1. INTRODUCTION
Melastoma malabathricum, commonly known as senggani, is a tropical flowering plant native to Southeast Asia [1]. Its flowers exhibit color variations ranging from white [2] to deep purple [3,4], with the purple variety particularly rich in phenolic compounds, including flavonoids such as rutin, quercetin 3-glucoside, and astragalin. Figure 1 illustrates the morphological characteristics of the purple senggani flower, which displays vibrant purple petals and a distinct floral structure reflecting its ornamental and medicinal significance. This striking appearance corresponds to its high content of bioactive pigments and phenolic constituents. Conventionally, senggani has been widely used in folk medicine due to its therapeutic properties, including anti-inflammatory, antimicrobial, and wound-healing effects [5]. These bioactivities are mainly attributed to the presence of secondary metabolites – primarily phenolic compounds – known for their strong antioxidant potential [6].
 | Figure 1: Purple flower of Melastoma malabathricum (senggani) collected from the special region of Yogyakarta, Indonesia. [Click here to view] |
Phenolic compounds play crucial roles in plant defense, contributing to color, taste, and stability in plant-derived products [7,8]. Among them, flavonoids, phenolic acids, tannins, and lignins exhibit diverse structural complexity and biological activities [9]. However, phenolic compounds are susceptible to environmental and processing conditions. Thermal treatments, such as blanching, can induce degradation, oxidation, or polymerization of phenolics, potentially reducing their antioxidant efficacy [10]. While some flavonoids remain relatively heat-stable, others, such as anthocyanins, are particularly sensitive to thermal processing [11]. For instance, heating can cleave glycosidic bonds in flavonoids, generating aglycones with altered bioactivity [12]. The extent of degradation largely depends on temperature and exposure time, where moderate heating (e.g., blanching at 70°C for short durations) may preserve phenolics, whereas prolonged or high-temperature treatment (e.g., boiling at 100°C for extended periods) often leads to considerable losses [13].
Blanching is commonly applied as a pretreatment before drying to inactivate enzymes, shorten drying time, and suppress microbial activities [14]. In particular, polyphenol oxidase (PPO) and peroxidase (POD) are key enzymes responsible for enzymatic browning and phytochemical degradation in plant materials [15]. Their inactivation during blanching is essential for maintaining the quality and bioactivity of phenolic-rich plants. Interestingly, while enzyme inactivation may help preserve target compounds during storage, blanching itself may also cause phenolic loss through thermal degradation or leaching into the blanching medium.
Previous studies suggest that blanched plant materials may yield higher metabolite extraction due to enzyme inactivation [16]; however, there remains a knowledge gap regarding how blanching influences specific flavonoid compounds and antioxidant capacity in M. malabathricum. Therefore, this study aimed to evaluate the effects of water blanching at different temperatures and times on PPO and POD activity, individual phenolic profiles, total phenolic content (TPC), and total antioxidant capacity (TAC) in purple senggani flowers. The findings are expected to provide evidence-based recommendations for optimal thermal pretreatment to preserve the functional quality of this underutilized but bioactive-rich plant.
2. MATERIALS AND METHODS
2.1. Chemicals and Reagents
High-performance liquid chromatography (HPLC)-grade methanol and acetic acid were purchased from Merck KGaA (Darmstadt, Germany). Water for injection was purchased from PT. Ikapharmindo Putramas Pharmaceutical Laboratories (Jakarta, Indonesia). Two flavonoid standards, quercetin 3-glucoside (CAS number: 482-35-9; purity: ≥98%) and rutin (CAS number: 207671-50-9; purity: ≥94%), were purchased from Sigma–Aldrich (St. Louis, MO, USA).
2.2. Plant Material and Sample Preparation
Purple M. malabathricum flowers were harvested from the Special Region of Yogyakarta, Indonesia (coordinates: 7°35’22.9”S 110°27’33.2”E). The flowers were picked in the morning and transported in sealed plastic bags for immediate processing. Water blanching was performed following the method described by Magangana et al. [16], with slight modifications, in a water bath (Memmert, Schwabach, Germany) at three temperatures: 70°C, 80°C, and 90°C for up to 3 min. Subsamples were taken every 30 s, rapidly cooled in ice water, and stored for further analysis. Unblanched flowers were used as the control.
All flower samples were frozen at −20°C for 2 days in a freezer (MDF-U55V-PE, Panasonic, Japan), followed by freeze-drying at −45°C for 2 days using a CoolSafe 4–15 L freeze dryer (LaboGene, Denmark). The freeze-dried flowers were ground into a fine powder using a laboratory grinder (Klaz, China) and stored at −20°C until further analysis.
All treatments were conducted using three independent biological replicates (n = 3), each consisting of flower samples collected on different days. For each biological replicate, all measurements (enzyme activity, HPLC analysis, TPC, and TAC) were performed in duplicate as technical replicates.
2.3. Determination of PPO Activity
PPO activity was determined according to the method described by Golan et al. [17], with slight modifications. A 15 g sample was blended with 60 mL deionized water for 30–60 s at 0°C, filtered, and kept in an ice bath. For analysis, 2.6 mL of 0.01 M acetate buffer (pH 5.0), 0.3 mL of 0.5 M pyrocatechol, and 0.1 mL enzyme extract were mixed, and the absorbance was monitored at 420 nm for 3 min. One unit of activity was defined as an absorbance change of 0.001/min/mL extract.
