1. INTRODUCTION
Lung cancer remains a leading cause of death worldwide, with non-small cell lung cancer (NSCLC) accounting for about 85% of global cases [1]. The global burden of lung cancer is increasing, with GLOBOCAN 2024 estimating 2.5 million cases by 2030, especially in high-pollution and resource-limited regions [2]. Primary treatment modalities encompass chemotherapy, radiation therapy, immunotherapy, and surgery [3]. While the 2018–2024 literature established the foundational role of the gut-lung axis through live microbial patterns [4,5], the clinical transition is hindered by significant safety concerns regarding the administration of live biotherapeutics to immunocompromised patients [6].
The gastrointestinal system harbors a complex consortium of trillions of microorganisms crucial for maintaining systemic immune homeostasis. Dysbiosis of this microbial equilibrium has been implicated in the pathogenesis of various multi-organ pathologies, including obesity, pneumonia, malnutrition, asthma, psychiatric disorders, and malignancy [7]. As lung cancer remains a leading cause of global mortality, research into the gut-lung axis directly supports Sustainable Development Goal (SDG) 3, which aims to reduce pre-mature mortality from non-communicable diseases (one-third) by 2030 through prevention and innovative treatment.
The bidirectional communication of the gut-lung axis is a component of a larger, systemic network of microbial signaling that includes the well-characterized microbiome-gut-brain axis. Recent research has underscored the multifunctional nature of this network, demonstrating that probiotic strains, such as Lactiplantibacillus plantarum AM2 can produce neuroactive compounds, such as acetylcholine to exert neuroprotective effects and modulate systemic oxidative stress [8]. Understanding the parallel mechanisms of the gut-brain axis provides vital context for lung cancer research; the same systemic immunological and metabolic signals that influence cognitive health and neuroinflammation are often involved in maintaining respiratory homeostasis and modulating the inflammatory drivers of NSCLC. By acknowledging these intersecting axes, research suggests that gut-derived metabolites act as long-range mediators that shape the tumor microenvironment across distal organs [8].
Building upon this systemic network, the gastrointestinal microbiome is further integrated into the gut-liver-lung and gut-kidney-lung axes, creating a multi-organ feedback loop that influences pulmonary health. Recent evidence emphasizes that the gut-liver axis, linked through portal circulation, acts as a critical checkpoint; dysbiosis-driven “leaky gut” allows the translocation of pathobionts that trigger hepatic inflammatory cascades, specifically involving toll-like receptor 4-mediated nuclear factor kappa B (NF-κB) activation and the subsequent systemic release of interleukin (IL)-6 and tumor necrosis factor (TNF)-α. These signals, when discharged into the systemic circulation, have been shown to modulate the pulmonary pre-metastatic niche, creating an environment conducive to NSCLC progression and colonization [9]. Simultaneously, the gut-kidney axis governs the clearance of microbial-derived metabolic byproducts. Recent 2024 investigations highlight the accumulation of gut-derived uremic toxins, such as indoxyl sulfate, that induces systemic oxidative stress. When these toxins remain unfiltered due to the microbial-renal imbalance, they directly exacerbate pulmonary endothelial damage and activate oncogenic signaling pathways, such as NF-κB and Janus Kinases-Signal Transducers and Activators of Transcription, within the lung microenvironment [10]. Collectively, these intersecting pathways suggest that the inflammatory drivers of NSCLC are not isolated pulmonary events but are part of a synchronized homeostatic collapse across the gut-liver-kidney network.
Unlike prior reviews on gut microbiota in general cancers, this narrative review explores dysbiosis across the triadic oral, lung, and gut microbiota elevates the risk of lung tumorigenesis. Furthermore, it proposes a “postbiotic-centric lung cancer framework,” which uniquely focuses on probiotic-derived metabolites, such as exopolysaccharides (EPS), bacteriocins, and non-ribosomal peptides (NRPs). By evaluating their lung-specific mechanisms, such as sulfur metabolism in EPS and addressing the translational obstacles of aerosolized delivery, this review offers a 2025 perspective on the integration of microbial innovations into clinical NSCLC management.
2. LITERATURE SEARCH STRATEGY AND SELECTION
To ensure a comprehensive and unbiased narrative synthesis, a structured literature search was conducted across PubMed, Scopus, and Web of Science for articles published from January 2015 to September 2025. The search terms included “probiotics AND lung cancer,” “gut-lung axis AND dysbiosis,” and “probiotic-derived compounds AND NSCLC.” While this work is a narrative review, we employed a transparent selection process involving the screening of 450 abstracts, of which 120 full-text articles were assessed. From the collection, approximately 60 high-impact studies were prioritized based on their mechanistic depth, focus on NSCLC, and relevance to probiotic-derived bioactive metabolites. The articles were excluded if they were not specific to microbiome-lung axis or targeted non-oncological respiratory pathologies. This approach ensures that the following synthesis represents the present state of the field while maintaining a critical postbiotic-centric study.
While several comprehensive reviews between 2018 and 2024 have established the foundational roles of gut dysbiosis and short-chain fatty acids (SCFAs) in lung cancer, a significant conceptual gap remains regarding the specific therapeutic potential of probiotic-derived metabolites. We uniquely evaluate their structural-functional relationships with the NSCLC microenvironment and provide a 2025 appraisal of the translational hurdles specifically associated with lung-targeted delivery. The comparative analysis in Table 1 reveals a significant evolutionary shift in microbiome oncology. While 2018–2024 literature successfully mapped the taxonomic landscape, the persistent gap in postbiotic centric framework suggests that the field is transitioning from broad ecological observation to targeted molecular pharmacology. This means that future research should deprioritize the cataloging of bacterial abundance in favor of characterizing the structural-functional relationships of specific metabolites, which offer more predictable safety profiles in clinical settings than live biotherapeutics.
Table 1: Comparative analysis of the present review with recent microbiome-lung cancer literature (2022–2025).
| Feature | Recent major reviews (2022–2025) | Present review (2025) |
|---|---|---|
| Primary scope | Overview of gut-lung axis and immunotherapy [11,12] | Mechanistic postbiotic centric lung cancer framework |
| Metabolite focus | Broad categories mostly SCFAs mediated immune modulation [13] | Detailed signaling of EPS, NRPs, and Bacteriocins, with mitochondrial disruption |
| Mechanistic depth | Immune checkpoints modulation [12,14] | Mechanism targeting sulfur-redox balance and mitochondrial-targeted apoptosis |
| Delivery insights | Oral probiotics/fecal microbial transplant [11,14] | Critical appraisal of aerosolized delivery challenges |
| Translational focus | Clinical markers and risk factors [13,15] | Synthesis of specialized metabolites as direct adjuvants |
| Critical appraisal | Primarily reports “positive” pre-clinical outcomes. | Engagement with failed findings, HGT risks, and limitations of 2D A549 models. |
SCFAs: Short-chain fatty acids, EPS: Exopolysaccharides, NRPs: Non-ribosomal peptides, HGT: Horizontal gene transfer.
