1. INTRODUCTION
Oxidative reactions are the most important factor in oils and fats deterioration which cause some unwanted changes in the taste, color, smell, and texture of lipid products and are due to the free radicals produced during the process. These free radicals can cause Alzheimer’s, stroke, diabetes, rheumatoid arthritis, poliomyelitis, cataracts, asthma, and heart failure [1]. Oxidation in oils is initiated by decomposition of fatty acids with multiple double bonds and production of free radicals which leads to a decrease in the amount of natural antioxidants, nutritional value, and sensory characteristics of oil [2]. In various industries, synthetic antioxidants are used to prevent lipid oxidation in food products and emulsions. However, due to the potential risks, the use of these antioxidants has been restricted or prohibited in some food products. Hence, the desire to use natural antioxidants has increased. Proteins are considered as one of these natural antioxidant sources. Therefore, in recent years, researchers’ studies have been conducted toward identifying bioactive peptides obtained from plant and animal sources. These peptides are specific protein sections produced by three methods of chemical synthesis, microbial fermentation, and enzymatic hydrolysis, containing 2–20 amino acids and their molecular mass is <6000 Dalton [3]. Bioactive peptides, according to their amino acid sequence and type, have a positive effect on the body’s function and condition, and thereby on the health of the individual, which include immunization, tranquilization, antimicrobial, antioxidant, anti-blood pressure, and anticancer effects [4]. The antioxidant activity of bioactive peptides can be attributed to the ability to free radical scavenging, to lipid peroxidation prevention, and to metallic ions chelating [5,6].
Among plant and animal sources suitable for producing hydrolyzed proteins, a lot of attention has been paid to the plant sources due to their lower prices and allergenicity. Cannabis sativa L. is considered as an important agricultural plant and a valuable source of nutrients, fiber, and medicines. Cannabis seeds are valuable sources of fiber, oil (about 30%), and protein (25%). Cannabis oil is also rich in polyunsaturated fatty acids. Furthermore, it is regarded as a rich source of high-quality protein and essential amino acids. Cannabis proteins are mainly composed of globulin and albumin [7].
Considering the evaluation of nanoliposomes loaded with antioxidant peptides obtained by enzymatic hydrolysis, Mohan et al. evaluated microencapsulation of anionic and cationic whey peptides and the effect of peptides load on the properties of the resulting nanovesicles [8]. Furthermore, Ramezanzade et al. investigated the microencapsulation effect of bioactive peptides derived from rainbow trout skin enzymatic hydrolysis on the structure of biopolymer-coated liposomes and they stated that the coated liposomes showed greater stability in releasing the peptide compounds in vivo [9]. Li et al. used the chitosan-coated liposomes to investigate the microencapsulation of hydrolyzed bioactive proteins in Salmon [10]. The results showed that coating with chitosan significantly improved the stability of nanoliposomes. Zavareze et al. used fluorescein enzyme to examine the hydrolysis and production of antioxidant peptides derived from the White croaker muscle and byproduct [11].
For as much as enzymes exclusive activity under mild reaction conditions is considered as one of their important features, the present study aimed at evaluating the possibility of protein extraction and the use of the pulp that obtained by cannabis oil-pressing with respect to the advantages and ever-increasing applications of producing and using bioactive peptides derived from enzymatic hydrolysis. Furthermore, regarding the challenges of using these peptides, such as instability and bitterness, the microencapsulation of these compounds will be accomplished using lipid nanocarriers such as nanoliposomes; and then the physical properties, encapsulation performance, stability, chemical structure, and morphology of nanoparticles will be evaluated.
2. MATERIALS AND METHODS
2.1. The Raw Materials Procurement
Cannabis seeds were purchased from the local market. The used chemicals were also provided by Merck, a German Company. Phospholipid (β-acetyl-γ-O-alkyl-L-α-phosphatidylcholine) obtained from lecithin of the cow was prepared as a lipophilic powder with a purity degree of more than 99% named Lipoid from Sigma Co (Germany).
