1. INTRODUCTION
Algae are one of the most potential sustainable future microbial cell factories which generate multiple forms of value added product biosynthesis. Macro and micro algae are the two very distinct domain of algae classification system. Macroalgae are the larger and multicellular aquatic photosynthetic plant-like living organisms while microalgae are comparatively smaller and unicellular aquatic photosynthetic plant like tiny microbial community in nature. Macroalgae are usually known to us as “seaweeds.” Red algae, green algae, and brown algal regimes belong to the macro algal taxonomy. On the other hand, microalgae have predominantly conferred as phytoplankton having diverse colorimetric appearance including blue-green, yellow, brown, or orange. There are two main group of microalgae do exist in natural system including diatoms and dinoflagellates. Microalgae are comparatively more economically sustainable because it requires less complicated platform to grown in the small scale or large scale production platform. Moreover, microalgae growth cycle is comparatively faster and homogeneous compared to macro algae growth pattern. Although algae are the major producers of algal bloom and induce pollution in the natural or oceanic water bodies, algae are most potential microbial cell factory which can biosynthesize diverse ranges of value added products including bioactive agents, biofuels, chemotherapeutic agents, and food supplements toward human health management in a sustainable manner within its cellular biomass [1]. Therefore, it would be an ideal idea if algal biomass production could be improved then it will indirectly induce the production rate and yield of these value added product generation per unit algal biomass. According to a recent United Nation (UN) survey, the global population has been projected to rise by 2 billion people in the upcoming 30 years, from 7.7 billion at present to 9.7 billion in 2050. To attain the demand of community high value added products including food and other biomolecules for the predicted population by 2050, it becomes ambitious on existing the agro-economic cycle. To retain current food production, putative models have been established to satisfy future global food demands to trap humanity in a vicious spiral of gradually decreasing agricultural outcome [2,3]. Hence, there is a circle of poverty leading to’ 4P’ that is over population, pollution, production and prodigality of reproduction [Figure 1]. Global production of macro-algae based on aquaculture is 30.51 Mt including few nations, that is, China, Indonesia, the Philippines, and South Korea. Global wild harvesting of microalgae is 1.12 Mt, out of which India has shared 0.019 Mt [4]. Among these countries, the USA ranks first in Spirulina-based products, primarily in the form of pills and spray-dried powder, followed by China, Israel, Japan, Mexico, Taiwan, and Thailand. There are so many merits to prioritize algal strains for value added production including high protein supplements as because of (1) potential photosynthetic conversion; (2) rapid growth rate; (3) high capacity to produce a wide variety of value added products; and (4) non-competitiveness for arable land. Predominantly, current scientific communities have been suggested that microalgae might be the key factor to solve many issues including pollution, hunger, energy, global warming, and diseases as an economically and environmentally sustainable arena. Microalgae have been used as food by humans for thousands of years [5-7]. A growing market for Spirulina sp. has been gradually establishing as pharmaceuticals, nutraceuticals, and protein supplements. There is a huge scope for both the small scale and large-scale algal farming provides a new dimension to solve another burning issue of unemployment in India [8]. Ulvarigida sp., Valoniaaegagropila sp., Chaetomorphaaerea sp., Gracilariaconfervoides sp., Microcystis sp., Chlorococcum infusionum sp., etc., algal species are cultivated for their various purposes [9]. Several studies have shown that micro algal growth can be improved by CO2 from the atmosphere or flue gases and they have better CO2 fixation abilities (10–50 times greater) than terrestrial plants. Spirulina sp. has 10 times more CO2 fixation rate than land plants [10]. Microalgae do have the capacity to fix CO2 from the three different sources namely from the atmosphere, from the industrial discharges and from the soluble carbonate. It is proved that 1.83 kg of CO2 is needed to produce 1 kg of algal biomass [11]. Microalgae can convert solar energy to chemical energy by fixing CO2, and its efficiency increases 10 times than terrestrial plants. The commercial production of microalgae is approximately 5000 tons/year of dry matter [12]. Microalgae growth rates and productivity are higher than any plant system. Dried biomass of algae would be the promising and alternative source of food as well as protein and other value-added products. Maximization of photosynthetic carbon capture efficiency and energy-return on investment in terms of food, fuel, and other value added products attributes of green algae attracted the scientists to produce large scale biomass production with low investment and extensive return [7,13-15].
