1. INTRODUCTION
Urbanization and industrialization are the chief routes of human development in multiple ways today. However, contaminants generated by anthropogenic activities and some natural incidences have endlessly polluted the environment. Arsenic is naturally found in the earth’s crust and is extensively distributed in various components of the environment. It has a 74.9 g/mol atomic weight, 5.73 g cm-3 specific gravity, and a boiling and melting point of 614°C and 817°C, respectively [1]. It is a firm, breakable crystalline solid with a silver-grey color [1]. Its high industrial value makes it inevitable to circumvent living in an arsenic-free environment. It is used in making semiconductors, herbicides, pesticides, fertilizers, paints, cosmetics, glass industry, fireworks, ammunition, etc. [2]. The International Agency for Research on Cancer has classified inorganic arsenic as a Group 1 carcinogen [3], and Agency for Toxic Substances and Disease Registry has considered it as genotoxic, neurotoxic, and embryotoxic with a half-life (plasma) of 3–4 h [4].
Soil health is essential as it is directly linked to food security, affecting plant growth, productivity, and human health. Soil-bound arsenic in the agroecosystems is transferred to the edible parts of crop plants through natural mineral uptake mechanism. However, prolonged consumption of such contaminated crops may pose health risks to the human population [5,6]. Another route for arsenic in the human body through the food chain is the intake of contaminated drinking water [7], consumption of seafood [8], and livestock-based products [9]. Food products such as milk, meat, eggs, fish, and other dairy and poultry products produced by animals feeding on arsenic contaminated crops and water or thriving in the arsenic contaminated environment could be another source of food chain contamination [10,11].
Arsenic toxicity in humans leads to multiple health risks such as skin disorders, cancers, melanosis, hyperkeratosis, lung disease, peripheral vascular diseases, gangrene, diabetes mellitus, hypertension, and ischemic heart disease [12,13]. Moreover, chronic exposure to inorganic arsenic increases the risk of diabetes mellitus, adverse pregnancy outcomes, and even skin cancers, lungs, and urinary bladder [14].
Therefore, it is crucial to focus on averting an increase in arsenic contamination in the food chain and exploring the novel methods to reduce it. Hence, the present study highlights the bioaccumulation of arsenic in soil and water and its subsequent transfer into the crop plants, including cereals, vegetables, seafood along with livestock-based food products, namely, milk, meat, and eggs. It emphasizes on food chain contamination and further outlines various health risks and disorders due to chronic arsenic toxicity. The study reflects the directions for future that may be helpful for global scientific communities, stakeholders, and policy makers to mitigate the arsenic contamination.
2. BIOAVAILABILITY AND MOBILITY OF ARSENIC IN SOIL-PLANT SYSTEM
Different soils have varying levels of arsenic depending on the parent material; in most cases, the baseline of arsenic in agricultural soils may range from 5 to 10 mg/kg soil [15]. However, agricultural soil quality is compromised by the escalation of urban and industrial activities globally. Arsenic is chiefly present as As(III) and As(V) in nature. The other main species of arsenic in natural environments include monomethylarsonite (MMA(III)), monomethylarsonate (MMA(V)), dimethyl arsenite (DMA(III)), dimethyl arsinate (DMA(V)), arsenocholine, arsenosugars, arsenobetaine, trimethylarsine oxide, and tetramethylarsonium ions [16]. Furthermore, the As(III)) has been found more toxic, mobile and soluble than organic arsenic because these can efficiently react with sulfhydryl (–SH) groups of different proteins and restrict the cellular functions that ultimately lead to the death of cells [17].
The mobility and transformation of arsenic in soil are determined by multiple factors such as its oxidation states, soil texture, and iron oxides [18], sorption desorption processes [19], soil pH [20], organic matter and metallic elements [21], redox potential and organic acids [22], oxic anoxic conditions [23], and microbial species [24]. Microbial species augment the interconversion of As(III) and As(VI) by solubilizing or immobilizing arsenic in the soil-plant system [25]. Arsenic dynamics in the soil-cabbage system showed that iron oxides increased arsenic mobilization in soil [18]. In contrast, aluminum oxides contributed strongly to the immobilization of arsenic. Moreover, an increased sand content promoted the mobility of arsenic, whereas increased silt and clay contents showed inhibitory effects [18]. These biotic and abiotic factors alter the biogeochemistry of arsenic in soil-plant system, making it bioavailable to the crop plants. The mechanism of arsenic uptake by plants varies with the chemical speciation. It has been reported that As(V) uses inorganic Phosphate (Pi) channels for its entry into the plant cell [26]. On the other hand, plants uptake As(III) via various nodulin-26-like intrinsic proteins and silicon transporters due to analogous chemical structures [27]. Most arsenic typically ends up in plants’ vacuolar compartments, whether in the root or shoot tissue. Arsenic is transported and accumulated to the next level consumer; prolonged consumption of such crops poses numerous health hazards.
3. BIOAVAILABILITY AND MOBILITY OF ARSENIC IN WATER
Chronic arsenic poisoning caused by drinking water is considered one of the world’s biggest environmental disasters recorded in the last century [28]. Water sources get contaminated either by natural processes or through anthropogenic activities. Natural processes include the leaching of minerals, the interaction of rocks with water, groundwater movement, geothermal activities, and mineralization [29]. The leaching of arsenic from aquifers depends on the geochemical characteristics of groundwater and arsenic speciation. In groundwater, As(V) is predominant in oxic environments, with significant forms of H3AsO4, H2AsO4, HAsO42-, and AsO43−, whereas, As(III) is more dominant in anoxic environments, with H3AsO3, H2AsO3, and HAsO32− being the common species [30]. Higher organic contents and alkaline conditions in groundwater and aquifers could also enhance the release of arsenic into groundwater [21,31]. Geogenic arsenic-contaminated groundwater has become a matter of concern in the Ganga delta region, especially for the human population living in India and Bangladesh [32]. Chakraborti et al. [7] reported health risks to more than 10 million Indians in the Ganga delta region from fluoride and arsenic due to dependency on groundwater resources. However, anthropogenic activities, including domestic and industrial effluent discharge, agricultural runoff, leaching of agrochemicals, coal power plants, smelting and mining activities [33] also contaminate the water resources. Besides drinking water, aquatic life thriving in arsenic-contaminated water resources also tends to accumulate in their organs. Many researchers have reported high arsenic concentrations in fish, crabs, eels, and other aquatic animals used as seafood [34,35].
4. ARSENIC ACCUMULATION IN CROPS
4.1. In Cereal Grains
Arsenic is a highly toxic metalloid and widely distributed contaminant in the food chain. Because of plants’ innate ability to uptake minerals from the soils, food crops such as cereals and vegetables have been regarded as important channels for arsenic exposure in humans. Arsenic contamination in cereals due to geogenic and anthropogenic sources has been reported in numerous studies [6,36] [Table 1]. Rice has a high arsenic accumulation efficiency than any other cereal due to anaerobic conditions that promote its mobilization and uptake by the rice plant [37]. Mining and smelting activities in China have contaminated paddy soils, resulting in high arsenic bioaccumulation in rice grains [38]. Besides, extensive and prolonged use of arsenic-based fertilizers and pesticides also contribute as a source of arsenic in agroecosystems [39]. Other factors that influence the arsenic content in rice include rice grain processing [40], rice variety [41], the region where it is grown [42], irrigation method [43], and the cooking method [44]. Arsenic contamination of the soil–wheat system has been documented as a result of excessive arsenic levels in groundwater used for irrigation in China [45]. In another study, high levels of arsenic in wheat grain (27 μg/kg) were reported from geo-genically contaminated areas of the mid-Gangetic plain of Bihar, India [46].
