1. INTRODUCTION
The genus Curcuma, belonging to Zingiberaceae family, order Scitaminae is native to tropical South-East Asia. Curcuma, vernacularly known as turmeric, cultivated for its underground rhizome, is used as spice, condiment, dye in cosmetics and drug industry in the preparation of anti-cancer medicines, textile, food, and confectionary industries. It forms an important adjuvant in Indian cuisine as it imparts color and aromatic flavor to various dishes. It is also used as herbal medicine “Amraharidra”, which gives a cooling, aromatic effect and promotes digestion. Turmeric has long been used in India for the treatment of sprains and inflammatory conditions. The turmeric rhizome contains the major pungent aromatic flavor “curcumin” responsible for the rhizome color [1]. Given the empirical usage of turmeric, out of 42 different species of Curcuma, few known species indicative of economic significance are tabulated as described in Table 1 [2]. Curcuma is enriched with macromolecules such as starch, minerals, cellulose, gum, volatile oils, and phenolic compounds like curcuminoids. In addition, curcuminoids being the principal ingredient of dried Curcuma with multiple medicinal properties has been exploited in nutraceutical and pharmaceutical industries. These are chemically related to the main component curcumin and are made up of diarylheptanoic nucleus with different oxidation and unsaturation properties, of which curcumin (CUR), dimethoxy curcumin (DEM), and bis-DEM make up the major rhizome constituents, comprehensively each distinct compounds having role in the ailment of human health.
Table 1: Economically important species of Curcuma [2].
| Species | Use |
|---|---|
| Curcuma longa L. syn Curcuma domestica Val. | Spice, medicine, dye, religious, local delicacies, insect repellent, aroma therapy and perfume |
| Curcuma amada Roxb., Curcuma mangga Val. and Zijp. | Spice, medicine, pickles and salads |
| Curcuma zedoaria Roxb. | Folk medicine, arrow root industry |
| Curcuma ochrorrhiza Val. and Van Zijp | Malayan traditional medicine |
| Curcuma pierreana Gagnep. | Vietnamese traditional medicine |
| Curcuma aromatica Salsb | Medicine, toiletry articles, insect repellent |
| Curcuma kwangsiensis S. G. Lec and C. F. Liang syn Curcuma chuanyujin, Curcuma phaeocaulis Val. | Chinese traditional medicine |
| Curcuma caesia Roxb. | Spice and medicine |
| Curcuma comosa Roxb. | Traditional medicine of Thailand |
| Curcuma angustifolia Roxb., Curcuma zedoaria Roxb. Curcuma caulina F. Grah., Curcuma psuedomontana F. Grah. Curcuma montana Roxb., Curcuma rubescens Roxb., Curcuma leucorrhiza, Curcuma xanthorrhiza Roxb., Curcuma decipiens Dalz., Curcuma malabarica Vel. et al., Curcuma raktakanta Mangaly and Sabu, Curcuma haritha Mangaly and Sabu, Curcuma aeruginosa Roxb. | Arrow root industry |
| Curcuma alismatifolia Gagnep, Curcuma thorelii, Curcuma roscoeana Wall. | Ornamental |
Curcumin, a well-known bioactive secondary metabolite, has been used in the treatment of Alzheimer’s disease [3]. The bioactivity of curcumin is well utilized in the treatment of liver diseases, eye diseases, and cancer, and displays anti-HIV, nematocidal, and mosquitocidal functions [4] [Figure 1]. The majority of medicinal plants harvested from wild populations (approximately 90%) cause the native populations to decline, eroding the genetic diversity. Commercial production of medicinal plants requires adequate standardization through agricultural practices. The traditional methods of crop multiplication followed for domestication require several years to harness the compounds with medicinal value. In contrast to traditional propagation techniques, biotechnology plays a significant role in medicinal plant improvement programs which was made possible due to plant tissue culture, which allows the rapid generation of genetically identical plants. In bioreactors, the production of the whole plant and organ cultures is more challenging to commercialize in comparison to cell and hairy root culture systems. Thus, tissue culture applications for the production of entire plants and organ cultures of medicinal plants have garnered little attention. This review will focus on Curcuma taxonomy, distribution, and plant and rhizome culture for the production of starter plants and the production of biomass that harbor biologically active secondary metabolites using various in vitro tissue culture techniques.