2.2. The Raw Materials Preparation
2.2.1. Protein extraction
To decrease, the resulting dry pulp was mixed with hexane at a ratio of 1:4 (w/v), and it was stirred at room temperature for 3 h. Then, hexane was separated using Büchner funnel, and the resulting flour was dried at room temperature, and it went through the sieve mesh 40. The operation of extracting protein from degreased powder was such that the powder was mixed with a 0.33 molar NaCl solvent (pH=9.25) with a ratio of 1:10 and it was stirred for 2 h; afterward, the resulting solvent will be centrifuged at ×4500 g for 30 min. In the next stage, the supernatant pH will be adjusted at pH=4.5 (isoelectric protein pH0). After that, to precipitate the proteins, the resulting solvent was centrifuged at ×4500 g for 20 min. The protein was then washed twice with distilled water and was centrifuged at ×4500 g for 5 min, and it would again turn into a solution state by setting pH=7.2 and adding 1molar NaOH. Afterward, the resulting protein isolate was dried by freeze drier and was kept at 4°?. All processes were carried out at room temperature [12].
2.2.2. Enzymatic hydrolysis of protein
In the enzymatic hydrolysis process, the protein extracted from the greased pulp of cannabis at 5% concentration (w/v) was solved into 0.2 m phosphate buffer (pH=8) and its complete hydration during continued stirring for 30 min was made possible. Then, the 2% Alcalase enzyme was added into a solvent containing cannabis protein at an enzyme to protein substrate ratio (w/w). The reaction temperature was 50°? and the reaction time was considered to be variable during continuous stirring with a 200 rpm for 60–300 min. When the hydrolysis process completed, to deactivate the reaction and the enzyme activity, the reaction medium was placed in a water bath at 90°? for 15 min. Afterward, the solvent was cooled down to ambient temperature. The solvent was centrifuged at 5000 rpm for 10 min. The supernatant was separated, lyophilized, and kept at 20°? until it would be used Sarabandi et al. [13].
2.2.3. Production of nanoliposomes containing hydrolyzed cannabis
Liposome was produced using thin-film hydration method and based on the method described by Li et al. with some changes [10]. The liposome production process involves dissolving 0.09 and 0.01, respectively, soybean and cholesterol in 10 ml of absolute ethanol. The process of evaporation and thin-film formation at the end of the round-bottom flask was carried out using a rotary evaporator at 60 rpm at 60°?. The round-bottom flask was kept at ambient temperature in a desiccator for at least 12–18 h to ensure complete removal of the solvent. Then, the dried lipid film using phosphate-buffered saline (pH 7.4) containing 5mg/ml peptide was re-hydrated using vortex for 1 min. The resulting solvent was stirred for 2 min at 60°? by using a rotary evaporator, and it was cooled down for 2 min at ambient temperature. The stirring process was repeated 3 times. Finally, the ultrasound probe (70% power and 0.5 cycles) was applied alternatively on the solvent for 10 steps, and each step lasted 1 min. To prevent temperature rise, ultrasonic process was performed under storage conditions of liposomes in cold water. Ultimately, the nanoliposome that was produced at a concentration of 10 mg/ml lipid (lecithin and cholesterol) was kept in the fridge until the tests were performed.
2.2.4. Determination of particle size, polydispersity index, and zeta potential
Particles size (as Z-average) and their polydispersity index with photon correlation spectroscopy were measured using a Zetasizer (Nanosizer 3000, Malvern Instruments, UK) at 90°? and 25°? inside the device-specific cell to a width of 0.01 m. Furthermore, the electrical charge of nanoliposomes (as zeta potential), special electrical conductivity and the electrophoresis movement were measured using this device.
2.2.5. Microencapsulation efficiency
The study on the microencapsulation efficiency of the peptides derived from cannabis enzymatic hydrolysis was conducted by Li et al. method with some modifications [10]. Moreover, the solvent that was passed through the filter was used to determine the amount of unloaded or free proteins by the Bradford method [14]. Finally, the hydrolyzed encapsulation efficiency cannabis was calculated using the following equation.
2.2.6. The effect of storage in ambient temperature and fridge on the stability and release of nanoliposomes
The amount of change in particle size and the microencapsulation efficiency at refrigerated temperature (4°?) as a stability index of nanocarriers was evaluated with dynamic light scattering (DLS) and Bradford method, respectively. To evaluate the release and change in the microencapsulation efficiency, the amount of peptide extracted from the nanocarriers was also calculated after separating the nanoliposomes from the continuous phase (buffer) using ultrafiltration (ammonia filter) through Bradford method, and it ultimately was measured relative to the total amount of peptide used in the formulation and the change in the encapsulation efficiency was reported as a result of releasing the loaded compounds [10].
2.2.7. Infrared spectroscopy Fourier-transform infrared (FTIR)
Infrared spectroscopy was performed by Shimadzu FTIR-8400, according to Li et al. [10].