3. THE ORAL-GUT-LUNG TRIPLE AXIS: A SYSTEMIC NETWORK OF LUNG TUMORIGENESIS
The dynamic interplay between the host microbiota and immune responses encompassing innate and adaptive arms profoundly shapes the functionality of multiple physiological systems, including the respiratory, gastrointestinal, and immune networks [16]. Contrary to the historical view of pulmonary sterility, the lung hosts a low-biomass but functionally significant microbiome. This pulmonary microbiome, though less dense than its gut counterpart, contributes to immune homeostasis and metabolic regulation, influencing overall health through microbial metabolites and immune crosstalk [17]. The gastrointestinal and respiratory tracts, despite their anatomical separation, share an embryological origin from the foregut and exhibit structural parallels, such as mucus production and immunoglobulin A secretion by goblet cells. These similarities facilitate a bidirectional gut-lung axis, where microbial and immune signals traverse through the systemic circulation, lymphatic drainage, or direct aspiration, as illustrated in Figure 1 [18]. Within this triadic framework, microbes from the oral and gut microbiota contribute to immune system enhancement, with gut-derived SCFAs playing a crucial role in suppressing lung cancer cell formation. In addition, microbial metabolites from both microbiota systems collaboratively sustain the lung’s ecological balance, highlighting their interconnected roles in overall health. This axis underscores the interconnectedness of distal microbial ecosystems and their collective impact on host physiology. From a clinical perspective, the existence of this triadic signaling network implies that successful NSCLC management cannot remain lung-centric. It necessitates a systemic ‘multi-organ’ approach where the oral and gut reservoirs are managed as primary drivers of pulmonary health.
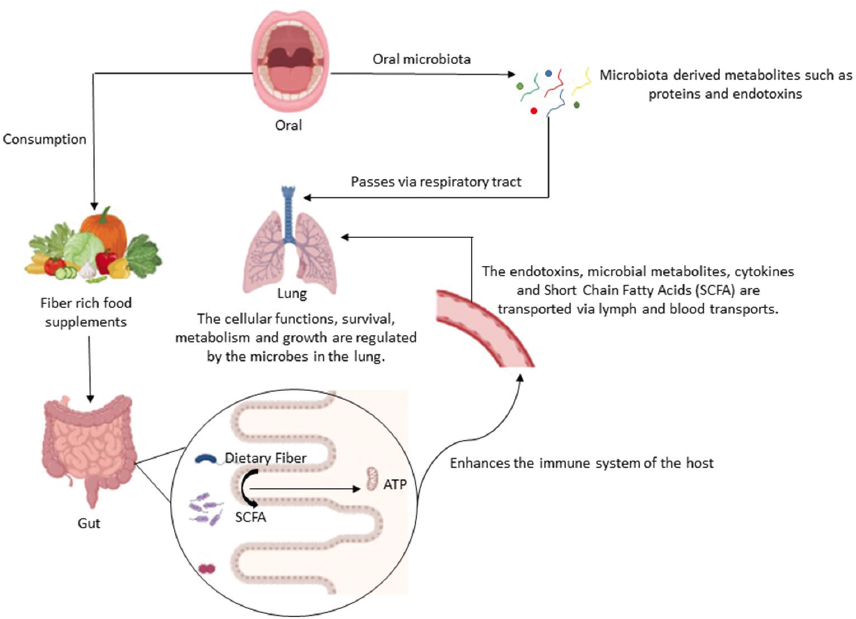 | Figure 1: The interplay between oral, gut, and lung microbiota. [Click here to view] |
The oral microbiome, a vibrant community of bacteria, viruses, and yeasts, occupies distinct ecological niches within the oral cavity, such as the tongue, buccal mucosa, and subgingival plaque, enabled by adhesion mechanisms mediated by fimbriae and adhesins [19]. This community generates bioactive derivatives, including proteins, lipopolysaccharides, and endotoxins, which can translocate to the respiratory tract through microaspiration, inhalation, or hematogenous dissemination [18]. The pharynx serves as a direct anatomical duct linking oral and lung microenvironments, with oral taxa, such as Streptococcus and Porphyromonas frequently isolated from bronchoalveolar lavage samples, suggesting a significant role in shaping the lung microbiome [20]. SCFAs, peptides, and signaling molecules are examples of bacterial products that influence intracellular and extracellular pathways in the lung, such as NF-κB and mitogen-activated protein kinase (MAPK) cascades, which control cellular survival, metabolism, and proliferation. These interactions highlight the lung microbiome’s role as a regulator of local homeostasis, though its low biomass and susceptibility to environmental perturbation complicate its characterization. The gut microbiota, a dense and intricate ecosystem, is a cornerstone of host physiology, orchestrating digestion, immune maturation, and pathogen resistance through metabolites, such as butyrate, acetate, and propionate. Dysbiosis is an imbalance in microbial composition or function that implicates the gut in a spectrum of diseases, including lung cancer [21]. Disruption of gut metabolic pathways, such as those involving SCFA production or tryptophan metabolism, may foster systemic inflammation or oncogenic signaling, contributing to tumorigenesis. In NSCLC patients, studies have revealed dysbiosis, particularly a decline in butyrate-producing bacteria, as a potential driver of disease progression. Butyrate, a histone deacetylase inhibitor, typically suppresses tumor growth by enhancing apoptosis and immune surveillance; its depletion may thus tilt the balance toward oncogenesis, conceivably altered T-regulatory cell function or heightened pro-inflammatory cytokine production (e.g., IL-6, TNF-α). The gut-lung crosstalk suggests that microbial metabolites act as long-range mediators, though causality remains elusive due to confounding factors, such as diet and chemotherapy [22]. Emerging evidence suggests a correlation between oral health and prognosis of lung cancer, with a pronounced association in postmenopausal women [23].
4. MOLECULAR MECHANISM AND TRANSLATIONAL SYNTHESIS OF PROBIOTIC-DERIVED COMPOUNDS
From an analytical perspective, the therapeutic potential of postbiotics lies in their ability to execute “multi-pathway” apoptosis. While conventional chemotherapy often fails due to single-point resistance, probiotic-derived metabolites converge on highly conserved pathways, specifically the induction of reactive oxygen species (ROS) and the activation of the caspase cascade through distinct biophysical and metabolic triggers. Instead of repeating these shared downstream events, the following sections highlight the unique molecular entry points and translational bottlenecks specific to each compound class, as illustrated in Figure 2.
