facilitates contact frequencies between the active site of the enzyme and their substrates, thus speeding up the reactions [17]. However, when used at concentrations that are higher than their respective CMC values, they form micelles, which interact with enzyme surfaces to form colloidal detergent-protein suspensions, which lower the catalytic function of enzymes by promoting: (a) conformational changes in their active sites; (b) partial unfolding of enzyme; or (c) disruption of substrate binding [9,17,36]. On the other hand, increasing the concentration of SDS resulted in a decrease in the activity of the enzyme from the onset until 0% at 1 mM SDS [Figure 4]. SDS is an anionic surfactant and may therefore have interacted strongly with the enzyme to induce a conformational change in its active site in a concentration-dependent manner that resulted in its inactivation. Similar results were obtained by other researchers [37,38]. The overall result shows that Tween 20, at a concentration of 0.05 mM, was the most compatible wetting agent for use with Amy LBW 5117.
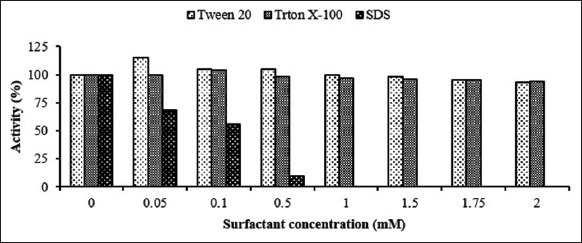 | Figure 5: Effect of different concentrations of various surfactants on the activity of Amy LBW 5117. Enzyme activity corresponding to 100% was 0.41 U/ml in the absence of the additives. [Click here to view] |
3.1.7. Thermostability of the enzyme
In addition to exerting a stimulatory effect on the activity of amylases, Ca2+ ions and Tween 20 have been reported to be good thermostabilizers of the enzyme [39,40]. For this reason, the stability of Amy LBW 5117 was studied with and without these additives (final concentrations of Ca2+ = 1.0 mM and Tween 20 = 0.05 mM) at optimized temperature and pH. As seen in Figure 6, the enzyme exhibited initial activities of 110, 115, and 190% in the presence of 1.0 mM Ca2+, 0.05 mM Tween 20, and both additives together, respectively, and retained 71, 76, and 96% of these activities 3 h later. Furthermore, it retained 70% of its original activity in the presence of both additives upon extending the incubation period to 20 h (data not shown). On the contrary, the control enzyme (no additive added) exhibited an initial activity of 100% but lost all the activity after 3 h [Figure 5]. This result shows that the enzyme is most stable in the presence of both Ca2+ and Tween 20 and can therefore be used to hydrolyze starch repeatedly for as long as it is active. This is cost-effective. Ca2+ ions contribute to the stability of amylases by: (a) interacting with the negatively charged amino acid residues (e.g., glutamic-and aspartic-acid), resulting in the maintenance of the enzyme conformation; and (b) salting out the hydrophobic amino acid residues to force the enzyme to acquire a compact structure that can resist extreme temperatures [32]. On the other hand, Tween 20 may have contributed to the stability of the enzyme by preventing it from dissociating into its respective monomers or inhibiting the removal of essential cofactors (or prosthetic groups) from its active sites [40].
