1. INTRODUCTION
Geographical regions positioned far from one another possess diverse climatic conditions which thereby impact the production of plant’s physiology. An extensive series of environmental aspects change with the varying elevation, for example, plants in high-altitude surroundings are exposed to numerous environmental strains, mainly in temperature and radiation extremes [1]. Elevation could be the main predictor of changes in phytochemical defense strategy of plants [2]. Altitude, in relation to several climatic trends, is accountable for adjustment in the biochemical and biological properties of the plant and also in determination of the phytochemical composition of the plant [3]. The temperature, pressure, and light regimes under which the plants grow affect various the plant attributes [4-6]. For instance, plants at higher altitudes face higher amount of light intensities as well as lower mean temperatures, suggesting that they might have established capability to prevent themselves from the damage created by factors such as chilling and photo destruction [7-10]. Provenance variations are significant in a wide range of traits in plants [11,12]. Variations could be beneficial or unprofitable to the plant [13]. Morphological and physiological variations can be associated with change in habitat conditions at higher elevation sites [14-17]. Along the challenging environmental gradients, plants modify their biological performances to indulge into a certain environmental situation [18]. For instance, in a study carried on different medicinal herbs, growing along an elevational gradient (3,000−4,600 m) in China, it was observed that the adaptive strategy of plants to adjust in environment was reflected by variation in their leaf traits, namely, leaf thickness, stomatal density, and mesophyll tissue thickness, at different altitudes thereby suggesting morphological and anatomical plasticity [19]. In a study carried in Italy, significant differences were observed in morphological and physiological traits of Crepis pygmaea and Isatis apennina as a result of differences occurred in their elevations [20]. A study of M. polymorpha revealed that several characteristics such as ecophysiological behavior and anatomical features were chiefly prompted by the environment and the other characteristics such as leaf size and internode length were seen decreasing with rise in elevation and were retained even when grown in similar environmental conditions suggesting alteration in their genetic background [21]. Plant develops well in its favorable environmental conditions. In a study, O. vulgare population exhibited larger and thicker leaves with vastly developed internal tissues, namely, palisade and spongy parenchymas and also the photochemical efficacy of PS II appeared to be highest in the mid altitude where environmental conditions were more favorable [22]. In a study carried on roots of Desmodium gangeticum L., the quantity of lupeol was estimated by HPTLC densitometric quantification and observed to be present in high amounts in plants that belonged to high altitude [23]. Significant variations in phenols, valerenic acid, and antioxidant activity in aerial and root portions were observed in inhabitants of Valeriana jatamansi sampled from Uttarakhand, West Himalaya [24]. These phytochemicals serve as a major source for pharmaceutical products [25]. In a study done on Adhatoda zeylanica in different phytogeographical regions of South India, four distinctive morphotypes were identified based and they exhibited significant variation in quantitative their vasicine content [26]. Plants may counter multiple stress conditions in various ways [27]. Explicitly, the plants are challenged to fight surplus abiotic and biotic stresses operating at different elevations and the worthwhile adaptation of plants to their sessile lifestyle can be ascribed to their capability to adapt and respond to various types of stresses. In this perspective, it is of utmost importance to highlight physiological mechanisms adapted by plants as a response to changing altitudes and coping with environmental stress.
2. MATERIALS AND METHODS
2.1. Study Site
The present study was performed on Justicia adhatoda L. which is an evergreen sub herbaceous bush mostly distributed up to altitude of 1300 m above sea level. The present study was done for two consecutive years in nine different provenances selected on the basis of different altitudes of Jammu, namely, 330 m, 336 m, 358 m, 661 m, 688 m, 696 m, 1302 m, 1311 m, and 1330 m (masl). The sampling was done from three different sites of each provenance. In case of morphological analysis, measurements of five plants from each sampling site were taken. The collected plant specimens were verified from Botanical Survey of India, Dehradun and submitted in the herbarium of Shoolini University under the voucher numbers, namely, SUBMS/BOT-4827 (Akhnoor, 330 m), SUBMS/BOT-4800 (Vijaypur, 336 m), SUBMS/BOT-4828 (Samba, 358 m) SUBMS/BOT-4801 (Harotkot, 688 m), SUBMS/BOT-4830 (Sarangdhar, 661 m), SUBMS/BOT-4829 (Salal, 696 m), SUBMS/BOT-4831 (Bashat, 1302 m), SUBMS/BOT-4832 (Mantalai, 1311 m), and SUBMS/BOT-4802 (Khetriar, 1330 m).
2.2. Morphological Analysis
To study the morphological variability among plants, following growth characteristics, namely, plant height, crown spread, inflorescence size, and leaf length as well as leaf width were measured and expressed in centimeters whereas number of leaves, number of branches, and number of inflorescences were counted.
2.2.1. Leaf area
To measure the leaf area, protocol described by Green-armytage [28] was followed, the collected leaves were placed on a graph paper having 1 cm2 square grid boxes and then boundary was outlined. After that following formula was applied:
Leaf area = No. of grids covered + No. of grids incompletely covered/2
2.2.2. Crown spread
The average crown spread was measured by the method described by Blozan [29]. Measurements of crown from both the directions of plant, that is, longest spread (A1) and longest cross spread (A2) were taken and average was determined.
Average crown spread = (A1 + A2)/2
2.3. Biochemical Analysis
For biochemical analysis, the leaves were hand harvested from each plant. Fifth and sixth mature leaves from the top of the plant were taken. Leaves were collected from all the four directions of each plant. The collected leaves were cleaned and shade dried and after that, the composite leaf samples were stored and analyzed in the laboratory for different parameters using standard techniques given below:
2.3.1. Carbohydrate content
Carbohydrates were estimated by following protocol of Thangaraj [30]. 100 mg of the leaf sample was placed in a boiling water bath to heat it along with 2.5 N hydrochloric acid for 3 h. The amount was kept 5 ml. After that, sodium carbonate was added to it to make its total volume up to 100 ml. The contents were further centrifuged at 10,000 rpm for 5 min. 4 ml of anthrone reagent was poured to the collected supernatant after centrifugation. On the other hand, different concentrations of standards were prepared with the help of glucose solution and it was treated with the same set of chemicals expect the leaf sample. All the contents were heated at least for 8 min inside the water bath and the absorbance was recorded at 630 nm in a spectrophotometer.
2.3.2. Protein content
The protein content was estimated by following Lowry’s method [31]. Leaf sample (100 mg) was ground well in 1−5 ml of the phosphate buffer. After that the centrifugation of the sample was performed for 5 min and at 7000 rpm. To the 1 ml of collected supernatant, 2 ml of alkaline copper solution was added and mixed. After that, 0.2 ml of Folin-Ciocalteu reagent was poured to it and the solutions were positioned at room temperature for incubation in the dark and the process took 30 min. OD was taken in spectrophotometer at 660 nm. A standard graph was drawn using BSA.
2.3.3. Amino acids
Amino acids were estimated by following protocol of Moore and Stein [32]. Leaf sample (100 mg) was taken and ground fine with acid washed sand. 2 ml of 80% ethanol was added to it. Centrifugation was performed for 5 min and at 7000 rpm. To 100 μl of extract, 900 μl of ninhydrin solution, and 1 ml distilled water was added and the contents were mixed. The solution was left for 15 min until the bluish-purple color appeared. OD was recorded in spectrophotometer taking lecine as standard at 570 nm.
2.3.4. Reducing sugar
The reducing sugars were estimated by the method described by Usunobun and Okolie [33]. 100 mg of powdered leaf sample was dissolved in 5 ml distilled water and thereafter filtered. The reducing sugar content was assayed by adding 2 ml of the 3,5-dinitrosalicyclic acid reagent in 1 ml of the sample. The dinitrosalicyclic reagent was made by adding 1.8 g of 3,5-dinitrosalicyclic acid in 20 ml of 1.0 M NaOH followed by further addition of 60 ml distilled water. After heating in water bath for 5 min and the solution was cooled swiftly. The absorbance was taken at 540 nm in a spectrophotometer. Glucose served as standard.
