1. INTRODUCTION
Shigellosis is a disease caused by Shigella sp. that is a Gram-negative and non-motile bacterium belonging to the Enterobacteriaceae family. The four most common species are Shigella dysenteriae, Shigella flexneri, Shigella boydii, and Shigella sonnei (designated as serogroups A, B, C, and D, respectively), with several serotypes. From an estimated 165 million shigella episodes of diarrhea each year, 99% of cases occur in low- and middle-income countries [1].
Salmonellosis is a disease caused by Salmonella sp. with the highest incidence and widest distribution [2]. In 2000, the number of salmonellosis cases globally reached 21.6 million, with 216,000 deaths, and more than 90% of them occurred in Asia [3]. The disease develops commonly in developing countries. An estimated 15.5 million cases of salmonellosis, with a mortality rate of 154,000 in 2016, were reported by the Global Burden of Disease (GBD) study conducted by the Institute for Health Metrics and Evaluation [4].
Rose myrtle, or (Rhodomyrtus tomentosa [Ait.] Hassk), is a plant that belongs to the Myrtaceae family. The rose myrtle plant is one of the biodiversity that must be developed sustainably because of its properties as an antidiabetic, diarrhea, burns, bleeding wounds, and stomachaches. Rose myrtle leaves contain rhodomyrtone antibiotic compounds. The rhodomyrtone compound belongs to the phloroglucinol derivatives and active against Escherichia coli and Staphylococcus aureus [5]. Rose myrtle leaves have been used by society to treat various diseases related to bacterial infections, such as dysentery and typhoid fever caused by S. dysenteriae and Salmonella Typhi. The antibacterial test results showed that n-hexane and ethyl acetate extracts were active against both bacteria, while ethanol extract was not. Isolates N1 and E1 were produced, respectively, from n-hexane extract and ethyl acetate extract. The MIC values of both N1 and E1 for S. dysenteriae and S. Typhi were the same, namely, 125 μg/mL. Isolate N1 was an essential oil containing menthol (59.60%), caryophyllene (25.77%), and cubenol (14.63%), while isolate E1 was essential oil containing (73.93%), pentanone (8.30%), alpha calacorene (7.58%), and calacorene (3.78%) [6].
The application of rose myrtle leaves as traditional medicine needs to be proven for its efficacy and safety. Fraction of n-hexane and ethyl acetate of rose myrtle leaves showed antibacterial activity against Streptococcus mutans (MIC of each fraction was 7.8 g/mL and 3.9 g/mL), Streptococcus salivarius (MIC of each fraction which was 15.6 g/mL and 62.5 g/mL, respectively), and Streptococcus gordonii (MIC of each fraction which were 62.5 g/mL and 15.6 g/m, respectively) [7]. The n-hexane and ethyl acetate extracts of rose myrtle leaves also showed potential as antibacterial for S. dysentriae (MIC for each extract, which was 250 g/mL) and S. Typhi (MIC for each extract, which was 250 g/mL and 500 g/mL) [8]. The research of the combination of n-hexane with ethyl acetate fraction of rose myrtle leaf as an antidiarrheal caused by S. Typhi and S. dysentriae bacteria showed that the best dose amount of rose myrtle leaves fraction combination in reducing the number of colonies of each bacterium was 100 mg/KgBW. A toxicology test is one effort to meet the safety requirements of traditional drugs. A toxicity test aims to detect the toxic effect of a substance on a biological system and obtain specific dose-response data from the administration of the sample [9].
Observations on toxicity tests are performed by observing symptoms of toxicity, body weight, hematological and biochemical parameters, and macroscopic and microscopic test of organs (histopathology) for a more extended period. An organ test is required to determine the effect of the drug by checking the biochemical levels used to determine the possibility of organ damage. A hematology test is required to help establish the diagnosis and monitor the tested rat’s toxicity. The results provide information on the effects of the test compound on blood and blood-forming tissues [10]. The n-hexane and ethyl acetate fractions from rose myrtle leaves contain antibacterial compounds that can be used as raw materials for infectious diarrhea drugs such as shigellosis and salmonellosis. This research is significant to prove the safety of medicinal raw materials from rose myrtle leaves. Natural drug preparations can be said to be safe if their safety has been tested for toxicity using test animals, including acute, subacute, chronic, and mutagenic toxicity tests, and proven safe for use in humans [11]. This subacute toxicity test study was conducted to see the safety of using n-hexane and ethyl acetate fractions in long-term use.
2. MATERIALS AND METHODS
2.1. Chemicals and Equipment
The materials that were used in this study were the n-hexane and ethyl acetate fractions of rose myrtle leaves, Na CMC, SGPT reagent kit (Dialab), SGOT reagent kit (Dialab), creatinine reagent kit (Dialab), urea reagent kit (Dialab), formalin buffer (Merck and Co), alcohol, xylol (Merck), paraffin (Merck), hematoxylin-eosin (HE), dye (Merck), and entellan fluid (Merck).
The types of equipment that are used in this study were a rat cage, analytical balance (Ohaus), sonde (Obsidi Medica), syringe injection (OneMed), hematocrit pipette (NescoTM), non-EDTA vacutainer tube (BioLab), Ethylenediamine Tetra-acetic Acid, 2K salt (EDTA-2K) vacutainer tube (BioLab), centrifugator (Hettich EBA21), A15 Analyzer (Biosystems), Sysmex KX-21 hematology analyzer (Sysmex), rat surgical kit (Gold Cross), fixation device (Kedee), water bath (Thermo Scientific), tissue embedding center (KD-BM), bismol, KD-BL cooling plate (Kedee), trimming (Kedee), KD-H Hot plate (Kedee), and microscope (Olympus).
