1. INTRODUCTION
A protein is a macromolecule consisting of a variety of amino acids and linked by peptide bonds [1]. Proteins can be hydrolyzed to produce protein hydrolysate through a hydrolytic degradation process with acids, bases, or proteolytic enzymes [2]. Protein hydrolysate can be derived from different sources such as insects, particularly black soldier fly (Hermetia illucens L.) larvae (BSFL) that have a high protein content of (40–50%), followed by a fat content of about 29–32% [3-6]. The highest protein content is observed in the early development of larvae particularly on days 4–6, then decreases until the adult larval stage [7]. Hence, the larvae phase may be valorized to produce protein hydrolysate [5].
The chemical and enzymatic processes are the most used method to hydrolyze proteins and produce protein hydrolysates. Most enzymes usually used for hydrolysis are derived from animals, plants, and microbes. Examples of enzymes widely used for hydrolysis are pancreatin, pepsin, papain, bromelain, and alcalase. Protein hydrolysis using alcalase enzymes derived from microbes occurs in the fermentation process [8]. One of the studies investigated that protein hydrolysis with the alcalase enzyme produced the highest antioxidant activity with a 2,2-diphenyl-1-picrylhydrazyl (DPPH) value of more than 3 mmol/mg protein and that there is no drastic difference between enzymes such as alcalase and papain on functional properties [9].
An optimum concentration of papain enzyme (6%) to synthesize protein hydrolysate from BSF prepupae has been determined with a yield of 31% [6]. In another study, the utilization of bromelain enzyme produced up to 10.7% protein hydrolysate from BSFL with an antioxidant activity to inhibit DPPH free radical up to 72.6% with an IC50 of 0.84% [5]. However, systematic studies on pre-treatment of the protein source before the protein hydrolysis of BSFL are indeed very rare. In this study, the protein was first extracted from the BSFL using acid hydrochloric acid and sodium hydroxide before the hydrolysis reaction to increase the degree of hydrolysis. This study aimed to explore the optimum conditions of hydrolysis of BSFL using bromelain enzyme in terms of degree of hydrolysis and antioxidant activity of the protein hydrolysate.
2. MATERIALS AND METHODS
2.1. Materials
Dried BSFL were obtained from a local community in Bandung, Indonesia, whereas bromelain enzyme was obtained in collaboration with PT Bromelain Enzim, Indonesia, as the primary provider of an enzyme with an enzyme activity of 200 CDU/mg. Aquades, NaOH, HCl, and methanol were purchased from Sakura Medical Bandung, Indonesia; formaldehyde and Coomassie blue from Nitra Kimia; and DPPH from Sigma-Aldrich (Singapore).
2.2. Extraction of Protein from Black Soldier Fly Larvae
BSFL were crushed and blend with cold aquades with a ratio of weight/volume (w/v) of 1:10 before being extracted with acid and alkaline solvent. For protein extraction with acids, 0.25 M HCl solution was mixed into the solution until it reached a pH of 2–3. For alkaline protein extraction, 0.25 M NaOH solution was mixed into the solution until a pH of 10–11 was reached. The solutions were incubated in a shaker at room temperature (25°C) and 50°C for 30 and 60 min. The solution was subsequently centrifuged at a speed of 4000 rpm for 15 min. The precipitate and supernatant were separated using filter paper and funnel glass. The supernatant obtained from the filtration process was then adjusted to pH 7 with 0.25 M HCl or 0.25 M NaOH solution [10].
2.3. Hydrolysis of Protein Solution Derived from Black Soldier Fly Larvae
Hydrolysis of the extracted protein solution was performed according to the procedures by Firmansyah and Abduh [5]. The extracted protein solution was dissolved in a phosphate buffer solution (pH 8) at a ratio of 1:10 (weight to volume, w/v). The solution was mixed with different concentrations of bromelain enzyme (enzyme activity of 200 CDU/mg) in 250 mL Erlenmeyer flasks and incubated using a water bath shaker at 150 rpm with various time and temperature. The enzyme in the solution was deactivated using water bath at 90°C for 30 min to stop the hydrolysis reaction. The solution was then separated at 4000 rpm for 15 min at 25°C with centrifuge. The supernatant was then filtered and stored at 4°C in a refrigerator for further analysis.