 | Figure 1: Poverty cycle encourages improvement on algae biomass production. (Lower biomass productivity and Lower waste bioremediation can be executed through the involvement of algal biomass generation). [Click here to view] |
2. VARIOUS CULTIVATION STRATEGIES TOWARDS IMPROVING ALGAL BIOMASS PRODUCTION
Topographical and seasonal variation can bring the change of algal biomass generation. In the year 1978, National Renewable Energy Laboratory (NREL) launched a program called Aquatic Species Program (ASP) where 3000 micro-algal strains were collected from continental regions of US and Hawaii for systemic screening for higher lipid production under normal and stress conditions [16]. For the production of high value microalgae derived products, light intensity and photoperiod cycle are important factors for growth rate and biomass composition The PBRs system of cultivation is more expensive than open ponds but more harvested biomass [17]. Spirulina sp. is used in food even the astronauts are used this algae during the time of space journey. It is estimated that about 72,500 microalgae out of them only around 44,000 had been described and only few of the species are cultivated commercially [18]. Phycoprospecting or screening of microalgae [Figure 2] aids to find out the desirable strains such as high growth rate or tolerance of stresses (high temperature, pH, salinity, etc.). For example, in laboratory cultivation of Spirulina sp. is influenced by eight factors to get maximum yield namely luminosity (photo-period 12/12, 4 luxes), 30°C temperature, inoculation size, stirring speed, dissolved solids (10–60 g/L), pH (8.5–10.5), water quality, and macro-nutrients exist (C, N, P, K, S, Mg, Na, Cl, Ca and Fe, Zn, Cu, Ni, Co, and Se) [19]. Furthermore, some biochemical and biomolecular characterization has been observed in diverse algal regimes [Table 1]. The using of various micronutrients (A5, B6, Co, V, and C13) in Nostoc sp. have been shown the various biomass productivity treatment basis (63, 74, 62, 72, 77, and 62 mg/dry weight) [20]. Genetically engineered algal biomass production is reduces light harvesting antenna complex in Chlamydomomonas reinharditii has been shown the improvement on photosynthetic rates and biomass yield about 10–30% in laboratory culture. Moreover, tlaA truncated engineered C. reinharditii strains has shown the reduction of antenna size in both photosystems I and II are about to 50% and 65%, respectively [21]. Diverse ranges of high value added product generations from diverse algal regimes have been demonstrated [Figure 3] as short summarization.
 | Figure 2: Phycoprospecting of microalgae impacts on high value added product generation and environment in conjunction with genetic engineering. [Click here to view] |