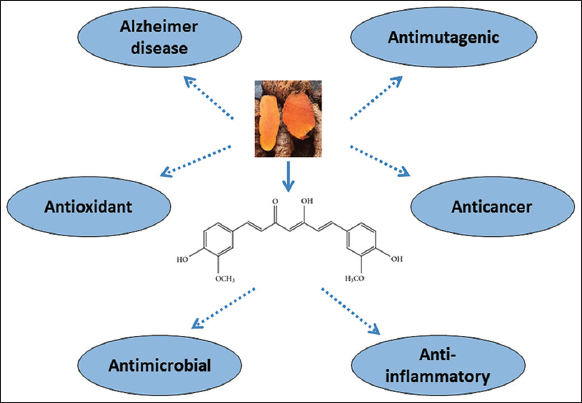 | Figure 1: Representation of biological activities of curcumin. [Click here to view] |
2. TAXONOMY
Curcuma is a monocotyledonous triploid (2n = 3× = 63) plant with functional sterility. As no sexually derived seeds are formed in this crop, rhizomes are the sole source of planting material. The pseudostem is tall and robust with narrowed oblonga-elliptical leaves at the base. Plant height is usually up to 1 m, with 8–10 leaves, an apical spike (length ranges between 10–18 cm) with pale yellow flowers, and starting root tubers are produced at the end of fibrous roots [Figure 2]. Rhizomes are larger in size and contain more curcumin, but only a moderate amount of volatile oil. The rhizomes formed in clusters underground are separated and used as planting material. Species belonging to the genus Curcuma, namely, Curcuma caesia, Curcuma rubescens, Curcuma amada, Curcuma leucorrhiza, Curcuma montana, Curcuma aromatica, Curcuma zedoaria, Curcuma angustifolia, Curcuma pseudomontana, and Curcuma longa are of high medicinal value [5] [Figure 3]. They are further differentiated by the color of their rhizomes, which appear as bluish-green in C. aeruginosa, bright yellow in C. aromatica, greyish- blue in C. caesia, and orange-yellow in C. longa. Over a period of time, accumulated somatic mutations have contributed to present-day heterogeneity. Heterozygosis observed in each one of the landraces has resulted in genetically different heterozygous populations of crop production unevenness. Furthermore, the prevalence of soil-borne diseases such as rhizome rot and foliar diseases has harmed rhizome quality and quantity, indicating the disheartened state of the Curcuma growers.
 | Figure 2: Important Curcuma species. (a) Curcuma longa; (b) Curcuma caesia; (c) Rhizome of C. longa; (d) Rhizome of C. caesia. [Click here to view] |
 | Figure 3: Rhizomes of Curcuma species. (a) C. longa; (b) C. Caesia; (c) C. aeruginosa; (d) C. zedoaria. [Click here to view] |
3. DISTRIBUTION
The genus Curcuma is Indo-Malayan in distribution and includes about 100 rhizomatous herb species. However, only a few of them are commercially important. C. longa L., alone contributes to 90% of commercial turmeric production and is extensively cultivated. Turmeric which originated in India, has spread throughout Far East and Polynesian Islands, South-east Asia [1]. Further, plants can be found throughout Asia’s tropics, extending from India to South China, South-east Asia, Papua New Guinea, and Northern Australia. Indian states producing the most turmeric include Maharashtra, Andhra Pradesh, Karnataka, Tamil Nadu, Kerala, Gujarat, Mizoram, Meghalaya, and Assam. Curcuma grown in Mizoram, Meghalaya, and Assam contain elevated curcumin accumulation in rhizomes due to vast genetic variations and geoclimatic conditions [6]. India has been ranked first in terms of turmeric exports, earning Rs. 385 crores in foreign revenue annually, which has encouraged the Curcuma cultivators to widen the turmeric cultivation by applying the latest technology through public domains.
4. PROPAGATION OF CURCUMA
Conventionally, the crop is raised by rhizomes in deep, fertile, and well-drained loamy soil with a pH range of 5.0–7.5. The crop prefers a warm and humid climate with well-distributed annual rainfall. Whole or split mother rhizomes, or “finger” rhizomes, are used for planting. Healthy rhizomes are selected at the time of planting and stored under the pit for the upcoming year’s planting. Mother rhizomes will commonly be treated with systemic fungicides along with drenching to avoid rhizome rot and other bacterial infections. Before the advent of present-day advanced breeding technologies, like marker-assisted plant breeding and bioinformatics-based tools, the selection criteria for high-value bioactive components and medicinally important aromatic oils dominated the production of local variants and conventionally bred cultivars. Furthermore, the process of harnessing these medicinally useful substances produced by Curcuma plants for their own defensive or protective uses was done in a traditional manner that required hundreds of years. Because sterility is the most significant limiting factor in genetic improvement programs, alternative approaches have been applied in a relatively exponential manner which could serve as a far-reaching move in increasing Curcuma productivity. Improvement and development of medicinally important crop plants predominantly follow biotechnology strategies for rapid plant-based medicine production. Tissue culture is one such biotechnology approach that offers a plethora of plant development potential and is an essential tool for identifying beneficial medicinal plant variants.