2.4. Experimental Design and Optimization
The hydrolysis of extracted protein solution was performed using design expert with a face-centered central composite design (CCD) with the factors and level for the experimental design is shown in Table 1. The data were simulated using a second-order polynomial model as coefficients, as shown in Equation (1).
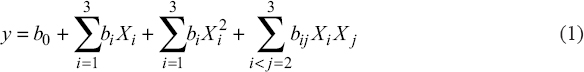 |
Where, y is degree of hydrolysis (%), b0 is a constant value, bi is coefficient of each variable, X1 is enzyme concentration (%), X2 is temperature (°C), and X3 is hydrolysis time (h). The regression equation was obtained by backward elimination of non-significant. The greatest conditions for the hydrolysis of extracted protein solution derived from BSFL were determined using the numerical optimization function available in Design-Expert 12.
Table 1: Factors and level for the hydrolysis of extracted protein solution using a face-cantered central composite design.
| Factors | Level | ||
|---|---|---|---|
| −1 | 0 | 1 | |
| Enzyme concentration, E (%) | 1 | 3 | 5 |
| pH | 7 | 8 | 9 |
| time, t (h) | 4 | 14 | 24 |
2.5. Amino Acid Analysis
The amino acid in the samples was determined at the Saraswanti Indo Genetech, Bogor, Indonesia, using a high-performance liquid chromatography (HPLC) instrument. The column used was Thermo Scientific ODS-2 Hypersil which was conditioned with a mobile phase of 1 mL/min. OPA reagents react with primary amino acids under alkaline conditions to form compounds with fluorescence which can be detected by a fluorescence detector.
2.6. Proximate Analysis
The composition of BSFL was determined by a proximate analysis at the Saraswanti Indo Genetech, Bogor, Indonesia. The protein content was determined using a standard Kjeldahl method (18-8-31/MU/SMM - SIG) whereas the total fat was determined using a Weibull method (18-8- 5/MU/SMM - SIG). The total ash, fiber, and water content were determined using a standard gravimetry method [6].
2.7. Degree of Hydrolysis Analysis
The degree of hydrolysis of the protein hydrolyzed solution was determined by calculating the ratio of α-amylase to the total nitrogen as shown in Equation (2).
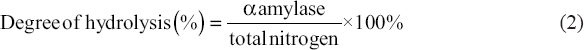 |
Total nitrogen was performed using the Kjeldahl method, whereas the total α-amylase was performed by the Sorensen method. Approximately 0.5 mL of hydrolysis solution was diluted to 10 mL of aquades. The pH of the solution was adjusted to pH 8 with a 0.25 M NaOH solution. An approximately 1 mL of formaldehyde liquid was added to the solution. The solution was then titrated using 0.02 M NaOH solution until it reached pH 8.
2.8. Antioxidant Activity Analysis
The antioxidant activity of the protein hydrolysate was analyzed from the scavenging ability of DPPH as indicated by the concentration of IC50. The solution was mixed with aquades to a concentration of 0.25–1.25%. After adding the DPPH solution, the solution was incubated in a dark room for 30 min. The absorbance (A) of the solution was then quantified using a spectrophotometer at a wavelength of 517 nm [11]. The total value of free radicals can be determined using Equation (3).
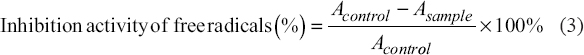 |
2.9. Statistical Analysis
The results were analyzed statistically using Minitab® 16 to determine the significance (P < 0.05) of temperature and incubation time on the protein recovery.
3. RESULTS AND DISCUSSION
3.1. Composition of Black Soldier Fly Larvae
The BSFL used in this study contains 48.90% lipid, 32.11% protein, 6.73% ash, 6.30% fiber, and 5.96% carbohydrate on a weight basis as determined by the proximate analysis. The protein substance within the BSFL is generally lower than the values that have been reported in the literature [12]. The BSFL used in this study were obtained from a nearby community that valorized coconut residue that may contribute to the relatively low amount of protein, but a higher lipid content as determined in this study. Based on nutrient analysis, coconut residue has a lower protein content of 1.69–2.19% compared to fat content of 12.58–17.26% [13,14]. Based on these data, the protein content resulted in this study will be lower due to the low amount of protein content compared to the fat content in the coconut residue.