2.2. Extract Preparation
Samples of rose myrtle leaves were collected from various areas in Universitas Sriwijaya, Palembang, Indonesia. The n-hexane fraction and the ethyl acetate fraction of rose myrtle leaves were obtained from the liquid fractionation of the ethanol extract of rose myrtle leaves. A main solution with a dose of 800 mg/KgBW is first made to prepare a suspension of n-hexane and ethyl acetate fractions combination. A total of 8 g fraction (80 mg/mL), consisting of 4 g of each fraction, was added with 50 mL of 0.5% Na-CMC. Then, the solution was added with distilled water up to the volume limit of the 100 mL volumetric flask. Finally, the main solution is used to produce suspensions with a dose of 400 mg/KgBW (40 mg/mL) and 200 mg/KgBW (20 mg/mL) by dilution.
2.3. Animals
Male rats (Wistar strain, aged 2–3 months) were housed in the Laboratory Pharmacology, Department of Pharmacy, Faculty of Mathematics and Natural Sciences, University of Sriwijaya. The experimental protocol was approved by the Ethics Commission of Universitas Ahmad Dahlan No.022012048 and carried out following recent guidance for animal nurture in the laboratory.
2.4. Subchronic Toxicity Test
This test used 24 male Wistar rats (200–250 g). Then, the rats were being acclimatized for 7 days. Rats were grouped into four groups. Each group consisted of five tested animals, which were given test preparations every day for 28 days. Group I was the control and received 0.5% Na CMC in place of the extract. Groups II-IV received the extract at doses of 200, 400, 600, and 800 mg/kgBW daily.
2.5. Observation
Observations were weight gain every week. Hematological parameters (erythrocytes, hemoglobin, and leukocytes) and clinical biochemical parameters (SGOT, SGPT, creatinine, and urea) were also observed. Hematological and biochemical profiles were carried out before and after the treatment, except for the satellite group, which was carried out only after observation. Meanwhile, observations of organs were carried out macroscopically and microscopically. Macroscopic organ observations were carried out on the liver, kidneys, and heart by examining the organs’ shape, color, and weight. The microscopic test was carried out on the histopathology of the liver and kidneys. The satellite group consisting of two tested animals of each group was tested 14 days later for delayed effects on rats. Observations of hematological, biochemical, and macroscopic levels of the liver, kidneys, and heart of the satellite group were carried out on the 43rd day.
2.6. Hematology Analysis
The blood samples of the tested animals were taken using the retro-orbital plexus method from the veins of the eye. 1 mL of blood was obtained to be placed in EDTA-2K vacutainer tube. Then, the blood is tested using a hematology analyzer. The data obtained are the number of erythrocytes, hemoglobin concentration, and leukocytes [9].
2.7. Biochemical Analysis
The test of biochemical levels was carried out using the A15 Analyzer at the Palembang Health Laboratory Center. The blood samples of the tested animals were taken using the retro-orbital plexus method from the veins of the eye. The blood was collected into a non-EDTA vacutainer tube. Blood was centrifuged for 5 min at 5000 rpm. The separated serum was put into the prepared cuvette using a micropipette. Each level of biochemical parameters (SGOT, SGPT, creatinine, and urea) was calculated using the A15 analyzer. The A15 analyzer was operated using a computer.
The operating procedure starts by opening the A15 analyzer application. Samples are differentiated using sample code. The parameters of SGOT, SGPT, creatinine, or urea are then chosen in the application. After that, the position of the cuvette on the A15 analyzer rack needs to be determined. The cuvette containing the serum should be placed in the A15 analyzer rack before running the task. The A15 analyzer will automatically take the serum sample in the cuvette to analyze the reagent from the indicator (SGOT, SGPT, creatinine, or urea). Then, the application will calculate the concentration of the calculated indicator. The measurement results can be seen on the monitor screen.
2.8. Microscopic Observation
Animals were euthanized by cervical dislocation and then dissected to remove the organs. Each organ that has been separated is immediately put into a 10% formalin buffer solution. Then, the organs were cut and placed in a tissue cassette for tissue processing, namely, fixation, dehydration, clearing, and paraffin infiltration. The fixed tissue was dehydrated with alcohol, gradually starting at 70, 80, 90, and 96% concentrations for 24 h, respectively. After dehydration, it was cleared using xylol 3 times in 1 h for each, followed by paraffin infiltration. After that, the tissue is planted in paraffin media.
The process was continued with a 4 mm thickness tissue incision using a microtome. The incision results were attached to a slide, then stained with HE. Entellan liquid was dropped on the staining results, then covered with a cover glass and gave the sample’s identity (labeling). A histopathological test was carried out under a microscope with a magnification of 10 × 10 and 10 × 40 to examine the damage to the organs.
The damage of hepatocytes and sinusoids around the central vein is an indicator of liver function, while the damage of the proximal tubule is an indicator of kidney function. In the liver, the damage parameters are hydropic degeneration, fat degeneration, and necrosis. The degree of organ damage of as many as 100 proximal tubules in the kidney was calculated with the parameters observed: Tubular cell dilatation, necrosis, loss of brush border, protein cast, and cell vacuolization. Histopathological analysis was performed using a scoring technique. The liver and kidney damages are scored as follows: No lesion (0), mild lesions (1), moderate lesions (2), and severe lesions (3).
2.9. Statistical Analysis
Statistical analysis was performed using paired t-test to determine the difference between groups of rats before and after administration of the combination of n-hexane and ethyl acetate rose myrtle leaf fractions. Comparisons between groups were performed using one-way analysis of variance (ANOVA) followed by Duncan Multiple Range Tests (DMRT) using SPSS statistical software. A value of < 0.05 was considered significant.