Table 1: Biochemical and biomolecular characterization of diverse algal regimes.
| Algal species | Biomass generation | Cultivation system | Protein % | Carbohydrate % | Lipid % | References |
|---|---|---|---|---|---|---|
| Anabaena cylindrical | - | - | 43–56 | 25–30 | 4–7 | [6] |
| Arthrospira platensis | 0.91–2.7 g.L-¹.day-¹ | - | - | - | 13.0–30.0 (DCW) | [96] |
| Botryococcus braunii | 0.02 g.L-¹.day-¹ | - | - | - | 25.0–75.0 (DCW) | [96] |
| C. vulgaris | 40 mg.L-¹.day-¹ | Tubular photobioreactors | 51–58 | 12–17 | 14–22 | [6,99] |
| C. zofingiensis | 58.40 mg. L-¹. day-¹ | Photobioreactors (PBR) | - | - | - | [99] |
| C. emersonii | 41 mg.L-¹.day-¹ | Tubular photobioreactors | - | - | - | [99] |
| Chlorella sp. | 561 mg.L-¹.day- ¹and 168 mg.L-¹.day-¹ | Mixotrophic fed batch cultivation | - | - | - | [101,102] |
| Chlorella protothecoides | 51.2 g.L-¹.day-¹ | High yielding Photobioreactors | - | - | 51.3 (DCW) | [96] |
| Chlorococcuminfusionum | 0.28 g.L-¹.day-¹ | - | - | - | 19.3 (DCW) | [96] |
| Scenedesmus obtusus | 20.20 g.L-¹.day-¹ | - | - | - | - | [100] |
| C. pyrenoidosa | 4.3 g.L-1 | - | - | - | - | [107,108] |
| Chlamydomonas reinhardtii | - | - | 48 | 17 | 21 | [6] |
| Dunaliella salina | - | - | 57 | 32 | 6 | [6] |
| Haematococcus pluvialis | 0.05–0.7 g.L-¹.day-¹ | - | - | - | 25.0 (DCW) | [96] |
| Monoraphidium contortum | 0.89 g.L-¹.day-¹ | - | - | - | 31.5 (DCW) | [96] |
| Nannochloropsis sp. | 0.051–0.27 g.L-¹.day-¹ | - | - | - | 20.0–53.0 (DCW) | [96] |
| Porphyridium cruentum | - | - | 28–39 | 40–57 | 9–14 | [6] |
| Scenedesmus obliquus | - | - | 50–56 | 10–17 | 12–14 | [6] |
| Spirulina maxima | - | - | 60–71 | 13–16 | 6–7 | [6] |
| Spirulina platensis | 0.046–0.4 g.L-¹.day-¹ | - | - | - | 4.0 (DCW) | [96] |
| Synechococcus sp. | - | - | 63 | 15 | 11 | [6] |
| Tetraselmis sp. | 0.4–0.6 g.L-¹.day-¹ | - | - | - | 12.0–23.0 (DCW) | [96] |
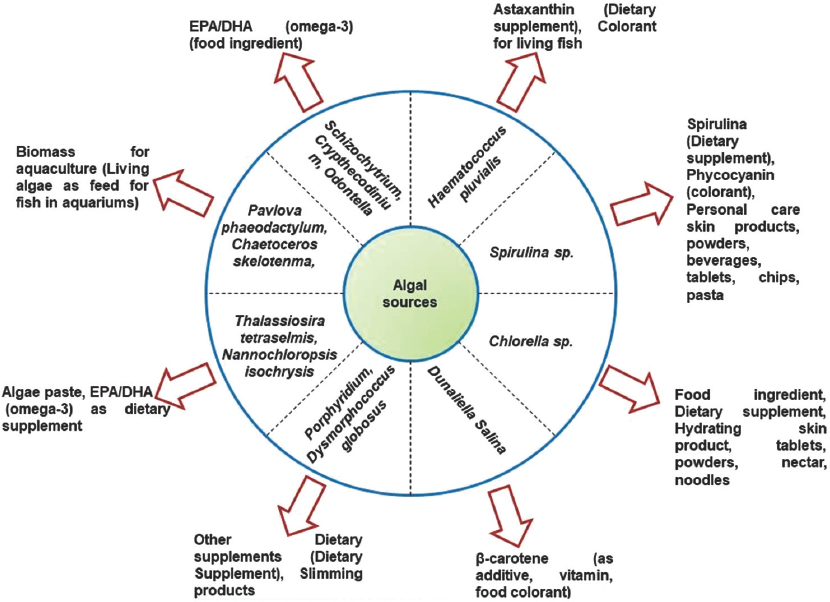 | Figure 3: High value added production derives from elevated algal biomass [4, 93, 114, 6] [Click here to view] |
3. ALGAE BASED VALUE ADDED PRODUCTS
Both microalgae and microalga are potential feedstock for production of different value added products (e.g., proteins, vitamins, minerals, fats, sugars, antioxidants, cosmetics, bio-pharmaceuticals, bioactive nutraceuticals, natural dyes, and colors in addition to animal feeds) to utilize their biomass. To balance the total biocatalytic efficacies from photon toward value added products, algae that are not only have the optimal carbon sequestration but also assimilate the extensive amounts of protein, carbohydrate, and fat contents. Algae have been widely used as a high-dietary supplement in human nutrition, aquaculture, nutraceuticals, fuel, and various purposes.