Table 1: Arsenic in cereal grains (μg/kg).
| Country | Crop | Sample size (n) | Arsenic (mean) | Arsenic (range) | References |
|---|---|---|---|---|---|
| China | Rice | 5 | 656.00 | 603–729 | Zheng et al.[51] |
| Pakistan | Maise | 4 | 3730.00 | 1470–3540 | Natasha et al.[52] |
| Wheat | 13 | 2310.00 | |||
| Malaysia | Paddy | 9 | 84.76 | Zulkafflee et al.[53] | |
| Bangladesh | Rice | 35 | 1370.00 | Proshad et al.[54] | |
| Brazil | Rice | 16 | 212.00 | 2–1830 | Ng et al.[55] |
| China | Maize | 18 | 90.00 | 60–260 | Cai and Li [38] |
| Rice | 22 | 130.00 | |||
| China | Wheat | 22 | 417.00 | 271–991 | Zhang et al.[56] |
| Saudi Arabia | Rice | 13 | 0.30 | 0.1–1 | Althobiti et al.[57] |
| Iran | Rice | 210 | 0.16 | 0.05–0.31 | Roya and Ali [58] |
| Italy | Paddy | 168 | 155.00 | 49–523 | Tenni et al.[59] |
| India | Wheat | 35 | 27.00 | 7.7–108 | Kumar et al.[46] |
| Green g | 6 | 23.00 | 7.9–49 | ||
| Maize | 31 | 13.00 | 4.8–43 | ||
| Paddy | 15 | 51.00 | 2.51–132 | ||
| China | Wheat | 25 | 66.90 | 22.8–154 | Tong et al.[45] |
| China | Wheat | 75 | 33.30 | 6.5–54.9 | Shi et al.[48] |
Food chain transfer of arsenic affects crop quality and productivity, threatening food security and further compromising human health. Prolonged consumption of such crops affects the health and wellness of human beings; moreover, such crops are not considered fit for export. Another study from the same region, Bihar, reported total arsenic in wheat grains (43.64 μg/kg) with a lifetime cancer risk (1.23 × 10−4), much higher than set by USEPA regulatory range (10−4–10−6) [10] for the rural population due to wheat consumption grown in that region. Since Bihar exports wheat across the country, arsenic exposure through wheat-based food consumption may occur in the non-endemic areas of the country with a similar wheat-based diet [10]. The other factors influencing the arsenic accumulation and bioavailability in wheat crop include, the wheat variety [47], industrial and mining activities [48], wastewater irrigation and sludge application [39], coal-fired thermal power plants [49], waste dumping in the vicinity of fields, and extensive use of agrochemicals [50].
4.2. In Vegetables
Vegetables are an essential component of the human diet; thus, they are also an important channel of arsenic exposure to humans. Vegetable cultivation in arsenic endemic regions threatens food security and human health to a greater extent. The arsenic contamination in vegetables depends on multiple factors, including the type of vegetable, soil type, irrigation water quality, vicinity area, anthropogenic pressure, and geogenic contamination [Table 2]. In a study from Samta village, West Bengal, India (arsenic endemic region), arsenic levels in vegetables surpassed the national and international limits. The contamination in vegetables was associated with arsenic-contaminated tube well water (0.24 mg/L) used for irrigation [60]. In another study from the mid-Gangetic plain of Bihar, India, the mean arsenic concentration in the vegetables was reported to be very high (452 μg/kg) and in the range of 37–3947 μg/kg, due to the geogenic source with some diffused anthropogenic activities [46]. The arsenic transfer to the food chain via irrigation water (contaminated ground water resources) has been well documented. Huq et al. [61] reported that excessive groundwater withdrawal for irrigating agricultural fields in Bangladesh has contributed to high arsenic levels in surface soils, further leading to contamination of crops. Industrial and mining activities also strongly influence arsenic availability in different components of environs and alter the quality of water, soil, and food crops [62]. Furthermore, wastewater irrigation-mediated arsenic food chain transfer is a significant pathway, particularly in developing countries with limited water resources and inadequate wastewater treatment facilities [52].
Table 2: Arsenic in vegetables (mean, μg/kg) and their contamination sources.
| Country | Crop type | Source | Arsenic | References |
|---|---|---|---|---|
| China | Spinach, broccoli, tomato, tine peas | Anthropogenic | 95.7, 112.0, 107.0, 115.0 | Zheng et al.[51] |
| Pakistan | Spinach, mustard, carrot, cabbage, fenugreek | Wastewater irrigation | 400.0, 390.0, 240.0, 200.0, 190.0 | Natasha et al.[52] |
| Bangladesh | Brinjal, potato, bottle gourd, pumpkin, green amaranth | Geogenic and anthropogenic contamination | 350.0, 280.0, 430.0, 320.0, 360.0 | Haque et al.[5] |
| China | Lettuce, rape, bitter, mustard, garlic, pakchoi | Mine water irrigation | 1990.0, 870.0, 3260.0, 3160.0, 1820.0 | Qin et al.[62] |
| Bangladesh | Bitter gourd, okra, bean, chilli, bottle gourd, cucumber, sponge gourd | Industrial activities | 2030.0, 2550.0, 1640.0, 2910.0, 2850.0, 1640.0, 3890.0 | Proshad et al.[54] |
| Zimbabwe | Tomato, okra, onion, pumpkin, leaves, spinach | Contaminated soil | 600.0, 1000.0, 1300.0, 5200.0, 4030.0 | Meck et al.[71] |
| China | Radish, carrot, cabbage, rice, celery | Industrial activities | 50.0, 20.0, 70.0, 130.0, 110.0 | Cai et al.[72] |
| Brazil | Beans, cabbage, carrot, garlic, lettuce | Mining activities | 50.0, 6.0, 7.0, 13.0, 17.0 | Ng et al.[55] |
| Nigeria | White yam, pumpkin, spinach, coriander, tomato | Mining activities | 100.0, 400.0, 1990.0, 2050.0, 560.0 | Obiora et al.[73] |
| China | Spinach, coriander, celery, pea, amaranthus | Mining activities | 110.0, 340.0, 170.0, 290.0, 280.0 | Luo et al.[74] |
| Pakistan | Spinach, coriander | Wastewater irrigation | 4180.0, 4030.0 | Khan et al.[75] |
| Pakistan | Radish, turnip | Wastewater irrigation | 3370.0, 3510.0 | Ahmad et al.[76] |
| India | Luffa, okra, cucumber, brinjal | Geogenic contamination | 800.0, 375.0, 399.0, 492.0 | Kumar et al.[46] |
The root-to-shoot translocation and accumulation of arsenic species differ between plant species and even between the variety of cultivars [63]. Arsenic translocation to the above-ground parts of the vegetables is generally limited [64] due to various tolerance mechanisms developed by plants [65]. Therefore, grains, seeds, lentils, and fruits end up with relatively lower levels of arsenic as compared to its concentration in roots and other belowground plant organs [66]. Overall, root tuber and leafy vegetables accumulate more arsenic content than fruity or fleshy vegetables of above-ground parts [67]. Due to the tolerance mechanisms, minimum arsenic translocation was found in wheat, and maximum tolerance was reported in Brinjal (38.80 mg/kg), followed by tomato (35.41mg/kg) [68]. In the xylem sap, arsenite predominated in tomato, cucumber, and rice while arsenate predominated in castor bean, wheat, and Indian mustard [69]. Besides, the external root skin on root vegetables has more arsenic concentration than within the root, implying that the washing and peeling process for edible tubers such as potatoes and carrot effectively reduces exposure for human beings [70]. The differential accumulation/tolerance ability of crop plants can provide target crops for cultivation in arsenic endemic regions with a high tolerance for arsenic, thereby reducing the chances of arsenic transfer to the food chain.