The technique involves growing tissues in vitro under aseptic conditions, mimicking the in vivo ambience environment, to facilitate growth and development without compromising the plant’s structural and functional integrity. The employment of tissue culture techniques for plant regeneration is a need of concern in the agriculture and horticulture fields based on commercial and exchange value of intended plant type. Approaches such as clonal selection, mutation breeding, and polyploidy induction-derived progenies were proposed as potential solutions for the production of stock plants for tissue culture-based genetic improvement programs. Due to their high commercial value, numerous in vitro regeneration strategies have been optimized with nutritional sources as well as plant growth regulators and other environmental factors, which are conducive for cellular growth and development. The rapidity with which tissue culture facilities have been established is due to the advancement and standardization of in vitro regeneration protocols for selective plant types, which has resulted in successful plant multiplication, disease-free plant types, high-value metabolites, and ex vivo germplasm conservation, indicating its role in reducing the burden on the farming community [7]. Large-scale propagation approaches coupled with in vitro regeneration under optimized conditions for Curcuma were further beneficial to the comprehensive improvement of this crop. In turn, all these efforts have proven to be beneficial for the production of therapeutic compounds and secondary metabolites.
4.1. Micropropagation
Micropropagation is the in vitro process of regenerating a whole plant using totipotent or pluripotent and self-renewal states of the cell, tissues, and organs by mimicking the favorable environmental conditions of in vivo conditions. Tissues and organ explants are cultured in a closed vessel using prescribed culture media under controlled and aseptic conditions. Despite the use of various explants, rhizome buds have been found to be very efficient material in Curcuma micropropagation. Several plant regeneration studies were successfully attempted in Curcuma through direct and indirect organogenesis using leaf sheath [8], leaf base [9], rhizome buds [10], leaf petiole [11], and through callus induction using apical and lateral buds [12] explants [Figure 4]. These results indicate that irrespective of the species studied, various explants cultured in MS medium supplemented with BAP and with or without other phytohormones or vitamins favored callus induction and multiple shoot production. Recently, an efficient and improved tissue culture approach was used for direct and callus-mediated regeneration of C. karnatakensis [13]. The summary of Curcuma micropropagation is represented in Table 2.
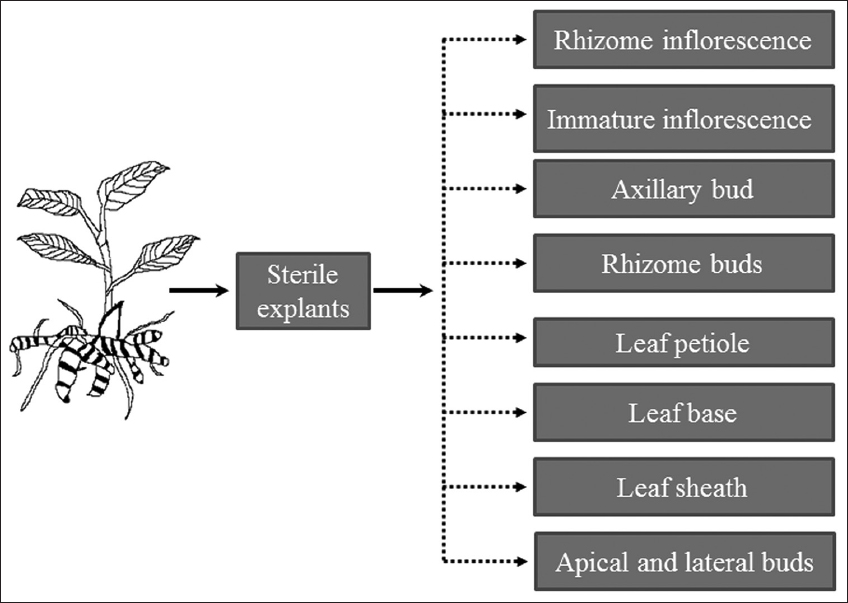 | Figure 4: Explants from Curcuma plant used in micropropagation study. [Click here to view] |
Table 2: Summary of micropropagation research in Curcuma spp.