2.4. Determination of POD Activity
POD activity was analyzed using the spectrophotometric method described by Flick Jr. et al. [18], with minor modifications. Five grams of fresh flowers were blended with 0.1 M phosphate buffer (pH 6.5) and filtered. The filtrate was centrifuged at 3000 rpm for 5 min. Then, 10 mL of 0.05 M phosphate buffer, 1 mL of 0.5% guaiacol in 5% ethanol, and 1 mL of 0.3% hydrogen peroxide were added to the supernatant. Absorbance at 470 nm was recorded, and activity was expressed as absorbance change per minute.
2.5. Ultrasound-assisted Extraction (UAE)
Phenolic compounds were extracted using an ultrasonic probe system (UP200St, Hielscher, Germany; 26 kHz, 200 W, 7 mm probe) following Mareta et al. [4]. Extraction used 60% methanol in water, a 1:10 g/mL sample-to-solvent ratio, 60% pulse-duty cycle, for 15 min at 70°C using a recirculating bath (Frigiterm-TFT-10, J.P. Selecta, Spain). Extracts were centrifuged at 4000 rpm for 15 min at 4°C (Sorvall ST-8R, Thermo Fisher, Germany), and the final volume adjusted to 25 mL with the extraction solvent.
2.6. HPLC-Diode Array Detector (DAD) Analysis of Phenolic Compounds
HPLC-DAD (Shimadzu Corp., Kyoto, Japan) system was used to analyze the senggani flower extract. The flavonoid compounds were isolated using a C18 reverse-phase column (5 μm, 4.6150 mm, Shimadzu Corp., Kyoto, Japan), maintained at a column oven temperature of 30°C. The method employed a mobile phase that included phase A (5% methanol, 2% acetic acid, and 93% water) and phase B (88% methanol, 2% acetic acid, and 10% water). The elution gradient (time, % solvent B) was programmed as follows: 0 min at 0%; 20 min at 100%; 25 min at 100%; and 27 min at 0%, with a flow rate of 1 mL/min [4]. To filter the extracts, a 0.45 μm nylon syringe filter was used. The chromatograms were analyzed by LabSolutions CS (Shimadzu Corp., Kyoto, Japan). DAD performed a complete scan for the spectrum (200–400 nm) to identify the flavonoid compounds. Identification was performed by comparing the spectrum and retention time of the compounds in the sample with those of standard compounds. The compounds (rutin and quercetin 3-glucoside) were quantified using a 260 nm wavelength. Only rutin and quercetin 3-glucoside were quantified using authentic standards. Other compounds (trifolin, astragalin, and flavonoid derivative) were tentatively identified based on literature spectra and ultraviolet visible (UV-Vis) characteristics, and quantified as rutin equivalents [4,19].
2.7. Determination of TPC
TPC was measured using a modified Folin-Ciocalteu spectrophotometric method described by Bobková et al. [20]. A 1 mL aliquot of senggani flower extract, diluted 100–fold, was combined with 0.5 mL of 10% Folin-Ciocalteu reagent. This mixture was allowed to sit for 10 min before adding 5 mL of 2% sodium carbonate solution. Following an incubation period of 40–60 min, the absorbance was recorded at 765 nm using a UV-Vis Spectrophotometer (UV-2450, Shimadzu Corporation, Kyoto, Japan).
To prepare a standard solution for gallic acid, 0.1 g of gallic acid was dissolved in distilled water to achieve a final volume of 100 mL, resulting in a stock solution. From this stock, 1 mL was diluted to 200 mL with distilled water to create a working solution. A calibration curve was established, covering a range of 20–100 mg/L of gallic acid, with a coefficient of determination (R2) of 0.996. Blanks were prepared using Folin-Ciocalteu reagent and distilled water without any standards or extracts. The TPC values were reported as milligrams of gallic acid equivalent (GAE) per gram of sample (mg GAE/g).
2.8. Determination of TAC
The TAC was assessed using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay, based on the method described by Brand-Williams et al., [21] with minor adjustments. A 0.1 mL sample of senggani extract, diluted 100-fold, was combined with a 0.1 mM DPPH solution. The mixture was allowed to stand for 40 min to reach equilibrium. The absorbance was measured at 517 nm with a UV-Vis Spectrophotometer (UV-2450, Shimadzu Corporation, Kyoto, Japan). The TAC was reported as % radical scavenging activity (% RSA), calculated using Equation 1.
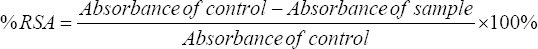 |
The limits of detection (LOD) of the DPPH assay used in this study was approximately 0.25% RSA. Values below this threshold were considered undetectable and are reported as
2.9. Replication and Statistical Analysis
All experiments were performed in triplicate using three independent biological replicates (n = 3). Each biological replicate represented a distinct batch of freshly harvested senggani flowers, individually processed for all blanching treatments and the control. Within each biological replicate, all analytical determinations – including PPO and POD enzymatic activities, HPLC-DAD quantification of individual phenolic compounds, TPC, and TAC – were conducted in technical triplicate. Results are expressed as mean ± standard deviation (SD). Statistical significance among treatments was evaluated by one-way analysis of variance followed by Tukey’s honestly significant difference post hoc test at a confidence level of P < 0.05.