5. ARSENIC BIOAVAILABILITY IN MILK, SEAFOOD, AND LIVESTOCK-BASED FOOD PRODUCTS
5.1. In Milk
Arsenic-contaminated water and feed for cattle is a well-established route of arsenic entry into the food chain. Arsenic is transferred to humans via the consumption of milk and meat from such animals. It may pose a carcinogenic and non-carcinogenic risk to the health of children and adults [11]. Arsenic contamination in milk is of particular concern because milk is primarily consumed by infants and children [77]. The arsenic concentration in cattle milk depends on their feeding habits, food, water, and environmental contamination. High arsenic concentration in raw milk (4.24–4.93 μg/L) was reported from the cows grazing on pastures near lava ground with high thermal activity in Turkey [78]. In a study from Pakistan (Tharparkar region), where elevated concentration of arsenic was found in milk samples of different milch animals, namely, cows, buffalos, sheep, goats, and camels (15.1–18.4, 2.6–7.7, 25.7–33.2, 10.5–37.3, and 6.6–13.7 μg/L, respectively) due to the contaminated drinking water (geogenic arsenic) given to cattle. A high carcinogenic risk to children was found in milk consumption from the said area [11].
According to the WHO recommendations, breastfeeding is essential for babies up to 6 months. An additional 2 years of breastfeeding and appropriate complementary foods are also much needed [79]. However, the composition of human milk is not constant and depends on the mother’s nutritional status, diet, lactation stage, socio-demographic status, lifestyle, and environmental contamination [80]. The presence of geogenic arsenic (50 mg/L) in drinking water contributed to the high arsenic (149 mg/L) in the breast milk of lactating women in West Bengal, India. Moreover, in the lack of mother’s milk, children were further exposed to arsenic from an early age due to their dependency on local cattle milk [81]. Newborns absorb metals to a greater extent than adults. They have a lower capacity to excrete arsenic compounds in the bile, decreasing the body clearance further leading to severe consequences of arsenic toxicity [82].
5.2. In Meat and Poultry Products
Meat and poultry products are nutritional diets necessary for human growth and development as they contain fat, protein, and other essential minerals [83] The use of organo-arsenic drugs as animal feed and antiparasitic agents promotes arsenic transfer in cattle and poultry products [84]. Roxarsone is an arsenic-containing component of chicken feed that supports growth, helps gain weight, improves feed utilization efficiency, and increases chicken meat pigmentation [84]. However, the elevated level of arsenic observed in different chicken tissues was strongly correlated with the use of roxarsone in chicken feed in Guangzhou, China [84]. Another study investigated the total arsenic in cooked chicken meat samples (3.0 μg/kg) due to roxarsone application; arsenic was detected in 50% of samples [85].
Contaminated groundwater has been reported to augment arsenic levels in cattle and poultry [Table 3]. High arsenic was reported in broiler meat (breast-0.633 mg/kg and liver-0.943 mg/kg) and Cock meat (breast meat-0.457 mg/kg and liver-0.379 mg/kg) in poultry farms of Bangladesh [95] due to drinking contaminated groundwater. In poultry farms, arsenic-rich drinking water and feed additives may accumulate a substantial concentration in their flesh and eggs, transfer to human beings upon prolonged consumption, and pose a health risk to the local population. Moreover, high arsenic levels have been found in poultry excreta in Bangladesh. Applying such excreta as fertilizer may cause an augmentation of arsenic contamination in soil, leading to recontamination of the food chain [96].
Table 3: Arsenic in milk (mean, μg/L), meat, and egg samples (mean, μg/kg).
| Country | Sample | Source | Arsenic | References |
|---|---|---|---|---|
| India | Cow milk | Geogenic | 6.37 | Das et al.[86] |
| Goat milk | <3 | |||
| Brazil | Cow milk | Gold mine | 3.00 | Ng et al. [55] |
| Bangladesh | Cow milk | Geogenic | 440.00 | Ahmed et al. [9] |
| United States | Breast milk | Geogenic | 0.62 | Carignan et al. [87] |
| Taiwan | Breast milk | Industrial waste incineration and coal power plant in the vicinity | 1.50 | Chao et al. [88] |
| United States | Breast milk | Lifestyle, Contaminated food | 0.01 | Gaxiola-Robles et al. [89] |
| Bangladesh | Cow milk | Chronic as exposure from drinking water | 51.0 | Islam et al. [90] |
| Sweden | Breast milk | Food habits | 0.55 | Björklund et al.[91] |
| Japan | Breast milk | 1.40 | Sakamoto et al.[92] | |
| Portugal | Breast milk | 5.80 | Almeida et al.[93] | |
| China | Pork meat | Anthropogenic | 482.00 | Zheng et al.[51] |
| Beef | 280.00 | |||
| Chicken meat | 318.00 | |||
| India | Chicken meat | Geogenic | 94.50 | Das et al.[86] |
| Goat meat | 107.00 | |||
| Eggs | 1.00 | |||
| Brazil | Chicken meat | Gold mine | 21.00 | Ng et al.[55] |
| Beef | 21.00 | |||
| Bangladesh | Duck egg | Geogenic | 76.00 | |
| Chicken meat | 33.00 | |||
| Bangladesh | Beef | Geogenic | 570.00 | Ahmed et al.[9] |
| Mutton | 140.00 | |||
| Chicken meat | 430.00 | |||
| Duck meat | 160.00 |
5.3. In Seafood
Among the food products, seafood supplies the majority of the total arsenic to humans; the US Food and Drug Administration indicated that seafood, including finfish, shellfish, seaweed, and other seafood, accounts for 90% of the total human arsenic exposure globally [8,97]. Seafood is the chief source of organic arsenic, such as arsenosugars, arsenobetaine, and arsenolipids [98]. Although more toxic form, that is, inorganic arsenic levels in seafood, are generally relatively low. But some aquatic organisms, such as mussels and algae like Sargassum (a higher alga), can have a high concentration of inorganic arsenic [34], which may transfer it to the human body upon consumption. Arsenic bioaccumulation and transfer in aquatic organisms depend on various biological and environmental factors such as arsenic species, organism type and species, body size, trophic level, and age [99,100]. For instance, arsenobetaine and arsenosugars are the most abundant arsenic compounds in finfish and seaweed [8]. The accumulation efficiency also varies from organism to organism; Crabs can efficiently accumulate the inorganic arsenic compared to finfish due to the presence of chitosan in shells containing charged amino acids which easily attach with the As(V) [100].
Many researchers have reported high arsenic contamination in seafood due to geogenic and anthropogenic sources [Table 4]. For example, Perry et al. [35] reported very high arsenic levels (7.3 mg/kg) in the crab’s muscles in the Gulf of Mexico. However, according to the national standard limit, inorganic arsenic should not exceed 0.5 mg/kg for aquatic crustaceans [105]. The population of coastal regions where the diet is mainly seafood based are more susceptible to arsenic toxicity. High seafood consumption is linked with more elevated arsenic in blood and urine [106], infant cord blood and breast milk [107], and arsenobetaine and dimethylarsinic acid in blood and urine [98].