3. RESULTS
The main purpose of this study is to evaluate the safety of n-hexane and ethyl acetate fractions from rose myrtles leaves in white male rats (Wister strain) by analyzing blood hematology and biochemistry data. Toxicity effects were also assessed by analyzing the ROW.
3.1. Rat Weight
Rat weight is one of the indicator data to see the effect of toxicity. The percentage of weight gaining and weight chart image per week showed as follows.
3.2. Hematological Parameters
The blood was collected by the retro-orbital plexus method from the eye vein. The eye organs of rats can regenerate quickly. Therefore, the blood can be taken back from the same organ for repeated measurements. In addition, the possibility of getting lysed blood is slight and easy to do. The hematology test for this study consisted of hemoglobin levels, erythrocyte levels, and leukocyte levels. The results of the rat hematology level can be seen in the following Tables 1, and 2.
Table 1: The average of hemoglobin and erythrocyte level before and after administration of sample.
| Group | Hemoglobin Level (g/dL) | Erythrocyte level (106/mm3) | Leukocyte level (103/mm3) | |||
|---|---|---|---|---|---|---|
| Before | After | Before | After | Before | After | |
| 0.5% Na-CMC | 11.40±0.10 | 15.20±0.10 | 6.07±0.21 | 8.58±0.24 | 15.13±1.90 | 14.03±2.78 |
| 200 mg/KgBW | 9.90±0.44 | 14.10±1.08 | 5.30±0.36 | 7.53±0.78 | 13.90±4.33 | 15.87±2.44 |
| 400 mg/KgBW | 10.17±0.68 | 14.30±2.03 | 5.23±0.15 | 7.76±0.58 | 11.27±0.59 | 11.43±1.88 |
| 800 mg/KgBW | 10.60±2.10 | 12.63±5.31 | 5.77±1.12 | 6.34±2.66 | 12.50±1.30 | 11.93±0.67 |
Table 2: The average of satellite group’s hematology.
| Group | Hemoglobin (g/dL) | Erythrocyte (106/mm3) | Leukocyte (103/mm3) |
|---|---|---|---|
| 0.5% Na CMC | 14.50±0.85 | 7.05±0.07 | 9.45±4.60 |
| 200 mg/KgBW | 14.25±1.63 | 7.95±0.07 | 11.95±3.61 |
| 400 mg/KgBW | 14.80±0.00 | 7.75±0.35 | 12.95±2.19 |
| 800 mg/KgBW | 13.45±1.63 | 7.30±1.27 | 14.10±1.13 |
3.3. Biochemical Parameters
Rat blood was collected by the retro-orbital plexus method from the eye veins. The blood is centrifuged to separate the plasma from the serum before the serum is used for biochemical tests. Determination of biochemical levels was carried out on SGOT, SGPT, creatinine, and urea parameters. The results are in following Tables 3-5.
Table 3: The average of SGOT and SGPT before and after administration of sample.
| Group | SGOT level (IU/L) | SGPT level (IU/L) | ||
|---|---|---|---|---|
| Before | After | Before | After | |
| 0.5% Na-CMC | 171.54±2.40 | 257.63±13.62a | 64.18±4.91 | 82.23±6.65 |
| 200 mg/KgBW | 186.54±58.82 | 224.80±16.89a | 51.78±9.41 | 69.62±2.95 |
| 400 mg/KgBW | 154.15±22.91 | 251.19±14.05a | 52.38±10.53 | 70.86±5.41 |
| 800 mg/KgBW | 247.72±34.24 | 306.92±30.86b | 66.28±10.61 | 65.56±18.55 |
a and b showed the significantly different in the DMRT (Duncan Multiple Range Test) Follow-up test at 0.05
Table 4: The average of creatinine and urea before and after administration of sample.
| Group | Creatinine level (mg/dL) | Urea level (mg/dL) | ||
|---|---|---|---|---|
| Before | After | Before | After | |
| 0.5% Na-CMC | 0.42±0.04 | 0.35±0.05 | 39.12±6.57 | 33.53±6.05 |
| 200 mg/KgBW | 0.39±0.06 | 0.32±0.20 | 37.19±0.22 | 30.16±3.75 |
| 400 mg/KgBW | 0.33±0.04 | 0.37±0.03 | 32.53±5.31 | 26.65±2.75 |
| 800 mg/KgBW | 0.43±0.07 | 0.35±0.11 | 36.62±5.08 | 43.29±13.09 |
Table 5: The average levels of satellite group’s biochemical test.
| Group | SGOT (IU/L) | SGPT (IU/L) | Creatinine (mg/dL) | Ureum |
|---|---|---|---|---|
| (mg/dL) | 251.92±76.35 | 58.18±11.03 | 0.65±0.14 | 43.11±0.85 |
| 200 mg/KgBW | 314.90±63.62 | 83.98±16.96 | 0.65±0.09 | 45.81±6.35 |
| 400 mg/KgBW | 275.91±8.49 | 80.98±12.72 | 0.58±0.11 | 42.52±8.46 |
| 800 mg/KgBW | 287.91±50.89 | 69.28±29.27 | 0.65±0.01 | 37.73±5.93 |
3.4. Organ Macroscopy
Organ macroscopy was carried out on the liver, kidneys, and heart. Figure 1 shows that the normal liver is dark red, the kidneys are brownish red, and the heart is red. The macroscopy of organs indicates liver damage in one or two rats in each group after the administration of the sample for 28 days. As for the satellite group, macroscopy showed normal liver, kidney, and heart organs with no signs of damage to any of these organs.
 | Figure 1: Normal organs (a) liver; (b) kidney; and (c) heart. [Click here to view] |
3.5. Weight of Liver, Kidney, and Heart Organ
Observation of organ weight aims to see the effect of giving a combination of rose myrtle leaf fraction on organs. Organ weight data can be used as support to see the damage to an organ in more detail.