3. RESULTS AND DISCUSSION
3.1. Effect of Water Blanching on PPO and POD Activity
PPO and POD are key enzymes that contribute to the degradation of phytochemicals in plants, leading to enzymatic browning and reduced antioxidant activity [15]. PPO catalyzes the oxidation of phenolic compounds into quinones, which polymerize into brown pigments, while POD uses hydrogen peroxide to oxidize a broad range of substrates [22,23]. The suppression of both enzymes is critical to maintaining the quality of phenolic-rich plant materials during processing.
The results [Figure 2] showed that PPO activity was significantly reduced following blanching at all temperature-time combinations. At 70°C for 30 s, PPO activity decreased by 37%, and this reduction progressed with increased temperature and time, reaching up to 93% inactivation at 90°C for 180 s. This pattern is consistent with previous studies indicating that PPO is thermolabile, with optimal activity at 30–40°C and rapid deactivation at higher temperatures [24,25].
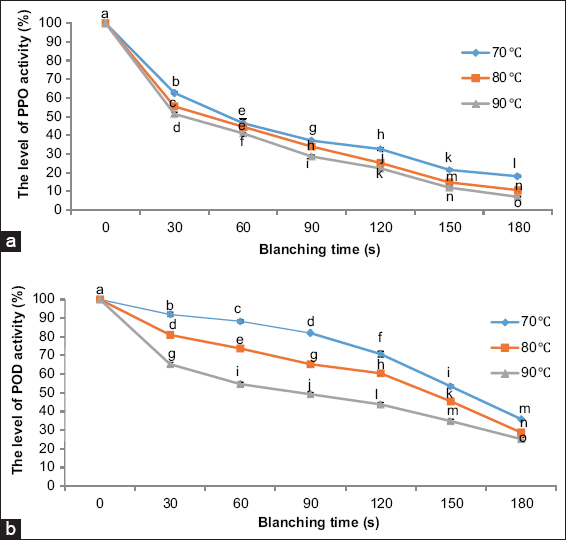 | Figure 2: Changes in (a) polyphenol oxidase (PPO) and (b) peroxidase (POD) activity in Melastoma malabathricum (purple senggani) flowers after water blanching at different temperatures (70, 80, and 90 °C) and durations (30–180 s). PPO and POD activity decreased significantly with increased blanching temperature and time. Different lowercase letters (a–o) above the markers indicate statistically significant differences (P < 0.05) according to Tukey’s HSD post hoc test. Error bars represent standard deviations. [Click here to view] |
POD activity followed a similar but less dramatic trend. Blanching at 70°C for 30 s caused an 8% reduction in activity, which increased to 75% inactivation at 90°C for 180 s. The relatively greater thermal stability of POD compared to PPO is in line with earlier reports [26,27]. These findings suggest that water blanching is effective in reducing enzymatic browning potential in senggani flowers. However, while enzyme inactivation is desirable for preventing oxidative degradation, the subsequent sections reveal that blanching has notable consequences on the chemical composition and functionality of the flowers.
3.2. Changes in Individual Phenolic Compounds
The individual phenolic compounds were identified using HPLC-DAD by matching each compound’s UV-Vis spectra and retention times with the standards. Five compounds were detected as rutin, quercetin 3-glucoside, trifolin, astragalin, and flavonoid derivative. Figure 3 shows the HPLC chromatogram of senggani extracts at 260 nm [Figure 3a] and the UV-Vis spectra of the five flavonoid compounds. UV-Vis spectra of rutin and quercetin 3-glucoside [Figure 3b and c] were obtained by matching the standards with the samples. Trifolin and astragalin [Figure 3d and 3e] were determined by matching the UV-Vis spectra obtained with the UV-Vis spectra in the study by Noh et al. [19]. This identification was corroborated by high-resolution mass spectrometry profiling of M. malabathricum (senggani) flower extracts, as documented in our prior study [4]. The last compound exhibited a UV-Vis spectral pattern similar to that of typical flavonoid compounds; however, its exact structure has not yet been identified and was therefore designated as a tentatively identified flavonoid derivative [Figure 3f].