Table 4: Arsenic in seafood (μg/kg).
| Country | Sample | Source | Arsenic (mg/kg) | References |
|---|---|---|---|---|
| Santa Catarina, Brazil | Oreochromis niloticus | Anthropogenic | 27500.00–49200.00 | Steckert et al.[108] |
| Paracatu, Brazil | Squalus acanthias | Anthropogenic | 233.00 | Ng et al.[55] |
| Bangladesh | Labeo rohita | Geogenic | 110.00 | Islam et al.[94] |
| Pangasius pangasius | 230.00 | |||
| Oreochromis niloticus | 940.00 | |||
| Channa panctatus | 230.00 | |||
| Augusta, Southern Italy | Pagellus erythrinus | Anthropogenic | 3620.00 | Di Bella et al. [109] |
| Pagellus acarne | 4720.00 | |||
| Mullus barbatus | 9940.00 | |||
| Sepia offcinalis | 2268.00 | |||
| Shandong Province, China | Ctenopharyngodon idella | Anthropogenic | 41.00 | Yang et al. [110] |
| Carassius carassius | 89.00 | |||
| Hypophthalmichthys nobilis | 118.00 | |||
| Scomberomorus niphonius | 995.00 | |||
| Trichiurus lepturus | 1130.00 | |||
| Larimichthys polyactis | 1400.00 | |||
| Lateolabrax japonicus | 852.00 | |||
| Sardina pilchardus | 2260.00 |
Physicochemical parameters, namely, pH, salinity, the presence of other elements, and dissolved organic matter along with food ingestion rate, gut passage time, gut environment, living prey/food composition, species, and density, also affect the assimilation of arsenic from food [101]. Physiological functions such as excretion, molting, and reproduction also affect aquatic organisms’ bioaccumulation rate by physically removing arsenic from their body [101,102]. The seafood cooking method also affects arsenic bioavailability; even the same cooking method can affect arsenic bioavailability in different seafood’s [103]. In conformity with this, Perelló et al. [104] reported that deep frying and baking increased the inorganic arsenic levels in bivalve shells and squids. However, more in-depth studies are needed to focus on various in vivo models to monitor the transformation of arsenic species during assimilation of seafood inside the gastrointestinal tract till its accumulation at the cellular level and excretion from the human body.
6. ARSENIC IN FOOD CHAIN AND HUMAN HEALTH RISK
Millions of people are chronically exposed to arsenic via food, air, water, and soil, leading to adverse long-term health consequences [32] [Figure 1]. In humans, arsenic exposure and toxicity arise due to environmental, occupational, or dietary exposure. It is a challenging task yet critical to monitor the food chain as human beings are the top consumers of the food chain. Thus contaminated food chain brings more degree of carcinogenic health risks on prolonged arsenic exposure through various dietary routes [Figure 2]. According to epidemiological studies, chronic arsenic exposure is associated with an increased risk of liver, kidney, prostate, skin, bladder, and lung cancers due to its ability to cause oxidative stress, DNA damage, cellular mutations, and enzyme inhibition [111]. Neuropathies of the central nervous system are attributed to its penetrability to the blood-brain barrier by altering the mitochondrial membrane instability, neurotransmitter impairment, and enzyme disruption [112]. Due to its capacity to cross placental barriers, it can reach fetuses from mothers, potentially causing adverse pregnancy outcomes [113]. It poses deleterious effects on reproductive health by increasing infant mortality and preterm birth and negatively influencing neonatal development [114]. Other reported outcomes of arsenic toxicity include skin disorders [53], hyperpigmentation, keratosis, bronchitis, chronic obstructive pulmonary disease, bronchiectasis, as noncirrhotic portal fibrosis, and others such as polyneuropathy, hypertension, peripheral vascular disorders, ischemic heart disease, diabetes, and edema of hands and feet [13,111,112]. In addition to these dermatological, lung, and heart illnesses, chronic arsenic toxicity also causes variable clinical consequences, the treatments of which are sometimes only indicative and are worse [13].
 | Figure 1: Health disorders and diseases caused by arsenic toxicity. [Click here to view] |
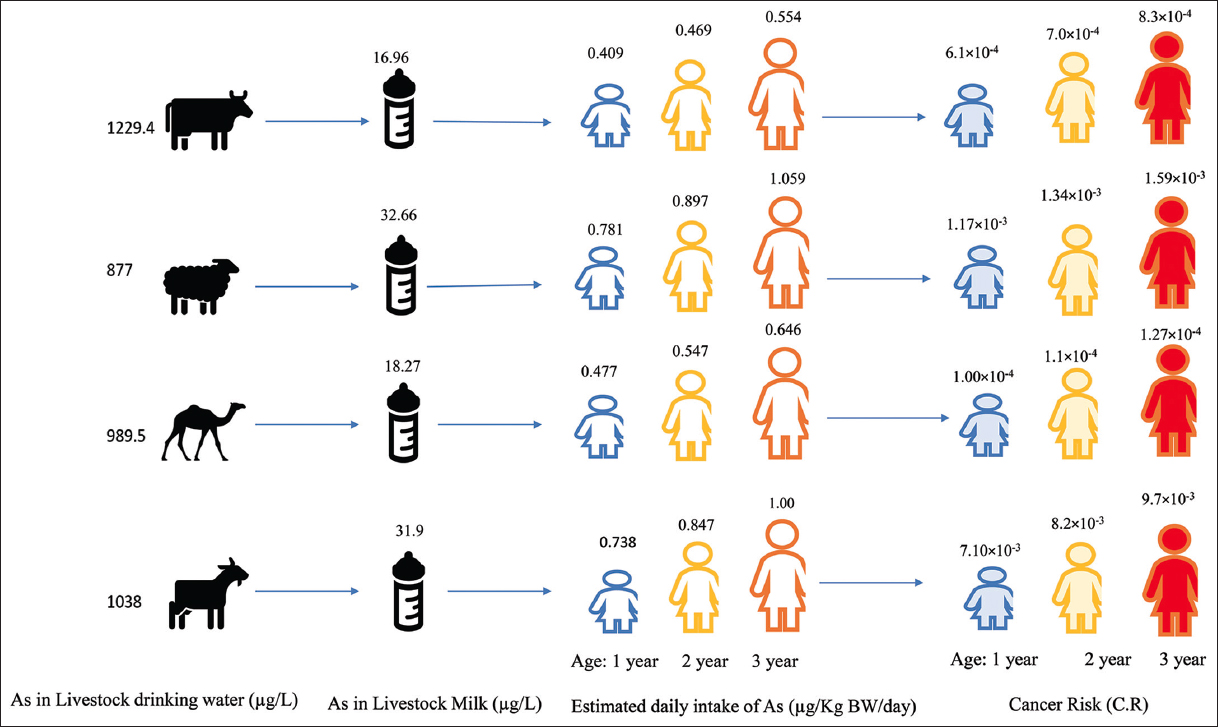 | Figure 2: Transfer of Arsenic in the food chain [(Arsenic in drinking water of livestock, milk produced by livestock, estimated daily intake of Arsenic and Cancer risks in children of three age groups); (Source: created from data reported by Kazi et al. [11])]. [Click here to view] |
Several studies have been conducted to evaluate arsenic toxicity and human health risk in various arsenic-contaminated regions [10,52]. Arsenic risk assessment studies have received massive attention in the recent past due to the fatal effects of its toxicity; these studies evaluate whether occupants of a specific area are exposed to arsenic from different media of the biosphere (soil/air/water/food). If so, whether those exposure levels pose an unacceptable health risk to the population or not. Risk assessment entails contaminant exposure, toxicity, and risk characterization. Many countries have used various models and indices to calculate the magnitude and likelihood of human health risk, namely, Iran [113], China [6,48], India [46,32], Pakistan [114], and Bangladesh [54] Consumption of highland barley irrigated with geothermally arsenic contaminated water in Tibet posed a cancer risk of 5.4 in 10,000 [6]. Kazi et al. [11] assessed the cancer risk in children from milk consumption of the local livestock. High arsenic in the milk samples of cows, sheep, goats, and camels was observed (range of 15.1–18.4, 25.7–33.2, 10.5–37.3, and 6.6–13.7 μg/L, respectively) due to drinking water. Arsenic in livestock drinking water of each farm/flock was found in the range of 238–2000 μg/L. The total hazard quotient for children consuming the milk of sheep, goats, cows, and camels was found >1 of the reference dose for arsenic, posing the adverse effects on the health of the local children [Figure 2].