2.3.5. Total sugar
The total sugar content was estimated by protocol given by Jain [34]. To 1 ml of the leaf extract, 1 ml of 5% phenol solution was added. The contents were further mixed with 5 ml of 96% sulfuric acid and left for 10 min. The solution was heated at 35°C in a water bath for 20 min. Its absorbance was taken in a colorimeter at 490 nm. Glucose served as standard solution.
2.3.6. Non-reducing sugar
Non reducing sugars were estimated by following the method of Otache [35]. It was estimated by subtracting the reducing sugars from that of the total sugars.
2.3.7. Crude fiber
Crude fiber was estimated by following the protocol of Bosha [36]. 2 g of leaf sample was taken and 1.25% sulfuric acid solution (100 ml) was added to it and further heated for 30 min. The contents were then filtered and 1.25% NaOH (100 ml), 1 % HNO3 solution was added. The residue obtained was heated to 105°C and weighed (M1). It was then treated at 55°C in a muffle furnace for 5 h. The ash was obtained and it was weighed (M2). The % crude fiber was calculated as:
Crude fiber (%) = 100 × (M2-M1)/weight of original sample
2.3.8. Crude fat
Crude fat was estimated by following the method of A.O.A.C. [37]. 5 g of dry leaf sample was extracted with petroleum ether in soxhlet apparatus. The extract obtained was further cooled and weighed. Washings with petroleum ether were also given 4−5 times. The contents were repeatedly dried and weight until the difference between first and last readings was <1 mg. Crude fat was estimated as:
Crude fat (%) = 100 × weight of flask containing dried extract − weight of flask/weight of obtained dried sample
2.3.9. Ash content
Ash content was measured by the protocol given by Kumar [38]. The leaf sample was weighed and heated at 600°C in muffle furnace for 5 h. The ash obtained was cooled swiftly and weighed. This procedure was subsequently repeated unless the weight of obtained ash became constant.
Ash (%) = 100 × weight of ash/weight of sample
2.3.10. Moisture content
The moisture content was estimated by following standard protocol of Kumar [38]. The fresh leaves were weighed after collection (B1) and then leaves were kept overnight at 150°C in an oven for drying. After that, the samples were cooled and weighed (B2).
Moisture (%) = 100 × (B1−B2)/B1
2.3.11. Potassium and sodium
Potassium and sodium were determined by following the protocol of Singh [39]. To 500 mg leaf sample, 10 ml of conc. HNO3 was added and left overnight. After that, 2−3 ml of HClO4 were also added after pre-digestion and the solution was kept on hot plate at 200°C. The solution was filtered using Whatman no. 42 filter paper and washings with distilled water were also given. NaCl and KCl were taken as standards and the concentrations were determined using flame photometer.
2.3.12. Calcium
Calcium was determined by following protocol of Oser [40]. 25 ml of ash solution was prepared by dry ashing of the leaf samples and 150 ml of water was added to it. Some amount of methyl red indicator was poured to it and the contents were neutralized awaiting the pale pink color turning to yellow. The contents were heated and 10 ml of ammonium oxalate was added which was left for boiling again for 5 min. Further, glacial acetic acid was added and the solution was filtered. Washings with warm water were given. 5−10 ml of dilute H2SO4 was also added and heated at 70°C. Finally, the obtained solution was titrated against 0.01 N KMnO4 to a permanent pale pink color.
Calcium mg/100 mg = Titre value × Normality of KMnO4 × 0.2004 × total volume of ash solution × 100/ml of ash solution taken for estimation× weight of sample taken for ashing.
2.3.13. Alkaloid content
Alkaloids were determined by following method of Harborne [41]. 0.5 g leaf sample was taken (D1) and was extracted with 50 ml of 10% acetic acid. The extract was filtered and transferred to a China dish. It was further boiled on a water bath for 2 h to reduce its volume. To the extract, concentrated ammonium hydroxide was added until the precipitates were formed. Further, washings with 1% ammonium hydroxide were given and the extract was dried at 80°C in oven. The content left at last was alkaloids (D2).
Alkaloid (%) = D2/D1 × 100
2.3.14. Flavonoid content
Total flavonoid content was determined using the protocol of Zao [42]. 1 ml leaf extract prepared in methanol and distilled water (4 ml) was added which was followed by further addition of 5% sodium nitrite (1 ml). After 5 min, 1 ml of 10 % aluminum chloride and 8 ml of sodium hydroxide were also added to the mixture. In this method, the aluminum chloride formed a complex with hydroxyl groups of flavonoids present in the sample and orange yellowish color was developed. Absorbance of that mixture was taken using spectrophotometer at 510 nm against blank containing all reagents except plant extract. The calibration curve of rutin was prepared to find out the flavonoid content.
2.3.15. Phenolic content
Phenolic content was determined by following method of Sethi and Sharma [43]. 1 ml leaf extract prepared in ethanol was taken and 1 ml of Folin-Ciocalteu reagent was added in it. 20% sodium carbonate solution (2 ml) was also added to it. After shaking the contents of the solution, it was boiled for 2 min and cooled swiftly. 25 ml of distilled water was added and O.D was taken in spectrophotometer at 750 nm. In the same way, gallic acid which was taken as a standard phenol was processed and O.D was taken. The blank solution was prepared by all reagents except the plant extract. Finally, total amount of phenol was calculated referring the O.D of standard phenol with standard curve.
2.3.16. Tannin content
Tannin content was estimated by following the protocol of Saxena [44]. 100 mg of the powdered leaf material was weighed and 1 ml of distilled water was added to it. The material was heated and centrifuged for a period of 20 min and at 2000 rpm and the supernatant was taken. Folin-Denis reagent (60 μl) and sodium carbonate solution (100 μl) were added and again diluted. For the preparation of standard curve, standard tannic acid solution was made. The absorbance was taken after 30 min and at 700 nm.
2.3.17. Saponin content
Saponins were determined by following the method described by Mir [45]. 5 g of leaf sample was taken (E1) and 20% of 50 ml ethanol was added to it. It was then heated for about 4 h at a temperature of 55°C and constant stirring was done. The volume of filtered extract was reduced up to 40 ml by keeping it at a temperature of 90°C. After that, 5 ml diethyl ether was added to it in a separating funnel and shaken well. The aqueous layer was recovered and ether layer was removed. Added to it n-butanol (15 ml). The contents were washed by 2.5 ml of 5% aqueous NaCl. After that, the contents of the solution were evaporated and the residue was obtained and weighed (E2).
Total saponin (%) = E2/E1 × 100
2.4. Statistical Analysis
All the data were subjected to two-way ANOVA analysis and Bonferroni multiple comparison test.