2.2.8. Surface structure evaluation
The surface properties of nanoliposomes were evaluated using a scanning electron microscope (SEM) (Hitachi PS-230, Japan). Eventually, each coated sample was transferred into the microscope, and the observation of particles’ surface structure was carried out.
2.3. Statistical Analysis
All tests were repeated 3 times. The average and standard deviation of the data were calculated. The effect of each treatment and variable was evaluated using one-way ANOVA and SPSS 19 for identifying the effective factors statistically. Then, the average comparison was done using Duncan’s multiple range test to examine the significance of variables affect (P < 0.05).
3. RESULTS AND DISCUSSION
In the present study, the production of antioxidant peptides was carried out using enzymatic hydrolysis of cannabis protein with Alcalase. Finally, these peptides were loaded into nanoliposomes, and the encapsulation effect of each peptide on the physical properties, encapsulation efficiency, and system stability during storage in different conditions was evaluated. Furthermore, the chemical structure and morphology of empty and loaded liposomal nanoparticles were evaluated.
3.1. Determination of Particle Size, Polydispersity Index, and Zeta Potential
Figure 1 shows the average particle size of empty nanoliposomes and those loaded with hydrolyzed cannabis protein. The average particle size for an empty nanoliposome was obtained 79 nm; but after loading with peptides, the value of this index increased and it varied between 95–101 nm. This change can be attributed to the placement of peptides within the structure of nanoliposomes and thus to the increase in the size of nanoparticles. Moreover, the liposomes loaded with peptides derived from enzymatic hydrolysis during a longer period had a smaller particle size. The reason for this finding may be attributed to the easier placement of lower molecular weight peptides in the nanoliposomal structure and the improvement of the monolayer vesicle’s arrangement compared to the larger peptides with higher molecular weight [13,15]. The particle size in a colloidal nanocarrier system plays an important role in determining its characteristics. The large size of particles leads to colloidal instability and higher gravitational dissociation, lower bioavailability, greater turbidity, and lower solubility of the active encapsulated compound in the nanocarrier structure.
Regarding the effects of the liposomal membrane compound on the particle size, in the liposomal membranes that are rich in lecithin, the arrangement of the acyl chain in a similar direction reduces the spaces created by the bulky polar groups in the lipid head, and consequently, it increases the contact and interactions between the chains. Cholesterol maintains the acyl chain that tilts toward a similar direction in a regular and direct manner, and it fills the spaces created between them [16].
Depending on the type of system, polydispersity index varies from 0 to 1 in determining the distribution and uniformity of the particle size and its value. The higher values of this index indicate the distribution of wide particle size and the existence of coarse and uneven particles and masses in the system [17]. In this research, the value of this index in empty and loaded nanoliposomes varied from 0.234 to 0.326 [Figure 2].
This slight amount indicates the uniformity and homogeneity of the particle size in the distribution system (Da Rosa Zavareze, 2014). Although the polydispersity is higher and approximate to 1, it shows the distribution of wide particle size as a result of the accumulation and deposition of nanovesicles in the system [10].
 | Figure 1: The average particle size of empty nanoliposomes and those loaded with hydrolyzed cannabis protein [Click here to view] |
 | Figure 2: Polydispersity index of empty nanoliposomes and those loaded with hydrolyzed cannabis protein [Click here to view] |
As shown in Figure 3, the amount of zeta potential was different in negative values and varied from −10mV to −15mV, depending on the degree of its loaded peptide hydrolysis. In this study, the lowest zeta potential amount was related to the sample loaded with the peptide obtained from hydrolysis for 300 min (−15.23 mV). This amount of surface loading in nanoliposomes is due to the presence of phosphatidylcholine and the position of its fatty acid and the ionization of phosphate and carboxylic groups. Furthermore, the difference in the amount of this index can be attributed to the release of more peptides with a lower hydrolysis degree than in the structure and their placement on the surface of the liposomal membrane, and consequently a greater change in the zeta potential [18].