3.6. Organ Microscopy
Organ microscopy was performed on the liver and kidneys of rats after 28 days. The organ was sampled from one rat for each group. The rats used as histology samples were the number one rats in all test groups. The results of the scoring of the damaged liver tissue are shown in Tables 6 and 7.
Table 6: Score results for liver damage.
| Group | Hydropic degeneration | Fat degeneration | Necrosis |
|---|---|---|---|
| Control | 0 (0%) | 0 (0%) | 0 (0%) |
| 200 mg/KgBW | 1 (20%) | 1 (40%) | 1 (30%) |
| 400 mg/KgBW | 2 (30%) | 2 (60%) | 2 (40%) |
| 800 mg/KgBW | 2 (60%) | 2 (70%) | 3 (80%) |
Table 7: Score results for kidney damage.
| Parameter | Control | 200 mg/KgBW | 400 mg/KgBW | 800 mg/KgBW |
|---|---|---|---|---|
| Tubular cell dilatation | 0 | 2 (60%) | 1 (10%) | 2 (60%) |
| Missing brush border | 0 | 2 (60%) | 1 (10%) | 2 (60%) |
| Protein cast (cylinder) | 0 | 1 (10%) | 0 (0%) | 1 (30%) |
| Cell vacuolization | 0 | 2 (60%) | 1 (5%) | 2 (60%) |
| Necrosis | 0 | 1 (20%) | 1 (10%) | 1 (10%) |
4. DISCUSSION
Based on Table 8, the control group shows that the average weight gaining after 28 days and after 42 days (rats in the satellite group) is still within normal limits. The expected growth of rats is 1.5–3% per day from their initial weight or 7.05–21% per week for rats under 5 months of age with adequate nutrition [12]. The group of rats before and after administration of n-hexane and ethyl acetate fraction showed no significant difference between groups (P > 0.05) in the one-way ANOVA test. The fraction combination does not affect the weight of rats. The paired t-test showed a non-significant difference (P > 0.05) for the group of rats before and after administration of the fraction combination. The combination n-hexane and ethyl acetate fraction of the rose myrtle leave had no effect on the weight gaining of the rats.
Table 8: The percentage of the average weight gaining of rats.
| Group | Weight Gaining (%) | |
|---|---|---|
| 28 days | Satellite | |
| 0.5% Na CMC | 22.74±11.68 | 12.15±0.52 |
| 200 mg/KgBW | 14.58±9.11 | 10.98±0.96 |
| 400 mg/KgBW | 14.04±6.26 | 11.09±0.16 |
| 800 mg/KgBW | 10.76±5.32 | 11.50±0.42 |
The group of rats had Hb levels below the normal range before the sample administration as shown in Table 1. The normal range of Hb levels in rats is 8–16 g/dL [13]. Hb levels can be influenced, among others, by age, sex, feed, environment, and physical activity. The low levels of Hb before treatment can be caused for environmental temperature factors and the physical activity of rats. Hb levels will increase at low ambient temperatures and will decrease at high ambient temperatures. Rat activity decreased during the day when compared to the night. After 28 days of the sample administration, there is an increase in Hb levels but they are still within normal limits.
The results of the ANOVA of Hb levels before and after the treatment showed no significant difference (P > 0.05) between the control group and the treated group. It indicates that the combination of rose myrtle leaf fraction did not affect the Hb levels of rats. The test indicates that the control group with 200 mg/KgBW dose experienced a significant increase in Hb levels. A significant increase in Hb levels in the control group and the group with a dose of 200 mg/KgBW showed that the Hb levels were still within the normal limits of the Hb level of the rat. The control group was the group that was not given any treatment except for 0.5% Na CMC. Therefore, the significant difference in Hb levels over time could be influenced by other factors such as rat activity and environmental temperature.
Table 1 shows an increase in erythrocyte levels, but levels were within the normal range, except for the group with a dose of 800 mg/KgBW. The erythrocyte levels were slightly below the normal range. As for the group before the administration of sample, the erythrocyte levels were below normal limits. The number of erythrocytes can decrease in conditions of anemia, which can cause a decrease in kidney function and hemolysis. Factors that affect the decrease of erythrocytes number include blood volume factors and environmental temperature. The number of erythrocytes will increase at low ambient temperatures and will decrease at high ambient temperatures. The normal range of erythrocyte levels is 7.2–9.6 × 106/mm3 [13]. Based on the ANOVA test, erythrocyte levels before and after sample administration showed no significant difference (P > 0.05) between the control and treatment groups. However, the average erythrocyte levels of the control group and the group with a dose of 200 and 400 mg/KgBW are still within normal limits. The significant increase that occurred was unlikely influenced by the combination of rose myrtle leaf fraction because there is no significant increase in erythrocyte levels at a dose of 800 mg/KgBW.
The leukocyte level in Table 1 shows that before the administration of the sample, the leukocyte level was slightly above the normal limit. After the sample administration, there were changes in leukocytes level. The decrease in leukocytes was still within the normal range, while the number of leukocytes was slightly above the normal limits. The normal range of rat leukocyte levels is 3–14.5 × 103/μL [13]. The high levels of leukocytes can be caused by bleeding, trauma, necrosis, toxins, leukemia, food, or stress. The high levels of leukocytes are affected by the trauma factor of rats due to fighting between rats, the presence of microorganisms that contaminate the food or intake, and the stress factors in rats that arise during blood sampling. ANOVA analysis of the leukocyte levels before and after the sample administration showed no significant difference (P > 0.05) between the control and treatment groups. The tests inferred that the n-hexane and ethyl acetate fraction combination did not affect the leukocyte levels of rats. The hematology level in Table 2 shows that hemoglobin, erythrocyte, and leukocyte were still in the normal range. The statistical analysis of the three parameters showed no significant difference (P > 0.05) between the control and treatment groups. The result indicates that the combination of rose myrtle leaf fraction also does not affect the hematological levels for the satellite group. Hematological levels can be used to help diagnose and monitor toxicity in tested animals [10]. Blood hematological parameters do not have a consistent pattern between increasing the dose with changes in hematological parameters and measurement time. This irregular pattern is considered due to a slight variation from experimental animals in one group [14].