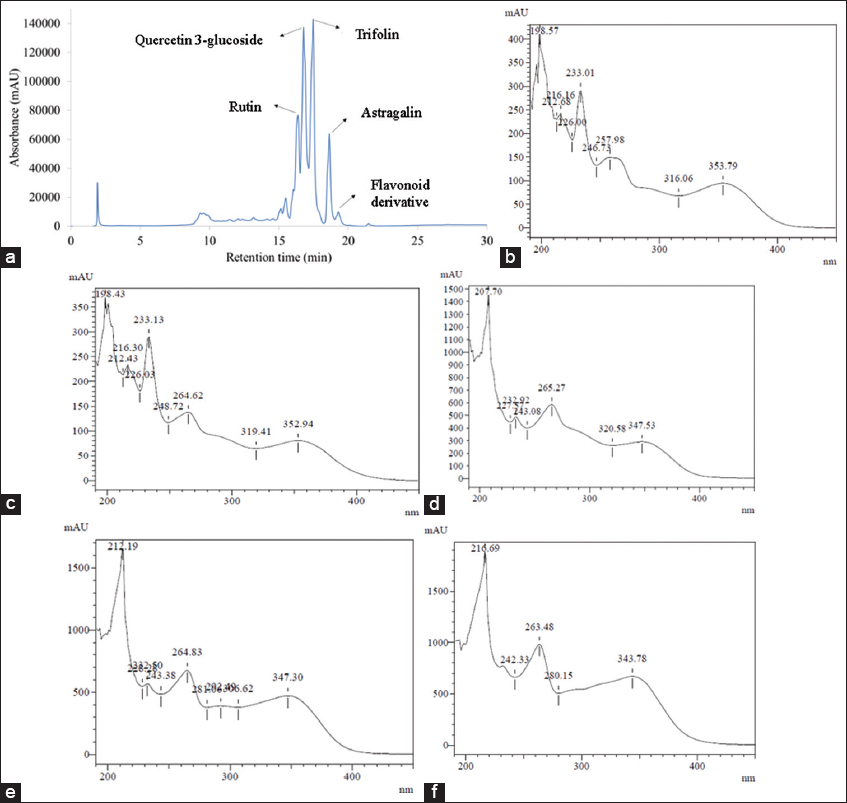 | Figure 3: High-performance liquid chromatography (HPLC) chromatogram of senggani extracts at 260 nm (a) and ultraviolet visible (UV-Vis) spectra of individual phenolic compounds detected in Melastoma malabathricum (purple senggani) flower extract using HPLC-diode array detector: rutin (b), quercetin 3-glucoside (c), trifolin (d), astragalin (e), and a flavonoid derivative (f). The spectra indicate the characteristic absorbance features of flavonoid compounds in the UV region. [Click here to view] |
Rutin and quercetin 3-glucoside standard calibration curves with the range of 0.5–40 ppm were arranged to cover the concentration range of the sample. Table 1 shows the performance of the HPLC-DAD method for determining rutin and quercetin 3-glucoside. Regression analysis was employed to quantify the individual phenolic compounds in the sample and to assess the coefficient of determination (R2) to demonstrate linearity within the examined range. The results showed high R2 values for rutin (0.994) and quercetin 3-glucoside (0.999). The LOD and limit of quantification were calculated using the slope and the SD at the origin of the regression analysis of the calibration curve. Therefore, the chromatographic method utilized in this study proved reliable for identifying the compounds in the senggani flowers.
Table 1: Analytical performance of the HPLC-DAD method for the quantification of rutin and quercetin 3-glucoside.
| Phenolic compounds | Range (0.5–40 ppm) | LOD (ppm) | LOQ (ppm) | |
|---|---|---|---|---|
| Linear equation | R2 | |||
| Rutin | y=26020x+4543.6 | 0.994 | 1.20 | 3.63 |
| Quercetin 3- glucoside | y=31823x–5579.7 | 0.999 | 0.73 | 2.22 |
HPLC-DAD: High-performance liquid chromatography-diode array detector, LOD: Limit of detection, LOQ: Limit of quantification
Blanching markedly decreased the concentration of these phenolics [Table 2]. Under control conditions (no blanching), rutin and quercetin 3-glucoside were the dominant flavonoids, with concentrations of 510.74 ± 8.96 and 686.43 ± 22.87 μg/g, respectively. However, as blanching time increased, the concentration of these flavonoids clearly reduced, particularly at higher temperatures. At 70°C, after 30 s, the rutin concentration dropped significantly to 119.90 ± 6.65 μg/g and quercetin 3-glucoside decreased to 121.88 ± 5.76 μg/g. Longer blanching durations and higher temperatures led to further losses, with most flavonoids falling below the LOD beyond 120 s of blanching.
Table 2: Concentration of individual phenolic compounds in purple senggani flowers after water blanching at various temperatures and durations.
| Blanching condition | Phenolic compounds (μg/g) | ||||
|---|---|---|---|---|---|
| Rutin | Quercetin 3-glucoside | Trifolin** | Astragalin** | Flavonoid derivative** | |
| Control | 510.74±8.96f | 686.43±22.87g | 569.34±13.49d | 590.35±11.53d | 414.26±30.06a |
| 70°C, 30 s | 119.90±6.65e | 121.88±5.76f | 54.75±6.43b* | 48.72±8.37bc* | |
| 70°C, 60 s | 105.40±1.97d | 91.30±0.15cd | 87.15±5.30c* | 43.84±5.99ab* | |
| 70°C, 90 s | 67.59±3.98b* | 42.23±2.22b* | |||
| 70°C, 120 s | |||||
| 70°C, 150 s | |||||
| 70°C, 180 s | |||||
| 80°C, 30 s | 112.57±5.05de | 105.05±0.92e | 53.27±3.78b* | 57.77±2.79c* | |
| 80°C, 60 s | 108.47±8.59d | 101.91±4.12de | 47.88±7.47b* | 50.04±8.26bc* | |
| 80°C, 90 s | 90.82±9.08c | 87.22±6.82c | 37.43±3.44a* | 36.57±7.60a* | |
| 80°C, 120 s | |||||
| 80°C, 150 s | |||||
| 80°C, 180 s | |||||
| 90°C, 30 s | 93.50±6.9c | 46.01±3.13b | |||
| 90°C, 60 s | 85.08±2.66c | 37.23±2.30ab* | |||
| 90°C, 90 s | 48.36±4.75a* | 28.89±1.25a* | |||
| 90°C, 120 s | |||||
| 90°C, 150 s | |||||
| 90°C, 180 s | |||||
Values are presented as mean±standard deviation. Different lowercase letters within the same column indicate statistically significant differences (P<0.05) based on Tukey’s honestly significant difference post hoc test. LOD: Limit of detection, LOQ: Limit of quantification. Values between LOD and LOQ are marked with an asterisk (*); compound concentration expressed as rutin equivalents are marked with two asterisks (***). LOD of rutin=24.00 µg/g; LOD of quercetin 3-glucoside=14.60 µg/g. Trifolin, astragalin, and the flavonoid derivative were reported as rutin equivalents and share the same detection threshold
At 80°C, a similar trend was observed. The rutin level fell to 112.57 ± 5.05 μg/g after 30 s of blanching, while quercetin 3-glucoside decreased to 105.05 ± 0.92 μg/g. Again, longer blanching times led to levels below LOD. At the higher temperature (90°C), the degradation of phenolic compounds was even more pronounced. After 30 s, the rutin concentration was reduced to 93.50 ± 6.94 μg/g, and quercetin 3-glucoside dropped to 46.01 ± 3.13 μg/g. Prolonging the time to 120 s or more resulted in undetectable levels of these compounds.