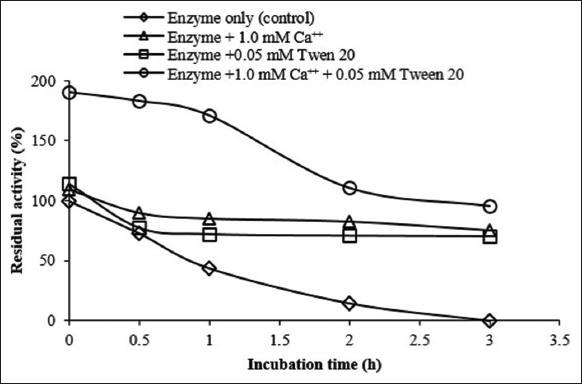 | Figure 6: Effect of 1.0 mM Ca2+ and 0.05 mM Tween 20 on the stability of the enzyme at 60oC and pH = 10. Enzyme activity corresponding to 100% was 0.41 U/ml (Enzyme only [control]). [Click here to view] |
3.1.8. Substrate specificity
The possibility of the enzyme to hydrolyze different starch and cellulosic products was studied semi-quantitatively in the presence of 1.0 mM Ca2+ and 0.05 mM Tween 20 on solid agar media under optimized pH and temperature. After 6 h of incubation and staining the starch plates with Gram’s iodine solution, halos were observed around the depressions on the plates to indicate that the enzyme hydrolyzed all the starch products tested [Figure 7a]. On the contrary, no halos were observed on plates containing CMC, avicel, and cellobiose, respectively, indicating that cellulosic enzymes (endoglucanase, exoglucanase, and β-glucosidase) could be absent in the crude enzymatic preparation [Figure 7a]. To confirm these results, both the starch and cellulosic products were subjected to enzymatic hydrolysis in liquid media, and the enzymatic activities obtained were determined quantitatively. As seen in Figure 7b, the enzyme hydrolyzed all the starch products tested. The highest activity was observed with the medium containing Irish potato starch as substrate, and this was therefore taken to represent 100% activity. Using this as a reference point, the activities of the enzyme in the presence of the remaining starch substrates were calculated. The activities varied from one product to another in the order: Irish potato > sweet potato > tapioca > rice > wheat or corn. This variation can be attributed to the differences in the chemical compositions of the various substrates (e.g., moisture, ash, lipid, protein, amylose, and amylopectin contents), which in turn affect the accessibility of the enzyme and its subsequent conversion to the product once bound [41,42]. On the contrary, no halos were observed on the CMC-, Avicel- and Cellobiose-plates, thus confirming that the crude enzymatic preparation was free from the cellulosic enzymes [Figure 6b].
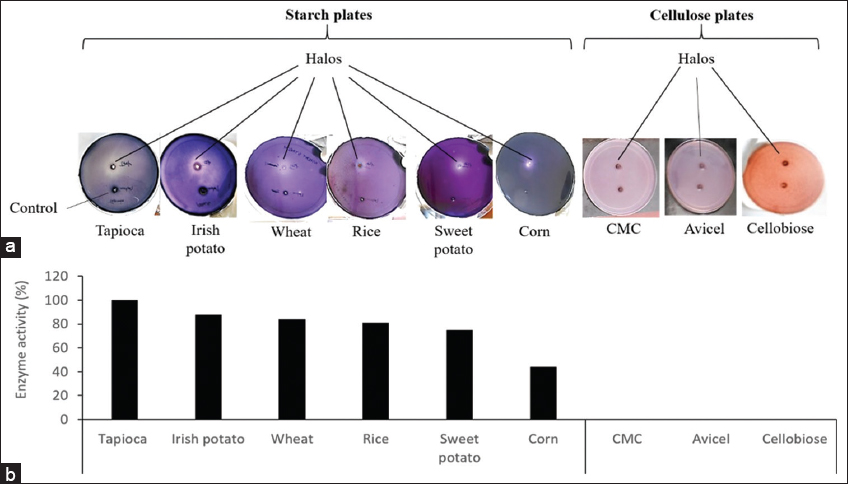 | Figure 7: Effect of different starch- and cellulosic- products on the activity of the enzyme. The activities were determined: (a) Semi-quantitatively, and are seen as halos around the depressions on solid starch plates, and (b) quantitatively, and are expressed as a percentage of that of Irish potato starch (activity corresponding to 100% was 0.41 U/ml). No halo formation around the depressions on cellulosic plates suggests the absence of cellulases in the crude enzymatic preparation. [Click here to view] |
This result confirms that Amy LBW 5117 has the potential to be used as a textile desizing agent, where the degradation and elimination of different types of starch-based sizing agents from woven cotton are required without damaging the cellulosic structures of the cloth [43]. Commercial desizing amylases exhibit a similar property (http://www.sunsonenzyme.com/Products/Textile/Desizing/) and (https://biosolutions.novozymes.com/en/textiles/products/desizing/aquazym).