The amino acid composition of the BSFL investigated in this study is shown in Table 2. The results show that glutamic acid was the most dominant amino acid (3.7%) and leucine was the most prevalent for essential amino acid (2.1%) which lies at the bottom edge of the values reported in the previous studies [5,15-17], as shown in Table 2. Both leucine and glutamic acid as the foremost abundant amino acid in the present study also presented by Zulkifli et al. [18] that reached 3.60% and 6.05% dry mass. Glutamate is the most abundant non-essential amino acid compared to other amino acids. Glutamic acid plays a crucial part in the stability of protein structure, nutrition, metabolism, and signaling. The abundance of glutamic acid is due to its chemical structure, which is more stable and negatively charged, thus stabilizing of the protein structure [19]. On the other hand, leucine is the foremost abundant essential amino acid that increases energy metabolism to synthesize protein, while inhibiting protein degradation [20]. A higher content of glutamic acid as opposed to other amino acids was observed when BSFL fed with vegetable and plant-based food residues were hydrolyzed with a bromelain enzyme [5].
Table 2: Amino acid composition (%) of black soldier fly (Hermetia illucens L.) larvae.
| Amino acid | This study | Literature [5, 15-17] |
|---|---|---|
| Serine | 1.5 | 1.6–4.5 |
| Glutamic acid | 3.7 | 4.0–18.1 |
| Phenylalanine | 1.2 | 1.5–6.2 |
| Isoleucine | 1.3 | 1.6–5.3 |
| Valine | 2.0 | 2.3–7.3 |
| Alanine | 1.9 | 2.5–12.1 |
| Arginine | 1.4 | 1.9–6.2 |
| Glycine | 2.0 | 1.9–6.1 |
| Lysine | 1.9 | 2.2–8.0 |
| Aspartic acid | 2.3 | 3.4–10.3 |
| Leucine | 2.1 | 2.6–7.7 |
| Tyrosine | 1.5 | 2.3–6.0 |
| Proline | 1.8 | 2.2–6.2 |
| Threonine | 1.5 | 1.4–5.0 |
| Histidine | 0.9 | 1.3–4.8 |
3.2. Extraction of Protein from Black Soldier Fly Larvae
The protein extraction was carried out using acid and alkaline treatment at different incubation times (30 and 60 min) and temperatures (25°C and 50°C), and the outcome is shown in Figure 1. The highest protein recovery of 38.42% was generated from the alkaline extraction at 50°C for 30 min, but this value was not significantly different from the alkaline extraction at 25°C for 60 min. In this study, alkaline extraction with various factors such as temperature and time was not significantly different (P < 0.05) because the temperature limit and extraction time were still within suitable range to produce optimum protein. According to the study by Mintah et al [21], there was a significant difference between extraction time and protein yield. At 30-50 min, protein yield significantly increased and over 60 min, there will be a decrease or increase and then decreased in longer extraction time. This is because most of the protein was extracted and dissolved at the initial 50 min [22].
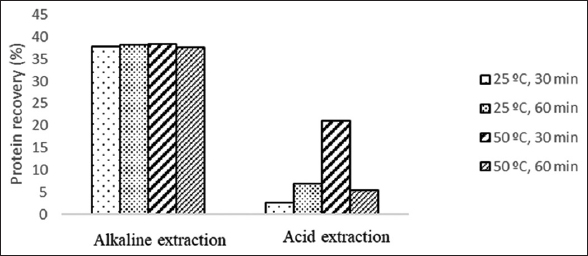 | Figure 1: Protein recovery for alkaline and acid extraction of black soldier fly (Hermetia illucens L.) larvae. [Click here to view] |
The effect of temperature on the protein extraction process also greatly influences the protein yield. Based on previous research by Choi et al. [23], an increase in high temperature will result in increasing protein denaturation. The temperature up to 60°C on protein extraction will produce more protein yield, while above that temperature will get lower yields due to denaturation. Hence, the temperature of 50°C is considered as the most suitable temperature for the extraction temperature [21,24,25]. In this study, the protein recovery with acid and base for each temperature and incubation time has been determined as significantly different (P < 0.05).