Zeta potential determination is a common method for specifying the electrostatic properties of liposomes, which represents the total loading of a particle in a fluid medium and it is a useful indicator of particles surface loading and colloidal repellency among them, and the physical stability of nanocarrier systems. Therefore, it is the best indicator for determining the surface electric state of colloidal solvents. High zeta potential amounts (positive or negative) increase the stability of liposomal dispersion by increasing the repulsive responses and decreasing the liposomal particles sticking [19]. Various studies have evaluated the effect of different compounds on designing and producing nanoliposomes, loading various bioactive compounds and coating nanocarriers with polymers. For instance, the microencapsulation of peptides derived from the collagen in bream scales fish was done using liposomal nanocarriers (using relatively pure phosphatidylcholine). Liposomes particle size was determined to be 66–214 nm with an average diameter of 90 nm and a polydispersity of 0.25 (which indicates the low polydispersity index in the liposome) [20]. da Rosa Zavareze et al. hydrolyzed and produced antioxidant peptides derived from the muscle and byproducts of White croaker using fluorescein enzyme [18]. The resulting antioxidant peptides were microencapsulated in phosphatidylcholine as a capsule wall material in the liposome structure. The particle size in the control sample (unhydrolyzed) and the sample containing muscle hydrolyzate and its byproducts were 208, 266, and 263 nm, respectively, indicating an increase in particle size due to active substance loading in the liposome structure. Furthermore, the polydispersity index for these nanocarriers was obtained in the range of 0.228, 0.298, and 0.197, respectively.
3.2. Encapsulation Efficiency
Encapsulation efficiency is considered as one of the major efficiency indicators of nanocarriers structure on maintaining the active compounds. The amount of this index was varied in the nanoliposomes loaded between 81 and 90%, depending on the peptide type [Figure 4].
The results indicate a decrease in the amount of encapsulation efficiency in nanoliposomes loaded with peptides that are derived from the higher degrees and duration of hydrolysis. This finding suggests the effect of hydrolysis degree, as a result, faster release of peptides with a molecular weight lower than the vascular structure, and ultimately the decreased amount of this index [9].
The factors affecting the encapsulation efficiency of liposomes include the nature of the active agent (its lipophilicity or hydrolocity), the nature of phospholipid (in terms of the type and arrangement of fatty acids), the ratio of phospholipid to the active substance, nanoliposome production method, the concentration, and type of stabilizer, such as cholesterol and environmental conditions such as temperature, pH, and ionic strength. The amounts of encapsulation efficiency obtained from this study were more than those reported by Were et al. [21], who studied on the encapsulation of nisin in the nanoliposomal structure by thin-film hydration and reported an encapsulation efficiency of about 54%. da Rosa Zavareze et al. conducted a study on peptides encapsulation of antioxidants produced by hydrolysis of muscle and the byproducts of White croaker. In their research, the encapsulation efficiency was about 80% [18]. According to Ramezanzade et al., the encapsulation efficiency in nanoliposomes containing peptides of antioxidants derived from rainbow trout skin enzymatic hydrolysis (0.2 mg/ml concentration), which coated with 0.4% (W/V) chitosan, was obtained at the highest level (80.2%) compared to the other treatments [9].
 | Figure 3: Zeta potential of empty nanoliposomes and those loaded with hydrolyzed cannabis protein [Click here to view] |
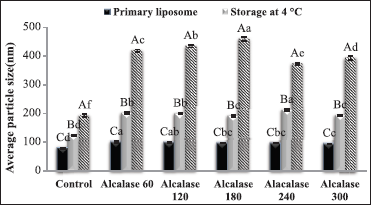 | Figure 4: Average particle size of primary nanoliposomes and nanoliposomes after storage at different temperatures [Click here to view] |
 | Figure 5: Encapsulation efficiency of empty nanoliposomes and those loaded with hydrolyzed cannabis protein [Click here to view] |
3.3. The Effect of Storage at Ambient and Fridge Temperature on the Particle Size
Particle size and particle size distribution (uniform or non-uniform) of colloid systems are of particular importance in determining their properties. The persistence of these two parameters over a long period of time reflects the stability of that system [22]. Figure 5 shows the effect of the type of loaded peptide and the storage condition on the physical stability and particle size of nanoliposomes. As shown in Figure 5, the slightest changes in the particle size are due to the empty nanoliposomes. These results indicate the effect of active agent and its degradation reaction in the system, which leads to instability of the loaded liposomal structure and massive changes in particle size, especially at high temperature [10]. Furthermore, the storage temperature significantly changed the particle size. These results indicated that most changes were in samples stored at ambient temperature. For example, in nanoliposomes loaded with the peptide obtained from enzymatic hydrolysis for 180 min, the average particle size increased from 97 nm, respectively, to 191 and 460 nm after 6 weeks of storage at the fridge and ambient temperatures. Increasing the particle size and the formation of large masses in nanoliposomes can be attributed to the neutralization of superficial loads, massification and the formation of bridge between the adjacent particles. The process of sticking particles to each other occurs as a result of the adsorbent reaction between particles with relatively negative and positive levels [19]. In a similar study, storing liposomes at 20°?, especially in chitosan-coated types after 3 and 4 weeks, resulted in an increase in particle size to 2000 and 6500 nm, respectively [10].