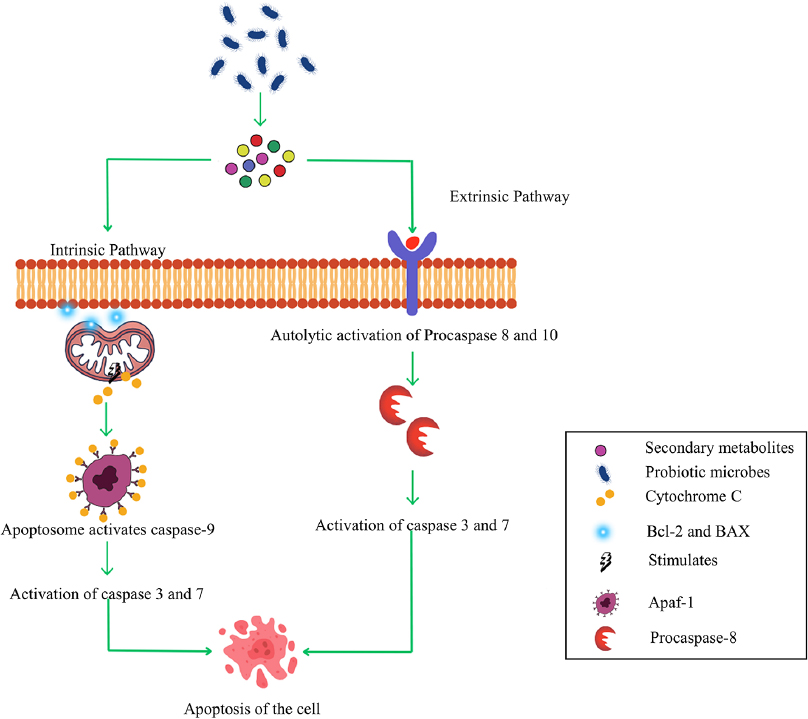 | Figure 2: Mechanistic depth of intrinsic and extrinsic apoptotic pathways induced by probiotic-derived metabolites. [Click here to view] |
4.1. Cell-Free Supernatant (CFS)
The CFS is a heterogeneous mixture of organic acids, such as lactic and acetic acids, short and long fatty acids, proteins, peptides, and bioactive substances, such as bacteriocins and EPSs harvested from probiotic cultures through centrifugation and filtration [24]. The aqueous extract of Bifidobacterium species specifically suppresses matrix metalloproteinase-9 through the p38 MAPK pathway, halting the cell cycle and limiting invasive potential [25]. These findings suggest that Bifidobacterium derived CFS may serve as a supplementary therapeutic agent, potentially modulating tumor microenvironments through both direct cellular effects and indirect immune-mediated mechanisms, though clinical translation requires further investigation into dosage, delivery, and specificity. A significant hurdle for the field is the presence of non-specific enterotoxins, such as Hemolysin BL (HBL), a three-component enterotoxin traditionally associated with the Bacillus cereus group, in the CFS of Bacillus toyonensis isolated from healthy human fecal samples demonstrates potent necrotic activity in vitro, using the A549 lung adenocarcinoma cell line, and in vivo, in murine models, its non-selective nature represents a “double-edged sword” that necessitates sophisticated fractionation or tumor-targeted delivery to avoid systemic toxicity [26].
4.2. EPSs
EPS are high molecular weight extracellular macromolecules secreted by microorganisms in the form of a thick capsule or slime layer. They are composed of reducing sugars, such as glucose, galactose, and mannose, often mixed with non-carbohydrates, such as acetyl groups or uronic acids. These polysaccharides serve critical functions in phagocytosis by modulating immune recognition, mediating cell-cell interactions, and defending against phage attacks. Specifically, the EPS confer protection against bacteriophage attacks by acting as a physical barrier, resistance to toxins, desiccation, antibiotics, and osmotic stress adaptation. In addition, EPS makes a substantial contribution to the antibacterial, anti-inflammatory, and anticancer qualities. The structural makeup of the microbe and the present environmental circumstances, however, determine the precise roles of EPS [27]. Emerging research has illuminated the anticancer efficacy of EPS derived from probiotic strains, with a particular focus on their activity against lung cancer cell lines. For instance, heteropolysaccharides extracted from Bacillus altitudinis, a species adapted to extreme altitudes, demonstrate potent antitumor effects against the A549 NSCLC cell line. These polysaccharides exhibit dose-dependent cytotoxicity across a range of concentrations, likely disrupting cancer cell viability through interference with membrane integrity or metabolic pathways, though the exact mechanisms remain under investigation [28]. Similarly, EPS produced by Bacillus albus DM-15, a probiotic isolate, displays a direct correlation between concentration and cytotoxic activity, eliciting significant antiproliferative effects. Microscopic analysis of treated A549 cells reveals hallmark features of apoptosis, including cellular shrinkage, nuclear fragmentation, and chromatin condensation, suggesting that EPS trigger programmed cell death by means of regulated pathways as illustrated in the mechanistic framework of Figure 2 [29]. These morphological changes highlight EPS’s capacity to selectively target cancer cells, a property that warrants further exploration for therapeutic specificity. In comparison, EPS from marine-derived Bacillus subtilis have shown antiproliferative activity against A549 cells, with an IC50 ranging from 123 to 148 μg/mL, surpassing the efficacy of cisplatin, a standard chemotherapeutic agent with an IC50 often exceeding 200 μg/mL in similar assays [30]. However, the superior potency may stem from the unique structural features of marine EPS, such as higher sulfate content or branching patterns, which enhance solubility and bioactivity. While high sulfate content is associated with enhanced bioactivity, the proposed role of sulfur-rich EPS in restoring redox balance within cancer cells should be interpreted as a hypothesis-generating framework for future pharmacological studies, rather than a definitively established clinical mechanism. The marine environment, characterized by high salinity and pressure, likely drives the evolution of such distinctive polysaccharide profiles, offering a rich reservoir for novel biotherapeutics. Furthermore, innovative approaches have amplified EPS’s potential through nanotechnology. When conjugated with nanoparticles derived from Weissella paramesenteroides, EPS induces apoptosis in A549 cells in both crude and partially purified forms, with efficacy bound to enhanced cellular uptake and targeted delivery. This synergy between EPS and nanomaterials suggests a multifaceted mechanism possibly involving ROS generation or disruption of oncogenic signaling that amplifies cytotoxicity while reducing off-target effects [31].