3.1. Algae as Protein Supplement Single Cell Proteins (SCPs)
As the world’s population grows day by day, there necessary food supply is becoming a major challenge. In this current, global scenario malnutrition deficiency is one the serious issue in India [22,23]. Overcome those foods and nutritional deficiencies dried biomass of algal species which are used as promising dietary supplements called single cell protein (SCP). In 1966, SCP was first termed by Carol L. Wilson [24]. Single cell protein (SCP) production from the algal sources is an inspirational field. Last few decades huge amount of algal cells have been used for feed additives due to their high nutritional value, high protein concentration. Spirulina sp. is the huge cultivated algal species for single cell protein production. Senedessmus sp., Chlorella has been cultivated as feed additives [24]. Spirulina sp. and Chlorella sp. are largely cultivated worldwide for feed purposes which yearly production 5000 and 2000 tons of dry matter/year, respectively [4]. Cyanophyceae groups of algal SCP contain of phycocyanin which has been used as antioxidant, anti-inflammatory, antitumor, and anticancerous agents. A study has been shown that Aphanizomenon sp. Arthrospira sp. and Chlorella sp. are one of the best algal sources of SCP production through extensive literature survey outcome [23]. Spirulina sp. contains 32.5 mg cholesterol/100 g algal dry biomass. Moreover, 10 g of Spirulina sp. powder contains 1.3 mg cholesterol and 36 kcal of energy, respectively [25]. On the other hand, some algal regimes (e.g., Anabaena cylindrical, Chlamydomonas reinhardtii, Chlorella vulgaris, and Dunaliella salina) are highly contains various yielded of protein, lipid, and carbohydrates like biomolecules [Table 1].
3.2. Algal Biomass Generation toward Biofuels Production
Since last few decades’ worldwide growth of population is one the major issue for energy crisis. Besides, global warming has gradually been increasing. In this mind many industries are focusing the production of algal derived biofuel. Mainly fossil fuels are hugely used in all over the world. At present, algae have been considered as potential microbial cell factories in various industrial sectors due to its high biomass generation, high productivity, CO2 sequestration, and higher efficiency of growth during various harsh environmental conditions. Dunaliella salina, Chlamydomonas reinhardtii, Haematococcus pluvialis, Selenastrum minutum, Ankistrodes musfalcatus, Haematococcus pluvialis, Chlorella vulgaris, Phaeodactylum tricornutum, Nannochloropsis oculata, Coccochlorispeniocystis, Botryococcus braunii, etc., algal species are the potential feedstock for biofuel production. Those are containing 20–50 % dry weight lipid which are very much effective substrate for fuel generation. Chlorella vulgaris contains starch (37% dry weight) which is used for ethanol fermentation [26]. B. braunii contains 80% oil in its biomass which is used for biodiesel production. Scenedesmus dimorphus produces 53.7 w/w carbohydrates which is a feasible component for bioethanol production during the hydrolysis process [27]. C. reinhardtii, Y67A algal strains have been shown hydrogen production in the presence of hydrogenase enzyme activities which is higher 10–15-folds which has been depicted in an extensive literature survey [28,29]. Zymobacter palmae produces maximum 0.38 g ethanol (gm.mannitol−¹) yield during the anaerobic condition [9]. Algal biomass produces 35–41 (MJ/kg) biodiesel during the transesterification process, Bioethanol 23.4 (MJ/kg) during fermentation, and 37.2 (MJ/kg) of biogas during anaerobic digestion, hydrothermal treatment, and 33–39 (MJ/kg) bio-oil during hydrothermal liquefaction process. Biological production of hydrogen, that is, 144 (MJ/kg) has shown the higher than another existing fuel producing feedstocks [30].
3.3. Algal Biomass Generation for Biochars and Algal Biofertilizers
Algal biochar has been produced through the thermo chemical methods. It contains higher amount of nitrogenous residues which enhance the soil quality, enhance the nutrient uptake efficiencies to the soil. Algal biochar has also been generated during the high temperature by pyrolysis process. Lyngbya sp. and Cladophora sp. are collected from Maumee bay of Lake Erie, in USA which produce (48 weight % per algal mass) biochar during the pyrolysis process at 510–600°C the calorific value was 25.6 (MJ/kg) [9]. In 1981, FAO has declared that algal biomass have been utilize as a potential biofertilizer replacement of the chemical fertilizer. The algal species are the best providers of nitrogen sources which are very useful for agricultural yield. Algal fertilizer increases the annual yield of rice production by an average 22% in India. Algal biomass has been potential to produce 25–30 kg of chemical nitrogen per acre. Spirulina sp. is one of the predominant algal species which are implemented as most potent group of bioferlizers due its low productivity cost, high availability, and higher biomass production. Spirulina platensis contains 10% of nitrogen content (weight/weight). Moreover, it releases carbon components, various micro and macronutrients which enhances the plant growths, increase the soil condition and fertility which indirectly most effective bioaugmentation approach for agricultural fields toward higher crop productivities.