Thus, it is evident that arsenic transfer from different environmental matrices may enter the food chain through dietary route, leading to severe risks to food security, and human health. Henceforth, focus on controlling the possible elevation of arsenic contamination in the food chain and its possible abatement is much needed. Novel methods such as transgenic approach and bioremediation along with conventional practices may help in mitigating the arsenic from various environmental metrics and food chain, further lowering the risks to human health [115].
7. CONCLUSION
Arsenic, being the first-class member of carcinogen, imparts high risks to the human population due to its characteristics such as toxicity, high persistence, and bioaccumulation capacity in different matrices of the environment. Arsenic contamination of soil, water resources, and food crops due to anthropogenic and geological sources is of profound environmental health concern. In this study, we found extensive evidence of elevated arsenic in the food chain, mainly in rice and vegetables. The uptake of arsenic by rice crops showed a significant exposure pathway to the population thriving in the arsenic endemic regions and where a rice-based diet is dominant. Arsenic in livestock-based food products such as milk, seafood, poultry, and meat products is a significant contributor to food chain contamination, especially in arsenic endemic regions. Chronic exposure can cause skin diseases, developmental, reproductive, respiratory, nephrological and endocrinological diseases, and various cancer types in human beings. Hence, in current scenario where arsenic has become a global menace, there is a dire need of an integrated approach to combat the arsenic contamination thus necessitating formulation of national and global action plan for arsenic contamination mitigation.
8. AUTHOR CONTRIBUTION
Siril Singh: Conceptualization of the idea, methodology, data curation, writing original draft, editing, and revisions Rajni Yadav and Sheenu Sharma: Data curation and editing Anand Narain Singh: Advising, supervision, and modifications.
9. FUNDING
The first author is thankful to the Department of Science and Technology, Ministry of Science and Technology, Government of India, under Women Scientist Scheme-B, WISE-KIRAN DIVISION, Project Grant No. DST/WOS-B/2018/1589.
10. CONFLICT OF INTEREST
All authors declare that they have no conflict of interest.
11. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
12. DATA AVAILABILITY
All the data reported are included within this review article.
13. ACKNOWLEDGMENTS
The authors gratefully acknowledge the Chairperson, Departments of Environment Studies and Botany, Panjab University Chandigarh for the facilities and contributions that motivated the drafting of this review.
REFERENCES
1. Zakhar R, Derco J, Cacho F. An overview of main arsenic removal technologies. Acta Chim Slov 2018;11:107-13. [CrossRef]
2. Briffa J, Sinagra E, Blundell R. Heavy metal pollution in the environment and their toxicological effects on humans. Heliyon 2020;6:e04691. [CrossRef]
3. International Agency for Research on Cancer. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans;1994. Available from:http://monographs.iarc.fr/ENG/Monographs/vol59/mono59.pdf [Last accessed on 2022 Jun 02].
4. Agency for Toxic Substances and Disease Registry. Toxicological Profile for Arsenic. Atlanta, GA:Division of Toxicology;2007. Available from:http://medbox.iiab.me/modules/encdc/www.atsdr.cdc.gov/toxprofiles/tp.asp_id=22&tid=3 [Last accessed on 2022 Jun 02].
5. Haque MM, Niloy NM, Khirul MA, Alam MF, Tareq SM. Appraisal of probabilistic human health risks of heavy metals in vegetables from industrial, non-industrial and arsenic contaminated areas of Bangladesh. Heliyon 2021;7:e06309. [CrossRef]
6. Xue L, Zhao Z, Zhang Y, Liao J, Wu M, Wang M, et al. Dietary exposure to arsenic and human health risks in western Tibet. Sci Total Environ 2020;731:138840. [CrossRef]
7. Chakraborti D, Rahman MM, Ahamed S, Dutta RN, Pati S, Mukherjee SC. Arsenic contamination of groundwater and its induced health effects in Shahpur block, Bhojpur district, Bihar state, India:Risk evaluation. Environ Sci Pollut Res Int 2016;23:9492-504. [CrossRef]
8. Taylor V, Goodale B, Raab A, Schwerdtle T, Reimer K, Conklin S, et al. Human exposure to organic arsenic species from seafood. Sci Total Environ 2017;580:266-82. [CrossRef]
9. Ahmed MK, Shaheen N, Islam MS, Habibullah-Al-Mamun M, Islam S, Islam MM, et al. Acomprehensive assessment of arsenic in commonly consumed foodstuffs to evaluate the potential health risk in Bangladesh. Sci Total Environ 2016;544:125-33. [CrossRef]
10. Suman S, Sharma PK, Siddique AB, Rahman MA, Kumar R, Rahman MM, et al. Wheat is an emerging exposure route for arsenic in Bihar, India. Sci Total Environ 2020;703:134774. [CrossRef]
11. Kazi TG, Brahman KD, Afridi HI, Arain MB, Talpur FN, Akhtar A. The effects of arsenic contaminated drinking water of livestock on its total levels in milk samples of different cattle:Risk assessment in children. Chemosphere 2016;165:427-33. [CrossRef]
12. Otle?S, Ca?indi O. Health importance of arsenic in drinking water and food. Environ Geochem Health 2010;32:367-71. [CrossRef]
13. Guha Mazumder DN. Chronic arsenic toxicity &human health. Indian J Med Res 2008;128:436-47.
14. Sanyal T, Bhattacharjee P, Paul S, Bhattacharjee P. Recent advances in arsenic research:Significance of differential susceptibility and sustainable strategies for mitigation. Front Public Health 2020;8:464. [CrossRef]
15. Basu A, Saha D, Saha R, Ghosh T, Saha B. A review on sources, toxicity and remediation technologies for removing arsenic from drinking water. Res Chem Intermed 2014;40:447-8. [CrossRef]
16. Joseph T, Dubey B, McBean EA. Human health risk assessment from arsenic exposures in Bangladesh. Sci Total Environ 2015;527-528:552-60. [CrossRef]
17. Armendariz AL, Talano MA, Travaglia C, Reinoso H, Wevar Oller AL, Agostini E. Arsenic toxicity in soybean seedlings and their attenuation mechanisms. Plant Physiol Biochem 2016;98:119-27. [CrossRef]
18. Chang C, Li F, Wang Q, Hu M, Du Y, Zhang X, et al. Bioavailability of antimony and arsenic in a flowering cabbage-soil system:Controlling factors and interactive effect. Sci Total Environ 2022;815:152920. [CrossRef]
19. Feng Q, Zhang Z, Chen Y, Liu L, Zhang Z, Chen C. Adsorption and desorption characteristics of arsenic on soils:Kinetics, equilibrium, and effect of Fe(OH)3 Colloid, H2SiO3 colloid and phosphate. Procedia Environ Sci 2013;18:26-36. [CrossRef]
20. Hussain MM, Bibi I, Niazi NK, Shahid M, Iqbal J, Shakoor MB, et al. Arsenic biogeochemical cycling in paddy soil-rice system:Interaction with various factors, amendments and mineral nutrients. Sci Total Environ 2021;773:145040. [CrossRef]
21. Wang S, Mulligan CN. Occurrence of arsenic contamination in Canada:Sources, behavior and distribution. Sci Total Environ 2006;366:701-21. [CrossRef]
22. Wang Y, Zhang G, Wang H, Cheng Y, Liu H, Jiang Z, et al. Effects of different dissolved organic matter on microbial communities and arsenic mobilization in aquifers. J Hazard Mater 2021;411:125146. [CrossRef]