4.3. Ribosomal Peptides
Ribosomal peptides represent a class of small molecules biosynthesized by the ribosomal machinery, increasingly reported in drug discovery and developmental biology due to their structural versatility and biological potential. Unlike non-ribosomal peptides, which are assembled by large enzymatic complexes, ribosomal peptides originate from short open reading frames and undergo extensive post-translational modifications, such as cyclization, hydroxylation, or glycosylation. These modifications generate a rich diversity of bioactive peptides, including metabolism, intracellular signaling, developmental pathways and gene expression [32]. The mechanism of action of bacteriocins with anticancer activity remains unclear. However, two primary modes of action are widely recognized: Selective membrane disruption (extracellular mechanism) and non-membrane disruption (intracellular mechanism). In selective membrane disruption, bacteriocins exploit the distinct differences between normal and cancerous cells. Normal cells are characterized by zwitterionic components, such as phospholipids, phosphatidylcholine, and sphingomyelin, which confer a neutral charge to their outer membrane. In contrast, cancerous cells exhibit an overexpression of anionic molecules on their surface, resulting in a negatively charged membrane. The positively charged bacteriocins are attracted to these negatively charged cancerous cells, facilitating a strong interaction. Furthermore, the high fluidity of cancer cell membranes enhances the ability of bacteriocins to penetrate these cells, triggering apoptosis [33]. While the electrostatic attraction between cationic bacteriocins and anionic cancer membranes is a primary driver of selectivity, clinical translation must account for the high degree of NSCLC heterogeneity. Factors, such as variations in membrane fluidity, cholesterol-mediated membrane stabilization, and the presence of specific surface receptors across different NSCLC subtypes (adenocarcinoma vs. squamous cell carcinoma) may influence bacteriocin binding efficiency. In addition, the acidic lung tumor microenvironment may alter the ionization of these peptides and reduce their target affinity. Therefore, a purely “charge-based” model may not account for the resistance patterns observed in highly heterogeneous tumor populations, necessitating the identification of specific molecular targets beyond lipid composition. The binding of bacteriocins to cancer cell membranes can occur through one of four distinct models: Barrel-stave interaction, Carpet model interaction, Toroidal pore interaction, and Wand edge-like model interaction. These interactions collectively highlight the targeted and efficient mechanism by which bacteriocins exert their anticancer effects [34]. Among ribosomal peptides, bacteriocins stand out as a specialized subset of antimicrobial peptides produced by bacteria, characterized by unique structural motifs, such as lanthionine bridges or amphipathic helices. Historically valued for their ability to inhibit bacterial growth by targeting cell membranes or intracellular processes, such as DNA replication, bacteriocins have recently revealed broader therapeutic potential. Beyond their antimicrobial role, they exhibit cytotoxic effects against various cancer cell lines and possess additional clinically relevant properties, including immunomodulation and anti-biofilm activity [35]. This expanded functionality likely arises from their ability to interact with eukaryotic membranes or signaling pathways, a feature that distinguishes them from narrow-spectrum antibiotics and highlights their evolutionary adaptability across microbial ecosystems. A compelling study by Saidumohamed et al. explored the anticancer effects of microcins, small, ribosomally synthesized bacteriocins derived from Bacillus amyloliquefaciens, a soil-dwelling probiotic species. Their findings demonstrated a striking reduction in lung cancer cell proliferation, exceeding 50% after 72 h of exposure, as assessed in vitro. This antiproliferative effect may stem from microcins’ ability to disrupt mitochondrial function or induce oxidative stress, though the precise molecular targets remain elusive. This suggests that microcins may complement existing chemotherapies, particularly for resistant lung carcinomas, pending further elucidation of their mechanism and selectivity [36]. Another standout example is Laterosporulin 10 (LS10), a bacteriocin produced by Brevibacillus laterosporus SKDU10, which demonstrates the dual antimicrobial-anticancer potential. LS10 not only inhibits a spectrum of pathogens likely through pore formation or membrane depolarization but also eradicates multiple cancer cell lines by compromising their membrane integrity. This membrane-disruption may be observed by electron microscopy as blebbing and lysis, mirrors the effects of cationic antimicrobial peptides, yet LS10’s efficacy against eukaryotic cancer cells hints at a broader specificity, possibly involving lipid composition differences between normal and malignant membranes [37].
4.3.1. Comparative translatability: Bacteriocin versus EPSs
A critical question for clinical translation is the comparative utility of different probiotic metabolites for lung-specific targeting [11]. Bacteriocins, as small, positively-charged peptides, offer a high degree of specificity for anionic cancer membranes and are particularly suited for aerosolized delivery. Their small molecular size allows for better penetration into the distal lung tissue when inhaled; however, their susceptibility to proteolytic degradation by pulmonary proteases remains a significant hurdle [34]. In contrast, EPS are high-molecular-weight polymers, resistant to glycosidases, exhibiting superior chemical stability compared to peptides. While their size may limit direct tissue penetration, their primary translatable value lies in systemic immune modulation. EPS are more effective in resetting the gut-lung axis by strengthening the intestinal barrier and inducing long-range cytokine signaling. Therefore, while bacteriocins show higher promise as direct, localized cytotoxic agents, EPS represent a more robust candidate for systemic adjuvant therapy by modulating the pre-metastatic niche and bolstering anti-tumor immunity [15].
4.4. Non-Ribosomal Peptides
NRPs, such as fengycin and surfactin, are biosynthesized by multifunctional enzyme complexes, allowing for cyclic and lipopeptide structures that bypass traditional genetic constraints [38]. A study by Shao et al. investigated the anticancer potential of NC16, an NRP derived from B. subtilis, which induces apoptosis through dual pathways: The extrinsic and the intrinsic mitochondrial pathway against the A549 lung adenocarcinoma cell line. In the former, NC16 likely activates death receptors through intrinsic pathway, cleaving vital cellular substrates, such as poly-ADP ribose polymerase to dismantle cellular architecture. Simultaneously, the mitochondrial pathway involves the release of cytochrome C, driven by membrane permeabilization, which activates the apoptosome and amplifies caspase signaling, which is detailed in Figure 2. This dual-mode action suggests a synergistic attack on cancer cell viability, potentially overcoming resistance mechanisms seen in single-pathway therapies, though specificity to malignant versus healthy cells remains a critical question for clinical translation [39]. Similarly, Yin et al. explored fengycin, a lipopeptide NRP from B. subtilis, revealing its capacity to modulate ROS generation in lung cancer cells. Elevated ROS levels disrupt redox homeostasis, inducing oxidative stress that stalls the cell cycle at the G0/G1 phases key checkpoints governed by cyclins and CDK inhibitors, such as p21. However, further mechanistic studies are required to establish whether this redox modulation serves as a primary therapeutic driver in vivo or acts as secondary to other apoptotic pathways. In addition, it facilitates ROS production through lipid peroxidation or interference with mitochondrial electron transport chains. These results establish fengycin as a versatile agent, combining cytotoxic effects with cell cycle regulation, but its therapeutic range, balancing efficacy and minimizing off-target oxidative damage, needs careful optimization [40]. Significantly, pre-clinical in vitro models suggest that this process may selectively target malignant cells, potentially due to differences in membrane surface charge, without significantly affecting surrounding healthy cells [41]. However, the biological relevance of this selectivity remains to be validated by comprehensive in vivo toxicity studies and pharmacokinetic (PK) profiling to determine whether the observed specificity is maintained within the systemic environment and the immunosuppressive lung tumor microenvironment.