3.1.9. Enzyme’s mode of hydrolysis
An investigation into the products formed by the action of Amy LBW 5117 on the hydrolysis of starch is shown in Figure 8. A mixture of low and high α-maltooligosaccharides and α-dextrins was formed after 2 h of reaction time, the yields of which increased and decreased, respectively, with time. After 24 h, another set of degradation products, i.e., moderate α-maltooligosaccharides and moderate α-dextrins were formed. In all cases, the main final product was maltose. This profile is typical of endo-α-1-4 amylase, which is a group of starch-degrading enzymes that randomly cleave multiple internal α-(1,4) glycosidic linkages in starch, thereby disintegrating it into water-soluble linear α-maltooligosaccharides of varying lengths, α- dextrins, and a small amount of glucose [44]. Based on this fact, Amy LBW 5117 is an endo-α-1-4 amylase (endo amylase or α-amylase). Commercially available desizing enzymes are also endo-α-1-4 amylases. They are used to degrade starch sizes present on the warp yarns of woven cotton into varying lengths of water-soluble α-maltooligisaccharide and α-dextrins, which can then easily be removed from the fabric through washes (desizing) (http://www.sunsonenzyme.com/Products/Textile/Desizing/) and (https://biosolutions.novozymes.com/en/textiles/products/desizing/aquazym).
 | Figure 8: Thin-layer chromatography analysis of starch hydrolytic products generated by Amy LBW 5117 over a 24 h incubation period. G1 and G2 are the standards of glucose and maltose, respectively. [Click here to view] |
3.2. Assessment of the Amy LBW 5117 Desized Fabric
The extent to which the starch sizing material was removed from the fabric by Amy LBW 5117 was assessed by determining the new properties of the fabric, e.g., (a) surface-handle or “feel,” (b) weight loss (%), (c) stainability with iodine dye solution, (d) TEGEWA rating, and (e) residual starch content (%), and then comparing the results with those from the control fabric. As seen in Table 3, the fabric that was treated with active enzyme acquired new surface-handle or “feel” properties to suggest the absence or near absence of starch compared to the control (treated with denatured enzyme), which exhibited the characteristics of a starched fabric [26]. Furthermore, it incurred a gross weight loss of 9.5% compared to that of the control, which lost only 1.3% [Table 3]. Assuming that the weight loss incurred by the latter was due to the loss of small particles as well as the removal of fringe yarns from its surface and edges, respectively, then the same can be argued for the former. This means that the fabric that was treated with the active enzyme incurred an additional weight loss of 8.2%. This relatively high weight loss was attributed to the removal of a significant amount of the starch from the fabric by Amy LBW 5117, which then went on to acquire the new surface handle properties observed earlier on. Although no reports are available on weight loss measurements incurred by enzymatically desized fabrics at the commercial level, a similar weight loss has previously been reported at the lab-scale level [29].
Table 3: Comparison of some properties of Amy LBW 5117 treated fabric with those of the control fabric.
| Property | Fabric treated with: | |
|---|---|---|
| Active enzyme | Denatured enzyme (control) | |
| Surface handling “feel” | Softer, smoother, and more flexible | Firm, rough and stiff |
| Weight loss (%) | 9.5 (8.2) | 1.3 |
That the enzyme was responsible for the removal of starch from the fabric was confirmed by performing the iodine stain test on the latter. As seen in Figure 9a, the fabric was presented with a light brown color to signify the absence or near absence of starch on it (desized), while the control fabric was presented with a black color to indicate the presence of starch (undesized) [Figure 9b]. This result confirms that Amy LBW 5117 removed the starch from the fabric by degrading it into soluble low molecular weight α-anomeric maltooligosaccharides and dextrins, which were subsequently eliminated from the fabric through several washes. The colors of the two fabrics were then compared with those on the TEGEWA violet scale [Figure 9c]. The color of the desized fabric matched with one that exhibited a high TEGEWA rating of 7-8 on the scale, thus confirming that most of the starch was removed from it and that it now had an estimate of only 0.0725% residual starch. On the other hand, the color of the control fabric matched with one that exhibited a low rating of 1 on the TEGEWA, implying that most of the starch was still intact on the fabric (residual starch > 1.0%). The recommended commercially acceptable amount of residual starch on desized fabrics is 0.125–0.085 (rating of 6–7) [45] This is important because improperly desized fabrics can develop different shades of color when subjected to subsequent processing steps, e.g., dyeing [17]. This result shows that Amy LBW 5117 can degrade and eliminate a significant amount of starch-sizing material from woven cotton. Therefore, it has the potential to be a good fabric-desizing agent.