At ambient temperature, liposomes become agglomeration due to the high fluidity of the membrane and form a clot that increases the particle size, resulting in a wide and heterogeneous particle size distribution. The followings can be considered as factors affecting the stability of nanoliposomes particles during storage: The nature and concentration of phospholipid in the system, the nanoliposome production method, the concentration, and type of stabilizer, such as cholesterol and environmental condition such as temperature, pH, and ionic strength [13]. Ko and Lee conducted some researches on the stability of retinol-containing nanoliposomes under dark conditions, ultraviolet light and at various temperatures (4, 25, 37, and 50 centigrade) [23]. The results indicated that the stability of the retinol-containing nanoliposomes increased under dark conditions. Regarding the storage temperatures, retinol was decomposed faster at higher temperatures, and the retinol-containing nanoliposome was most protected at a temperature of 4°? and under dark conditions, which is probably due to the reduced membrane permeability compared to oxygen, light and reduced oxidation and phospholipid hydrolysis.
3.4. The Effect of Storage at Ambient and Fridge Temperature on the Efficiency of Encapsulation
Different temperatures and storage conditions of nanoliposomes lead to a change in the crystalline structure of the lipid, to soften the membrane, and to decrease the zeta potential, and thus reduce the dispersion between particles and their massification [24]. The results indicating the effect of loaded peptide type and the storage temperature on the efficiency of the nanoliposomes encapsulation are shown in Figure 6. Under the same conditions, nanoliposomes loaded with peptides derived from the hydrolysis during a longer period (higher hydrolysis degree) were of higher release, and consequently of a greater drop in encapsulation efficiency. Moreover, among different storage conditions, nanoliposomes stored at the fridge temperature maintained a greater degree of initial encapsulation stability and showed a lower drop in the amount of this index. This finding represents higher stability of the nanoliposomes stored at the fridge temperature than the ambient temperature. For example, regarding the nanoliposomes loaded with peptides derived from hydrolysis for 300 min, the amount of encapsulation efficiency decreased from about 81% to, respectively, 62% and 42% after storage at the fridge and ambient temperature.
 | Figure 6: Encapsulation efficiency of primary nanoliposomes and nanoliposomes after storage at different temperatures [Click here to view] |
Similar results also indicated that refrigerate temperature was the best condition for storage of nanoliposomes. Further stability and lower release of nanoliposomes at low temperature can be attributed to the lower membrane permeability at low temperature, the reduced massification due to the low molecular mobility and the delay in the oxidative process of unsaturated fatty acids in phospholipids [24].
In a similar study, the effect of long-term storage on the release and encapsulation efficiency of hydrolyzed bioactive proteins of salmon in chitosan-coated liposomes were investigated. During storage of liposomes for 4 weeks at 4°?, there was no change in the size of liposome particles; however, the amount of hydrolyzed loss was reached nearly 87% after storing for 4 weeks. Furthermore, the liposomal coating with chitosan resulted in an 11% loss of active agent during the storage. Furthermore, the amount of active agent release in the liposomes coated with 0.4 and 0.6% chitosan was reached, respectively, 16% and 65% after 4 weeks of storage at the above temperature [10].
3.5. Infrared Spectroscopy (FTIR)
Figure 7a-c shows the chemical structure and FTIR spectroscopy in the pure peptides, empty nanoliposomes, and peptide-loaded nanoliposomes. In the structure of hydrolyzed proteins, the related peaks are shown in the following spectra; 3738 (O-H stretch), 3435 (N-H stretch), 1457 (C-N stretch), and 671 (N-H bending). The peaks in the range of 1645 cm−1 and 11535 cm−1 are, respectively, related to the amide I region (C-O stretch) and the amide II region in random spirals [13].