Flavonoid compounds such as rutin, quercetin 3-glucoside, trifolin, and astragalin are known for their antioxidant properties and contribute significantly to the health benefits of plant extracts [19]. However, these compounds are susceptible to thermal treatment [28], as demonstrated in this study, which analyzed the effects of blanching at various temperatures and durations on the phenolic content in senggani flowers.
This rapid degradation can be attributed to both thermal decomposition and leaching into the blanching water. Although flavonoids are generally poorly soluble in water, heat increases their solubility by weakening hydrogen bonding and enhancing molecular motion [29-31]. Consequently, flavonoids may dissolve more readily into the blanching medium. Previous studies on other plant matrices have reported similar trends, where prolonged boiling or steaming results in the loss of water-soluble antioxidants [32].
Furthermore, heat may cleave glycosidic bonds in flavonoid glycosides such as quercetin 3-glucoside and rutin, potentially forming aglycones with reduced solubility or reactivity [12]. Similar declines in flavonoid content following thermal treatment have been documented in other phenolic-rich plants [28]. These results affirm that while blanching suppresses enzymatic degradation, it concurrently accelerates the loss of valuable phenolic compounds.
In this study, trifolin, astragalin, and flavonoid derivatives were quantified using a standard calibration curve for rutin (equivalent to rutin). Although their relative changes are meaningful, absolute quantification should be interpreted cautiously. Future work needs to include Liquid chromatography–tandem mass spectrometry (LC-MS/MS) analysis and standard-based quantification to increase confidence in compound determination.
3.3. Impact on TPC and Antioxidant Capacity
Phenolic compounds, particularly flavonoids, contribute substantially to the antioxidant properties of plant extracts [7,33,34]. While some plants have shown increased phenolic levels after blanching due to enhanced release of bound phenolics [35,36], others experienced a decrease [37] including purple senggani that consistently exhibited reductions [Figure 4]. This may be due to the predominance of free flavonoids in senggani, which are more prone to degradation or solubilization, rather than thermal release from conjugated forms.
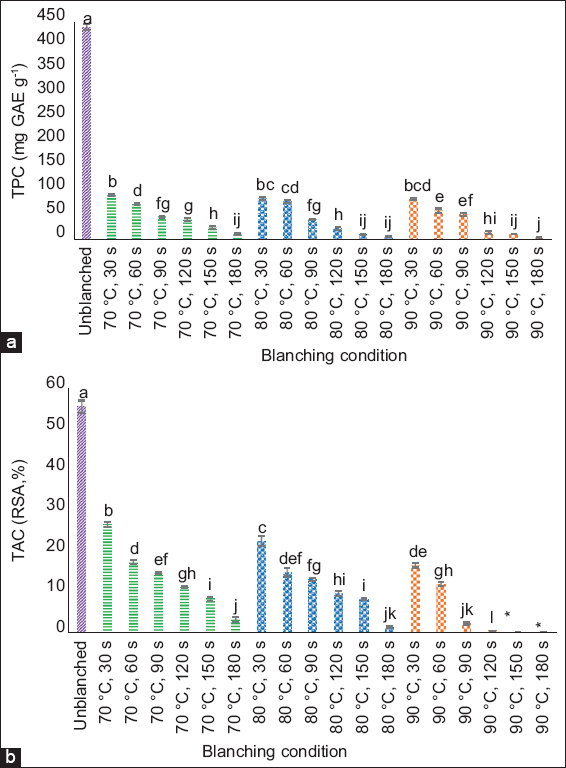 | Figure 4: Changes in (a) total phenolic content (TPC) and (b) total antioxidant capacity (TAC) of Melastoma malabathricum (purple senggani) flowers after water blanching at 70, 80, and 90°C for 30–180 s. Different lowercase letters (a-l) above the bars indicate statistically significant differences (P < 0.05) based on Tukey’s honestly significant difference post hoc test. Error bars represent standard deviations. Values below the limit of detection (LOD) are marked with an asterisk (*). [Click here to view] |
The TPC in unblanched samples was 441.12 ± 5.95 mg GAE/g, but dropped to 92.52 ± 1.98 mg GAE/g after blanching at 70°C for 30 s. At the highest treatment (90°C for 180 s), TPC declined sharply to 4.21 ± 0.00 mg GAE/g. A similar trend was observed for TAC, which dropped from 55.60 ± 1.53% in unblanched flowers to below the detection limit at the most intense blanching condition (90°C for 150 s).