4.5. Miscellaneous
Extracellular vesicles (EVs) secreted by the probiotic strain Faecalibacterium prausnitzii, a key commensal bacterium in the human gut, have been evaluated for their therapeutic potential against the A549 lung adenocarcinoma cell line. These nanoscale vesicles, enriched with bioactive cargo, such as lipids, proteins, and nucleic acids, exert a more pronounced immunomodulatory effect than the corresponding CFS. Specifically, EVs disrupt cytokine expression profiles by upregulating pro-inflammatory mediators, such as TNF-α and TNF-β, which can enhance immune surveillance and trigger tumor cell apoptosis through receptor-mediated pathways. Simultaneously, they downregulate anti-inflammatory cytokines, such as IL-6, a known promoter of cancer cell survival and proliferation through Signal Transducers and Activators of Transcription 3 activation. This dual modulation suggests that F. prausnitzii-derived EVs may shift the tumor microenvironment toward an anti-tumorigenic state, potentially outstripping CFS by delivering concentrated payloads directly to target cells through membrane fusion or endocytosis [42]. The superior efficacy of EVs highlights their potential as precision therapeutics, though their stability, scalability, and specificity in vivo require further optimization. In a parallel investigation, the immunomodulatory effects of a fermented milk product, kefir, were assessed using the 4T1 breast cancer cell line in a murine model. Kefir, rich in probiotic bacteria, such as Lactobacillus and Bifidobacterium spp., significantly bolstered adaptive immunity, evidenced by increased populations of CD4+ helper T cells and CD8+ cytotoxic T cells in treated mice. These immune enhancements correlated with a marked reduction in metastatic burden in bone marrow and lungs, likely driven by heightened T-cell-mediated cytotoxicity and improved recognition of tumor antigens. The fermentation process, which generates bioactive peptides and organic acids, may amplify kefir’s anti-metastatic effects by disrupting cancer cell adhesion or invasion. However the precise molecular interplay between kefir metabolites and immune activation remains under-explored [43]. These findings underscore kefir’s promise as a dietary adjuvant in cancer management, bridging gut microbiota modulation with systemic anti-tumor immunity. A clinical study involving lung cancer patients further demonstrated the benefits of probiotic intervention, utilizing Clostridium butyricum administered as per a standardized protocol during chemotherapy. Monitoring of fecal microbiota revealed that C. butyricum supplementation mitigated chemotherapy-induced dysbiosis, reducing the incidence and severity of diarrhea, a common side effect linked to gut barrier disruption and microbial imbalance. The probiotic also attenuates systemic inflammatory responses, possibly through SCFA production of organic acids, such as butyrate, supports epithelial integrity and diminishes pro-inflammatory cytokine storms. By maintaining microbial homeostasis, C. butyricum likely bolstered patient resilience to chemotherapy’s toxic effects, suggesting a role in supportive care that could improve treatment tolerance and quality of life [44]. An innovative strategy targeting lung tumor growth has emerged through modulation of the pulmonary microbiota using aerosolized probiotics or antibiotics. This approach aims to reshape the local microbial ecosystem, potentially suppressing tumor-promoting bacteria, while enriching anti-tumorigenic species. Probiotics may exert direct effects, such as metabolite-mediated cytotoxicity or indirect effects through immune priming, while antibiotics may eliminate oncogenic microbial drivers. Preliminary evidence suggests this method disrupts tumor progression, possibly by altering hypoxia or inflammation within the lung microenvironment yet, the lung’s delicate microbial balance and the risk of aerosol-induced resistance or off-target effects demand rigorous pre-clinical testing to refine this cutting-edge intervention [45].
5. INTEGRATED SYNTHESIS: LINKING DYSBIOSIS TO MOLECULAR COUNTERMEASURES
The gut-lung axis is governed by a complex interplay of microbial translocation and metabolic signaling. It is essential to map these taxonomic shifts directly to their underlying pathogenic drivers and corresponding bioactive solutions. The integration of multi-niche dysbiosis with targeted molecular countermeasures detailed in Table 2 provides a roadmap for the transition from general probiotic supplementation to precision postbiotic therapy. The significance of this framework is threefold. First, it identifies the oral reservoir as a distinct therapeutic target; the localized interventions may proactively reduce the “microaspiration load” into the lower respiratory tract. Second, the linkage between lung-resident and advanced staging/TP53 mutations suggests that these taxa may serve as prognostic biomarkers to guide the intensity of adjuvant therapy. Finally, the framework underscores the necessity of niche-specific delivery systems. By mapping specific microbial failures to targeted molecular solutions, this framework moves toward a pharmacological model where postbiotics are used as precision tools to disrupt the pro-tumorigenic signaling of the triadic axis.
Table 2: Integrated framework: Multi-niche dysbiosis and targeted postbiotic solutions.
| Niche | Taxonomic shift | Pathogenic mechanism | Bioactive countermeasure |
|---|---|---|---|
| Gut (Fecal) | Reduction of Faecalibacterium prausnitzii, Clostridial Cluster I, Clostridium leptum, and Ruminococcus [22,46] | Loss of Butyrate/SCFA signaling: Leads to systemic inflammation (↑IL-6, ↑TNF-α) and blunted regulatory T-cell (Treg) differentiation. | The EPS and butyrate produced by the probiotics could lead to the restoration of the gut barrier integrity and systemically suppresses protumorigenic inflammation. |
| Gut (Fecal) | Elevation of the microbes, such as Enterococcus, Veillonella, Bacteroides, Anaerotruncus, and Proteobacteria [47-50] | Pathogenic enrichment drives gut-barrier disruption and provides long-range oncogenic signals to the lung microenvironment | Bacteriocin produced by the probiotics specifically inhibits pathogen proliferation and disrupts pro-tumorigenic biofilms |
| Oral | Elevation of the oral pathogens, such as Streptococcus, Neisseria, Capnocytophaga, and Selenomonas [51-53] | Oral taxa translocates to lower airways, triggering Th17-mediated inflammation and PI3K pathway activation | Localized bacteriocins to reduce the oral reservoir of lung-translocating pathogens, using oral wash |
| Oral | Elevation of the pathogens, such as Granulicatella adiacens, Acinetobacter junii, Sphingomonas, and Blastomonas [54,55] | These shifts reflect an altered local redox state and serve as markers for diagnostic monitoring in smokers/non-smokers | Non-Ribosomal peptides, localized delivery of superoxide dismutase to mitigate oral oxidative damage |
| Lung | Elevation of Thermus, Legionella, Acidovorax, and Prevotella [56-58] | Associated with advanced staging and TP53 mutations; drives NF-κB activation and extracellular matrix degradation | Aerosolized Non-Ribosomal Peptides may be a direct delivery to the tumor niche to interrupt signalling and induce apoptosis |
| Lung | Decrease in alpha diversity, Staphylococcus, and Coriobacteriaceae [59,60] | Loss of protective commensals reduces the lung’s natural resistance to colonization by oncogenic pathogens | Broad-spectrum postbiotics could be used to sustain ecological stability and baseline immune priming |
| Lung | Elevation of lung pathogens, such as Veillonella parvula, Firmicutes, and Bacteroidetes [61] | Direct interaction with lung epithelium triggers unchecked proliferation and suppresses apoptosis. | Targeted peptides could be used for direct membrane disruption of Veillonella to halt localized protumorigenic signaling. |
SCFA: Short-chain fatty acids, EPS: Exopolysaccharides, IL-6: Interleukin-6, TNF-α: Tumor necrosis factor-α, NF-κB: Nuclear factor kappa B.
6. CRITICAL APPRAISAL: SAFETY, MICROBIAL RISKS, AND CONTRADICTORY FINDINGS
The therapeutic application of HBL is a double-edged sword. As a potent enterotoxin, its necrotic activity is non-specific, posing a high risk of systemic toxicity and off-target damage to healthy lung parenchyma. Utilizing such metabolites requires highly sophisticated, tumor-targeted delivery systems to ensure that their cytotoxic effects are restricted solely to malignant cells [26]. On the other hand, the marine-derived EPS exhibit promising IC50 values against A549 cell lines; these pre-clinical findings must be interpreted with significant caution. Direct comparisons to established chemotherapeutic agents, such as cisplatin may be misleading, as values IC50 are laboratory surrogates for cytotoxic potential that do not account for disparate assay conditions or the systemic complexities that dictate clinical efficacy. A critical translational gap persists; unlike the well-characterized profiles of cisplatin, the PKs and pharmacodynamics (PD) of microbial-derived EPS specifically regarding their interaction with gut-associated lymphoid tissue, serum half-life, and the ability to navigate the immunosuppressive lung tumor microenvironment remain largely uncharacterized. Consequently, while in vitro assays provide a valuable baseline for biological screening, they are insufficient for predicting the safety or therapeutic performance of postbiotic compounds in NSCLC patients [31].