 | Figure 9: Photograph of the iodine-stained fabrics that were treated with; (a) active enzyme and (b) denatured enzyme (control). The TEGEWA violet scale showing the numerical value (rating) for each shade of color (1–9) and the corresponding approximate residual starch content (%) is shown in (c) [28,29]. [Click here to view] |
4. CONCLUSION
Like commercial desizing amylases, Amy LBW 5117 is an endo-α-1-4-amylase with good storage and operational properties. The enzyme can be produced, stored in liquid form at 4, 20, or 30°C, and used within 6 weeks, during which it is expected to have retained at least 70% of its activity. This can help reduce the cost of its production. Furthermore, the enzyme can be used to hydrolyze starch under alkaline conditions (pH= 7.0–10.5 [optimum 10]) and relatively low temperatures (50–63°C [optimum 60°C]) without being negatively affected by metal ions. This can help reduce contamination from neutrophilic microorganisms, lower the cost of energy consumption, and render the purchase of specialized heat-resistant equipment and metal ion chelators null and void. The implication is reduced operational costs. Moreover, it is thermostable (in the presence of Ca2+ and Tween 20 surfactant) and can hydrolyze different types of starch-based products under optimized pH and temperature. This shows that it has the potential to be used repeatedly to eliminate different types of starch-based sizing agents from woven cotton. A preliminary application study of the enzyme revealed that it could eliminate up to 8.2% corn-starch sizing material from woven cotton under its established optimum operating conditions and yield a fabric with a high TEGEWA rating of 7–8, which corresponds to a residual starch content of only 0.0725%. This shows that the enzyme possesses good operational and storage properties that potentially make it a good desizing agent. This is, however, subject to optimization of its desizing conditions.
5. ACKNOWLEDGMENTS
The authors would like to thank the Department of Biochemistry, University of Nairobi, and Dr. Joseph M. Kamau of the Institute of Primate Research, Nairobi, Kenya, for providing the laboratory spaces used for the study. We also extend our gratitude to Rivatex (EA) Ltd., Eldoret, Kenya, for providing the woven cotton used in the study and to Mr. Faustus Mmbaya of the Department of Biochemistry, University of Nairobi, for his useful suggestions with the TLC experiment.
6. AUTHORS’ CONTRIBUTION
KRO, EKM, YWM, and FJM conceived and designed the experiments. KRO performed all the experiments with assistance from YWM. EKM and FJM supervised the execution of the research. KRO analyzed the data with guidance from EKM and FJM. KRO processed the data and wrote the manuscript. All the authors revised and approved the manuscript.
7. FUNDING
There is no funding to report.
8. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
9. ETHICAL APPROVALS
The study did not involve experiments on animals or human subjects.
10. DATA AVAILABILITY
All generated and analysed data is included in the article.
11. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Danilova I, Sharipova M. The practical potential of Bacilli and their enzymes for industrial production. Front Microbiol 2020;11:1782. [https://doi.org/10.3389/fmicb.2020.01782]
2. Sharma S. Alkalophiles and industrial enzymes. Int Res J Eng Technol 2016;3:1805-8.
3. Preiss L, Hicks DB, Suzuki S, Meier T, Krulwich TA. Alkaliphilic bacteria with impact on industrial applications, concepts of early life forms, and bioenergetics of ATP synthesis. Front Bioeng Biotechnol 2015;3:75. [https://doi.org/10.3389/fbioe.2015.00075]
4. Nielsen P, Fritze D, Priest FG. Phenetic diversity of alkaliphilic Bacillus strains:Proposal for nine new species. Microbiology 1995;141:1745-61. [https://doi.org/10.1099/13500872-141-7-1745]
5. Mei YZ, Chen YR, Zhai R, Liu Y. Cloning, purification and biochemical properties of a thermostable pectinase from Bacillus halodurans M29. J Mol Catal B Enzym 2013;94:77-81. [https://doi.org/10.1016/j.molcatb.2013.05.004]
6. Murakami S, Nishimoto H, Toyama Y, Shimamoto E, Takenaka S, Kaulpiboon J, et al. Purification and characterization of two alkaline, thermotolerant α-amylases from Bacillus halodurans 38C-2-1 and expression of the cloned gene in Escherichia coli. Biosci Biotechnol Biochem 2007;71:2393-401. [https://doi.org/10.1271/bbb.60666]
7. Kumar V, Satyanarayana T. Production of thermo-alkali-stable xylanase by a novel polyextremophilic Bacillus halodurans TSEV1 in cane molasses medium and its applicability in making whole wheat bread. Bioprocess Biosyst Eng 2014;37:1043-53. [https://doi.org/10.1007/s00449-013-1075-3]
8. Oluoch KR, Okanya PW, Hatti-Kaul R, Mattiasson B, Mulaa F. Protease-, pectinase-and amylase-producing bacteria from a Kenyan Soda Lake. Open Biotechnol J 2018;12:33-45. [https://doi.org/10.2174/1874070701812010033]
9. Yahya S, Sohail M, Khan SA. Characterization, thermal stabilization and desizing potential of amylase from A. tubingensis SY 1. J Text Inst 2021;113:993-1000. [https://doi.org/10.1080/00405000.2021.1914393]
10. Paul JS, Gupta N, Beliya E, Tiwari S, Jadhav SK. Aspects and recent trends in microbial α-amylase:A review. Appl Biochem Biotechnol 2021;193:2649-98. [https://doi.org/10.1007/s12010-021-03546-4]
11. Hussain I, Siddique F, Mahmood M, Ahmed SI. A review of the microbiological aspect of α-amylase production. Int J Agric Biol 2013;15:1029-34.