3. RESULTS AND DISCUSSION
In morphological analysis, there was a significant change in plant height, the plants from high altitudes were comparatively short in size [Figure 1]. The maximum plant height was recorded in mid altitude (661 masl), namely, 169.6 cm and minimum at 1311 masl, that is, 13.1 cm. Accordingly, crown spread also decreased with increasing altitude with maximum value, namely, 205.2 cm at 661 masl and minimum value at 1311 masl (139.4 cm) [Figure 1]. Plant’s capability to adjust to the troubling environmental circumstances can be related to the morphological or physiological properties displayed by its organs [46-48]. The results reveal that altitudinal gradients and concomitant variables hold a significant impact on plant’s height. The height is a crucial component of a plant, various traits such as ability to compete for light, metabolic rate, and carbon gain strategy are dependent on the height of a plant [49]. Low altitude plants have to tolerate the high temperature and dry climatic conditions, harsh radiations, and limited precipitation whereas high altitude plants deal with low temperatures, high irradiance, etc., [50]. This significant reduction is in agreement with several other studies that have found decreasing plant height along environmental gradients [51] as higher elevations are often considered more stressful for plant growth and survival. Nutritional requirements and competition pose a remark on plants of all the sizes for instance the competition for light possess a noteworthy consequence on the growth of plants. In a study, growth of mountain beech trees was affected because of the competition for light and nutrients, tree growth dropped with increasing elevation [52]. This can bargain with the fact that trees become undersized at high elevations [53,54]. However, short stature of plants is also known to possess some benefits for instance, provides protection against stresses like winds [55-57]. Reduction in size is also considered as an important policy of plants at high altitude to endure stress, namely, reduced nutrient availability and low temperature. Reduction in cell size and intercellular spaces that ultimately led to reduction in whole plant size has been observed at high altitudes [58-61]. The decrease in temperature with elevation adds to less availability of prerequisite heat that is to be utilized by plants during the period of short summer and as a consequence of which the rates of many temperature dependent reactions get hindered [62]. The small stature could also be a genetic adaptation of the plant [63] and it could be possible that when a heat tolerant plant like Justicia adhatoda L. grows under low temperature at high altitudinal regions, it retains the changes in its morphological and physiological features to thrive in unsuitable conditions. Thus, the small height could be a strategy to cope up with the prevailing environmental conditions for instance staying near the warmer microclimatic conditions close to the ground and safeguard from the wind [64]. The crown spread also varied and showed a significant reduction at the higher elevation in comparison to the lower ones. Maximum value was observed in 661 masl, that is, 205.2 cm and minimum value was observed in 1311 masl, that is, 139.4 cm. Similarly, in a study, noticeable variations in the morphological traits of Boswellia trees in Eritrea were observed. Tree height, crown diameter, and depth all decreased with increasing altitude [65,66]. The number of primary branches was found to be significantly low at 1330 masl, that is, 94.4 and high at 336 masl, that is, 139.8 and maximum number of secondary branches were found at 1330 masl, namely, 122.6. No significant change was observed in number of tertiary branches [Table 1]. With decreasing height, mechanical damage in branches and trunks of the trees was also observed in some studies suggesting that regardless of the thermal conditions, mechanical damage for instance, by strong winds also contributes to these variations [67]. Wind, although gentle but can obligate an important effect on plant’s height, plant’s growth can also be chiefly influenced by the amount of water available in the soil [68]. Number of inflorescence were observed to be highest in 688 masl, namely, 148.2 whereas 1330 masl had lowest number of inflorescence count, that is, 92.4. This significant reduction in the total number of inflorescences at high altitude is in agreement with the study done on Polylepis indicana where the number of inflorescences showed a marginally significant decrease at higher altitude [69]. The leaf width and inflorescence size showed statistically significant variation [Table 1] [70-79]. The amount of solar radiation captured by plants is intensely affected by the leaf morphology [73]. The previous studies have revealed decrease in leaf size with the increasing altitude [74,75]. In a study, leaf traits of Pleioblastus amarus were evaluated along varying altitudes where leaf length and width decreased significantly with increasing altitude and increase in environmental stresses, that is, solar radiation, nutrient loss, low temperatures, and other abiotic pressure were held responsible for those variations [70]. In a study, all the morphological characters were affected with increasing altitude but a few of them were retained when the plants were maintained in their common environment thereby indicating heritable variation for those morphological characters [76]. The number of leaves decreased as altitude increased, maximum number of leaves were found at 358 masl, namely, 2855 and minimum number of leaves were present at 661 masl, namely, 1566.2 [Table 1]. Several other studies have found decrease in number of leaves [19,74]. The environment in which the leaves are produced has a significant influence on leaf morphology [77,78].
Table 1: Variation in morphological attributes of Justicia adhatoda L. growing in different altitudes.
| Morphological analysis | Year | Study Site 1 (330 masl) | Study Site 2 (336 masl) | Study Site 3 (358 masl) | Study Site 4 (661 masl) | Study Site 5 (688 masl) | Study Site 6 (696 masl) | Study Site 7 (1302 masl) | Study Site 8 (1311 masl) | Study Site 9 (1330 masl) |
|---|---|---|---|---|---|---|---|---|---|---|
| Plant height (cm) | 1st Year | 143.6±16.5 | 154±12.94 | 152.4±9.96 | 169.6±19.93 | 163.2±7.66 | 159.2±7.46 | 140±19.65 | 138.8±7.04 | 143.2±4.14 |
| 2nd Year | 156.8±9.88 | 157.8±7.85 | 149.4±8.44 | 155.2±9.038 | 160.8±6.22 | 159.6±9.07 | 149.4±10.784 | 131.4±9.343 | 134.2±9.36 | |
| Crown spread (cm) | 1st Year | 198.6±3.2 | 200.8±1.64 | 178.2±30.17 | 205.2±33.83 | 198.6±6.76 | 177.8±19.4 | 164.6±13.84 | 152.2±16.39 | 155.8±9.06 |
| 2nd Year | 180.2±27.26 | 190.6±8.2 | 169.8±6.09 | 178.4±30.07 | 193±8.18 | 184±19.22 | 179.4±20.18 | 139.4±10.06 | 147.6±5.94 | |
| Primary branches | 1st Year | 95.6±19.26 | 139.8±32.08 | 111.2±28.78 | 112.8±13.62 | 123.6±19.88 | 139±15.5 | 100.2±23.56 | 101.4±25.10 | 118±15.92 |
| 2nd Year | 133±23.01 | 138.6±17.41 | 98.8±17.07 | 113±23.28 | 119.8±13.73 | 133.4±19.69 | 137.6±11.63 | 118.8±14.61 | 94.4±13.22 | |
| Secondary branches | 1st Year | 127±30.14 | 156.4±43.04 | 130.2±23.38 | 134.4±25.85 | 141.8±20.71 | 164.6±41.48 | 135.6±23.89 | 123.8±22.39 | 129.8±25.38 |
| 2nd Year | 150.2±38.44 | 156±26.58 | 133.2±14.30 | 135.8±13.49 | 136.8±14.41 | 149.6±13.55 | 150±22.80 | 135.6±14.87 | 122.6±12.17 | |
| Tertiary branches | 1st Year | 47.8±10.96 | 46.6±14.92 | 37.8±12.55 | 40±9.08 | 47.6±10.59 | 35.6±15.46 | 27.8±14.34 | 35.8±18.45 | 39±9.92 |
| 2nd Year | 52±18.22 | 47.8±11.47 | 43.2±7.04 | 46.4±9.93 | 51.8±12.09 | 43±8.15 | 35±13.61 | 40.8±17.49 | 33.8±2.77 | |
| Total number of leaves | 1st Year | 1962.4±661.79 | 2247±384.72 | 2855±760.73 | 1762.8±233.21 | 2486.6±776.08 | 2420.8±598.16 | 1852.8±444.53 | 1749.2±379.99 | 2328±558.87 |
| 2nd Year | 1993.8±406.55 | 1963.8±286.28 | 1750±222.50 | 1566.2±374.42 | 2087.8±444.21 | 1994.2±537.94 | 2064.8±67.18 | 1754.6±293.30 | 1728.4±198.58 | |
| Leaf length (cm) | 1st Year | 12.96±0.76 | 13.695±0.49 | 13.63±0.82 | 13.705±1.29 | 13.975±1.25 | 14.525±0.52 | 13.435±1.41 | 13.87±1.03 | 14.3±1.38 |
| 2nd Year | 14.301±1.4 | 13.84±0.32 | 14±0.67 | 14.1±0.71 | 14.175±0.85 | 14.105±0.47 | 14.24±0.88 | 13.785±1.09 | 14.11±0.71 | |
| Leaf width (cm) | 1st Year | 5.54±0.50 | 5.17±0.25 | 5.18±0.26 | 4.805±0.79 | 4.89±0.72 | 5.08±0.45 | 4.69±0.59 | 4.78±0.33 | 5.035±0.29 |
| 2nd Year | 5.143±0.23 | 5.18±0.15 | 4.931±0.29 | 5.045±0.29 | 5.29±0.21 | 4.945±0.13 | 5.1248±0.24 | 5.079±0.17 | 5.175±0.20 | |
| Leaf area | 1st Year | 47±1.58 | 44.2±4.08 | 47.8±3.96 | 47.2±3.34 | 44.4±1.51 | 45±3.7 | 41.8±4.65 | 45.9±4.44 | 43.2±2.16 |
| 2nd Year | 45.6±1.14 | 47±3.93 | 45.8±0.49 | 45.8±0.44 | 46±0.70 | 43.8±4.38 | 44.8±4.54 | 42.7±5.02 | 44.6±1.14 | |
| Inflorescence size (cm) | 1st Year | 17.365±2.49 | 12.6±0.55 | 13.19±0.80 | 13.235±1.24 | 13.71±0.69 | 13.885±0.61 | 12.5±1.03 | 13.215±0.41 | 13.355±0.98 |
| 2nd Year | 14.035±1.53 | 12.274±0.48 | 13.24±0.57 | 13.11±0.45 | 12.04±3.75 | 13.32±1.123 | 13.385±0.46 | 12.605±0.75 | 13.81±0.93 | |
| Total number of Inflorescences | 1st Year | 100.8±16.48 | 99.4±26.08 | 106.8±15.44 | 140.6±13.68 | 148.2±50.34 | 108.8±16.63 | 96.6±14.74 | 107.2±10.42 | 92.4±38.82 |
| 2nd Year | 115.6±18.60 | 120.4±15.46 | 107.4±25.16 | 108.6±20.41 | 132.4±8.20 | 111.8±14.54 | 115.2±17.09 | 111.6±12.97 | 104.8±11.81 |
Data is represented as mean±standard deviation.