In the empty nanoliposomes, the bands near the 3439 cm−1 are related to hydroxyl stretch (O-H). Furthermore, some information on a regular or irregular state (such as the acyl chain flexibility) in the lipid membrane is obtained by examining the symmetric or asymmetric stretching vibrations of CH2, which are in the range of 2828 cm−1 and 3008 cm−1, respectively [1].
The peaks in the range of 1737 cm−1 in an empty nanoliposome indicate C=O stretching vibrations of an aliphatic stearic bond at the linkage between the hydrocarbon chain and the head group [25]. These vibrations were reported in a research carried out by Ramazanzadeh et al. (2017) in the 1754 cm−1 region, which possibly is due to differences in the formulation of the primary nanoliposome and the phosphatidylcholine type. Bands at the spectra of 1249 cm−1 and 1095 cm−1 represent the PO2- symmetric stretching vibrations of phospholipids [9].
 | Figure 7: Fourier-transform infrared spectroscopy in the pure peptides (a), empty nanoliposomes (b), and peptide-loaded nanoliposomes (c) [Click here to view] |
In addition, the asymmetric stretching vibrations of the choline group (N+CH3) were detected in the range of 966 cm−1. These vibrations were reported in nanoliposomes, evaluated by Ramezanzade et al. and Da Rosa Zavareze et al. (2014), respectively, at wavelengths of 995 cm−1 and 989 cm−1.
In peptide-loaded nanoliposomes, the 3439 cm−1 band (in the empty nanoliposome) was transferred to a lower frequency of 3438 cm−1 after loading with the peptide. The increased volume and the extended peaks in this range, in addition to its increased severity, indicate the presence of reactions and hydrogen bonds between hydroxyl groups (O-H) and N-H [26]. Displacement of the band to the higher or lower wavelengths suggests a reaction in the structure and between the functional groups existent in the encapsulated compounds with the liposomal membrane.
The peals related to the symmetric or asymmetric stretching vibrations of CH2, respectively, ranging from 2828 cm−1 to 3008 cm-1, were transferred to 2864 cm−1 and 2928 cm−1 after loading with peptides. This displacement in the peaks represents the effect of peptides and their placement in the inner region of the monolayer nanoliposomal membrane. Furthermore, the results of this study indicate the ionic complexation between phosphatidylcholine and peptides derived from enzymatic hydrolysis [18].
The peaks in the range of 1737 cm−1 in an empty nanoliposome, which indicate C=O stretching vibrations of an aliphatic stearic bond at the linkage between the hydrocarbon chain and the head group, after loading the nanoliposomes with peptides, were transmitted to 1739 cm−1 at higher frequencies. This indicates the reaction through hydrogen bonding between carbonyl (C=O) phosphatidylcholine group and the active compound [18].
The FTIR spectroscopy process was conducted to evaluate the reaction between phosphatidylcholine and hydrolysates. The FTIR test is performed to investigate the presence of minor changes in the structure of lipid and active compounds by analyzing the frequency, intensity, and changes in bandwidth in different vibrations indicating the acyl chain, interfacial, and superficial regions. In the structure of liposome, the orientation of the lipophilic regions is toward the monolayer center, and the polar regions are oriented toward the inner region and the outer surface of the capsule. The FTIR test measures the asymmetric axial stretching in the double bond of phosphate groups (PO2), the axial stretching of the carbonyl group (C=O), and the axial stretching of the CH2 group in the nanoliposomes [27]. The displacement of the C=O stretching bands represents reactions such as hydrogen bonds between the monolayer carbonyl regions of liposome and the active compound (such as peptides derived from enzymatic hydrolysis). The phosphate group frequency is also useful for investigating the water supplying state of the phospholipid polar group.
Increasing and decreasing the frequency are, respectively, occurred as the result of water loss and more water supplying in the phosphate group. The frequency of this bond determines the presence or absence of hydrogen bonds between the phosphate group and the hydrogen atom of water or large biological molecules [27].