Clinical translation faces critical safety hurdles, particularly for immunocompromised patients, where live probiotic translocation may lead to bacteremia or fungemia; cases of central line-associated Saccharomyces cerevisiae infections are already documented in NSCLC contexts [62]. In addition, horizontal gene transfer (HGT) risks allow probiotic strains to act as reservoirs for antibiotic resistance genes. This genetic exchange can exacerbate existing dysbiosis by compromising the stability of the resident lung and gut microbiota and potentially promoting the multidrug-resistant pathobionts [35]. Moreover, recent clinical evidence suggests a “probiotic paradox” where the unguided use of over-the-counter probiotic supplements has been associated with reduced gut microbial diversity and a diminished response to immune checkpoint inhibitors in some cancer cohorts, contradicting the traditional view of probiotics as universally beneficial adjuvants [63]. This highlights that while specific probiotic-derived metabolites show therapeutic promise, “one-size-fits-all” supplementation may paradoxically interfere with immunotherapy efficacy, further emphasizing the need for targeted, postbiotic-centric interventions over unguided microbial administration. Furthermore, innovative aerosolized delivery carries the risk of localized immune overstimulation or acute inflammatory responses in the fragile lung microenvironments. These safety concerns underscore the necessity of “next-generation” postbiotics, non-viable derivatives that offer therapeutic efficacy without the inherent risks of live microbial administration.
7. CLINICAL CHALLENGES AND FUTURE PERSPECTIVES
The triadic dysbiosis across oral, lung, and gut environments significantly influences NSCLC progression, positioning probiotic-derived compounds as promising low-toxicity adjuvants to conventional therapy. However, a significant knowledge gap remains regarding their interaction with oncogenic drivers, such as epidermal growth factor receptor and KRAS. A major methodological hurdle is the over-reliance on A549 monolayer cultures, which fail to replicate the structural complexity or the immunosuppressive signaling of the human tumor microenvironment. To bridge the translational gap, research must shift toward patient-derived organoids and orthotopic murine models that better simulate clinical bioavailability.
Furthermore, the present scarcity of lung-specific clinical trials necessitates a rigorous investigation into optimal strain-specific dosages and the safety of administering live biotherapeutics to immunocompromised patients at risk for systemic infections. Beyond molecular mechanisms, the integration of standardized probiotic protocols into oncological supportive care guidelines directly supports SDG 3. By mitigating chemotherapy-induced toxicities and reducing the economic burden on public health systems, these cost-effective interventions can enhance treatment adherence in high-risk, resource-limited regions, effectively bridging the gap between advanced research and the global mission of “Good Health and Well-being.”
8. CONCLUSION
This review explores the transition from broad gut-lung axis characterizations to a specialized postbiotic-centric framework for NSCLC management. We have delineated the triadic interplay of oral, gut, and lung microbiomes that shape the tumor microenvironment through a pharmacological repertoire of bioactive metabolites, including sulfur-rich EPS, bacteriocins, and NRPs. These compounds function as multifaceted molecular adjuvants, triggering intrinsic and extrinsic apoptosis specifically through the Caspase-3/7/9 cascades while simultaneously disrupting oncogenic PI3K/Akt and Wnt/β-catenin signaling.
Unlike conventional cytotoxic agents, probiotic-derived compounds offer a pleiotropic approach to cancer therapy, with the potential to enhance chemotherapy efficacy and mitigate treatment-related toxicities. This observed specificity in vitro, which appears to spare healthy cells possessing neutral membranes, characterizes probiotic-derived compounds as potential low-toxicity candidates for adjunctive therapy. Nevertheless, it is pre-mature to classify them as safer than conventional drugs until their safety profiles are rigorously validated in clinical trials to ensure they do not interfere with the efficacy of conventional oncological regimens. However, a critical synthesis of present data reveals a significant translational gap. While the observed specificity of bacteriocins for anionic cancer membranes is promising in vitro, the heavy reliance on A549 monolayer models necessitates caution. The biological relevance of these findings must be validated through rigorous in vivo PK and PD profiling to ensure that therapeutic efficacy is maintained within the complex, immunosuppressive human lung niche.
Moving forward, the successful clinical integration of postbiotics depends on resolving biophysical hurdles, such as the proteolytic degradation of peptides and the high molecular weight delivery challenges of EPS. Furthermore, robust safety protocols are required to address risks involving HGT and the administration of microbial derivatives to immunocompromised patients. Aligning with SDG 3 (Target 3.4), these scalable and cost-effective innovations represent a vital pathway toward equitable cancer care and improved treatment adherence in resource-limited regions. Ultimately, the transition from laboratory screening to clinical oncology will require interdisciplinary efforts to standardize production and develop advanced lung-targeted delivery systems, potentially extending these microbial interventions to a broader spectrum of malignancies.
9. ACKNOWLEDGMENT
Adeline Celina Rufus expresses gratitude for Founder-Chancellor Shri. N.P.V. Ramaswamy Udayar Fellowship, awarded by Sri Ramachandra Institute of Higher Education and Research, supports her doctoral research endeavors. The authors extend their thanks to the management of Sri Ramachandra Institute of Higher Education and Research for providing the essential resources for conducting the study.
10. AUTHOR’S CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work. All the authors are eligible to be author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
11. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
12. ETHICS STATEMENT
This study does not involve experiments on animals or human subjects.
13. DATA AVAILABILITY STATEMENT
The authors confirm that the data supporting this review are available within the article.
14. INFORMED CONSENT STATEMENT
This study did not involve human participants, and therefore, informed consent was not required.
15. DECLARATION OF AI
The authors confirm that the manuscript was written by the authors without the use of AI tools for content generation. However, AI-based language tools, such as Quillbot and Gemini, were used solely to improve English clarity and readability, ensuring that the content is effectively communicated to the scientific community.