From Figure 1, it can be observed that the extraction using alkali resulted in higher protein recovery than using acid. This is because extraction above pH 10–11 increases the protein hydrophobicity that affects the increase of dissolved protein [26]. At the isoelectric pH, protein aggregate myofibrils increase due to hydrophobic interactions [26,27]. However, there is a possibility that the protein conformation will open, and that the protein will denature. Extraction of protein with a strong alkaline up to pH 11 is not recommended for food due to potential synthesis of unsafe compound for consumption during the hydrolysis reaction [28].
The values of protein recovery obtained in this study are lower than those reported that reaching up to 96% [29]. In that study, the BSFL were first defatted followed by protein extraction with NaOH and protein recovery using trichloroacetic acid solution in acetone. In this study, the protein was directly extracted using acid and alkaline without removal of the fat. Nevertheless, the values of protein recovery in this work are slightly higher than the protein recovery of 28% for protein extraction from Azolla microphylla using NaOH [30]. Alkaline solution to sample ratio of 20:1 on protein extraction from Hermetia illucens resulted in a sharp increase of protein yield up to 60% [21].
3.3. Effects of Enzyme Concentration, pH, and Time on the Degree of Hydrolysis of Protein Hydrolysate
The results of the degree of hydrolysis are revealed in Table 3, while the coefficients of the regression model equation are presented in Table 4. The highest degree of hydrolysis (58.52%) was obtained at 3% concentration of enzyme, an incubation time of 24 h, and pH 8. The degree of hydrolysis obtained in this study is slightly higher than the degree of hydrolysis (47.4%) reported by Firmansyah and Abduh [5]. The differences in the degree of hydrolysis may be contributed by the pre-treatment step introduced in this study where the protein was first extracted with alkaline (NaOH) at an elevated temperature (50°C) to facilitate the hydrolysis of the extracted protein solution.
Table 3: Degree of hydrolysis and antioxidant activity of protein hydrolysate from black soldier fly (Hermetia illucens L.) larvae at different concentration of enzyme, temperature, and hydrolysis time.
| Running | Reaction condition | Degree of hydrolysis (%) | Antioxidant activity, IC50 (v/v) | ||||
|---|---|---|---|---|---|---|---|
| Enzyme (%) | Time (h) | pH | Experiment | Model | Experiment | Model | |
| 1 | 3 | 14 | 8 | 32.12 | 34.66 | 5.23 | 5.43 |
| 2 | 4 | 4 | 7 | 20.47 | 18.67 | 8.06 | 7.81 |
| 3 | 3 | 14 | 8 | 32.12 | 34.66 | 5.23 | 5.43 |
| 4 | 3 | 14 | 8 | 32.12 | 34.66 | 5.23 | 5.43 |
| 5 | 2 | 24 | 9 | 34.82 | 37.95 | 3.28 | 5.41 |
| 6 | 4 | 14 | 8 | 40.71 | 36.75 | 2.77 | 4.62 |
| 7 | 3 | 14 | 8 | 32.12 | 34.66 | 5.23 | 5.43 |
| 8 | 2 | 14 | 8 | 38.94 | 34.09 | 2.69 | 4.44 |
| 9 | 3 | 4 | 8 | 25.18 | 27.95 | 3.34 | 3.93 |
| 10 | 3 | 14 | 8 | 32.12 | 34.66 | 5.23 | 5.43 |
| 11 | 3 | 14 | 9 | 28.24 | 25.14 | 4.18 | 5.82 |
| 12 | 4 | 24 | 9 | 39.41 | 39.74 | 1.17 | 3.57 |
| 13 | 2 | 4 | 9 | 22.35 | 19.99 | 4.07 | 3.78 |
| 14 | 3 | 14 | 8 | 32.12 | 34.66 | 5.23 | 5.43 |
| 15 | 3 | 24 | 8 | 58.52 | 49.36 | 1.15 | 4.93 |
| 16 | 4 | 24 | 7 | 38.82 | 43.52 | 3.27 | 5.62 |
| 17 | 3 | 14 | 7 | 27.06 | 24.61 | 5.81 | 7.77 |
| 18 | 2 | 24 | 7 | 41.4 | 40.15 | 4.85 | 6.96 |
| 19 | 4 | 4 | 9 | 21.53 | 21.93 | 3.18 | 2.90 |
| 20 | 2 | 4 | 7 | 15.29 | 15.14 | 3.34 | 3.07 |
Table 4: Regression model coefficient for predicting of the degree of hydrolysis and antioxidant activity of protein hydrolysate from black soldier fly (Hermetia illucens L.) larvae.