SGOT is mainly found in the liver, heart, and muscles and has moderate levels in the kidneys, skeletal muscles, and pancreas. Therefore, SGOT is less specific to indicating liver damage. The normal level of SGOT in male rats is 60–300 IU/L [14]. Table 3 shows an increase in each group and in the test group of 800 mg/KgBW, the levels of SGOT are above the normal limit. The increasing levels of SGOT can occur in liver disease, acute pancreatitis, trauma, and acute hemolytic anemia. Rats are actively moving and often fight among each other, which can cause trauma or muscle injury that increases SGOT levels. In addition, the stress condition of rats can also increase SGOT levels [15]. Stress in rats can occur due to repeated treatment in rats and occurs when rat blood is drawn.
The one-Way ANOVA test showed a significant difference in the group after the administration of the sample. Based on the post hoc DMRT follow-up test, there was a significant difference (P < 0.05) between the 800 mg/KgBW test group and the control group, and the 200 mg/KgBW test group and also the 400 mg/KgBW test group. The groups with doses of 200 and 400 mg/KgBW showed no difference (P > 0.05) compared with the control group. The paired t-test of SGOT levels showed a significant difference (P < 0.05) in the control group, the 400 mg/KgBW group, and the 800 mg/KgBW group. The significant increase in the control and 400 mg/KgBW groups was still within the normal limits of SGOT levels in male rats. In comparison, the increase in SGOT in the 800 mg/KgBW group could be associated with SGPT levels and the results of liver histology examination.
Table 3 shows that there is an increase after the administration of the sample. The significant increase is twice of normal value. The increase in SGPT levels was not twice as the levels before the treatment and the SGPT levels were still within normal limits. SGPT is more abundant in the liver than cardiac muscle tissue, and it is more specific for liver function than SGOT. The normal limit of SGPT levels for white rats is 65.12−111.50 IU/L [16]. SGPT levels based on the results of statistical analysis showed no significant difference (P > 0.05) between the control group and the test group, both for levels before and after administration of sample. The result indicates that the combination of rose myrtle leaf fraction does not affect SGPT levels. Likewise, the paired t-test showed no significant difference (P > 0.05) before and after administration of the combination of rose myrtle leaf fraction. However, the control group showed a significant increase in SGPT levels (P < 0.05). However, the significant increase in SGPT levels was still within the normal range of SGPT levels in rats.
Table 4 shows that there is an increase and a decrease in creatinine levels. Creatinine levels may increase in conditions such as impaired renal function due to urinary tract obstruction, nephritis, muscle disease, or acute dehydration. In contrast, it may decrease in conditions of muscle dystrophy, malnutrition, atrophy, or decreasing muscle mass due to aging. Creatinine levels that increase and decrease indicate that they are still within the normal range of creatinine in Wistar rats, namely, 0.4–0.8 mg/dL [15]. Based on statistical analysis results, creatinine levels showed no significant difference among the groups (P > 0.05), which indicates that the combination of rose myrtle leaf fraction does not affect creatinine levels. The paired t-test showed a significant difference (P < 0.05) against the control group, but creatinine increased still within the normal range.
The normal range of urea levels in rats is 12–42 mg/dL [17]. Table 4 shows that the levels are still within normal limits, except for the 800 mg/KgBW group after sample administration. The level of urea in the test group of 800 mg/KgBW was slightly above the normal limit. Urea levels before and after administration of sample, based on the results of ANOVA analysis, showed no significant difference (P > 0.05) between the control group and the test group. The result indicates that the combination of the rose myrtle leaf fraction did not affect the urea level.
All biochemical parameters were still within normal limits, except for SGOT levels in the 200 mg/KgBW group and urea levels in the control group, 200 mg/KgBW group, and 400 mg/KgBW group, which were slightly above the normal limits for rat levels. The high levels of SGOT can be caused by trauma or muscle injury and stress conditions related to repeated treatment and blood sampling. The high levels of urea can be influenced by protein in the diet. High protein intake can increase urea levels. The results of the ANOVA statistical analysis for levels of creatinine, urea, SGOT, and SGPT indicate that the data are normally distributed (P > 0.05). ANOVA analysis also found no significant difference (P > 0.05) between groups. This result means that after 14 days of administration of the sample, it showed that the combination of rose myrtle leaves fraction did not affect any parameters of biochemical levels in all groups of satellite rats.
Figure 1 shows that the normal liver has a flat and smooth surface and a dark red color, while the abnormal liver has a mottled surface. There are cysts and changes in color, such as yellow or black. However, the visible damage to the liver can be caused by disease exposure and previous infection, as not all rats from each group showed macroscopic signs of damage. Figure 2 shows that liver damage indicated by the formation of masses and spots on the liver.