The decrease in the TPC and TAC values of senggani flowers due to the blanching treatment results from the reduction in the value of individual phenolic compounds, which are damaged due to high temperatures and dissolve in water during the blanching treatment. The phenolic compounds in senggani flowers, most of which are flavonoids (rutin, quercetin 3-glucosides, trifolin, astragalin), are polar, so they easily dissolve in polar solvents such as water [38]. These results aligned with our previous research, which stated that the highest TPC value was produced when senggani flowers were extracted using water as a solvent, compared to methanol, ethanol, and hexane [4].
The reduction in TPC and TAC values may also result from the degradation of other phenolic components, such as anthocyanins and tannins present in senggani flowers [3,39]. These compounds are highly water-soluble and thermally labile, rendering them susceptible to loss during blanching processes [40-43]. However, given the focus of this study on flavonoid compounds, future investigations should examine the stability of anthocyanin and tannin pigments in senggani flowers.
3.4. Interpreting the Trade-off Between Enzyme Inactivation and Phenolic Loss
The dual role of blanching – suppressing enzyme activity while risking phytochemical degradation – poses a significant challenge in optimizing postharvest treatment. Although enzyme inactivation is beneficial for preventing browning and prolonging shelf life, excessive thermal exposure it also reduces antioxidant capacity and phenolic content, which are important indicators of phytochemical preservation. The results of this study affirm that higher blanching temperatures and longer durations should be avoided for phenolic-rich materials like senggani flowers.
Although this study did not compare different processing methods, previous studies have reported better phenolic retention under milder or non-thermal treatments. Future research is recommended to investigate techniques such as steam blanching, freeze-drying, or cold storage to evaluate their potential in enhancing flavonoid retention while minimizing thermal degradation, as observed in other botanical matrices [10,12,32]. This study did not include blanching-only (non-immersion heat) or blanching-water analysis controls, which limits our ability to distinguish between thermal degradation and leaching as mechanisms of flavonoid loss. Future studies should address this gap to clarify compound fate during blanching.
Taken together, this study offers new insights into the processing sensitivity of M. malabathricum flowers. By systematically evaluating enzyme activity and phytochemical composition under various blanching conditions, we provide practical recommendations for preserving their nutritional and functional properties in future applications.
4. CONCLUSION
Water blanching effectively inactivated PPO and POD enzymes in purple M. malabathricum (senggani) flowers, with up to 93% and 75% reduction in activity, respectively. However, this thermal treatment also led to a significant decline in individual flavonoid compounds – such as rutin, quercetin 3-glucoside, trifolin, and astragalin – as well as in TPC and TAC. Most flavonoids fell below detection limits after blanching at higher temperatures and longer durations, indicating their thermal sensitivity and solubility in hot water.
These findings highlighted a critical trade-off between enzyme inactivation and phytochemical preservation. Although water blanching was effective in reducing enzyme activity, it also led to considerable losses in phenolic compounds and antioxidant capacity. Thus, blanching as a pretreatment should be applied cautiously, and future comparative studies are needed to evaluate alternative techniques such as freeze-drying or cold storage for optimal preservation of phenolic-rich plant materials. The results of this study provide evidence-based guidance for optimizing postharvest processing strategies for phenolic-rich plant materials.
5. ACKNOWLEDGMENTS
The authors gratefully acknowledge the financial support provided through the Indonesian Education Scholarship program, administered by the Center for Higher Education Funding and Assessment under the Ministry of Higher Education, Science, and Technology of the Republic of Indonesia, in collaboration with the Indonesian Endowment Fund for Education (LPDP), Ministry of Finance of the Republic of Indonesia (Grant No. 202101121028). This funding substantially supported the completion of this research. The first author was awarded a doctoral (PhD) scholarship under this program, for which the authors express their sincere appreciation.
6. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
7. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
8. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
9. DATA AVAILABILITY
All data are available from the authors and will be provided upon reasonable request.
10. PUBLISHER’S NOTE
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors and the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
11. USE OF ARTIFICIAL INTELLIGENCE (AI)-ASSISTED TECHNOLOGY
The authors declare that they have not used artificial intelligence (AI)-tools for writing and editing of the manuscript, and no images were manipulated using AI.
REFERENCES
1. Joffry SM, Yob NJ, Rofiee MS, Affandi MM, Suhaili Z, Othman F, et al. Melastoma malabathricum (L.) smith ethnomedicinal uses, chemical constituents, and pharmacological properties:A review. Evid Based Complement Alternat Med. 2012;2012:258434. [CrossRef]
2. Lee CH, Lee TH, Ya'akoba H, Wong S, Ben Jannet H. Optimization of ultrasound-assisted extraction of total flavonoids content from the white flowering variety of Melastoma malabathricum. JKejuruter. 2019;2(1):91-102. [CrossRef]
3. Janna OA, Khairul A, Maziah M, Mohd Y. Flower pigment analysis of Melastoma malabathricum. Afr J Biotechnol. 2006;5(2):170-4.