| Variable | Coefficient | |
|---|---|---|
| Degree of hydrolysis | Antioxidant activity | |
| Constant | −627.1 | 46.6 |
| X1 | - | 20.0 |
| X2 | 1.37 | 2.06 |
| X3 | 160.4 | −17.9 |
| X1 X2 | −0.04 | −0.60 |
| X1 X3 | −0.40 | −1.66 |
| X2 X3 | - | −0.18 |
| X12 | 0.77 | −0.90 |
| X22 | 0.04 | −0.01 |
| X32 | −9.78 | 1.36 |
| X1 X2X3 | - | 0.06 |
X1: Enzyme concentration (%), X2: Hydrolysis time (h), and X3: pH
The results obtained in this study resemble the optimum degree of hydrolysis of 58% [31], for the hydrolysis of Anadara granosa that was obtained using a bromelain concentration of 2.5%. Another study reported an optimum degree of hydrolysis of 19.83% at an enzyme concentration of 3% and pH 8 for the hydrolysis of monkfish [32]. However, a higher concentration of bromelain extract (11%) was reported by Bahri et al. [33], to produce protein hydrolysate with a degree of hydrolysis of 22.8% in 120 min for tofu dregs hydrolysis.
The response surface diagrams that show the interaction between enzyme concentration, pH, and hydrolysis time toward the degree of hydrolysis are presented in Figure 2. From the figure, it can be observed that the degree of hydrolysis is highly influenced by the hydrolysis time. The degree of hydrolysis increases as the hydrolysis time increases from 4 to 24 h. The degree of hydrolysis was also positively influenced by the pH with an optimum value observed at pH 8. Enzyme concentration of 2–4% did not influence the degree of hydrolysis. Hence, the result obtained in this study suggests that a bromelain concentration of 2% is sufficient to increase the degree of hydrolysis.
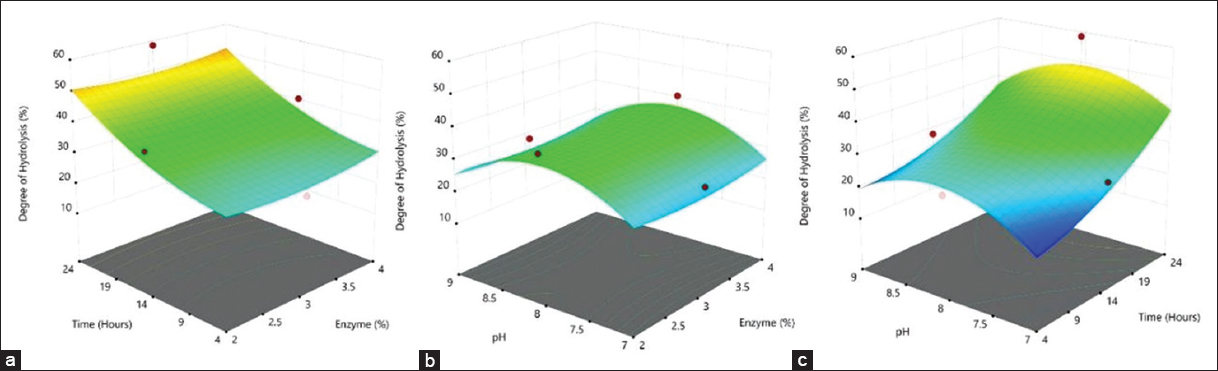 | Figure 2: Response surface diagram showing the interaction of (a) enzyme concentration and time; (b) pH and enzyme concentration; and (c) pH and time on the degree of hydrolysis. [Click here to view] |
3.4. Effects of Enzyme Concentration, pH, and Time on the Antioxidant Activity of Protein Hydrolysate
The antioxidant activity was determined using the DPPH method. The results showed that the optimum conditions for the antioxidant activity also occurred at an enzyme concentration of 3%, pH 8, and hydrolysis time of 24 h to inhibit free radicals 2,2-diphenyl-2-picrylhydrazyl with IC50 of 1.15%, as shown in Table 3, while the coefficients of the regression model equation are shown in Table 4. The result resembles the previous finding with an IC50 value of 0.84% for protein hydrolysate from black soldier fly prepupae [5]; 1.22% for protein hydrolysate from defatted hemp [34]; and 2.3% for protein hydrolysate from oyster [35].