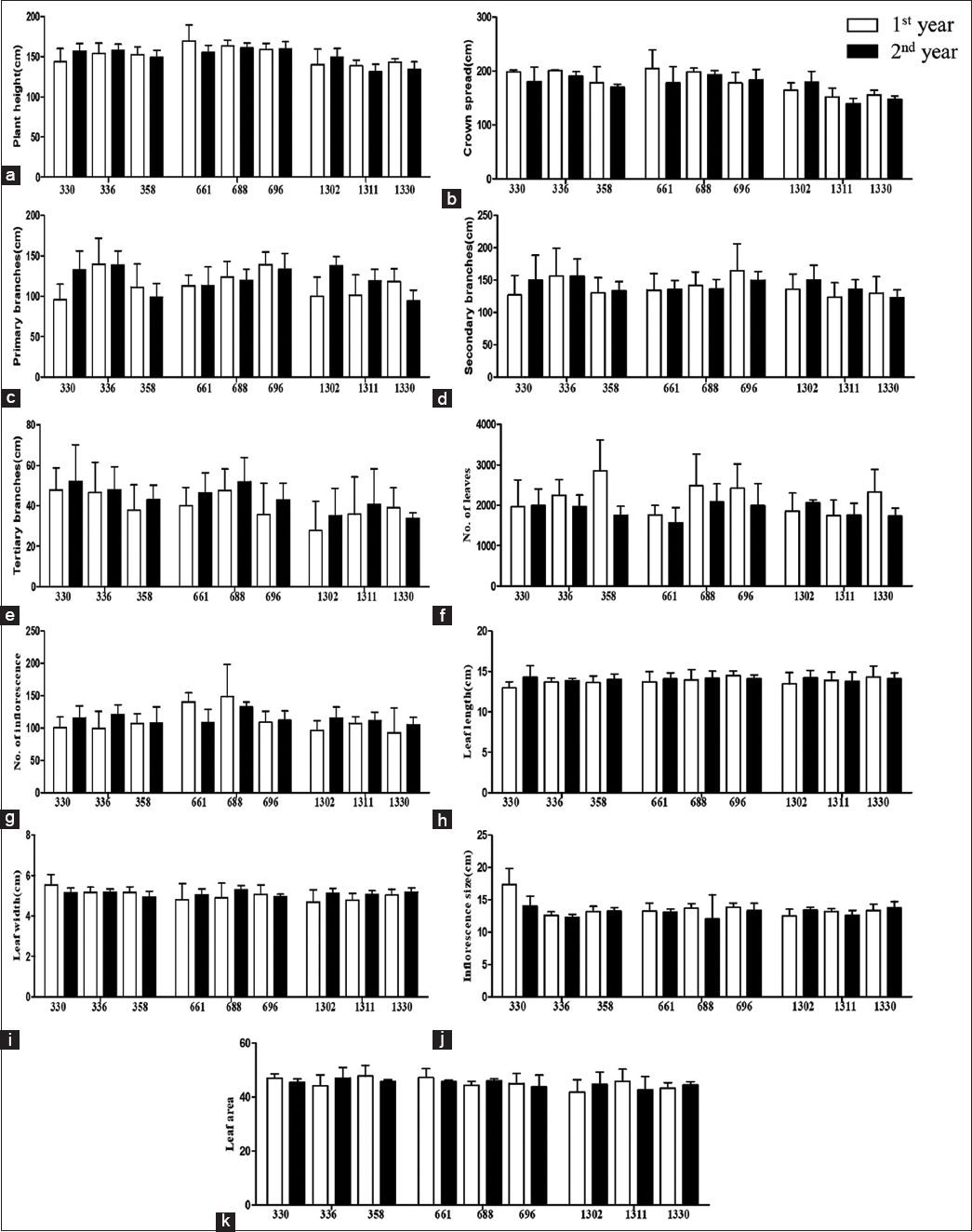 | Figure 1: Plant height (a), crown spread (b), primary branches (c), secondary branches (d), tertiary branches (e), number of leaves (f), number of inflorescence (g), leaf length (h), leaf width (i), inflorescence size (j), and leaf area (k) of Justicia adhatoda L. growing in different altitudes of Jammu. [Click here to view] |
Total carbohydrates were seen decreasing significantly with increasing altitude for both the years with maximum value at 330 masl (31.465 mg/g) during 1st year and minimum value at 1311 masl (19.264 mg/g) during 2nd year, that is, the carbohydrate content was observed to be highest in low and mid elevations and lowest in high elevation [Table 2] [Figure 2]. However, the previous studies have also reported that plants have adopted to stress by altering several metabolic pathways for instance, carbohydrate synthesis and increased sugar content that act in osmoregulation, accumulation of sucrose during cold stress [60,79,80]. Hence, it is possible that the amount of carbohydrates is less which are to be invested for metabolic processes, hence, altering plant growth at high elevations.
Table 2: Variation in physico and phytochemical elements of Justicia adhatoda L. growing in different altitudes.