In this research, the peaks related to the phosphate phosphatidylcholine group were moved from 1124 cm−1 in the empty nanoliposome to 1099 cm−1 in nanoliposomes loaded with peptides. In a similar study, in capsules containing hydrolysates, a displacement from 1082 cm−1 to 1084 cm−1 was observed in the phosphate band in phosphatidylcholine group [18]. The band related to the stretching in the choline group (N+CH3) in the polar region of phosphatidylcholine was displaced from 966 cm−1 to 965 cm−1 in the hydrolyzed capsules. FTIR spectroscopy shows the reactions of peptides with groups C=O and PO2 in phosphatidylcholine, indicating the placement of these peptides in the polar regions of the lipid and the formation of hydrogen bonds with PO2 [18].
 | Figure 8: Scanning electron microscopy of pure peptides (a), empty nanoliposomes (b), and peptide-loaded nanoliposomes (c) [Click here to view] |
3.6. The Particles Morphology Evaluation SEM
Figure 8a, b and c shows SEM images of the structural properties of empty nanoparticles loaded with peptides derived from the enzymatic hydrolysis of cannabis protein. The resulting images represent dense and compact masses of relatively spherical nanoparticles with flat surfaces. These images also confirmed the results of the particle size evaluation by DLS.
In a similar study, Vitamin D was loaded into the chitosan-coated nanostructured lipid carriers, and the physical stability was investigated. The encapsulation efficiency, physical stability, particle size, polydispersity index, and the zeta potential were investigated. The morphological characteristics of the coated and non-coated nanoparticles were compared using transmission electron microscopy and SEM electron microscopy. These images implied the compacted structures of particles. Moreover, the layered structures were observed in the coated nanoparticles. Furthermore, the non-coated particles were deformed during the drying process and preparing the samples for analysis; however, these changes were not observed in the coated samples [28].
4. CONCLUSION
In recent years, the ever-increasing incidence of various diseases, such as cardiovascular disease, cancers, and diabetes, has led researchers to understand the relationship and the role of food in the health of the consumer and in decreasing the incidence of these disorders. Among the functional compounds, antioxidant peptides with many characteristics, such as low allergenicity, blood pressure reducer, antidiabetes, and anticancer, are natural and efficient sources of antioxidants with the chelating activity of the metal ions and many other health-giving benefits. The present research aimed at developing a lipid conduction system using nanoliposomes to load antioxidant peptides. The results showed the effect of hydrolysis degree and peptide type on the characteristics of the loaded nanoliposomes, in particular, encapsulation efficiency, and zeta potential. Furthermore, the stability and maintenance of the encapsulation efficiency were affected by the storage temperature. FTIR spectroscopy evaluation also indicated the placement of peptides in the vesicle structure and the formation of an effective ionic complex between functional groups in peptides with phosphatidylcholine. The microstructure evaluation of nanoparticles using SEM also suggests the existence of dense clusters containing particles with flat surfaces and spherical structure in nanodimensions.
5. REFERENCES
1. Bilge D, Sahin I, Kazanci N, Severcan F. Interactions of tamoxifen with distearoyl phosphatidylcholine multilamellar vesicles: FTIR and DSC studies. Spectrochim Acta A Mol Biomol Spectrosc 2014;130:250-6. CrossRef
2. Gordon MH. The development of oxidative rancidity in foods. In: Antioxidants in Food. Netherlands: Elsevier; 2001. p. 7-21. CrossRef
3. Sun J, He H, Xie BJ. Novel antioxidant peptides from fermented mushroom ganoderma lucidum. J Agric Food Chem 2004;52:6646-52. CrossRef
4. Xie Z, Huang J, Xu X, Jin Z. Antioxidant activity of peptides isolated from alfalfa leaf protein hydrolysate. Food Chem 2008;111:370-6. CrossRef
5. Chen HM, Muramoto K, Yamauchi F, Fujimoto K, Nokihara K. Antioxidative properties of histidine-containing peptides designed from peptide fragments found in the digests of a soybean protein. J Agric Food Chem 1998;46:49-53. CrossRef