| Species | Explant | Medium | Findings | References |
|---|---|---|---|---|
| Curcuma longa | Vegetative rhizome buds | MS+BAP, Kinetin Riboside, Zeatin, DMAP+Adenine+Adenine Sulphate+Metatopolin (10 µM)+ IAA (1 µM) | Shoot multiplication more efficient in liquid medium | [14] |
| Curcuma amada | Leaf sheath explants | MS+2.0 mg/L 2,4-D+0.5 mg/L BAP+3% sucrose | Embryonic callus induction | [8] |
| Curcuma longa | Leaf base explants | MS+4.49 µM 2,4-D+1.32, 2.20+2.64 µM BAP. | Plant regeneration from somatic embryos | [9] |
| Curcuma angustifolia | Axillary bud explants | MS+4.4–22.2 µM BAP+2.9–5.7 µM IAA+2.3–23.2 µM Kn+2.7–5.4 µM NAA+67.8-271.5 µM Adenine Sulphate | Multiple shoot production | [28] |
| Curcuma caesia | Rhizome buds | MS+3.0 µM BA+0.5 µM NAA | Shoot production | [35] |
| Curcuma longa | Leaf, petiole, stem, and root explants embryogenic cell-suspension derived from roots | Schenk and Hildebrandt (SH)+ 0.5 mg/L TDZ+various concentrations of BAP | Shoot and root production | [11] |
| Curcuma zedoaria | Axillary bud | MS+BAP (2.2–22.2 µM)+ Kn (2.3–23.2 µM)+ IAA (2.9–11.4 µM)+ NAA (2.7–10.2 µ NAA)+ Adenine Sulphate (33.9–203.6 µ) | Shoot and root production | [10] |
| Curcuma longa | Leaf sheath, leaf lamina, leaf base, apical buds, and lateral buds | MS+3 mg/L 2,4-D+0.8 mg/mL Kn | Callus induction | [12] |
| Curcuma caesia | Shoot buds | MS+B5+15 µ of BAP d 6 µ of IBA | Shoot and root production | [36] |
4.2. Microrhizome Production
Micro-rhizomes produced for germplasm conservation are also very convenient for packing and transportation, thus attracting the attention of researchers for improvements in Curcuma biotechnology. A significant degree of achievement had been documented with the cultural modification technique under in vitro conditions, wherein increased levels of sucrose in liquid media revealed a corresponding elevation in the induction of rhizome size. Furthermore, percent survival rate and physiological acclimatization of the micro-rhizome during the field hardening process had significantly improved [14]. Based on the above cues, it could be inferred that, with a modification in the cultural condition along with physiological parameters, in vitro production of micro-rhizomes could be well achieved. Several studies have shown that microrhizomes in Curcuma species such as C. longa and C. caesia can be induced by sucrose concentrations as low as 5% [15], 6% [16], 9% [17]. The microrhizome production in Curcuma species is depicted in Table 3.
Table 3: Summary of microrhizome production in Curcuma spp.
| Species | Explant | Media | Results | References |
|---|---|---|---|---|
| Curcuma aromatica | Plantlets | MS+5 mg/L BAP+60 g/L sucrose and | 8 h photoperiod microrhizome | [37] |
| Curcuma longa | Plantlets | MS+BAP (0, 0.32, 1 µM) + methyl jasmonate (MeJa) (0, 5 and 16 µM) + 6% sucrose | Microrhizome induction | [16] |
| Curcuma longa | Plantlets | Liquid MS + (NH4) 2SO4 (3 µM) + 5 g/L sucrose+BAP (3 µM) | Microrhizomes produced in high concentrations of sucrose in liquid medium. | [15] |
| Curcuma caesia | plantlets | BAP (1.0 mg/l) + 9 g/L sucrose | Microrhizome induction was observed at16 h photoperiod | [17] |
5. LIQUID CULTURE MEDIUM
Micropropagation of plants using a liquid culture medium is more favorable, advantageous, and efficient for large-scale production than a semi-solid culture medium. Liquid culture media allows for the modification of replenishment technique, making it easier for the immersed explants to acquire water, nutrients, and growth regulators efficiently. Given conditions described above, it is possible that it enhanced the cultural growth and development, somatic embryogenesis, regenerative capacity, shoot, and root proliferation rates, ultimately increasing the microrhizomes in zinger and turmeric [14]. Furthermore, the constant contact of the tissues with the liquid culture medium facilitates the uptake of nutrients and phytohormones, resulting in uniform shoot production and rapid growth with increased biomass (fresh and dry weight) yield. Micropropagation in liquid culture medium facilitates for greater water availability allowing for large nutrients concentrations that can be replenished as needed. Plant regeneration under in vitro conditions is a well-orchestrated, sequential, and process where the plant tissues are placed over the liquid media to be in direct contact, leading to large-scale automated bioreactor contained tissue culture system coupled with a shaking liquid culture facility.