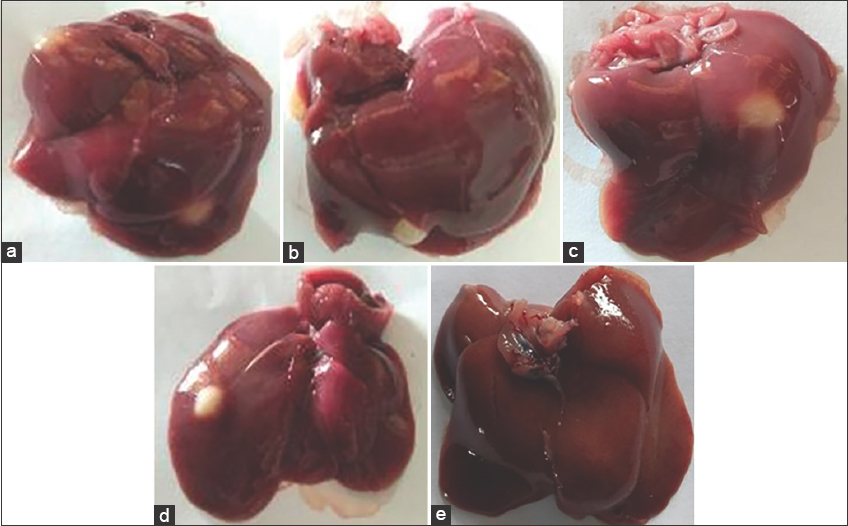 | Figure 2: Liver mass formed (a) control, (b) 200 mg/KgBW, (c) 200 mg/KgBW, (d) 800 mg/KgBW, and freckles (d) 800 mg/KgBW. [Click here to view] |
Based on Table 5, all levels were still within normal limits, except for SGOT levels in the 200 mg/KgBW test group, urea levels in the control group, 200 mg/KgBW, and 400 mg/KgBW doses, which were slightly above the normal limits for rat levels. The high levels of SGOT can be caused, among others, due to trauma or muscle injury and stress conditions in rats due to repeated treatment in rats and occurs when rat blood is drawn. The high levels of urea can be influenced by protein in the diet. The higher the protein intake, the urea levels can increase. The results of the ANOVA statistical analysis for levels of creatinine, urea, SGOT, and SGPT obtained data that were normally distributed (P > 0.05). Based on the ANOVA analysis, it was found that there was no significant difference (P > 0.05) between groups. After 14 days of administration of the test preparation, it showed that the combination of rose myrtle leaf fraction did not affect any parameters of biochemical levels in all groups of satellite rats.
Tables 9 and 10 show that the kidneys and heart are still within normal limits, while the liver is above the normal limit for all groups, including the control group, which were only given 0.5% Na CMC. However, statistical results showed that there was no significant difference (P > 0.05) between the treatment group and the normal group, which indicates that there is no effect of giving the combination of rose myrtle leaf fraction to the liver, kidney, and heart of rats. The relative weight of the rat liver is 2.3–3.10% body weight, and the kidney is 0.4–0.9% body weight of rats. At the same time, the relative weight of the heart organ is 0.26–0.58 [18].
Table 9: The average of relative organ weight after administration of sample.
| Group | Organ Weight (%) | ||
|---|---|---|---|
| Liver | Kidney | Heart | |
| 0.5% Na-CMC | 3.25±0.46 | 0.72±0.05 | 0.51±0.03 |
| 200 mg/KgBW | 3.81±0.51 | 0.77±0.08 | 0.52±0.01 |
| 400 mg/KgBW | 3.67±0.27 | 0.74±0.08 | 0.54±0.03 |
| 800 mg/KgBW | 4.03±0.70 | 0.78±0.07 | 0.56±0.02 |
Table 10: The average of satellite group’s relative organ weight.
| Group | Organ Weight (%) | ||
|---|---|---|---|
| Liver | Kidney | Heart | |
| 0.5% Na-CMC | 4.36±0.37 | 0.77±0.03 | 0.48±0.01 |
| 200 mg/KgBW | 4.09±0.19 | 0.75±0.03 | 0.50±0.11 |
| 400 mg/KgBW | 4.13±0.33 | 0.74±0.05 | 0.56±0.02 |
| 800 mg/KgBW | 3.66±0.35 | 0.78±0.02 | 0.55±0.01 |
Table 6 shows that the liver histology in the control group was in normal condition, meaning that the liver tissue showed no degeneration or necrosis. While in the test group, histology showed that the higher the dose of the combination of rose myrtle leaf fraction, the greater the damage. The test group experienced liver cell damage characterized by necrosis, hydropic degeneration, and fat degeneration. In contrast, the control group had no liver tissue damage. It is consistent with the levels of SGOT and SGPT and the levels of Hb, erythrocytes, and leukocytes, which is in the normal range. The histology result shows that the damage in the 200 mg/KgBW test group was conflicting with SGPT and SGOT levels, which are still within the normal limits. In addition, the results of hematological examinations show that the hemoglobin and leukocyte levels are also within normal limits. Only the erythrocyte levels are slightly below normal limits. However, statistically, the levels of SGPT, SGOT, and levels of Hb, leukocytes, and erythrocytes showed that they were not significantly different (P > 0.05) from the control group after administration of the sample. This means that there is no effect of the combination of rose myrtle leaf fraction at a dose of 200 mg/KgBW on liver damage.
The damage to liver tissue in the test group of 400 mg/KgBW was unlikely to be caused by the combination of the rose myrtle leaf fraction. The SGPT and SGOT levels were in the normal range. The levels of Hb, erythrocytes, and leukocytes were also normal. Statistically, there was no significant difference between the 400 mg/KgBW test group and the control group after the administration of sample on the levels of the SGPT, SGOT, and levels of Hb, erythrocytes, and leukocytes. Histological results also showed liver damage in the 800 mg/KgBW test group. The results conflict with SGPT, SGOT, and leukocyte levels, which are within the normal range. However, the Hb and erythrocyte levels were slightly below the normal limit in rats. The tested rats were suspected of having anemia. Anemia can occur due to liver tissue damage. Therefore, liver damage is suspected to be caused by infection or other disorders before the treatment. Factors affecting tissue damage include stress factors in rats, microorganisms that may contaminate feed and drinking water and free radicals [12]. Stress conditions in rats can increase the formation of free radicals that can cause oxidative stress. Oxidative stress can cause lipid peroxidation to cause cell damage and cause degenerative diseases, such as liver disease [18].