23. Park S, Kim SH, Chung H, An J, Nam K. Effect of organic substrate and Fe oxides transformation on the mobility of arsenic by biotic reductive dissolution under repetitive redox conditions. Chemosphere 2022;305:135431. [CrossRef]
24. Biswas R, Sarkar A. Microbes:Key players of the arsenic biogeochemical cycle. In:Hurst CJ, editors. Microbial Metabolism of Metals and Metalloids. Advances in Environmental Microbiology. Cham:Springer;2022. 197-221. Available from:[CrossRef]
25. Darma A, Yang J, Bloem E, Mo?d?en K, Zandi P. Arsenic biotransformation and mobilization:The role of bacterial strains and other environmental variables. Environ Sci Pollut Res Int 2022;29:1763-87. [CrossRef]
26. Cao GH, Wang XF, Li ZD, Zhang X, Li XG, Gu W, et al. APanax notoginseng phosphate transporter, PnPht1;3, greatly contributes to phosphate and arsenate uptake. Funct Plant Biol 2022;49:259-71. [CrossRef]
27. Khan I, Awan SA, Rizwan M, Ali S, Zhang X, Huang L. Arsenic behavior in soil-plant system and its detoxification mechanisms in plants:A review. Environ Pollut 2021;286:117389. [CrossRef]
28. Kapaj S, Peterson H, Liber K, Bhattacharya P. Human health effects from chronic arsenic poisoning –A review. J Environ Sci Health A Tox Hazard Subst Environ Eng 2006;41:2399-428. [CrossRef]
29. Machiwal D, Jha MK. Characterising rainfall-groundwater dynamics in a hard-rock aquifer system using time series, geographic information system and geostatistical modelling. Hydrol Process 2014;28:2824-43. [CrossRef]
30. Cullen WR, Reimer KJ. Arsenic speciation in the environment. Chem Rev 1989;89:713-64. [CrossRef]
31. Fendorf S, Kocar BD. Biogeochemical processes controlling the fate and transport of arsenic. Implications for South and Southeast Asia. Adv Agron 2009;104:137-64. [CrossRef]
32. Mondal D, Rahman MM, Suman S, Sharma P, Siddique AB, Rahman MA, et al. Arsenic exposure from food exceeds that from drinking water in endemic area of Bihar, India. Sci Total Environ 2021;754:142082. [CrossRef]
33. Simeonov V, Stratis JA, Samara C, Zachariadis G, Voutsa D, Anthemidis A, et al. Assessment of the surface water quality in Northern Greece. Water Res 2003;37:4119-24. [CrossRef]
34. Modestin E, Devault DA, Baylet A, Massat F, Dolique F. Arsenic in Caribbean bivalves in the context of Sargassum beachings:A new risk for seafood consumers. Environ Monit Assess 2022;194:553. [CrossRef]
35. Perry H, Isphording W, Trigg C, Riedel R. Heavy metals in red crabs, Chaceon quinquedens, from the Gulf of Mexico. Mar Pollut Bull 2015;101:845-51. [CrossRef]
36. Chakraborti D, Rahman MM, Das B, Murrill M, Dey S, Chandra Mukherjee S, et al. Status of groundwater arsenic contamination in Bangladesh:A 14-year study report. Water Res 2010;44:5789-802. [CrossRef]
37. Su YH, McGrath SP, Zhao FJ. Rice is more efficient in arsenite uptake and translocation than wheat and barley. Plant Soil 2010;328:27-34. [CrossRef]
38. Cai K, Li C. Street dust heavy metal pollution source apportionment and sustainable management in A typical city-Shijiazhuang, China. Int J Environ Res Public Health 2019;16:2625. [CrossRef]
39. Tarvainen T, Reichel S, Müller I, Jordan I, Hube D, Eurola M, et al. Arsenic in agroecosystems under anthropogenic pressure in Germany and France compared to a geogenic As region in Finland. J Geochem Explor 2020;217:106606. [CrossRef]
40. Suriyagoda LD, Dittert K, Lambers H. Mechanism of arsenic uptake, translocation and plant resistance to accumulate arsenic in rice grains. Agric Ecosyst Environ 2018;253:23-37. [CrossRef]
41. Niazi NK, Hussain MM, Bibi I, Natasha N, Shahid M, Ali F, et al. The significance of eighteen rice genotypes on arsenic accumulation, physiological response and potential health risk. Sci Total Environ 2022;832:155004. [CrossRef]
42. Signes-Pastor AJ, Carey M, Meharg AA. Inorganic arsenic in rice-based products for infants and young children. Food Chem 2016;191:128-34. [CrossRef]
43. Rahman MA, Hasegawa H, Rahman MA, Rahman MM, Miah MA. Influence of cooking method on arsenic retention in cooked rice related to dietary exposure. Sci Total Environ 2006;370:51-60. [CrossRef]
44. Menon M, Dong W, Chen X, Hufton J, Rhodes EJ. Improved rice cooking approach to maximise arsenic removal while preserving nutrient elements. Sci Total Environ 2021;755:143341. [CrossRef]
45. Tong J, Guo H, Wei C. Arsenic contamination of the soil-wheat system irrigated with high arsenic groundwater in the Hetao Basin, Inner Mongolia, China. Sci Total Environ 2014;496:479-87. [CrossRef]
46. Kumar M, Rahman MM, Ramanathan AL, Naidu R. Arsenic and other elements in drinking water and dietary components from the middle Gangetic plain of Bihar, India:Health risk index. Sci Total Environ 2016;539:125-34. [CrossRef]
47. Kamrozzaman MM, Khan MA, Ahmed S, Sultana N, Quddus AF. Evaluation of five wheat varieties on arsenic contaminated soils of Bangladesh. Int J Appl Sci Biotechnol 2016;4:330-7. [CrossRef]
48. Shi GL, Lou LQ, Zhang S, Xia XW, Cai QS. Arsenic, copper, and zinc contamination in soil and wheat during coal mining, with assessment of health risks for the inhabitants of Huaibei, China. Environ Sci Pollut Res Int 2013;20:8435-45. [CrossRef]
49. Xiao X, Qin K, Sun X, Hui W, Yuan L, Wu L. Will wheat be damaged by heavy metals on exposure to coal fly ash?Atmos Pollut Res 2018;9:814-21. [CrossRef]
50. Rilwan U, Abbas AA, Abdulrahman H. Heavy metal contamination in swampy agricultural soils of Kokona, Nasarawa, Nigeria. Asian J Appl Chem Res 2020;5:28-33. [CrossRef]
51. Zheng C, Yang ZB, Xu XX, Cheng Z. Assessing the risk of human exposure to bioaccessible arsenic from total diet through market food consumption in Chengdu, China. Environ Geochem Health 2022;[CrossRef]
52. Natasha N, Shahid M, Khalid S, Niazi NK, Murtaza B, Ahmad N, et al. Health risks of arsenic buildup in soil and food crops after wastewater irrigation. Sci Total Environ 2021;772:145266. [CrossRef]
53. Zulkafflee NS, Redzuan NA, Selamat J, Ismail MR, Praveena SM, Razis AF. Evaluation of heavy metal contamination in paddy plants at the northern region of Malaysia using ICPMS and its risk assessment. Plants (Basel) 2020;10:3. [CrossRef]
54. Proshad R, Kormoker T, Islam MS, Chandra K. Potential health risk of heavy metals via consumption of rice and vegetables grown in the industrial areas of Bangladesh. Hum Ecol Risk Assessment 2020;26:921-43. [CrossRef]
55. Ng JC, Ciminelli V, Gasparon M, Caldeira C. Health risk apportionment of arsenic from multiple exposure pathways in Paracatu, a gold mining town in Brazil. Sci Total Environ 2019;673:36-43. [CrossRef]
56. Zhang Q, Wang S, Nan Z, Li Y, Zang F. Accumulation, fractionation, and risk assessment of mercury and arsenic in the soil-wheat system from the wastewater-irrigated soil in Baiyin, northwest China. Environ Sci Pollut Res Int 2018;25:14856-67. [CrossRef]
57. Althobiti RA, Sadiq NW, Beauchemin D. Realistic risk assessment of arsenic in rice. Food Chem 2018;257:230-6. [CrossRef]