6. Pihlanto, A. Antioxidative peptides derived from milk proteins. Int Dairy J 2006;16:1306-14. CrossRef
7. Yin SW, Tang CH, Cao JS, Hu EK, Wen QB, Yanga XQ. Effects of limited enzymatic hydrolysis with trypsin on the functional properties of hemp (Cannabis sativa L.) protein isolate. Food Chem 2008;106:1004-13. CrossRef
8. Mohan A, Rajendran SR, Thibodeau J, Bazinet L, Udenigwe CC. Liposome encapsulation of anionic and cationic whey peptides: Influence of peptide net charge on properties of the nanovesicles. LWT 2018;87:40-6. CrossRef
9. Ramezanzade L, Hosseini SF, Nikkhah M. Biopolymer-coated nanoliposomes as carriers of rainbow trout skin-derived antioxidant peptides. Food Chem 2017;234:220-9. CrossRef
10. Li X, Deng J, Shen S, Li T, Yuan M, Yang R, et al. Antioxidant activities and functional properties of enzymatic protein hydrolysates from defatted Camellia oleifera seed cake. J Food Sci Technol 2015;52:5681-90. CrossRef
11. Zavareze ED, Telles AC, El Halal SL, da Rocha M, Colussi R, de Assis LM, et al. Production and characterization of encapsulated antioxidative protein hydrolysates from Whitemouth croaker (Micropogonias furnieri) muscle and byproduct. LWT Food Sci Technol 2014;59:841-8. CrossRef
12. Feyzi S, Varidi M, Zare F, Varidi MJ. Fenugreek (Trigonella foenum-graecum) seed protein isolate: Extraction optimization, amino acid composition, thermo and functional properties. J Sci Food Agric 2015;95:3165-76. CrossRef
13. Sarabandi K, Mahoonak AS, Hamishehkar H, Ghorbani M, Jafari SM. Protection of casein hydrolysates within nanoliposomes: Antioxidant and stability characterization. J Food Eng 2019;251:19-28. CrossRef
14. Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976;72:248-54. CrossRef
15. Sarabandi K, Mahoonak AS, Hamishekar H, Ghorbani M, Jafari SM. Microencapsulation of casein hydrolysates: Physicochemical, antioxidant and microstructure properties. J Food Eng 2018;237:86-95. CrossRef
16. Nagle JF, Tristram-Nagle S. Structure of lipid bilayers. Biochim Biophys Acta 2000;1469:159-95. CrossRef
17. Romero-Pérez A, García-García E, Zavaleta-Mancera A, Ramírez-Bribiesca JE, Revilla-Vázquez A, Hernández-Calva LM, et al. Designing and evaluation of sodium selenite nanoparticles in vitro to improve selenium absorption in ruminants. Vet Res Commun 2010;34:71-9. CrossRef
18. da Rosa Zavareze E, Telles AC, El Halal SL, da Rocha M, Colussi R, de Assis LM, et al. Production and characterization of encapsulated antioxidative protein hydrolysates from Whitemouth croaker (Micropogonias furnieri) muscle and byproduct. LWT Food Sci Technol 2014;59:841-8. CrossRef
19. Mun S, Decker EA, McClements DJ. Influence of droplet characteristics on the formation of oil-in-water emulsions stabilized by surfactant-chitosan layers. Langmuir 2005;21:6228-34. CrossRef
20. Mosquera M, Giménez B, da Silva IM, Boelter JF, Montero P, Gómez-Guillén MC, et al. Nanoencapsulation of an active peptidic fraction from sea bream scales collagen. Food Chem 2014;156:144-50. CrossRef
21. Were LM, Bruce B, Davidson PM, Weiss J. Encapsulation of nisin and lysozyme in liposomes enhances efficacy against listeria monocytogenes. J Food Prot 2004;67:922-7. CrossRef
22. Wu L, Zhang J, Watanabe W. Physical and chemical stability of drug nanoparticles. Adv Drug Deliv Rev 2011;63:456-69. CrossRef
23. Ko S, Lee SC. Effect of nanoliposomes on the stabilization of incorporated retinol. Afr J Biotechnol 2010;9:6158-61.
24. Gibis M, Zeeb B, Weiss ZJ. Formation, characterization, and stability of encapsulated hibiscus extract in multilayered liposomes. Food Hydrocoll 2014;38:28-39. CrossRef
25. Hasan M, Ben Messaoud G, Michaux F, Tamayol A, Kahn CJ, Belhaj N, et al. Chitosan-coated liposomes encapsulating curcumin: Study of lipid polysaccharide interactions and nanovesicle behavior. RSC Adv 2016;6:45290-304. CrossRef
26. Ferraresi TM, Ferreira EP, Silva WT, Neto L. Application of near and medium infrared spectroscopy in the assessment of soil microbial biomass. Empresa Bras Pesqui Agropecu Ministério Agric 2012;6:5-36.
27. Toyran N, Severcan F. Competitive effect of Vitamin D2 and Ca2+ on phospholipid model membranes: An FTIR study. Chem Phys Lipids 2003;123:165-76. CrossRef
28. Rabelo RS, Oliveira IF, da Silva VM, Prata AS, Hubinger MD. Chitosan coated nanostructured lipid carriers (NLCs) for loading Vitamin D: A physical stability study. Int J Biol Macromol 2018;119:902-12. CrossRef