12. Sundarram A, Murthy TP. α-amylase production and applications:A review. Appl Environ Microbiol 2014;2:166-75.
13. Martins RF, Davids W, Al-Soud WA, Levander F, Rådström P, Hatti-Kaul R. Starch-hydrolyzing bacteria from Ethiopian Soda Lakes. Extremophiles 2001;5:135-44. [https://doi.org/10.1007/s007920100183]
14. Martin MF, Okpo EA, Andy IE. Microbial amylases:A review. World News Nat Sci 2019;22:174-9.
15. Putri AZ, Nakagawa T. Microbial α-amylases in the industrial extremozymes. Rev Agric Sci 2020;8:158-69. [https://doi.org/10.7831/ras.8.0_158]
16. Sultana S, Haque MZ, Nur HP. Preparation and application of different size materials on the cotton yarn and investigating the effect of sizing on the tensile properties of cotton yarn. Bangladesh J Sci Ind Res 2014;49:25-30. [https://doi.org/10.3329/bjsir.v49i1.18850]
17. Agrawal BJ. Desizing of cotton fabrics with enzymes for improved performance. Int J Ind Biotechnol Biomater 2016;2:11-6.
18. Horikoshi K. Alkaliphiles:Some applications of their products for biotechnology. Microbiol Mol Biol Rev 1999;63:735-50. [https://doi.org/10.1128/MMBR.63.4.735-750.1999]
19. Wang G, Michailides TJ, Bostock RM. Improved detection of polygalacturonase activity due to Mucor piriformis with a modified dinitrosalicylic acid reagent. Phytopathology 1997;87:161-3. [https://doi.org/10.1094/PHYTO.1997.87.2.161]
20. Gomori G. Preparation of Buffers for use in Enzyme Studies. Netherlands:Elsevier;1955. 138-46. [https://doi.org/10.1016/0076-6879(55)01020-3]
21. Yassin SN, Jiru TM, Indracanti M. Screening and characterization of thermostable amylase-producing bacteria isolated from soil samples of Afdera, Afar region, and molecular detection of amylase-coding gene. Int J Microbiol 2021;2021:5592885. [https://doi.org/10.1155/2021/5592885]
22. Sinza EM, Mwakilili AD, Mpinda CB, Lyantagaye SL. Cellulase-producing bacteria isolated from Mufindi paper mill industrial effluent, Iringa Tanzania. Tanz J Sci 2021;47:204-13.
23. Kiio IK, Jackim MF, Munyali WB, Muge EK. Isolation and characterization of a thermostable cellulase from Bacillus licheniformis strain vic isolated from geothermal Wells in the Kenyan Rift Valley. Open Biotechnol J 2016;10:198-207. [https://doi.org/10.2174/1874070701610010198]
24. Wang G, Luo M, Lin J, Lin Y, Yan R, Streit WJ, et al. Anew extremely halophilic, calcium-independent and surfactant-resistant alpha-amylase from Alkalibacterium sp. SL3. J Microbiol Biotechnol 2019;29:765-75. [https://doi.org/10.4014/jmb.1901.01038]
25. Chinnammal KV, Arunkumar SK. Production and application of amylase enzyme for bio-desizing. J Environ Nanotechnol 2013;2:6-12. [https://doi.org/10.13074/jent.2013.06.132006]
26. Grineviciute D, Gutauskas M. The comparison of methods for the evaluation of woven fabric hand. Mater Sci 2004;10:97-100.