4. Mareta DT, Santoso U, Supriyadi S, Setyaningsih W. Optimized ultrasound-assisted extraction for phenolic and antioxidant analysis in purple senggani (Melastoma malabathricum) flowers:Influence of developmental stages. Food Chem Adv. 2024;7:100949. [CrossRef]
5. Mayasari D, Murti YB, Sudarsono S, Pratiwi SU. Phytochemical, antioxidant and antibacterial evaluation of Melastoma malabathricum L.:An indonesian traditional medicinal plant. Trop J Nat Prod Res. 2021;5(5):819-824. [CrossRef]
6. Isnaini I, Permatasari IN, Mintaroem K, Widodo MA. Analysis of quercetin and kaempferol levels in various phase of flowers melastoma malabathricum L. Int J Plant Biol. 2018;9(1):1-4. [CrossRef]
7. Pratyusha S. Phenolic compounds in the plant development and defense:An overview. In:Plant Stress Physiology Perspectives in Agriculture. London:Intech Open;2016. 225-40.
8. Sulusoglu M. Phenolic compounds and uses in fruit growing. Turk J Agric Nat Sci Spec Issue. 2014;1:947. https://dergipark.org.tr/en/download/article-file/142206
9. Ozcan T, Delikanli B. Phenolics in human health. Int J Chem Eng Appl. 2014;5(5):393-6. [CrossRef]
10. De Lima Marsiglia WI, Conrado Oliveira LS, Santos NC, Da Silva Neto JM, Santos NC, et al. Thermal stability of total phenolic compounds and antioxidant activities of jaboticaba peel:Effect of solvents and extraction methods. J Indian Chem Soc. 2023;100(5):100995. [CrossRef]
11. Cao H, Saroglu O, Karadag A, Diaconeasa Z, Zoccatelli G, Adam Conte-Junior C, et al. Available technologies on improving the stability of polyphenols in food processing. Food Front. 2021;2(2):109-39. [CrossRef]
12. ElGamal R, Song C, Rayan AM, Liu C, Al-Rejaie S, ElMasry G. Thermal degradation of bioactive compounds during drying process of horticultural and agronomic products:A comprehensive overview. Agronomy. 2023;13(6):1580. [CrossRef]
13. Gonçalves EM, Pinheiro J, Alegria C, Abreu M, Brandão TR, Silva CL. Degradation kinetics of peroxidase enzyme, phenolic content, and physical and sensorial characteristics in broccoli (Brassica oleracea L. ssp. Italica) during blanching. J Agric Food Chem. 2009;57(12):5370-5. [CrossRef]
14. An NN, Lv WQ, Li D, Wang LJ, Wang Y. Effects of hot-air microwave rolling blanching pretreatment on the drying of turmeric (Curcuma longa L.):Physiochemical properties and microstructure evaluation. Food Chem. 2022;398:133925. [CrossRef]
15. Mai TH, Tran TT, Le VV. Protection of antioxidants in pitaya (Hylocereus undatus) peel:Effects of blanching conditions on polyphenoloxidase, peroxidase and antioxidant activities. Food Sci Technol. 2022;42:e112921. [CrossRef]
16. Magangana TP, Makunga NP, Grange C, Stander MA, Fawole OA, Opara UL. Blanching pre-treatment promotes high yields, bioactive compounds, antioxidants, enzyme inactivation and antibacterial activity of 'wonderful'pomegranate peel extracts at three different harvest maturities. Antioxidants (Basel). 2021;10(4):1119. [CrossRef]
17. Golan A, Kahn V, Sadovski AY. Relationship between polyphenols and browning in avocado mesocarp. Comparison between the fuerte and lerman cultivars. J Agric Food Chem. 1977;25(6):1253-60. [CrossRef]
18. Flick GJ Jr., Ory RL, St. Angelo AJ. Comparison of nutrient composition and of enzyme activity in purple, green, and white eggplants. J Agric Food Chem. 1977;25(1):1972-5. [CrossRef]
19. Noh HR, Kang JY, Kim BG. Biosynthesis of trifolin, a bioactive flavonoid by biotransformation. J Appl Biol Chem. 2021;64(3):309-16. [CrossRef]
20. Bobková A, Hudácek M, Jakabová S, Belej L, Capcarová M, Curlej J, et al. The effect of roasting on the total polyphenols and antioxidant activity of coffee. J Environ Sci Health Part B. 2020;1234:495-500. [CrossRef]
21. Brand-Williams W, Cuvelier ME, Berset C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci Technol. 1995;30(28):25-30. [CrossRef]
22. Araji S, Grammer TA, Gertzen R, Anderson SD, Mikulic-Petkovsek M, Veberic R, et al. Novel Roles for the Polyphenol Oxidase Enzyme in Secondary Metabolism and the Regulation of Cell Death in Walnut. Plant Physiol. 2014;164:1191-203. [CrossRef]
23. Liu L, Cao SQ, Qi XY, Yang ZF. The effect of pH on the activity, thermokinetics and inhibition of polyphenol oxidase from peach. J Food Sci Technol. 2015;52(11):7465-71. [CrossRef]