| Biochemical Analysis | Year | Study Site 1 (330 masl) | Study Site 2 (336 masl) | Study Site 3 (358 masl) | Study Site 4 (661 masl) | Study Site 5 (688 masl) | Study Site 6 (696 masl) | Study Site 7 (1302 masl) | Study Site 8 (1311 masl) | Study Site 9 (1330 masl) |
|---|---|---|---|---|---|---|---|---|---|---|
| Carbohydrate (mg/g) | 1st Year | 31.4±4.5 | 27.1±3.13 | 30±4.88 | 29.6±5.85 | 20.8±3.96 | 21±1.33 | 20.3±2.94 | 21.3±2.29 | 19.4±0.99 |
| 2nd Year | 30.5±1.57 | 28±2.34 | 25.8±2.9 | 28.6±1.32 | 24.7±4.08 | 27.18±3.08 | 23±1.32 | 19.2±1.81 | 21±5.28 | |
| Protein (mg/g) | 1st Year | 19.4±0.96 | 17.6±2.37 | 15.9±1.87 | 18±1.94 | 16.7±3.64 | 18.6±0.68 | 16.5±2.28 | 15.9±1.37 | 17.1±2.83 |
| 2nd Year | 17.8±0.67 | 15.7±1.14 | 19.1±2.07 | 16.9±1.96 | 17.3±1.33 | 17.8±1.75 | 16.8±0.78 | 15.1±5.23 | 17.2±1.41 | |
| Crude Fiber (%) | 1st Year | 5.3±0.61 | 5.5±0.55 | 5.6±0.75 | 6.1±0.79 | 5.4±1.15 | 6.2±0.91 | 5.4±0.7 | 5.7±0.9 | 5.5±0.77 |
| 2nd Year | 5.6±0.8 | 5.9±1.02 | 4.9±0.58 | 5.03±0.66 | 5.1±1.34 | 5.9±0.62 | 5.5±0.77 | 4.7±0.9 | 5.6±0.57 | |
| Ash Content (%) | 1st Year | 3.7±0.45 | 3.6±1.03 | 3.4±1.1 | 4.1±0.75 | 4.3±1.25 | 3.7±0.11 | 5.4±0.3 | 4.3±1.17 | 4.9±0.23 |
| 2nd Year | 4.6±0.76 | 5.4±0.8 | 4.6±0.65 | 5.2±0.55 | 4.7±0.45 | 4.9±0.55 | 5±0.72 | 4.3±0.2 | 4.93±0.55 | |
| Moisture content (%) | 1st Year | 4.7±0.45 | 5.3±0.5 | 4.9±0.2 | 5.8±0.83 | 5.7±0.9 | 6.1±0.6 | 5.6±0.45 | 4.4±0.69 | 5±0.23 |
| 2nd Year | 5±1.41 | 5.3±1.24 | 5.8±0.83 | 4.4±1.85 | 5±0.66 | 3.7±0.9 | 3.8±1.15 | 5.3±2.57 | 5.7±0.72 | |
| Crude fat (%) | 1st Year | 5.3±0.61 | 5.5±0.55 | 5.6±0.75 | 6.1±0.79 | 5.4±1.15 | 6.2±0.91 | 5.4±0.7 | 5.7±0.9 | 5.5±0.75 |
| 2nd Year | 5.6±0.8 | 5.9±1.02 | 4.9±0.5 | 5±0.66 | 5.1±1.34 | 5.9±0.62 | 5.5±0.77 | 4.7±0.9 | 5.6±0.57 | |
| Reducing sugar (mg/g) | 1st Year | 32±3.33 | 38.4±0.96 | 39.2±4 | 30±2.61 | 33.1±2.35 | 35.2±6.88 | 26.1±2.07 | 33.2±6.12 | 35.1±9.94 |
| 2nd Year | 40.3±5.34 | 38.6±2.17 | 43.9±1.85 | 34.2±6.14 | 33.9±2.5 | 36.3±10.2 | 27±9.8 | 26.6±2.12 | 24.6±2.88 | |
| Non-reducing sugar (mg/g) | 1st Year | 20.8±1.52 | 20.5±2.4 | 21.3±2.67 | 19.1±6.49 | 19.7±8.09 | 15±4.15 | 11.2±3.18 | 12.7±12.66 | 10.6±11.79 |
| 2nd Year | 20.7±6.98 | 13.2±2.46 | 18.8±5.6 | 9.3±19.37 | 23±7.59 | 19±13.27 | 16±15.23 | 19.1±9.21 | 14.1±11.7 | |
| Amino acid (mg/g) | 1st Year | 13.1±1.98 | 13.4±0.7 | 14.3±0.69 | 12.7±0.48 | 13.9±0.94 | 14.7±0.54 | 12±1.53 | 14.7±0.2 | 13.7±1.1 |
| 2nd Year | 13±1.39 | 13.9±0.79 | 13.8±0.7 | 13.8±1.07 | 13.7±1.28 | 13.9±0.94 | 13.9±0.44 | 14.4±0.3 | 14±0.44 | |
| Alkaloid (%) | 1st Year | 46.9±0.8 | 44.5±0.87 | 43.8±2.9 | 47.2±1.27 | 47±3.95 | 45.5±4.28 | 45±3 | 46.4±1.35 | 45.9±3 |
| 2nd Year | 41.4±5.06 | 45.2±2.35 | 45.5±0.6 | 49.4±8.65 | 46.2±2.68 | 45.9±3.35 | 49±9.81 | 50.6±2.06 | 46.4±6.46 | |
| Flavonoid (mg/g) | 1st Year | 3.1±0.49 | 2.9±1.69 | 1.7±1.88 | 4.2±1.57 | 3.3±1.44 | 4.5±0.36 | 2.8±1.34 | 4.4±0.51 | 4.5±0.16 |
| 2nd Year | 3.2±0.73 | 3.2±1 | 2.7±0.67 | 4.4±0.32 | 4.1±0.19 | 4.2±0.47 | 2.7±1.2 | 4.4±1 | 4.4±0.85 | |
| Tannin (mg/g) | 1st Year | 3.7±0.07 | 3.9±0.255 | 3.68±0.05 | 3.7±0.04 | 3.7±0.06 | 3.7±0.03 | 3.8±0.06 | 3.8±0.06 | 3.9±0.22 |
| 2nd Year | 4.1±0.38 | 3.8±0.14 | 3.9±0.19 | 3.8±0.09 | 3.8±0.07 | 3.7±0.05 | 3.6±0.46 | 3.8±0.14 | 3.9±0.11 | |
| Phenol (mg/g) | 1st Year | 1.5±0.18 | 1.8±0.17 | 1.2±0.18 | 1.8±0.51 | 1.1±0.06 | 2.1±0.27 | 2.5±0.07 | 2.1±0.47 | 2.3±0.13 |
| 2nd Year | 1.3±0.45 | 1.5±0.15 | 1.2±0.03 | 2.5±0.45 | 1.7±0.29 | 1.4±0.11 | 2.7±0.33 | 2.4±0.14 | 3±0.08 | |
| Saponin (%) | 1st Year | 22.9±2.79 | 23.1±6.1 | 31.9±5.1 | 33.1±1.91 | 26.6±4.96 | 31.1±6.74 | 30.3±8.95 | 31.5±2.36 | 29.2±2.13 |
| 2nd Year | 28.4±5.71 | 31.1±7.49 | 29.1±7.57 | 28.7±5.31 | 32.4±3.13 | 29.2±7.03 | 29.2±6.27 | 26.4±5.67 | 29.1±5.49 | |
| Calcium (mg/100 g) | 1st Year | 13.6±0.3 | 13.5±1.15 | 14±2.49 | 13.6±2.59 | 14.2±2.7 | 13.5±2.15 | 13.2±1.41 | 14.3±2.02 | 15.8±1.45 |
| 2nd Year | 14.5±3.38 | 14.5±1.4 | 14.8±0.8 | 14.7±1.3 | 13.9±2.68 | 15.6±2.3 | 13.4±2.53 | 13.6±1.98 | 14.5±3.33 | |
| Potassium (ppm) | 1st Year | 2.1±0.51 | 2.9±0.61 | 2.3±0.42 | 2.2±0.46 | 2.6±0.35 | 1.3±0.37 | 2±0.16 | 1.9±0.03 | 2.3±0.4 |
| 2nd Year | 2.3±0.71 | 2±0.4 | 2.7±0.1 | 2.5±0.58 | 2.2±0.4 | 2.6±0.4 | 2.4±0.3 | 3±0.32 | 2.5±0.69 | |
| Sodium (ppm) | 1st Year | 0.14±0 | 0.14±0 | 0.26±0.2 | 0.26±0.2 | 0.26±0.2 | 0.14±0 | 0.14±0 | 0.14±0 | 0.26±0.2 |
| 2nd Year | 0.14±0 | 0.26±0.2 | 0.14±0 | 0.26±0.2 | 0.14±0 | 0.26±0.2 | 0.14±0 | 0.26±0.2 | 0.26±0.2 | |
| Phosphorus (mg/g) | 1st Year | 1.9±0.45 | 1.9±0.04 | 1.7±0.47 | 1.6±0.26 | 1.6±0.3 | 1.4±0.14 | 1.7±0.53 | 1.6±0.04 | 1.5±0.2 |
| 2nd Year | 1.8±0.22 | 1.8±0.17 | 1.3±0.26 | 1.5±0.39 | 1.4±0.2 | 1.4±0.12 | 1.5±0.12 | 1.5±0.2 | 1.7±0.28 | |
| Total sugar (mg/g) | 1st Year | 52.9±4.63 | 59±1.71 | 60.5±3.66 | 49.2±6.23 | 52.9±8.9 | 50.3±7.4 | 37.4±1.22 | 45.9±7.77 | 45.8±12.3 |
| 2nd Year | 61.1±5.94 | 51.8±1.03 | 62.8±3.95 | 43.6±15.2 | 57±5.1 | 55.4±3.05 | 43.1±9.17 | 45.8±11.2 | 38.8±8.8 |
Data are represented as mean±standard deviation.