The response surface diagrams that show the interaction between enzyme concentration, pH, and hydrolysis time toward the antioxidant activity are presented in Figure 3. As shown in the figure, the antioxidant activity is affected by the enzyme concentration, hydrolysis time, and pH. The antioxidant activity tends to increase as the enzyme concentration decreases while the hydrolysis time increases. The figure also suggests that there is an optimum pH for the antioxidant activity of protein hydrolysate from BSFL which is indicated by lower values of the IC50. The capability of the protein hydrolysate to prevent DPPH free radical is affected by the amino acid composition. The BSFL sample used in this study contains aromatic amino acids, particularly phenylalanine (11.9%) and tyrosine (14.5%) that can stabilize DPPH free radicals by donating a proton to electron-deficient free radicals [36] that lead to the discontinuation of free radical reaction [37].
 | Figure 3: Response surface diagram showing the interaction of (a) time and enzyme concentration; (b) pH and enzyme concentration; and (c) pH and time on the antioxidant activity. [Click here to view] |
4. CONCLUSION
The BSFL used in this study contain 32.11% protein. The protein contains essential amino acids particularly leucine (2.1%) as well as aromatic amino acids; phenylalanine (1.2%); and tyrosine (1.5%). A maximum protein recovery of 38.42% was obtained after the BSFL were extracted with NaOH at 50°C for 30 min. The extracted protein solution had been hydrolyzed using a bromelain enzyme with the degree of hydrolysis lies in the range of 15.29–58.52%. At the optimum conditions (3% enzyme, pH 8, and 24 h), the degree of hydrolysis was 58.52% with an IC50 of 1.15%. Thus, BSFL have the high potential to be developed as an alternative source of protein to produce a bioactive hydrolysate. Nevertheless, further studies may be carried out to further increase the protein recovery and degree of hydrolysis of the protein hydrolysate.
5. ACKNOWLEDGMENT
The authors would like to thank PT Bromelain Enzyme for providing enzyme material during this research.
6. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
7. FUNDING
Research grant from Institut Teknologi Bandung (Grant number: LPPM.PN-6-32-2022).
8. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
9. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
10. DATA AVAILABILITY
Additional data is available upon request.
11. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Hou Y, Wu Z, Dai Z, Wang G, Wu G. Protein hydrolysates in animal nutrition:Industrial production, bioactive peptides, and functional significance. J Anim Sci Biotechnol 2017;8:24. [CrossRef]
2. Hashim H, Maskat M, Wan mustapha W, Mamot S. Process development for the production of protein hydrolysate from cockle (
3. Wardhana AH. Black soldier fly (
4. Bosch G, Zhang S, Oonincx DG, Hendriks WH. Protein quality of insects as potential ingredients for dog and cat foods. J Nutr Sci 2014;3:e29. [CrossRef]
5. Firmansyah M, Abduh MY. Production of protein hydrolysate containing antioxidant activity from
6. Triani R, Alfianny R, Manurung R, Abduh MY. Synthesis of protein hydrolysate from the prepupae of
7. Liu X, Chen X, Wang H, Yang Q, Rehman K, Li W,
8. Tapal A, Tiku PK. Nutritional and nutraceutical improvement by enzymatic modification of food proteins. Enzymes Food Biotechnol 2019;471-81
9. Batish I, Brits D, Valencia P, Miyai C, Rafeeq S, Xu Y,
10. Zhao X, Vázquez-Gutiérrez JL, Johansson DP, Landberg R, Langton M. Yellow mealworm protein for food purposes-extraction and functional properties. PLoS One 2016;11:e0147791. [CrossRef]
11. Gangwar M, Gautam MK, Sharma AK, Tripathi YB, Goel RK, Nath G. Antioxidant capacity and radical scavenging effect of polyphenol rich