Statistically, the levels of SGPT and Hb, erythrocytes, and leukocytes showed no significant difference (P > 0.05) with the control group after sample administration, indicating no effect of the combination of rose myrtle leaf fraction on the liver. Meanwhile, the levels of SGOT showed a significant difference with the control group. SGOT level difference is caused by muscle injury or trauma due to fights between rats and stress related to the repeated treatment and blood sampling. SGPT levels are more specific than SGOT to determine liver damage because SGPT is more abundant in the liver. Therefore, the normal level of SGPT in the 800 mg/KgBW test group was suspected to not be related to the combination of the rose myrtle leaf fraction. Another factor that can cause tissue damage is the process of fixation of rat organ tissue. Fixation is a critical step in determining the success of the readable indicators from tissue microscopy preparations. The tardiness in the fixation process can cause autolysis. Autolysis can cause disturbances in histopathology because autolysis has characteristics that resemble necrosis, such as cells undergoing pyknosis that are characterized by hyperchromatic (a shrinking of cell nucleus) [18]. Microscopic observations were also carried out to see the damage to kidney tissue. The results of scoring kidney damage are shown in Table 7 and Figure 3.
 | Figure 3: Liver histology (a) control, (b) 200 mg/KgBW, (c) 400 mg/KgBW, and (d) 800 mg/KgBW. [Click here to view] |
Histological results of the control group showed no damage to kidney tissue [Figure 4]. It is consistent with the normal level of creatinine and urea. The hematological levels of the control group rats were also within the normal range. Tissue damage in the group with a dose of 200 mg/KgBW was suspected to not be related to the combination of rose myrtle leaf fractions because the creatinine and urea levels of the rats were still in the normal range. Hematology test also shows that the rats have a normal level, except for the erythrocyte levels, which were slightly below the normal limit. However, the erythrocyte difference is not statistically different (P > 0.05) from the control group. Creatinine and urea levels also showed no significant difference (P > 0.05) with the control group.
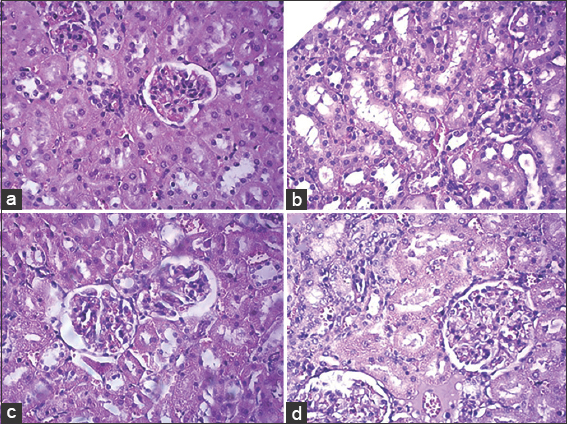 | Figure 4: Kidney histology (a) control (×400); (b) 200 mg/KgBW (×400); (c) 400 mg/KgBW (×400); and (d) 800 mg/KgBW (×400). [Click here to view] |
The test group of 400 mg/KgBW showed kidney damage on histology results. However, the levels of creatinine and urea and the hematological levels of rats showed were normal. Statistical analysis also showed no significant difference (P > 0.05) with the control group. Tissue damage in the group with a dose of 400 mg/KgBW was assumed not to be related to the combination of rose myrtle leaf fractions. Kidney histology results for the 800 mg/KgBW test group also showed damage. Based on the results, the creatinine levels of the rats were within normal limits, while the urea levels were slightly above normal limits. Creatinine levels are more precise than urea in showing kidney damage because they are not affected by protein content in a rat’s diet. High protein intake can increase urea levels. The damage in the test group was suspected to be due to infection or other disorders before the treatment and was not caused by the combination of the rose myrtle leaf fraction. Factors that can cause damage to kidney tissue include stress conditions in tested animals. Stress conditions cause a decrease in blood flow to the kidneys [19]. It can lead to decreased kidney function. Another factor that can affect the histology of organ tissue is the fixation process. Therefore, an error in the fixation process can cause an error in histology readings so that the histological results are different from the results of the rat blood examination.
The n-hexane and ethyl acetate fractions of rose myrtle leaves contain antibacterial compounds in the form of essential oils, phloroglucinol derivatives, rhodomyrtone compounds, and flavonoid compounds that have the potential to be used as raw materials for herbal medicines for salmonellosis and shigellosis. The n-hexane and ethyl acetate fractions were low toxic at a dose of 800 mg/KgBW, a dose of 400 mg/KgBW, and below did not affect the toxicity parameters, so it was safe to use. The results of this study are almost the same as nanoherbals from rose myrtle leaves. Nanoherbal rose myrtle contains flavonoids, steroids, glycosides, saponins, and tannins. Its LC50 and LD50 values, respectively, were 2,961,535 ppm and 10.4 ± 0.135 mg/kg BW. Histology of the heart, kidneys, lungs, heart, and brain is altered and affected by rose myrtle nanoherbal treatment in each dose level. Rose myrtle nanoherbals have strong antioxidant activity and small size and can be used effectively as a drug in the future because it contains secondary metabolites compounds that can be developed as drugs. However, it has mild toxicity [20].