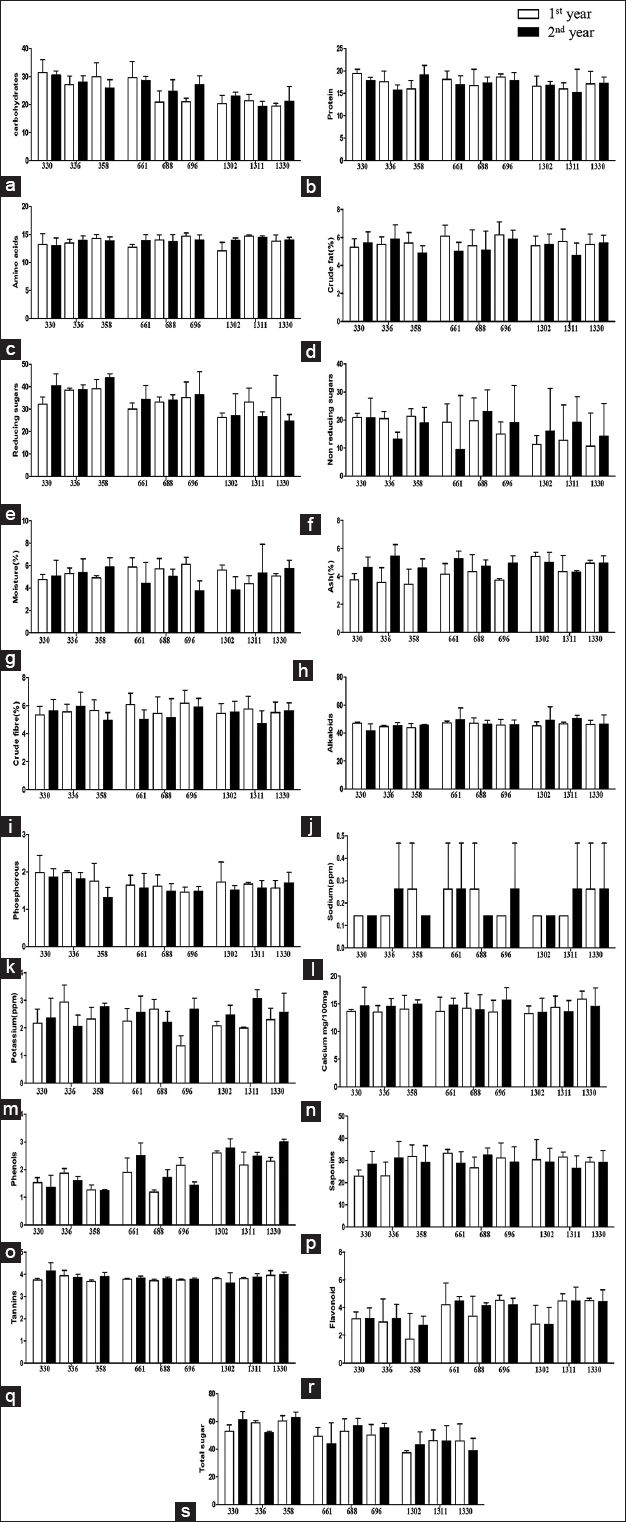 | Figure 2: Carbohydrate (a), protein (b), amino acid (c), crude fat (d), reducing sugar (e), non-reducing sugar (f), moisture (g), ash (h), crude fiber (i), alkaloid (j), phosphorus (k), sodium (l), potassium (m), calcium (n), phenol (o), saponin (p), tannin (q), flavonoid (r), and total sugar (s) of Justicia adhatoda L. growing in different altitudes of Jammu. [Click here to view] |
Plants can turn an ample amount of assimilated carbon and energy into the synthesis of other organic molecules that are included in the plant defense [81]. Plants produce secondary metabolites by undergoing various metabolic pathways as a response to stress conditions and these pathways are in turn prompted from the primary metabolite pathways which create the crucial precursors of secondary metabolites [82]. Total sugar, reducing sugar, non-reducing sugar, and ash also significantly varied with elevation [Table 2]. In some studies, significant variation in total amino acids and total proteins among different provenances has been observed [83,84] but in our study, variations were not statistically significant [11,85,86]. Insignificant changes in calcium content, alkaloid content, saponin content, phosphorus, potassium, and sodium were observed in the study [Table 2] [Figure 2] [87,88]. Crude fiber and crude fat in our study were observed in higher amounts at middle altitude in comparison to the higher and low elevation which was in agreement with the study done on red clover cultivars, where crude fat content was higher in mountain region in comparison to the low land region [1,89] but the outcomes in our study were not statistically significant [Table 1].
In comparison to low altitude, concentration of phenols increased at high altitudes in our study. The highest amount of phenols was observed to be 3.006 mg/g present in 1330 masl, the phenol content of mid and high altitude area was comparatively high as compared to the low altitude area and minimum phenols were found to be present at 688 masl, that is, 1.186 mg/g [Table 2]. This is in agreement with the fact that phenols were found in high levels in the plants collected from high altitudes in a study and that increase in phenol concentration with altitude was ascribed as a response to increasing intensity of UV radiations [90]. The previous studies also suggest that decreased temperatures and increase in intensity of UV radiation activate the amplified production of UV-absorbing and various antioxidant compounds, namely, phenols in plants, this could be probably the phenomenon happening to the phenols there [91-93]. Plants at high altitudes possess UV protective systems to cope up with the increasing harmful UV radiations with altitude. Phenols play a vital part in defense and other mechanisms of the plant for instance, hydrogen scavenging system [94] which could function less effectively when the temperature remains less and therefore, more phenolic compounds are required to avoid the loss happening to plants at low temperatures [95]. Total flavonoid content increased in high altitude areas. Total flavonoid content of the plant also significantly increased with increasing altitude, maximum amount of flavonoids was observed at 696 masl, that is, 4.543 mg/g and least present at 358 masl, namely, 1.71 mg/g [Table 2]. Some studies have revealed that due to the various environmental factors happening at high altitudes, flavonoid accumulation was induced as a consequence of UV radiations. Flavonoid is considered as a foremost UV protectant and free radical scavenging compound [96]. Phenolic acids and flavonoids are considerably produced in plants at high elevation sites [97]. It is possible that other abiotic and biotic factors could also be contributing to the observed altitudinal variations in phenols. There could be various other factors like exposure to strong wind regimes at high altitudes [98,99], air temperature [100,101], soil mineralization and nitrification with increasing altitude [102-104], transpiration rates [105,106], rate of photosynthesis [107,108], and disturbances in light intensity as the photoperiod holds an impact on the biosynthesis of several secondary metabolites in a number of plant species [109] which can contribute to these variations for instance, in a study, soil and leaves of herbs, shrubs, and trees of Ecuadorian tropical montane forest were accessed at various altitudes where foliar nutrient concentrations were observed to be highest in low altitude and also there was slow mineralization of plant litter and less availability of nutrients at high altitudes [110]. Plant trait variations are mostly environmentally based but sometimes, these can have a genetic background. In a study, variations were found in the morphological attributes and still retained when the plants were grown under similar environmental conditions which suggested a possible genetic background for those traits [111]. Hence, the findings of the present study illustrated the general phenomenon that the plants possess the ability to deal with the environmental stress by alternate mechanisms be it morphological, physicochemical, or phytochemical.
4. CONCLUSION
Plants are not randomly distributed in a particular elevational site, their distribution depends on the environment in which they grow and are able to functionally adapt themselves. Plant’s ability to adapt to various ecological conditions relies on the morphological and physiological properties exhibited by its organs. Phytochemical variability helps plants to cope up with unsuitable environment for instance, in this study, phenols and flavonoids which are also powerful antioxidants increase their levels in Justicia adhatoda L. plants growing at higher elevations to combat stress. Justicia adhatoda L. chemically adapted itself along the altitudinal range by regulation of its phytochemicals. Plants at high altitudes are equipped with defense system which activates biosynthesis of secondary metabolites, and the results demonstrate that plants can adapt to varying environmental circumstances happening along altitudinal gradient.
5. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
6. FUNDING
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
7. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
8. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
9. DATA AVAILABILITY
All data generated and analyzed are included within this research article.
10. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Dongdong C, Li Q, Liu Z, Fuquan H, Xin C, Shixiao X,
2. Glassmire AE, Jeffrey CS, Forister ML, Parchman TL, Nice CC, Jahner JP,
3. Gouvinhas I, Pinto R, Santos R, Saavedra MJ, Barros AI. Enhanced phytochemical composition and biological activities of grape (
4. Hatfield JL, Prueger JH. Temperature extremes:Effect on plant growth and development. Weather Clim Extrem 2015;10:4-10. [CrossRef]
5. Schumann T, Paul S, Melzer M, Dormann P, Jahns P. Plant growth under natural light conditions provides highly flexible short-term acclimation properties toward high light stress. Front Plant Sci 2017;8:681. [CrossRef]
6. Wang M, Zhang J, Guo Z, Guan Y, Qu G, Liu J,
7. Bosch MS, Cotado A, Morales M, Fleta-Soriano E, Villellas J, Garcia MB. Adaptation of the long-lived monocarpic perennial saxifraga longifolia to high altitude. Plant Physiol 2016;172:765-75.
8. Zhang T, Qiao Q, Yu P, Novikova, Wang Q, Yue J,
9. Mattila H, Mishra KB, Kuusisto I, Mishra A, Novotna K, Sebela D,
10. Roeber VM, Bajaj I, Rohde M, Schmulling T, Cortleven, A. Light acts as a stressor and influences abiotic and biotic stress responses in plants. Plant Cell Environ 2020;44:645-64. [CrossRef]
11. Singh B, Bhatt BP, Prasad P. Altitudinal variation in nutritive value of adult-juvenile foliage of
12. Meller S, Frossard E, Spohn M, Luster J. Plant nutritional status explains the modifying effect of provenance on the response of beech sapling root traits to differences in soil nutrient supply. Front For Glob Change 2020;3:535117. [CrossRef]
13. Saxena RK, Edwards D, Varshney RK. Structural variations in plant genomes. Brief Funct Genomics 2014;13:296-307. [CrossRef]
14. Classen A, Dewenter SI, Kindeketa WJ, Peters MK. Integrating intraspecific variation in community ecology unifies theories on body size shifts along climatic gradients. Funct Ecol 2016;31:768-77. [CrossRef]
15. Roux D, Alnaser O, Garayev E, Baghdikian B, Elias R, Chiffoleau P,
16. Schneider FD, Morsdorf F, Schmid B, Schimel DS, Schaepman ME, Petchey OL,
17. Henn JJ, Buzzard V, Enquist BJ, Halbritter AH, Klanderud K, Maitner BS,
18. Read QD, Moorhead LC, Swenson NG, Bailey JK, Sanders NJ. Convergent effects of elevation on functional leaf traits within and among species. Funct Ecol 2014;28:37-45. [CrossRef]
19. Liu W, Zheng L, Qi D. Variation in leaf traits at different altitudes reflects the adaptive strategy of plants to environmental changes. Ecol Evol 2020;10:8166-75. [CrossRef]
20. Gratani L, Catoni R, Pirone G, Frattaroli AR, Varone L. Physiological and morphological leaf trait variations in two Apennine plant species in response to different altitudes. Photosynthetica 2012;50:15-23. [CrossRef]
21. Cordell S, Goldstein G, Mueller-Dombois D, Webb D, Vitousek PM. Physiological and morphological variation in
22. Kofidis G, Bosabalidis AM, Moustakas M. Contemporary seasonal and altitudinal variations of leaf structural features in oregano (
23. Jayanthy A, Kumar PU, Remashree AB. Seasonal and geographical variations in cellular characters and chemical contents in
24. Jugran AK, Bahukhandi A, Dhyani P, Bhatt ID, Rawal RS, Nandi SK. Impact of altitudes and habitats on valerenic acid, total phenolics, flavonoids, tannins, and antioxidant activity of
25. Khaleefa A, Nawchoo IA, Bashir GA. Altitudinal variation in some phytochemical constituents and stomatal traits of
26. Raja SS, Unnikrishnan KP, George S, Remashree AB, Udayan PS, Tushar KV,
27. Gurung PD, Upadhyay AK, Bhardwaj PK, Sowdhamini R, Ramakrishnan U. Transcriptome analysis reveals plasticity in gene regulation due to environmental cues in
28. Green-Armystage S. Extraordinary leaves. Color Res Appl 2008;31:253-69. [CrossRef]
29. Blozan, W. Tree measuring guidelines of the eastern native tree society. Bull East Native Tree Soc 2006;1:3-10.
30. Thangaraj, P. Proximate composition analysis. Prog Drug Res 2016;71:21-31. [CrossRef]
31. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the folin phenol reagent. J Biol Chem 1951;193:265-75. [CrossRef]
32. Moore S, Stein WH. Photometric ninhydrin method for use in the chromatography of amino acids. J Biol Chem 1984;176:367-88. [CrossRef]
33. Usunobun U, Okolie N. Phytochemical, trace and mineral composition of
34. Jain VM, Karibasappa GN, Dodamani AS, Mali GV. Estimating the carbohydrate content of various forms of tobacco by phenol sulphuric acid method. J Educ Health Promot 2017;6:90. [CrossRef]
35. Otache M, Agbajor G, Akpovona A, Ogoh B. Quantitative determination of sugars in three varieties of cassava pulp. Asian J Chem Sci 2017;3:1-8. [CrossRef]
36. Bosha JA, Anaga AO, Asuzu IU. Chemical composition of
37. AOAC Official Methods of Analysis. The Association of Official Analytical Chemists. 17th ed. Gaithersburg, MD, USA:AOAC Official Methods of Analysis;2000.
38. Kumar M, Dandapat S, Kumar A, Sinha MP. Determination of nutritive value and mineral elements of five-leaf chaste tree (
39. Singh D, Chhonkor PK, Dwivedi BS. Manual on Soil, Plant and Water Analysis. New Delhi:Westville;2005. 82-4.
40. Oser BL. Hawks Physiological Chemistry. 14th ed. New Delhi, India:Tata McGraw-Hill Publishing Co. Ltd.;1965. 1263-5.
41. Harborne, JB. Phytochemicals Methods. London:Chapman and Hall Ltd.;1973.
42. Zao Y, Lu Y, Wei D. Antioxidant activity of a flavonoid-rich extract of
43. Sethi, A, Sharma, RA. Antioxidant activity with total phenolic constituents from
44. Saxena V, Mishra G, Saxena A, Vishvakarma KK. A comparative study on quantitative estimation of tannins in
45. Mir MA, Sawhney SS, Jassal MM. Qualitative and quantitative analysis of phytochemicals of
46. Corenblit D, Baas A, Balke T, Bouma T, Fromard F, Garofano-Gomez V,
47. Perez-Harguindeguy N, Diaz S, Garnier E, Lavorel S, Poorter H, Jaureguiberry P,
48. Heilmeier H. Functional traits explaining plant responses to past and future climate. Flora 2019;254:1-11. [CrossRef]
49. Liao H, Pal RW, Niinemets U, Bahn M, Cerabolini BE, Peng S. Different functional characteristics can explain different dimensions of plant invasion success. J Ecol 2021;109:1524-36. [CrossRef]
50. Paridari IC, Jalali SG, Sonboli A, Zarafshar M, Bruschi P. Leaf macro-and micro-morphological altitudinal variability of
51. Mao L, Chen S, Zhang J, Zhou G. Altitudinal patterns of maximum plant height on the Tibetan Plateau. J Plant Ecol 2018;11:85-91.