Since the system is automated, it eases handling and operations and reduces human interference in the bioreactor production strategy, thereby overcoming a greater degree of contamination. Through continual shaking of the liquid medium, the aeration mechanism in the bioreactors facilitates higher levels of plant development and multiplication rates of shoots. Shoots grown in vitro are more directly influenced by the liquid medium’s ingredients, such as the concentration of growth hormones, inorganic ions, and sugar, than those grown on agar medium. Culture media could be easily replenished by fresh media by draining out the used-up media in sophisticated ways in liquid culture systems. Thus, liquid culture systems could overcome the limitation of high production cost at the industrial level by minimizing the time and number of subcultures with reduced labor requirements as compared to the conventional micropropagation method.
5.1. Bioreactors
Bioreactors are vessel systems that enable the growth of cells through constant monitoring and manipulation without compromising asepsis. Micropropagation and direct phytochemical synthesis have both been established using bioreactor systems that generate an enormous amount of aeration when a liquid culture medium is used. Bioreactors are commonly used to cultivate organisms to create cells or metabolites and can also be applicable to plant regeneration using the shake culture technique for micropropagation. Bioreactors designed for in vitro culturing range in size from 0.5 to 500 liters and can be perforated in one specifically designed liquid culture or a refined, miniaturized, air-compressed, and steam-based microbial fermenter. To manufacture bioactive compounds, large amount of plant material is required, and this can be achieved by either employing large containers with liquid medium or by adding additional starting plant material during the culture cycle in a fed-batch manner [15]. Bioreactor micropropagation allows culturing of plant tissues such as plantlets and or organs such as microtubers and bulblets in liquid media under constant aeration, resulting in propagule establishment and multiplication.
The following are the major advantages of employing micropropagation in a bioreactor, (i) the effortless generation and scaling-up of plantlets in large numbers in a short period of time, (ii) culture handling procedures such as inoculation and harvesting is simple with a reduced cost of labor, (iii) explants remain in close vicinity in liquid cultures, allowing for rapid nutrient uptake and regeneration, (iv) increased growth rate and multiplication due to sufficient oxygen supply from forced aeration, and (v) culture movement in the bioreactors results in the loss of apical dominance and stimulates the fast multiplication of shoots and plantlets. Explants in the bioreactors can be cultured under continuously submerged conditions or under partially or temporarily immersed conditions in a liquid medium. Numerous types of bioreactors are employed in plant biotechnology industries, however, only a few bioreactor types possess a light conditioning control unit for culturing explants. Bioreactors used for in vitro propagation purposes can be operated in an agitated or non-agitated manner by mechanical or pneumatic means or can be of the temporarily immersed type. The utility of a bioreactor is determined by its cost-effectiveness and ease of operation, as well as its ability to maintain culture quality. Bioreactor culture systems have shown the feasibility of growing bulblets in liquid media, as well as explore the parameters that influence the formation and enlargement of bulblets when creating a bioreactor culture system for the production of Lilium [18].
5.2. Temporary Immersion System (TIS)
Propagation of plant and organ cultures can be achieved well in a temporary or partial immersion systems as there is less mechanical shearing and good oxygen supply as compared to stirred bioreactors. Temporary immersion culture is used for three main reasons: Delivery and nutrient transfer to the plants, thereby preserving correct oxygenation, maintaining low levels of mechanical shearing and hydrodynamic force, and cost-effectiveness. TIS has improved the gaseous and solute transport properties of several crop species, allowing for the production of large, and numerous plants with increased dry weight. Many TIS system casts are designed to accommodate plant nutrient constituents with hyperhydricity while incurring costs. The TIS system devised could be effective in semi-automated in vitro propagation by predominantly controlling contamination, optimal delivery of oxygen, nutrient concentration, and proper blending of nutrient sources. Additionally, it enables an adequate relative humidity level with complete propagule and liquid medium separation, reduces the frequency of subculturing, and has the advantage of flexibility in changing the medium with minimal shearing damage. Among various forms of mechanically devised TIS systems, RITA (automated temporary immersion recipient) is found to be a standard of reference that consists of two compartments [19]. The liquid culture medium is kept in the bottom compartment, while the explants are placed in the upper compartment. The upper chamber and lower chamber are connected with each other, and the medium is pumped into the upper chamber by exerting extra pressure on the lower chamber. When the overpressure reduces, the liquid medium flows from the upper into the lower chamber.