24. Moon KM, Bin Kwon E, Lee B, Kim CY. Recent trends in controlling the enzymatic browning of fruit and vegetable products. Molecules. 2020;25(12):2754. [CrossRef]
25. Lv Y, Cai L, Yang M, Liu X, Hui N, Li J. Purification, characterisation, and thermal denaturation of polyphenoloxidase from prawns (Penaeus vannamei). Int J Food Prop. 2018;20(3):S3345-59. [CrossRef]
26. De Oliveira FK, Santos LO, Buffon JG. Mechanism of action, sources, and application of peroxidases. Food Res Int. 2021;143:110266. [CrossRef]
27. Sarika D, Kumar PS, Arshad S, Sukumaran MK. Purification and evaluation of horseradish peroxidase activity. Int J Curr Microbiol Appl Sci. 2015;4(7):367-75. https://www.ijcmas.com/vol-4-7/D.%20Sarika,%20et%20al.pdf
28. Tanwar B, Modgil R. Flavonoids:Dietary occurrence and health benefits. Spat DD. 2012;2(1):59. [CrossRef]
29. Setyaningsih W, Saputro IE, Palma M, Barroso CG. Stability of 40 phenolic compounds during ultrasound-assisted extractions (UAE). AIP Conf Proc. 2016;1755:080009. [CrossRef]
30. Jabbari M, Khosravi N, Feizabadi M, Ajloo D. Solubility temperature and solvent dependence and preferential solvation of citrus flavonoid naringin in aqueous DMSO mixtures:An experimental and molecular dynamics simulation study. RSC Adv. 2017;7(24):14776-89. [CrossRef]
31. Dong X, Li X, Ruan X, Kong L, Wang N, Gao W, et al. Adeep insight into the structure-solubility relationship and molecular interaction mechanism of diverse flavonoids in molecular solvents, ionic liquids, and molecular solvent/ionic liquid mixtures. J Mol Liq. 2022;385:122359. [CrossRef]
32. Nwanak S. Impact of temperature on the solubility of ionic compounds in water in cameroon. J Chem. 2024;3(2):42-51. [CrossRef]
33. Kiliç C, Can Z, Yilmaz A, Yildiz S, Turna H. Antioxidant properties of some herbal teas (green tea, senna, corn silk, rosemary) brewed at different temperatures. Int J Second Metab. 2017;4(3):148-54. [CrossRef]
34. Croho M, Bouymajane A, Oulad Y, Majdoub E, Cacciola F, Mondello L, et al. Phenolic composition, antioxidant and antibacterial activities of extract from flowers of rosa damascena from morocco. Separations. 2022;9(9):247. [CrossRef]
35. Kim AN, Lee KY, Rahman MS, Kim HJ, Chun J, Heo HJ, et al. Effect of water blanching on phenolic compounds, antioxidant activities, enzyme inactivation, microbial reduction, and surface structure of samnamul (Aruncus dioicus var Kamtschaticus). Int J Food Sci Technol. 2020;55(4):1754-62. [CrossRef]
36. Kiptiyah SY, Harmayani E, Santoso U, Supriyadi S. The effect of blanching and extraction method on total phenolic content, total flavonoid content and antioxidant activity of Kencur (Kaempferia galanga. L) extract. IOP Conf Ser Earth Environ Sci. 2021;709(1):012025. [CrossRef]
37. Meena SK, Agrawal M. Impact of blanching and drying on antioxidant contents and antioxidant activity of Indian mustard leaves (Brassica juncea) Sumitra. J Oilseed Brassica. 2017;8:158-62. https://epubs.icar.org.in/index.php/JOB/article/view/158832
38. Munu PS, Puspita G, Supu RD. The effect of solvent type on flavonoid levels of moringa leaf extract (Moringa oleifera L.). J Heal Technol Sci. 2023;4(4):45-55. https://journals.ubmg.ac.id/index.php/JHTS/article/download/1619/702/
39. Lee TH, Lee CH, Ong PY, Wong SL, Hamdan N, Akob HY, et al. Comparison of extraction methods of phytochemical compounds from white flower variety of Melastoma malabathricum. South African JBot. 2022;148:170-9. [CrossRef]
40. Loypimai P, Moongngarm A, Chottanom P. Thermal and pH degradation kinetics of anthocyanins in natural food colorant prepared from black rice bran. J Food Sci Technol. 2016;53:461-70. [CrossRef]
41. Fokuo MK, Aggrey WN, Ama M, Rockson D. Tannin-based deflocculants in high temperature high pressure wells: A comprehensive review. Adv Chem Eng Sci. 2021;11:263-89. [CrossRef]
42. Go E, Ryu BR, Ryu SJ, Kim HB, Lee HT, Kwon JW, et al. An enhanced water solubility and stability of anthocyanins in mulberry processed with hot melt extrusion. Int J Mol Sci. 2021;22:12377. [CrossRef]
43. Xue H, Zhao J, Wang Y, Shi Z, Xie K, Liao X, et al. Food chemistry: X factors affecting the stability of anthocyanins and strategies for improving their stability:A review. Food Chem X. 2024;24:101883. [CrossRef]