12. Abduh MY, Nadia MH, Manurung R, Putra RE, Syaripudin. Factors affecting the bioconversion of Philippine tung seed by black soldier fly larvae for the production of protein and oil-rich biomass. J Asia Pac Entomol 2018;21:836-42. [CrossRef]
13. Nor NN, Abbasiliasi S, Marikkar MN, Ariff A, Amid M, Lamasudin DU,
14. Hanafi FN, Kamaruding NA, Shaharuddin S. Influence of coconut residue dietary fiber on physicochemical, probiotic (
15. Tschirner M, Simon A. Influence of different growing substrates and processing on the nutrient composition of black soldier fly larvae destined for animal feed. J Insects Food Feed 2015;4:249-59. [CrossRef]
16. Müller A, Wolf D, Gutzeit HO. The black soldier fly
17. Kawasaki K, Hashimoto Y, Hori A, Kawasaki T, Hirayasu H, Iwase S,
18. Zulkifli NF, Seok-Kian AY, Seng LL, Mustafa S, Kim YS, Shapawi R. Nutritional value of black soldier fly (
19. Brosnan JT, Brosnan ME. Glutamate:A truly functional amino acid. Amino Acids 2013;45:413-8. [CrossRef]
20. Duan Y, Li F, Li Y, Tang Y, Kong X, Feng Z,
21. Mintah B, He R, Agyekum A, Dabbour M, Golly MK, Ma H. Edible insect protein for food applications:Extraction, composition, and functional properties. J Food Process Eng 2020;43:e13362. [CrossRef]
22. Kain R, Chen Z, Sonda T, Abu-Kpawoh J. Study on the effect of control variables on the extraction of peanut protein isolates from peanut meal (
23. Choi I, Choi SJ, Chun JK, Moon TW. Extraction yield of soluble protein and microstructure of soybean affected by microwave heating. J Food Processing Preserv 2006;30:407-19. [CrossRef]
24. Durling NE, Catchpole OJ, Grey JB, Webby RF, Mitchell KA, Foo LY,
25. Spigno G, De Faveri DM. Antioxidants from grape stalks and marc:Influence of extraction procedure on yield, purity and antioxidant power of the extracts. J Food Eng 2007;78:793-801. [CrossRef]
26. Omana D, Moayedi V, Xu Y, Betti M. Alkali-aided protein extraction from chicken dark meat:Textural properties and color characteristics of recovered proteins. Poult Sci 2010;89:1056-64. [CrossRef]
27. Undeland I, Kelleher S, Hultin H. Recovery of functional proteins from herring (
28. Betti M, Fletcher DL. The influence of extraction and precipitation pH on the dry matter yield of broiler dark meat. Poult Sci 2005;84:1303-7. [CrossRef]
29. Caligiani A, Marseglia A, Leni G, Baldassarre S, Maistrello L, Dossena A,
30. Abduh MY, Ono JM, Khairani M, Manurung R. The influence of light intensity on the protein content of
31. Hashim H, Maskat M, Wan mustapha W, Mamot S. The effects of enzyme concentration, temperature and incubation time on nitrogen content and degree of hydrolysis of protein precipitate from cockle (
32. Chi C, Cao Z, Wang B, Hu X, Li Z, Zhang B. Antioxidant and functional properties of collagen hydrolysates from Spanish mackerel skin as influenced by average molecular weight. Molecules 2014;19:11211-30. [CrossRef]
33. Bahri S, Hadati K, Satrimafitrah P. Production of protein hydrolysate from tofu dregs using the crude extract of bromelain from pineapple core (
34. Tang F, Barbacioru C, Wang Y, Nordman E, Lee C, Xu N,
35. Umayaparvathi S, Saravanan M, Vinayagam V, Muthuvel A, Gunasekaran S, Thangavel B. Antioxidant activity and anticancer effect of bioactive peptide from enzymatic hydrolysate of oyster (
36. Dávalos A, Miguel M, BartoloméB, López-Fandiño R. Antioxidant activity of peptides derived from egg white proteins by enzymatic hydrolysis. J Food Prot 2004;67:1939-44. [CrossRef]
37. Auwal SM, Zarei M, Hamid AA, Saari N. Response surface optimisation for the production of antioxidant hydrolysates from stone fish protein using bromelain. Evid Based Complement Altern Med 2017;2017:4765463. [CrossRef]