16. PUBLISHER’S NOTE
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors and the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Thandra KC, Barsouk A, Saginala K, Aluru JS, Barsouk A. Epidemiology of lung cancer. Contemp Oncol (Pozn). 2021;25:45-52. [CrossRef]
2. Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022:GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74:229-63. [CrossRef]
3. Jones GS, Baldwin DR. Recent advances in the management of lung cancer. Clin Med (Lond). 2018;18:S41-6. [CrossRef]
4. Budden KF, Gellatly SL, Wood DL, Cooper MA, Morrison M, Hugenholtz P, et al. Emerging pathogenic links between microbiota and the gut-lung axis. Nat Rev Microbiol. 2016;15:55-63. [CrossRef]
5. Enaud R, Prevel R, Ciarlo E, Beaufils F, Wieërs G, Guery B, et al. The gut-lung axis in health and respiratory diseases:A place for inter-organ and inter-Kingdom crosstalks. Front Cell Infect Microbiol. 2020;10:9. [CrossRef]
6. Gurunathan S, Thangaraj P, Kim JH. Postbiotics:Functional food materials and therapeutic agents for cancer, diabetes, and inflammatory diseases. Foods. 2023;13:89. [CrossRef]
7. Sharma A, Viswanath B, Park YS. Role of probiotics in the management of lung cancer and related diseases:An update. J Funct Foods. 2018;40:625-33. [CrossRef]
8. Lotfy WA, Ali AM, Abdou HM, Ghanem KM. Neuroprotective and probiotic potential of Lactiplantibacillus plantarum AM2 in cognitive impairment. Sci Rep. 2025;15:20186. [CrossRef]
9. Zhu X, Zhang X, Shen J, Zheng S, Li H, Han B, et al. Gut microbiota-dependent modulation of pre-metastatic niches by Jianpi Yangzheng decoction in the prevention of lung metastasis of gastric cancer. Phytomedicine 2024;128:155413. [CrossRef]
10. Sun E, Meng X, Kang Z, Gu H, Li M, Tan X, et al. Zengshengping improves lung cancer by regulating the intestinal barrier and intestinal microbiota. Front Pharmacol. 2023;14:1123819. [CrossRef]
11. Sudaarsan AS, Ghosh AR. Appraisal of postbiotics in cancer therapy. Front Pharmacol. 2024;15:1436021. [CrossRef]
12. Liu L, Yang L, Zhang H, Li H, Shang T, Liu L. Lung cancer and the Gut-microbiota-lung Axis: Emerging evidence and potential clinical implications. Front Med (Lausanne). 2025;12:1655780. [CrossRef]
13. Ashique S, Mishra N, Garg A, Kumar N, Khan Z, Mohanto S, et al. A critical review on the role of probiotics in lung cancer biology and prognosis. Arch Bronconeumol. 2024;60:S46-S58. [CrossRef]
14. Shi M, Wang LF, Hu WT, Liang ZG. The gut microbiome in lung cancer: From pathogenesis to precision therapy. Front Microbiol. 2025;16:1606684. https://doi.org/10.3389/fmicb.2025.1606684 [CrossRef]
15. Balendra V, Rosenfeld R, Amoroso C, Castagnone C, Rossino MG, Garrone O, et al. Postbiotics as adjuvant therapy in cancer care. Nutrients. 2024;16:2400. https://doi.org/10.3390/nu16152400 [CrossRef]
16. McLean AE, Kao SC, Barnes DJ, Wong KK, Scolyer RA, Cooper WA, et al. The emerging role of the lung microbiome and its importance in non-small cell lung cancer diagnosis and treatment. Lung Cancer. 2022;165:124-32. [CrossRef]
17. Fabbrizzi A, Amedei A, Lavorini F, Renda T, Fontana G. The lung microbiome: Clinical and therapeutic implications. Intern Emerg Med. 2019;14:1241-50. [CrossRef]
18. Ma Q, Li X, Jiang H, Fu X, You L, You F, et al. Mechanisms underlying the effects, and clinical applications, of oral microbiota in lung cancer:Current challenges and prospects. Crit Rev Microbiol. 2023;50:631-52. [CrossRef]
19. Deo PN, Deshmukh R. Oral microbiome: Unveiling the fundamentals. J Oral Maxillofac Pathol. 2019;23:122-8. [CrossRef]
20. Zeng XT, Xia LY, Zhang YG, Li S, Leng WD, Kwong JS. Periodontal disease and incident lung cancer risk: A meta-analysis of cohort studies. J Periodontol. 2016;87:1158-64. [CrossRef]
21. Zheng Y, Fang Z, Xue Y, Zhang J, Zhu J, Gao R, et al. Specific gut microbiome signature predicts the early-stage lung cancer. Gut Microbes. 2020;11:1030-42. [CrossRef]
22. Gui Q, Li H, Wang A, Zhao X, Tan Z, Chen L, Xu K, Xiao C. The association between gut butyrate-producing bacteria and non-small-cell lung cancer. J Clin Lab Anal. 2020;34:e23318. [CrossRef]
23. Nwizu NN, Marshall JR, Moysich K, Genco RJ, Hovey KM, Mai X, et al. Periodontal disease and incident cancer risk among postmenopausal women: Results from the women's health initiative observational cohort. Cancer Epidemiol Biomarkers Prev. 2017;26:1255-65. [CrossRef]
24. Mani-López E, Arrioja-Bretón D, López-Malo A. The impacts of antimicrobial and antifungal activity of cell-free supernatants from lactic acid bacteria in vitro and foods. Compr Rev Food Sci Food Saf. 2022;21:604-41. [CrossRef]
25. An J, Kim H, Yang KM. An aqueous extract of a Bifidobacterium species induces apoptosis and inhibits invasiveness of non-small cell lung cancer cells. J Microbiol Biotechnol. 2020;30:885-93. [CrossRef]
26. Chen J, Hu S, Ji D, Gao Z, Wang H, Yang Y, et al. Hemolysin BL from novel Bacillus toyonensis BV-17 induces antitumor activity both in vitro and in vivo. Gut Microbes. 2020;12:1782158. [CrossRef]
27. Angelin J, Kavitha M. Exopolysaccharides from probiotic bacteria and their health potential. Int J Biol Macromol. 2020;162:853-65. [CrossRef]
28. Mohamed SS, Amer SK, Selim MS, Rifaat HM. Characterization and applications of exopolysaccharide produced by marine Bacillus altitudinis MSH2014 from Ras Mohamed, Sinai, Egypt. Egypt J Basic Appl Sci. 2019;5:204-9. [CrossRef]
29. Vinothkanna A, Sathiyanarayanan G, Rai AK, Mathivanan K, Saravanan K, Sudharsan K, et al. Exopolysaccharide produced by probiotic Bacillus albus DM-15 isolated from ayurvedic fermented dasamoolarishta:Characterization, antioxidant, and anticancer activities. Front Microbiol. 2022;13:832109. [CrossRef]
30. Abdel-Wahab BA, Abd El-Kareem FH, Alzamami A, Fahmy CA, Elesawy BH, Mostafa Mahmoud M, et al. Novel exopolysaccharide from marine Bacillus subtilis with broad potential biological activities:Insights into antioxidant, anti-inflammatory, cytotoxicity, and anti-Alzheimer activity. Metabolites. 2022;12:715. [CrossRef]
31. Amer M, Eldiwany A, Elgammal E, Atwa N, Dawoud I, Rashad F. Nano-exopolysaccharide from the probiotic Weissella paramesenteroides MN2C2: Production, characterization and anticancer activity. Egypt J Chem. 2021;64:7023-33. [CrossRef]
32. Sukmarini L. Marine bacterial ribosomal peptides: Recent genomics- and synthetic biology-based discoveries and biosynthetic studies. Mar Drugs. 2022;20:544. [CrossRef]