27. Aly AS, Sayed SM, Zahran MK. One-step process for enzymatic desizing and bioscouring of cotton fabrics. J Nat Fibers 2010;7:71-92. [https://doi.org/10.1080/15440478.2010.481086]
28. Au CK, Holme I. The alkali desizing of woven cotton fabrics. Res J Text Appar 1999;3:16-30. [https://doi.org/10.1108/RJTA-03-01-1999-B003]
29. Halim AF, Zhou W. Study on the comparison of the effects of pH buffer in single stage preparatory process with that of conventional for CVC woven fabric. J Text Sci Eng 2018;8:1000345.
30. El Mageed BM, Ali SG, Hammouda O, Raslan M. Optimization of α-amylase enzyme activity produced by Bacillus subtilis and Aspergillus niger. Egypt J Chem 2022;66:421-6.
31. Pandey A, Nigam P, Soccol CR, Soccol VT, Singh D, Mohan R. Advances in microbial amylases. Biotechnol Appl Biochem 2000;31:135-52. [https://doi.org/10.1042/BA19990073]
32. Sudha. Effect of different concentrations of metal ions on alpha-amylase production by Bacillus amyloliquefaciens. Res Biotechnol 2012;3:67-71.
33. Chang CT, Lo HF, Chi MC, Yao CY, Hsu WH, Lin LL. Identification of essential histidine residues in a recombinant Þ-amylase of thermophilic and alkaliphilic Bacillus sp. strain TS-23. Extremophiles 2003;7:505-9. [https://doi.org/10.1007/s00792-003-0341-8]
34. Lévêque E, Janecek Š, Haye B, Belarbi A. Thermophilic archaeal amylolytic enzymes. Enzyme Microb Technol 2000;26:3-14. [https://doi.org/10.1016/S0141-0229(99)00142-8]
35. Brushwood DE, Perkins HH. Determining the metal content of cotton. Text Chem Color 1994;26:32-5.
36. Sethi BK, Nanda PK, Sahoo S, Sena S. Characterization of purified α-amylase produced by Aspergillus terreus NCFT 4269.10 using pearl millet as substrate. Cogent Food Agric 2016;2:1158902. [https://doi.org/10.1080/23311932.2016.1158902]
37. Hou H, He H, Wang Y. Effects of SDS on the activity and conformation of protein tyrosine phosphatase from Thermus thermophilus HB27. Sci Rep 2020;10:3195. [https://doi.org/10.1038/s41598-020-60263-4]
38. Ikemiya M. The Effect of Synthetic Detergents on Malt Amylase. Bulletin of the Institute for Chemical Research. Vol. 35. Kyoto University;1957. 81-8. Available from:https://repository.kulib.kyoto-u.ac.jp/dspace/bitstream/2433/75587/1/chd035_3-4_081.pdf
39. Deb P, Talukdar SA, Mohsina K, Sarker PK, Sayem SA. Production and partial characterization of extracellular amylase enzyme from Bacillus amyloliquefaciens P-001. Springerplus 2013;2:154. [https://doi.org/10.1186/2193-1801-2-154]
40. Komori Y, Chiang KT, Fukuto JM. The effect of nonionic detergents on the activity and/or stability of rat brain nitric oxide synthase. Arch Biochem Biophys 1993;307:311-5. [https://doi.org/10.1006/abbi.1993.1594]
41. Adeleye OO, Ologhobo AD, Iji P. Prediction of starch hydrolysis in native starches studied in vitro. Starch Starke 2013;65:1-6. [https://doi.org/10.1002/star.201300129]
42. Soto LG, Garcia JL, Gonzalez LM, Nicanor VJ, Cruz AB. Influence of starch source in the required hydrolysis time for the production of maltodextrins with different dextrose equivalent. Afr J Biotechnol 2012;11:13428-35. [https://doi.org/10.5897/AJB12.2257]
43. Saha P, Khan MF, Patra S. Truncated α-amylase:An improved candidate for textile processing. Prep Biochem Biotechnol 2018;48:635-45. [https://doi.org/10.1080/10826068.2018.1479863]
44. Savaner S, Sohani S. Review on microbial α-amylase, types and their industrial application. Kala Sarovar 2020;23:105-12.
45. Harane RS, Adivarekar RV. Sustainable processes for pre-treatment of cotton fabric. Text Cloth Sustain 2017;2:2. [https://doi.org/10.1186/s40689-016-0012-7]