58. Roya AQ, Ali MS. Heavy metals in rice samples on the Torbat-Heidarieh market, Iran. Food Addit Contam Part B Surveill 2017;10:59-63. [CrossRef]
59. Tenni D, Martin M, Barberis E, Beone GM, Miniotti E, Sodano M, et al. Total as and as speciation in Italian rice as related to producing areas and paddy soils properties. J Agric Food Chem 2017;65:3443-52. [CrossRef]
60. Alam MG, Snow ET, Tanaka A. Arsenic and heavy metal contamination of vegetables grown in Samta village, Bangladesh. Sci Total Environ 2003;308:83-96. [CrossRef]
61. Huq ME, Fahad S, Shao Z, Sarven MS, Khan IA, Alam M, et al. Arsenic in a groundwater environment in Bangladesh:Occurrence and mobilization. J Environ Manage 2020;262:110318. [CrossRef]
62. Qin J, Niu A, Liu Y, Lin C. Arsenic in leafy vegetable plants grown on mine water-contaminated soils:Uptake, human health risk and remedial effects of biochar. J Hazard Mater 2021;402:123488. [CrossRef]
63. Samal AC, Bhattacharya P, Biswas P, Maity JP, Bundschuh J, Santra SC. Variety-specific arsenic accumulation in 44 different rice cultivars (O. sativa L.) and human health risks due to co-exposure of arsenic-contaminated rice and drinking water. J Hazard Mater 2021;407:124804. [CrossRef]
64. Aslan S, Ozturk M, Demirbas A. Evaluation of arsenic and nutrients uptake of tomato plant at various arsenic concentrations of irrigation waters. Commun Soil Sci Plant Anal 2021;52:2388-400. [CrossRef]
65. Palma JM, Gupta DK, Corpas FJ. Metalloenzymes involved in the metabolism of reactive oxygen species and heavy metal stress. In:Gupta D, Corpas F, Palma J, editors. Heavy Metal Stress in Plants. Berlin, Heidelberg:Springer;2013. 1-17. Available from:[CrossRef]
66. Senanayake N, Mukherji A. Irrigating with Arsenic contaminated groundwater in West Bengal and Bangladesh:A review of interventions for mitigating adverse health and crop outcomes. Agric Water Manag 2014;135:90-9. [CrossRef]
67. Roychowdhury T, Uchino T, Tokunaga H, Ando M. Survey of arsenic in food composites from an arsenic-affected area of West Bengal, India. Food Chem Toxicol 2002;40:1611-21. [CrossRef]
68. Pandey D, Kehri HK, Zoomi I, Chaturvedi S, Chaudhary KL. In-vitroevaluation of arsenic accumulation and tolerance in some agricultural crops growing adjacent to the Ganga River. J Appl Biol Biotech 2022;10:10-7. [CrossRef]
69. Ye WL, Wood BA, Stroud JL, Andralojc PJ, Raab A, McGrath SP, et al. Arsenic speciation in phloem and xylem exudates of castor bean. Plant Physiol 2010;154:1505-13. [CrossRef]
70. Norton G, Deacon C, Mestrot A, Feldmann J, Jenkins P, Baskaran C, et al. Arsenic speciation and localization in horticultural produce grown in a historically impacted mining region. Environ Sci Technol 2013;47:6164-72. [CrossRef]
71. Meck ML, Mudimbu D, Davies TC. Accumulation of potentially harmful elements in edible parts of vegetables grown on two different geological substrates in Zimbabwe. J Geochem Explor 2020;208:106392. [CrossRef]
72. Cai LM, Wang QS, Luo J, Chen LG, Zhu RL, Wang S, et al. Heavy metal contamination and health risk assessment for children near a large Cu-smelter in central China. Sci Total Environ 2019;650:725-33. [CrossRef]
73. Obiora SC, Chukwu A, Chibuike G, Nwegbu AN. Potentially harmful elements and their health implications in cultivable soils and food crops around lead-zinc mines in Ishiagu, Southeastern Nigeria. J Geochem Explor 2019;204:289-96. [CrossRef]
74. Luo L, Shen Y, Wang X, Chu B, Xu T, Liu Y, et al. Phytoavailability, bioaccumulation, and human health risks of metal(loid) elements in an agroecosystem near a lead-zinc mine. Environ Sci Pollut Res Int 2018;25:24111-24. [CrossRef]
75. Khan ZI, Ugulu I, Ahmad K, Yasmeen S, Noorka IR, Mehmood N, et al. Assessment of trace metal and metalloid accumulation and human health risk from vegetables consumption through spinach and coriander specimens irrigated with wastewater. Bull Environ Contam Toxicol 2018;101:787-95. [CrossRef]
76. Ahmad K, Ashfaq A, Khan ZI, Bashir H, Sohail M, Mehmood N, et al. Metal accumulation in Raphanus sativus and Brassica rapa:An assessment of potential health risk for inhabitants in Punjab, Pakistan. Environ Sci Pollut Res Int 2018;25:16676-85. [CrossRef]
77. Tripathi RM, Raghunath R, Sastry VN, Krishnamoorthy TM. Daily intake of heavy metals by infants through milk and milk products. Sci Total Environ 1999;227:229-35. [CrossRef]
78. Ulman C, Gezer S, Anal O, Tore IR, Kirca U. Arsenic in human and cow's milk:A reflection of environmental pollution. Water Air Soil Pollut 1998;101:411-6. [CrossRef]
79. WHO. Indicators for Assessing Infant and Young Child Feeding Practices:Part 1 De?nition:Conclusions of a Consensus Meeting Held 6-8 November 2007 (in Washington DC, USA). Geneva, Switzerland:World Health Organization;2007. Available from:https://apps.who.int/iris/handle/10665/43895 [Last accessed on 2022 May 30].
80. Ballard O, Morrow AL. Human milk composition:Nutrients and bioactive factors. Pediatr Clin North Am 2013;60:49-74. [CrossRef]
81. Samanta G, Das D, Mandal BK, Chowdhury TR, Chakraborti D, Pal A, et al. Arsenic in the breast milk of lactating women in arsenic-affected areas of West Bengal, India and its effect on infants. J Environ Sci Health A Tox Hazard Subst Environ Eng 2007;42:1815-25. [CrossRef]
82. Oskarsson A, Palminger Hallén I, Sundberg J, Petersson GrawéK. Risk assessment in relation to neonatal metal exposure. Analyst 1998;123:19-23. [CrossRef]
83. Godswill AC, Somtochukwu IV, Ikechukwu A, Kate EC. Health benefits of micronutrients (Vitamins and Minerals) and their associated deficiency diseases:A systematic review. Int J Food Sci 2020;3:1-32. [CrossRef]
84. McDougald LR, Gilbert JM, Fuller L, Rotibi A, Xie M, Zhu G. How much does roxarsone contribute to coccidiosis control in broilers when used in combination with ionophores?J Appl Poult Res 1992;1:172-9. [CrossRef]
85. Nachman KE, Baron PA, Raber G, Francesconi KA, Navas-Acien A, Love DC. Roxarsone, inorganic arsenic, and other arsenic species in chicken:A U.S.-based market basket sample. Environ Health Perspect 2013;121:818-24. [CrossRef]
86. Das A, Joardar M, Chowdhury NR, De A, Mridha D, Roychowdhury T. Arsenic toxicity in livestock growing in arsenic endemic and control sites of West Bengal:Risk for human and environment. Environ Geochem Health 2021;43:3005-25. [CrossRef]
87. Carignan CC, Cottingham KL, Jackson BP, Farzan SF, Gandolfi AJ, Punshon T, et al. Estimated exposure to arsenic in breastfed and formula-fed infants in a United States cohort. Environ Health Perspect 2015;123:500-6. [CrossRef]
88. Chao HH, Guo CH, Huang CB, Chen PC, Li HC, Hsiung DY, et al. Arsenic, cadmium, lead, and aluminium concentrations in human milk at early stages of lactation. Pediatr Neonatol 2014;55:127-34. [CrossRef]
89. Gaxiola-Robles R, Labrada-Martagón V, Celis de la Rosa Ade J, Acosta-Vargas B, Méndez-Rodríguez LC, Zenteno-Savín T. Interaction between mercury (Hg), arsenic (As) and selenium (Se) affects the activity of glutathione S-transferase in breast milk;possible relationship with fish and sellfish intake. Nutr Hosp 2014;30:436-46.