33. Molujin AM, Abbasiliasi S, Nurdin A, Lee PC, Gansau JA, Jawan R. Bacteriocins as potential therapeutic approaches in the treatment of various cancers: A review of in vitro studies. Cancers (Basel). 2022;14:4758. [CrossRef]
34. Wang Y, Wang Y, Sun T, Xu J. Bacteriocins in cancer treatment: Mechanisms and clinical potentials. Biomolecules. 2024;14:831. [CrossRef]
35. Benítez-Chao DF, León-Buitimea A, Lerma-Escalera JA, Morones-Ramírez JR. Bacteriocins: An overview of antimicrobial, toxicity, and biosafety assessment by in vivo models. Front Microbiol. 2021;12:630695. [CrossRef]
36. Saidumohamed BE, Johny TK, Raveendran AT, Sheela UB, Sreeranganathan M, Sasidharan RS, et al. 3D structure elucidation and appraisal of mode of action of a bacteriocin BaCf3 with anticancer potential produced by marine Bacillus amyloliquefaciens BTSS3. Re GEN Open. 2022;2:45-56. [CrossRef]
37. Huang F, Teng K, Liu Y, Cao Y, Wang T, Ma C, et al. Bacteriocins: Potential for human health. Oxid Med Cell Longev. 2021;2021:5518825. [CrossRef]
38. Dincer S, Takci HA, Ozdenefe MS. Nonribosomal peptide synthesis. In: Molecular Cloning. London: Intech Open;2022. [CrossRef]
39. Shao Y, Wang XY, Qiu X, Niu LL, Ma ZL. Isolation and purification of a new Bacillus subtilis strain from deer dung with anti-microbial and anti-cancer activities. Curr Med Sci. 2021;41:832-40. [CrossRef]
40. Yin H, Guo C, Wang Y, Liu D, Lv Y, Lv F, et al. Fengycin inhibits the growth of the human lung cancer cell line 95D through reactive oxygen species production and mitochondria-dependent apoptosis. Anticancer Drugs. 2013;24:587-98. [CrossRef]
41. Zhao H, Xu X, Lei S, Shao D, Jiang C, Shi J, et al. Iturin A-like lipopeptides from Bacillus subtilis trigger apoptosis, paraptosis, and autophagy in Caco-2 cells. J Cell Physiol. 2019;234:6414-27. [CrossRef]
42. Jafari B, Khavari Nejad RA, Vaziri F, Siadat SD. Evaluation of the effects of extracellular vesicles derived from Faecalibacterium prausnitzii on lung cancer cell line. Biologia. 2019;74:889-98. [CrossRef]
43. Zamberi NR, Abu N, Mohamed NE, Nordin N, Keong YS, Beh BK, et al. The antimetastatic and antiangiogenesis effects of kefir water on murine breast cancer cells. Integr Cancer Ther. 2016;15:NP53-66. [CrossRef]
44. Tian Y, Li M, Song W, Jiang R, Li YQ. Effects of probiotics on chemotherapy in patients with lung cancer. Oncol Lett. 2019;17:2836-48. [CrossRef]
45. Le Noci V, Guglielmetti S, Arioli S, Camisaschi C, Bianchi F, Sommariva M, et al. Modulation of pulmonary microbiota by antibiotic or probiotic aerosol therapy: A strategy to promote immunosurveillance against lung metastases. Cell Rep. 2018;24:3528-38. [CrossRef]
46. Qin X, Bi L, Yang W, He Y, Gu Y, Yang Y, et al. Dysbiosis of the gut microbiome is associated with histopathology of lung cancer. Front Microbiol. 2022;13:918823. [CrossRef]
47. Zhuang H, Cheng L, Wang Y, Zhang YK, Zhao MF, Liang GD, et al. Dysbiosis of the gut microbiome in lung cancer. Front Cell Infect Microbiol. 2019;9:112. [CrossRef]
48. Zhang WQ, Zhao SK, Luo JW, Dong XP, Hao YT, Li H, et al. Alterations of fecal bacterial communities in patients with lung cancer. Am J Transl Res. 2018;10:3171-85. https://pubmed.ncbi.nlm.nih.gov/30416659/
49. Liu F, Li J, Guan Y, Lou Y, Chen H, Xu M, et al. Dysbiosis of the gut microbiome is associated with tumor biomarkers in lung cancer. Int J Biol Sci. 2019;15:2381-92. [CrossRef]
50. Wei YF, Huang MS, Huang CH, Yeh YT, Hung CH. Impact of gut dysbiosis on the risk of non-small-cell lung cancer. Int J Environ Res Public Health. 2022;19:15991. [CrossRef]
51. Vogtmann E, Hua X, Yu G, Purandare V, Hullings AG, Shao D, et al. The oral microbiome and lung cancer risk: An analysis of 3 prospective cohort studies. J Natl Cancer Inst. 2022;114:1501-10. [CrossRef]
52. Yan X, Yang M, Liu J, Gao R, Hu J, Li J, et al. Discovery and validation of potential bacterial biomarkers for lung cancer. Am J Cancer Res. 2015;5:3111-22. https://pmc.ncbi.nlm.nih.gov/articles/PMC4656734/
53. Zhang W, Luo J, Dong X, Zhao S, Hao Y, Peng C, et al. Salivary microbial dysbiosis is associated with systemic inflammatory markers and predicted oral metabolites in non-small cell lung cancer patients. J Cancer. 2019;10:1651-62. [CrossRef]
54. Cameron SJ, Lewis KE, Huws SA, Hegarty MJ, Lewis PD, Pachebat JA, et al. A pilot study using metagenomic sequencing of the sputum microbiome suggests potential bacterial biomarkers for lung cancer. PLoS One. 2017;12:e0177062. [CrossRef]
55. Yang J, Mu X, Wang Y, Zhu D, Zhang J, Liang C, et al. Dysbiosis of the salivary microbiome is associated with non-smoking female lung cancer and correlated with immunocytochemistry markers. Front Oncol. 2018;8:520. [CrossRef]
56. Yu G, Gail MH, Consonni D, Carugno M, Humphrys M, Pesatori AC, et al. Characterizing human lung tissue microbiota and its relationship to epidemiological and clinical features. Genome Biol. 2016;17:163. [CrossRef]
57. Greathouse KL, White JR, Vargas AJ, Bliskovsky VV, Beck JA, Von Muhlinen N, et al. Interaction between the microbiome and TP53 in human lung cancer. Genome Biol. 2018;19:123. [CrossRef]
58. Liu Y, O'Brien JL, Ajami NJ, Scheurer ME, Amirian ES, Armstrong G, et al. Lung tissue microbial profile in lung cancer is distinct from emphysema. Am J Cancer Res. 2018;8:1775-87. https://pmc.ncbi.nlm.nih.gov/articles/PMC6176189/
59. Liu HX, Tao LL, Zhang J, Zhu YG, Zheng Y, Liu D, et al. Difference of lower airway microbiome in bilateral protected specimen brush between lung cancer patients with unilateral lobar masses and control subjects. Int J Cancer. 2018;142:769-78. [CrossRef]
60. Peters BA, Hayes RB, Goparaju C, Reid C, Pass HI, Ahn J. The microbiome in lung cancer tissue and recurrence-free survival. Cancer Epidemiol Biomarkers Prev. 2019;28:731-40. [CrossRef]
61. Zeng W, Zhao C, Yu M, Chen H, Pan Y, Wang Y, et al. Alterations of lung microbiota in patients with non-small cell lung cancer. Bioengineered. 2022;13:6665-77. [CrossRef]
62. Lee YM. Central line-related bloodstream infection by Saccharomyces cerevisiae following probiotic use in a patient with Clostridioides difficile colitis:A case report. Microorganisms. 2026;14:70. [CrossRef]
63. Spencer CN, McQuade JL, Gopalakrishnan V, McCulloch JA, Vetizou M, Cogdill AP, et al. Dietary fiber and probiotics influence the gut microbiome and melanoma immunotherapy response. Science. 2021;374:1632-40. [CrossRef]