The TIS system applied for scaled-up process has achieved a maximal rate of propagule generation in banana, pineapple, and coffee plants than with semi-solid media; however, minimal differences were noticed due to variation in the scale-up process of the TIS system. In comparison to agar-derived plantlets, TIS system-based plantlets have demonstrated greater photosynthetically active growth capability, eventually yielding higher biomass when transferred to the soil at a lower cost. In vitro shoot proliferation was reported in wild lingonberry and clones of Vaccinium vitis-idaea ssp. in submerged and TIS added with liquid medium showing 2–3 times lower shoot proliferation on semi-solid medium [20]. When grown in TIS along with a shake flask method, C. zedoaria and Zingiber zerumbet produced significantly more shoots than agar-gelled cultivated cultures [21]. The recent upsurge in TIS technology from the previous studies by scaling up the process with the deployment of reactor coupled conditions has benefited to a greater extent in achieving shoot culture in a wide variety of commercially important plants. The micropropagation of a wide range of plants has been achieved through shoot cultures using TIS reactors [19,22]. Recently, an efficient in vitro mass propagation of C. longa was demonstrated using a TIS system with different LED spectra [23].
5.3. Rocker System
The bioreactor coupled rocker system for plant explant cultures, especially plant organ culture (POC) has gained a lot of insight into plant tissue culture. POC is a well-organized structure with individual rectangular culture boxes made of polycarbonate with diffusive ventilation and a rocker platform built for a thin-film liquid rocker system suitable for monocotyledonous species culturing. Simple TIS, known as “rocker systems,” is used to aerate plantlets without the requirement for direct connections between mechanical parts such as the motor and vessels. One such device is the “Liquid Lab Rocker,” which occasionally moistens plant tissues by employing the slow-pitched motion of big rectangular containers on an articulated shelf [16]. A greater degree of success with multiplication rate, higher biomass, and deployment of high sugar content has been achieved in Alocasia, Colocasia, Hemerocallis, Hosta, and Miscanthus x giganteus monocotyledonous crops grown under a rocker system on a thin film liquid medium as compared to semi-solid agar medium [24]. The rocker technique has been tried and successfully accomplished in different plant species for propagation, such as micro-rhizomes in C. longa [16] and microtubers of Solanum tuberosum [25].
6. CLONAL FIDELITY STUDIES
In medicinal plants, significant intraspecific variation is frequently observed in relation to a variety of complex traits, including their phytochemical profile. True-to-type propagules and genetic stability are crucial aspects in commercial micropropagation, which results in physiological and somaclonal heterogeneity because in vitro generated plant cells, tissues, organs, and undifferentiated calli are linked with genetic alterations throughout the process.The variation occurring in micropropagated plants is influenced by the source of the tissue or plant regeneration system. Somaclonal variations are both heritable (genetic) and non-heritable (epigenetic), and these phenomena are primarily influenced by factors including genotype, the availability of chimeral tissue, type, and origin of explants, type of medium, the concentration of plant growth regulators, duration, and environment of the culture such as light and temperature. These factors may limit the broad applicability of micropropagation systems. Although somaclonal variation is beneficial for selecting new genotypes, it is undesirable for the preservation of germplasm, necessitating the multiplication of such elite genotypes to preserve distinct genotypes. Evaluation of genetic diversity and purity is critical for developing appropriate conservation programs. The genetic homogeneity of in vitro propagated plants could be validated at various levels, namely, genetic, morphological, physiological, and biochemical analysis.
DNA-based markers are promising tools for validating the clonal fidelity of micropropagated plants as they are unaffected by environmental factors. PCR-based DNA markers such as arbitrary (RAPD), semi-arbitrary (Inter-Simple Sequence Repeat, ISSR; Amplified Fragment Length Polymorphism, and AFLP), and sequence-based (Simple Sequence Repeat, SSR) techniques are used in genetic fidelity studies [26]. RAPD markers are frequently employed for genetic affinity assessment as they have the ability to amplify many genomic locations enabling improved analysis. No evidence of genetic variation established in the callus-derived plants with the parental plant was reported when RAPD and inter-simple sequence repeat (ISSR) analysis methods were adopted, indicating that these protocols could be applied for mass multiplication and true-to-type propagation of superior clones in C. caesia [27]. In a far-reaching move, ISSR technology is being used for ascertaining genetic integrity in several regenerated plantlets cultured in vitro belonging to Zingiberaceae family, such as Alpinia calcarata [28]. Subsequently, corroborative studies have also revealed similar inheritance patterns with ISSR markers in the species of C. angustifolia and C. zedoaria which displayed a lack of genetic variation [29,30]. Conservation of shoot primordia through synthetic seed and RAPD fingerprinting of in vitro generated C. amada reported that randomly chosen synthetic seed-derived plantlets maintained 84.62% of their genetic makeup [30].