52. Coomes DA, Allen RB. Effects of size, competition and altitude on tree growth. J Ecol 2007;95:1084-97. [CrossRef]
53. Reif JC, Xia XC, Melchinger AE, Warburton ML, Hoisington DA, Beck D,
54. Xu Z, Hu T, Zhang Y. Effects of experimental warming on phenology, growth and gas exchange of treeline birch (
55. Korner C. Alpine Plant Life:Functional Plant Ecology of High Mountain Ecosystems. Berlin:Spinger;2003.
56. Korner C, Paulsen J. A world-wide study of high altitude treeline temperatures. J Biogeogr 2004;31:713-32. [CrossRef]
57. Pellissier L, Fournier B, Guisan A, Vittoz P. Plant traits co-vary with altitude in grasslands and forests in the European Alps. Plant Ecol 2010;211:351-65. [CrossRef]
58. Orshan G, Montenegro G, Avila G, Aljaro ME, Walckowiak A, Mujica AM. Plant growth forms of chilean matorral a monocharacter growth form analysis along an altitudinal transect from sea level to 2000 M.A.S.L. Bulletin de La Societe Botanique de France. Actual Bot 1984;131:411-25. [CrossRef]
59. Goldstein G, Rada F, Azocar A. Cold hardiness and supercooling along an altitudinal gradient in andean giant rosette species. Oecologia 1985;68:147-52. [CrossRef]
60. Korner, C. Alpine plant life. Geographical J 1999;31:662. [CrossRef]
61. Bajpai PK, Warghat AR, Yadav A, Kant A, Srivastava RB, Stobdan T. Hightypic phenotypic variation in
62. Tranquillini W. The physiology of plants at high altitudes. Annu Rev Plant Physiol 1963;15:345-62. [CrossRef]
63. Akhatar J, Goyal A, Kaur N, Atri C, Mittal M, Singh MP,
64. Wang B, Wu L, Chen D, Wu Y, Hu S, Li L,
65. Ogbazghi W, Bongers F, Rijkers T, Wessel M. Population structure and morphology of the frankincense tree
66. Pi HQ, Quan QM, Wu B, Lv XW, Shen LM, Huang SQ. An altitude-related shift of relative abundance from insect to sunbird pollination in
67. Miyajima Y, Takahashi K. Changes with altitude of the stand structure of temperate forests on Mount Norikura, Central Japan. J For Res 2007;12:187-92. [CrossRef]
68. Korner C, Erika H. Why is the alpine flora comparatively robust against climatic warming?Diversity 2021;13:383. [CrossRef]
69. Cierjacks A, Ruhr NK, Wesche K, Hensen I. Effects of altitude and livestock on the regeneration of two tree line forming
70. Guo Z, Lin H, Chen S, Yang Q. Altitudinal patterns of leaf traits and leaf allometry in bamboo
71. Nam KJ, Lee EJ. Variation in leaf functional traits of the Korean maple (
72. Paudel BR, Dyer AG, Garcia JE, Shrestha M. The effect of elevational gradient on alpine gingers (
73. Ye M, Zhu X, Gao P, Jiang L, Wu R. Identification of quantitative trait loci for altitude adaptation of tree leaf shape with
74. Joel G, Aplet G, Vitousek PM. Leaf morphology along environmental gradients in Hawaiian
75. Taguchi Y, Wada N. Variations of leaf traits of an alpine shrub Sieversia pentapetala along an altitudinal gradient and under a simulated environmental change. Polar Biosci 2001;14:79-87.
76. Hoffmann AA, Griffin, PC, MacRaild, RD. Morphological variation and floral abnormalities in a trigger plant across a narrow altitudinal gradient. Aust Ecol 2009;34:780-92. [CrossRef]
77. Berli FJ, Alonso R, Bressan-Smith R, Bottini R. UV-B impairs growth and gas exchange in grapevines grown in high altitude. Physiol Plant 2012;149:127-40. [CrossRef]
78. Baumgartner A, Donahoo M, Chitwood DH, Peppe DJ. The influences of environmental change and development on leaf shape in Vitis. Am J Bot 2020;107:676-88. [CrossRef]
79. Loewus FA, Tanner W. Plant Carbohydrates. I. Intracellular Carbohydrates. Vol. 13. Encyclopedia of Plant Physiol;1983. [CrossRef]
80. Guy CL, Joan LA, Huber SC. Sucrose phosphate synthase and sucrose accumulation at low temperature. Plant Physiol 1992;100:502-8. [CrossRef]
81. Bhaduri D, Rakshit R, Chakraborty K. Primary and secondary nutrients-a boon to defense system against plant diseases. Int J Bioresource Stress Manage 2014;5:461-6. [CrossRef]
82. Movahedi A, Amir AZ, Hui W, Paul R, Weibo S, Mohaddeseh M,
83. Shuguang W, Xiaolan P, Yulong D, Xianchong W. Aboveground biomass and bamboo shoot nutrients of high altitude bamboos (
84. Han WY, Huang JG, Li X, Li ZX, Ahammed GJ, Yan P,
85. Singh B, Todaria NP. Nutrients composition changes in leaves of
86. Rochana A, Indriani NP, Wiradimadja R, Ayuningsih B, Winaryanto S, Dhalika T,
87. Singh JS. Sustainable development of the Indian Himalayan region:Linking ecological and economic concerns. Curr Sci 2006;90:784-8.
88. Asmare B, Demeke S, Tolemariam T, Tegegne F, Haile A, Wamatu J. Effects of altitude and harvesting dates on morphological characteristics, yield and nutritive value of desho grass (
89. Leto J, Knezevic M, Bosnjak K, Macesic D, Stafa Z, Kozumplik V. Yield and forage quality of red clover (
90. Rochana A, Indriani NP, Ayuningsih B, Hernaman I, Dhalika T, Rahmat D,
91. Spitaler R, Winkler A, Lins I, Yanar S, Stuppner H, Zidorn, C. Altitudinal variation of phenolic contents in flowering heads of
92. Markham KR, Ryan KG, Bloor SJ, Mitchell KA. An increase in the luteolin:Apigenin ratio in
93. Bilger W, Rolland M, Nybakken L. UV screening in higher plants induced by low temperature in the absence of UV-B radiation. Photochem Photobiol Sci 2007;6:190-5. [CrossRef]
94. Takahama U, Oniki T. A peroxidase/phenolics/ascorbate system can scavenge hydrogen peroxide in plant cells. Physiol Plant 1997;101:845-52. [CrossRef]
95. Jin XD, Wu X, Liu X. Phenolic characteristics and antioxidant activity of merlot and cabernet sauvignon wines increase with vineyard altitude in a high-altitude region. S Afr J Enol Vitic 2017;38:132-43. [CrossRef]
96. Murai Y, Setoguchi H, Kitajim J, Iwashina T. Altitudinal variation of flavonoid content in the leaves of
97. Murai Y, Takemura S, Takeda K, Kitajima J, Iwashina T. Altitudinal variation of UV-absorbing compounds in
98. Cordero RA. Ecophysiology of
99. Gardiner B, Berry P, Moulia B. Review:Wind Impacts on plant growth, mechanics and damage. Plant Sci 2016;245:94-118. [CrossRef]
100. Grubb PJ. Control of forest growth and distribution on wet tropical mountains:With special reference to mineral nutrition. Annu Rev Ecol Syst 1977;8:83-107. [CrossRef]
101. Korner C, Erika H. The 90 ways to describe plant temperature. Ecol Evol Syst 2017;30:16-21. [CrossRef]
102. Marrs RH, Proctor J, Heaney A, Mountford MD. Changes in soil nitrogen-mineralization and nitrification along an altitudinal transect in tropical rain forest in Costa Rica. J Ecol 1988;76:466-82. [CrossRef]
103. Weaver PL, Murphy PG. Forest structure and productivity in Puerto Rico's Luquillo Mountains. Biotropica 1990;22:69-82. [CrossRef]
104. Baldos AP, Corre MD, Veldkamp E. Response of N cycling to nutrient inputs in forest soils across a 1000-3000 m elevation gradient in the ecuadorian andes. Ecology 2015;96:749-61. [CrossRef]
105. Gale J. Plants and altitude-revisited. Ann Bot 2004;94:199. [CrossRef]
106. Van Kanten R, Vaast P. Transpiration of arabica coffee and associated shade tree species in sub-optimal, low-altitude conditions of Costa Rica. Agroforest Syst 2006;67:187-202. [CrossRef]
107. Bruijnzeel LA, Veneklaas EJ. Climatic conditions and tropical, montane forest productivity:The fog has not lifted yet. Ecology 1998;79:3-9. [CrossRef]
108. Wang H, Prentice IC, Davis TW, Keenan TF, Wright IJ, Peng C. Photosynthetic responses to altitude:An explanation based on optimality principles. New Phytol 2016;213:976-82. [CrossRef]
109. Jaakola L, Hohtola A. Effect of latitude on flavonoid biosynthesis in plants. Plant Cell Environ 2010;33:1239-47. [CrossRef]
110. Soethe N, Lehmann J, Engels C. Nutrient availability at different altitudes in a tropical montane forest in Ecuador. J Trop Ecol 2008;24:397-406. [CrossRef]
111. Byars SG, Parsons Y, Hoffmann AA. Effect of altitude on the genetic structure of an Alpine grass,