5. CONCLUSIONS
Combination n-hexane and ethyl acetate fraction of rose myrtle leaf (R. tomentosa [Ait.] Hassk) up to a dose of 800 mg/KgBW after 28 days and 42 days did not affect hematological parameters (Hb, erythrocytes, and leukocytes) and biochemical parameters (SGPT, creatinine, and urea). The combination n-hexane and ethyl acetate fraction of rose myrtle leaf after 28 days did not cause any damage to the liver, kidneys, or heart. The combination of n-hexane and ethyl acetate fraction of rose myrtle leaf did not cause toxicity, so it can be used as raw material for infectious diarrhea treatment up to 400 mg/KgBW.
6. ACKNOWLEDGMENTS
We express our gratitude to the Pharmacology Laboratory of the Department of Pharmacy, Faculty of Mathematics and Natural Sciences, Universitas Sriwijaya, Palembang Health Laboratory Center, and Special Laboratory of Dyatnitalis Clinical Pathology Palembang.
7. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
8. FUNDING
There is no funding to report.
9. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
10. ETHICAL APPROVALS
The experimental protocol was approved by the Ethics Commission of Universitas Ahmad Dahlan No. 022012048 and carried out following recent guidance for animal nurture in the laboratory.
11. DATA AVAILABILITY
All data generated and analyzed are included within this research article.
12. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Ashkenazi S. Shigella infections in children:New insights. Semin Pediatr Infect Dis 2004;15:246-52. [CrossRef]
2. Ochiai LR. A study for typhoid fever in five asean countries:Disease burden and implications for controls. Bull World Health Organ 2008;86:260-8. [CrossRef]
3. Crump JA. The global burden of typhoid fever. Bull World Health Organ 2004;82:346-53.
4. Global Burden of Disease. Global Burden of Disease Results Tool. GBD Results Tool GHDx;2017. Available from:http://www.ghdx.healthdata.org/gbd-results-tool [Last accessed on 2021 Oct 05].
5. Dachriyanus, Salni, Sargent MV, Skelton BW, Soediro I, Sutisna M. Rhodomyrthon, an antibiotic from Rhodomythus tomentosa. Australian J Chem 2002;55:229-32. [CrossRef]
6. Salni S, Marisa H. Antibacterial activity of essential oil from rose myrtle leaves (Rhodomyrtus tomentosa (Ait) Hassk). Molekul 2020;15:158-65. [CrossRef]
7. Limsuwan S, Trip EN, Kouwen TR, Piersma S, Hiranrat A, Mahabusarakam W, et al. Rhodomyrtone:A new candidate as natural antibacterial drug from Rhodomyrtus tomentosa, Phytomedicine 2009;16:645-51. [CrossRef]
8. Salni S, Marisa H. Evaluation of antibacterial activity of rose myrtle leaf extract (Rhodomyrthus tomentosa (Ain.) Hassk) with various solvents Shigella dysentriae and Salmonella typhi. Mal J Fun Appl Sci 2019;15:671-4.
9. Food and Drug Supervisory Agency. Regulation of the Head of the Food and Drug Supervisory Agency of the Republic of Indonesia Number:13 of 2014 concerning Guidelines for In Vivo Nonclinical Toxicity Testing, Food and Drug Supervisory Agency of the Republic of Indonesia, Jakarta, Indonesia. Indonesia:Food and Drug Supervisory Agency;2014.
10. Mitruka BM, Rawnsly HM. Clinical, Biochemical, and Hematological Refference Values in Normal Experimental Animals and Normal Humans. 2nd ed. Chicago, USA:Year Book Medical Publisher, Inc.;1981.
11. Food and Drug Supervisory Agency. Use natural ingredients wisely. in Nature Cost. III. Jakarta. Food and Drug Supervisory Agency;2008.
12. Levine S, Saltzman A. Effect of coprophagy on serum urea and the weight of the gastrointestinal tract of fed or fasted rats. Lab Anim 1999;33:265-8. [CrossRef]
13. Giknis ML, Clifford CB. Clincal Laboratory Parameter for Crl:WI (Han) Rats. Wilmington:Charles River Laboratories;2008.
14. Li B, Jin Y, Xu Y, Wu Y, Xu J, Tu Y. Safety evaluation of tea (Camellia sinesis (L.) O. Kuntze) flower extract:Assesment of mutagenicity, and acute and subchronic toxicity in rats. J Ethnopharmacol 2011;1:90-583. [CrossRef]
15. Schoeffner DJ, Warren DA, Muralidhara S, Bruckner JV, Simmons JE. Organ weights and fat volume in rats as a function of strain and age. J Toxicol Environ Health 1999;56:449-62. [CrossRef]
16. Nagmoti DM, Yeshwante SB, Wankhede SS, Juvekar AR. Hepatoprotective effect of Averrhoa Bilimbi Linn. Against carbon tetrachloride induced hepatic damage in rats. Pharmacologyonline 2010;3:1-6.
17. Sen S, Chakraborty R, Sridhar C, Reddy YS, De B. Free radicals antioxidant, diseases and phytomedicines:Current status and future prospect. Int J Pharm Sci Rev Res 2010;3:91-100.
18. Kroemer G, Deiry WS, Golstein P, Peter ME, Vaux D, Vandenabeele P, et al. Glstein Classification of cell death:Recommendations of the nomenclature committe on cell death. Cell Death Differ 2005;12:1463-7. [CrossRef]
19. Boron WF, Boulpep EL. Medical Physiology. Philadelphia, PA:Saunders;2003.
20. Situmorang PC, Syafruddin Ilyas S, Hutahaean S, Rosidah R. Components and acute toxicity of nanoherbal haramonting (Rhodomyrtus tomentosa). J Herbmed Pharmacol 2021;10:139-48. [CrossRef]