Chemical fidelity investigations are also attempted in Curcuma, in addition to DNA-based genetic fidelity research based on essential oil content. Essential oils also known as Curcuma oil are produced during oleoresin processing as a byproduct of curcumin extraction from fresh or dry rhizomes of Curcuma species. These essential oils indicate the chemical profile which varies due to genotype and geographical differences [31]. The chemical components analyzed by GC-MS (Gas chromatography-Mass spectroscopy) displayed homogeneity of essential oil extracted from leaf and rhizome from in vitro derived C. longa plants that were found to be higher than conventional field grown plants [32]. Similarly, the essential oil of in vitro propagated C. amada plants possessed the same GC-MS pattern suggesting biochemical fidelity when compared to their mother plants [33]. Chemical uniformity of in vitro regenerated and conventionally raised C. angustifolia plantlets analyzed through high performance thin layer chromatography (HPTLC) had similarity in metabolic profiles when compared [29]. Alpinia calcarata, a member of Zingiberaceae family, revealed identical qualitative and quantitative chemical profiles in different parts (leaves, rhizome, and roots) of in vitro developed and conventional field-grown plants [34].
7. ADVANTAGES OF MICROPROPAGATION
Micropropagation has several advantages over traditional methods of propagation, including a rapid rate of multiplication and the production of disease-free plants. Therefore, elite genotypes of Curcuma species highly susceptible to various diseases can be produced using in vitro micropropagation techniques. Plant micropropagation under in vitro conditions can be mainly approached through direct organogenesis or by callus formation. Callus cultures established in vitro from explants could be used to induce organs such as shoots, roots, and storage organ structures such as bulbs, tubers, microrhizomes, and flowers through the de-differentiation process. Another method of producing in vitro plants is by culturing of apical meristematic tissue originating from auxiliary buds as explants. In vitro-cultured plants have the potential to eliminate harmful pathogens that may have been present in the donor plant.
In addition, meristematic cells maintain plant growth and give rise to different plant organs. Another advantage of this technique is that organs are formed directly from meristems, ensuring genetic stability and minimizing somaclonal variation due to the prevention of callus formation and adventitious organogenesis. Somatic embryogenesis is another method applied in micropropagation, that produces an embryo from a single cell, leading to complete plant development. Since it employs the embryogenic pathway in vitro, this method can be a rapid and efficient means to propagate at a large scale with a reduction in multiplication time. The micropropagation of Curcuma by in vitro induced microrhizomes is considered an ideal method for the generation of disease-free plantlets for propagation and also for plant conservation and the exchange of germplasm [37]. This method may be sufficient for the production of genetically viable planting material because it requires a small number of growth regulators and fewer subculture cycles.
8. CONCLUSION
The in vitro propagation of Curcuma plants is achieved through direct organogenesis using auxiliary buds and rhizome sprouts and indirect organogenesis through callus induction using various rhizome explants. Plant growth and proliferation rates are found to be high in liquid systems than those obtained in solid media. Furthermore, increased levels of sucrose containing liquid media had resulted in significant increase in the micro-rhizome production over solid media. The bioreactor-based in vitro propagation method using liquid culture media offers economic feasibility with respect to efficacy, cost, a span of time, and space. Nevertheless, some degree of hyperhydricity (vitrification) is still of concern, which can be rectified by keen vigilance of the explants with liquid culture corrections followed by stabilizing optimal conditions of immersion time in TIS. Molecular markers have been proven to be efficient for assessing the micropropagated plants for clonal fidelity. Further, another way to reduce the risk of somaclonal variation is to use fewer growth hormones in the culture media, regular subculturing, and selection of suitable genotypes. Overall, this review summarized the potential benefits of using a liquid culture system for mass propagation and microrhizome production.
9. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
10. FUNDING
There is no funding to this report.
11. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
12. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
13. DATA AVAILABILITY
All data generated and analyzed are included within this research article.
14. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Sasikumar B. Genetic resources of
2. Velayudhan KC, Muralidharan VK, Amalraj VA, Gautam PL, Mandal S, Kumar D.
3. Goozee KG, Shah TM, Sohrabi HR, Rainey-Smith SR, Brown B, Verdile G,
4. Sharifi-Rad J, Rayess YE, Rizk AA, Sadaka C, Zgheib R, Zam W,
5. Pemba HB, Sharangi AB. Promising
6. Singh S, Joshi RK, Nayak S. Identification of elite genotypes of turmeric through agroclimatic zone based evaluation of important drug yielding traits. Indian Crop Prod 2013;343:165-71. [CrossRef]
7. Debnath SC, Usha A.
8. Raju CS, Kathiravan K, Aslam A, Shajahan A. An efficient regeneration system via somatic embryogenesis in mango ginger (
9. Raju CS, Aslam A, Shajahan A. High-efficiency direct somatic embryogenesis and plant regeneration from leaf base explants of turmeric (
10. Jena S, Ray A, Sahoo A, Sahoo S, Dash B, Kar B,
11. Jie EY, Ahn MS, Lee J, Cheon YI, Kim CY, Kim SW. Establishment of a high-frequency plant regeneration system from rhizome-derived embryogenic cell-suspension cultures of
12. Gurav SS, Gurav N, Patil AT, Duragkar NJ. Effect of explant source, culture media, and growth regulators on callogenesis and expression of secondary metabolites of
13. Shanthala AA, Dilkalal A, Umesh TG. An efficient
14. Salvi ND, George L, Eapen S. Micropropagation and field evaluation of micropropagated plants of turmeric. Plant Cell Tiss Org Cult 2002;68:143-51. [CrossRef]
15. El-Hawaz RF, Bridges WC, Adelberg JW.
16. Cousins MM, Adelberg JW. Short-term and long-term time course studies of turmeric (
17. Sarma I, Deka AC, Sarma TC. A protocol for rapid clonal propagation and micro-rhizome production of
18. Lian ML, Piao XC, Park SY. Mass production of
19. Georgiev V, Schumann A, Pavlov A, Bley T. Temporary immersion systems in plant biotechnology. Eng Life Sci 2014;14:607-21. [CrossRef]
20. Arigundam U, Variyath AM, Yaw LS, Marshall D, Debnath SC. Liquid culture for efficient
21. Stanly C, Bhatt A, Keng CL. A comparative study of
22. Watt MP. The status of temporary immersion system (TIS) technology for plant micropropagation. Afr J Biotechnol 2012;11:14025-35. [CrossRef]
23. Marchant MJ, Molina P, Montecinos M, Guzmán L, Balada C, Fassio C,
24. Adelberg JW. Micropropagation in Liquid Culture using Partial Immersion Systems. In:XXIX International Horticultural Congress on Horticulture:Sustaining Lives, Livelihoods and Landscapes (IHC2014). Vol. 1113;2014. 35-46. [CrossRef]
25. Kämäräinen-Karppinen T, Virtanen E, Rokka VM, PirttiläAM. Novel bioreactor technology for mass propagation of potato microtubers. Plant Cell Tiss Org Cult 2010;101:245-9. [CrossRef]
26. Singh S, Dalal S, Singh R, Dhawan AK, Kalia R. Evaluation of genetic fidelity of
27. Jose S, Thomas TD. High-frequency callus organogenesis, large-scale cultivation and assessment of clonal fidelity of regenerated plants of
28. Das BS, Basu A, Sahoo L. Direct shoot organogenesis from rhizomes of medicinal
29. Jena S, Ray A, Sahoo A, Sahoo S, Kar B, Panda PC,
30. Banerjee S, Singh S, Pandey H, Pandey P, Rahman L. Conservation and storage of
31. Dosoky NS, Setzer WN. Chemical composition and biological activities of essential oils of
32. Kar B, Kuanar A, Singh S, Mohanty S, Joshi RK, Subudhi E,
33. Behera S, Monalisa K, Meher RK, Mohapatra S, Madkami SK, Das PK,
34. Sudha CG, George M, Rameshkumar KB, Nair GM. Improved clonal propagation of
35. Shahinozzaman M, Ferdous M, Faruq M, Azad M, Amin M. Micropropagation of black turmeric (
36. Haida Z, Sinniah UR, Nakasha JJ, Hakiman M. Shoot induction, multiplication, rooting and acclimatization of black turmeric (
37. Nayak S.