3.4. Algal Biomass Generation toward Therapeutics and Bioadsorbent
Algal biomass is the predominant reservoir of diverse ranges of bioactive compounds which having great impact on human health. Algae based bioactive biomolecules has been biosynthesized through the advent of algal primary metabolic activities. In parallel, secondary metabolic networks have been associated with proteins, fatty acids, vitamins, and bio-pigments productions. Interestingly, these entire metabolisms derived bioactive compounds they do have extensive impacts on human health as antifungal, antiviral, algal toxins, anti-enzymatic and chemotherapeutic agents (i.e., antibiotics). Basic composition of algae derived bioactive molecules include polysaccharides, C-phycocyanin, phenolic acids, tocopherols (Vitamin E), neophytadiene, phytol, PUFAs (n-3) fatty acids, oleic acid, linolenic acid, palmitoleic acid, diacylglycerols, astaxanthin, lutein, zeaxanthin, canthaxanthin, b-carotene, carotenoids, sulfated polysaccharides, sterols, peptide, eicosapentaenoic acid (EPA), violaxanthin, trans-betacarotene, cis-betacarotene, palmitic acid, linear alkadienes (C25, C27, C29, and C31), triene (C29), borophycin, and cryptophycin. These bioactive molecules have been predominantly biosynthesized and/or exist in algal biomass including following algal regimes, that is, Spirulina platensis, Spirulina fusiformis, Haematococcus pluvialis, Chlorella vulgaris, Chlorella minutissima, Chlorella ellipsoidea, Dunaliella salina, Botryococcus braunii, Chlorella zofingiensis, Chlorella protothecoides, Chlorella pyrenoidosa, Nostoc linckia, and Nostoc spongiaeforme, respectively [31-33]. Moreover, these bioactive compounds are usually exploited and characterized utilizing different advanced biochemical tests (Folin–Ciocalteu, Bradford assay, and Kjeldahl assay, etc.) and high through put instruments, that is, GC-MS (gas chromatography-mass spectroscopy), LC-MS (liquid chromatography-mass spectroscopy), etc. [34]. Even these algal bioactive compounds do have profound impacts on human health issues as algal bioactive compounds act as antiproliferative agents, cancer growth inhibition, anti-inflammatory agents, chemotherapeutic agents, antiviral agents, and cholesterol lowering drugs [35]. Moreover, algae are the most valuable components in pharmaceutical industries. Various types of algal biomass have been utilized for various therapeutic purposes. It contains different value added products (e.g., omega-3 fatty acids, vitamins, phycobiliproteins, amino acid, and poly unsaturated fatty acids) which are being used as antioxidant, anti-inflammatory, anticancer, anti-aging, ant-tanning, and antitumor agents. Different algal species produces huge amount of biomass which are utilized for various therapeutic purposes. Spirulina sp. (3000 tonnes per dry biomass weight); Chlorella sp. (2000 tonnes/dry biomass weight), Dunaliella sp. (1200 tonnes per/dry biomass weight); Aphanizomenon sp. (500 tonnes/dry biomass weight); Haematococcus (300 tonnes/dry biomass weight); Crypthecodinium sp. (240 tonnes/dry biomass weight from DHA oil); and Schizochytrium sp. (10 tonnes/dry biomass weight from DHA oil). Phaeodactylum tricornutum has been shown the higher productivities for EPA (i.e., eicosapentaenoic acid) 430 kg EPA/year having purity of 96% (w/w) [5]. Chlorococcum infusionum and Leptolyngbya foveolaurum have been used as bioadsorbent during the wastewater treatment cultivation. Dried algal biomass has been collected as bio-adsorbent which have been used as in chemical adsorbent enhancer. Aforementioned algal regimes have been shown to bear the adsorbent capacity of 34.36 mg/g dry algal biomass at temperature of 30°C for 3.5 g/L dosage of adsorbent [9].