90. Islam MS, Ahmed MK, Habibullah-Al-Mamun M, Islam KN, Ibrahim M, Masunaga S. Arsenic and lead in foods:A potential threat to human health in Bangladesh. Food Addit Contam Part A Chem Anal Control Expo Risk Assess 2014;31:1982-92. [CrossRef]
91. Björklund KL, Vahter M, Palm B, Grandér M, Lignell S, Berglund M. Metals and trace element concentrations in breast milk of first time healthy mothers:A biological monitoring study. Environ Health 2012;11:92. [CrossRef]
92. Sakamoto M, Chan HM, Domingo JL, Kubota M, Murata K. Changes in body burden of mercury, lead, arsenic, cadmium and selenium in infants during early lactation in comparison with placental transfer. Ecotoxicol Environ Saf 2012;84:179-84. [CrossRef]
93. Almeida AA, Lopes CM, Silva AM, Barrado E. Trace elements in human milk:Correlation with blood levels, inter-element correlations and changes in concentration during the first month of lactation. J Trace Elem Med Biol 2008;22:196-205. [CrossRef]
94. Islam MS, Ahmed MK, Habibullah-Al-Mamun M, Eaton DW. Arsenic in the food chain and assessment of population health risks in Bangladesh. Environ Syst Decis 2017;37:344-52. [CrossRef]
95. Mottalib MA, Zilani G, Suman TI, Ahmed T, Islam S. Assessment of trace metals in consumer chickens in Bangladesh. J Health Pollut 2018;8:181208. [CrossRef]
96. Ghosh A, Awal MA, Majumder S, Mostofa M, Khair A, Islam MZ, et al. Arsenic in eggs and excreta of laying hens in Bangladesh:A preliminary study. J Health Popul Nutr 2012;30:383-93. [CrossRef]
97. Bentley K, Soebandrio A. Arsenic and mercury concentrations in marine fish sourced from local fishermen and fish markets in mine-impacted communities in Ratatotok Sub-district, North Sulawesi, Indonesia. Mar Pollut Bull 2017;120:75-81. [CrossRef]
98. Navas-Acien A, Francesconi KA, Silbergeld EK, Guallar E. Seafood intake and urine concentrations of total arsenic, dimethylarsinate and arsenobetaine in the US population. Environ Res 2011;111:110-8. [CrossRef]
99. Has-Schön E, Bogut I, Vukovi?R, Galovi?D, Bogut A, Horvati?J. Distribution and age-related bioaccumulation of lead (Pb), mercury (Hg), cadmium (Cd), and arsenic (As) in tissues of common carp (Cyprinus carpio) and European catfish (Sylurus glanis) from the Buško Blato reservoir (Bosnia and Herzegovina). Chemosphere 2015;135:289-96. [CrossRef]
100. Zhang H, Schiewer S. Arsenic (V) Sorption on Crab Shell Based Chitosan. World Water and Environmental Resources Congress;2005. Available from:http://dx.doi.org/10.1061/40792(173)296 [Last accessed on 2022 May 30]. [CrossRef]
101. Zhang W, Miao AJ, Wang NX, Li C, Sha J, Jia J, et al. Arsenic bioaccumulation and biotransformation in aquatic organisms. Environ Int 2022;163:107221. [CrossRef]
102. Tsui MT, Wang WX. Biokinetics and tolerance development of toxic metals in Daphnia magna. Environ Toxicol Chem 2007;26:1023-32. [CrossRef]
103. Cao H, Wang Z, Meng J, Du M, Pan Y, Zhao Y, et al. Determination of arsenic in Chinese mitten crabs (Eriocheir sinensis):Effects of cooking and gastrointestinal digestion on food safety. Food Chem 2022;393:133345. [CrossRef]
104. PerellóG, Martí-Cid R, Llobet JM, Domingo JL. Effects of various cooking processes on the concentrations of arsenic, cadmium, mercury, and lead in foods. J Agric Food Chem 2008;56:11262-9. [CrossRef]
105. National Health Comission of the PRC. National Standards for the Food Safety. PRC, NHCo., editors. GB 2762-2017, National Food Safety Standard Maximum Levels of Contaminants in Food. Beijing, China:Standards Press of China;2016.
106. Birgisdottir BE, Knutsen HK, Haugen M, Gjelstad IM, Jenssen MT, Ellingsen DG, et al. Essential and toxic element concentrations in blood and urine and their associations with diet:Results from a Norwegian population study including high-consumers of seafood and game. Sci Total Environ 2013;463-464:836-44. [CrossRef]
107. Miklav?i?A, Casetta A, Snoj Tratnik J, Mazej D, Krsnik M, Mariuz M, et al. Mercury, arsenic and selenium exposure levels in relation to fish consumption in the Mediterranean area. Environ Res 2013;120:7-17. [CrossRef]
108. Steckert LD, Furtado WE, Jerônimo GT, Pereira SA, Jesus GF, Mouriño JL, et al. Trace elements and microbiological parameters in farmed Nile tilapia with emphasis on muscle, water, sediment and feed. J Environ Sci Health B 2019;54:237-46. [CrossRef]
109. Di Bella C, Traina A, GiosuèC, Carpintieri D, Lo Dico GM, Bellante A, et al. Heavy metals and PAHs in meat, milk, and seafood from augusta area (Southern Italy):Contamination levels, dietary intake, and human exposure assessment. Front Public Health 2020;8:273. [CrossRef]
110. Yang L, Guo M, Xin C, Ren X, Wang L, Liu Y. Comparison of trace element concentrations in freshwater fish and marine fish consumed in Shandong Province, China, and their potential risks to human health. Mar Pollut Bull 2021;165:112114. [CrossRef]
111. WHO. EHC 224. Arsenic and Arsenic Compounds. 2nd ed. Geneva, Switzerland:World Health Organization;2001. Available from:https://apps.who.int/iris/handle/10665/42366 [Last accessed on 2022 May 30].
112. NRC. Arsenic in Drinking Water:2001 Update. Washington, DC:National Academy Press;2001.
113. Ahmadi M, Akhbarizadeh R, Haghighifard NJ, Barzegar G, Jorfi S. Geochemical determination and pollution assessment of heavy metals in agricultural soils of south western of Iran. J Environ Health Sci Eng 2019;17:657-69. [CrossRef]
114. Alamdar A, Eqani SA, Hanif N, Ali SM, Fasola M, Bokhari H, et al. Human exposure to trace metals and arsenic via consumption of fish from river Chenab, Pakistan and associated health risks. Chemosphere 2017;168:1004-12. [CrossRef]
115. Devi R, Kaur T, Kour D, Hricovec M, Mohan R, Yadav N, et al. Microbes-mediated alleviation of heavy metal stress in crops:Current research and future challenges. J Appl Biol Biotech 2022;10 Suppl 2:25-37. [CrossRef]