4. IMPACT OF VARIOUS PHYSICAL AND CHEMICAL STRESS FACTORS FOR ENHANCING ALGAL BIOMASS GENERATIONS THROUGH GENETIC ENGINEERING
4.1. Role of Phytohormones for Elevating Algal Biomass
It has been observed that phytohormones bring positive impact in vascular plants, so phytohormones could be a promising element that can reduce the inputs necessary to grow the potential microalgae for the highest biomass generation. The research on the role of phytohormones in algal growth is in the stage of infancy in comparison with vascular plants. A few works had been done but it was too scanty to get the valuable data for large scale algal biomass generation. In general, phytohormones are classified into three groups’ namely natural, artificial and postulated hormones. Source of auxins may be natural or artificial. Biological role of auxins in vascular plants and algae is likely similar. Auxins control the mitotic cell cycle, the transition of cells from dormancy to active proliferation, stimulates respiration, and influence biosynthetic processes [36]. Abscisic acid, Cytokinins (CKs), Auxins (IAA), and Gibberellins (GA) are the main phytohormones for algae as well as higher plants. IAA and CKs are used to stimulate the algal growth and enlarge the metabolites formation such as proteins, carbohydrates, lipid, and carotenoids [37]. IAA is synthesized in the apical shoot meristem and, it is able to form conjugates with glucose, aspartic acid, oligosaccharides, proteins, and nucleic acids [38]. Along with natural auxins there are many synthetic auxins which have similar physiological function. Like auxins, gibberellins promote growth and cytokinins promote cell division.ABA is a key hormone which helps to adapt with adverse climatic environment. Application of phytohormone and its concentration in the algal growth medium is also a vital parameter for algal biomass generation. In an experiment it has been shown that biomass production of Nannochloropsis salina has been elevated in presence of 0.1 mg/L of IAA [39] but higher concentration inhibits growth. By an experiment it has been shown that exogenous application of IAA in the culture medium of C. vulgaris brings maximum number of cell in compared to control after 48 h of cultivation [40]. Hence, the use of auxins in the field of algal biomass generation could enhance the large scale biomass generation. In an experiment it has been shown that at a concentration of 100 μM of natural auxins such as IAA, PAA, IBN, and synthetic auxins NAA inhibit the growth of C. vulgaris when applied them exogenously in the growth medium. However, at a concentration of 0.1 μM for IAA and IBA displayed the highest biological activity and PAA and NAA showed its highest stimulatory effect at 1 μM concentration and all auxins stimulate enzymatic (ascorbate peroxidase, catalase, and superoxide dismutase) and non-enzymatic antioxidant (ascorbate and glutathione) systems in C. vulgaris. Suppression of lipid peroxidation and hydrogen peroxide accumulation [40,41] has shown that addition of NAA and BAP (6-benzylaminopurine) in the growth medium of Chlorella pyrenoidosa separately brings 2.2 and 1.26 fold biomass in compared to the control but addition of BAP and kinetin in the growth medium of Chlorella pyrenoidosa separately brings 2.94 and 3.03 fold alpha-linolenic acid in compared to control. Again exogenous phytohormones reduce the toxic effect of heavy metal (Pb) on green algae Acutodesmus obliquus. Toxic metal (Pb) decreases endogenous auxins, cytokinins, and gibberellins level and increases absicisic acid [42]. In an experiment, it has been proved that exogenous brassinosteroids (brassinolide, 24-epibrassinolide, and castasterone) at the concentrations of 10–12–10–8 M accelerated growth of the cultured green microalga Chlorella vulgaris, increased the content of protein and nucleic acids in the cells, and stimulated photosynthetic processes [43]. In an experiment, Scenedesmus obliquus was used to analyze the coupled effect of benzyl amino purine (BAP) and gibberellic acid (GA) in N2 deficient growth media on cell growth, biomass and fatty acid generation. The result showed that the concentration of 10–5 M BAP increased the biomass by 1.44-fold, and 10–6 M GA by 1.35-fold. Fluvic acid has been triggered for lipid accumulation and gene expressed with Monoraphidium sp. FXY-10 strain toward augmented the ROS production, acetyl-CoA carboxylase activation, malic enzyme emanation for algal biomass enhancement [44,45].
4.2. Impact of Light Intensity on Algae Based Biomass Development
Lightning has an eminent impact on algal biomass cultivation. Algal regimes have been avowed as photosynthetic organism also. A major group of studies has been reported that algal species has higher potentiality to produce huge amount of biomass concentration during the autotrophic growth condition. Through the photosynthesis in microalgae and macroalgae both capture the solar energy and it has been converted into chemical energy and also transferred two large proteins such as photosystems PSI and PSII. CO2 has been rigid through the Calvin-Benson (CB) cycle during the dark reaction. Nannochloropsis sp. Monoraphidium sp., Scenedesmus obliquus, Pavlova lutheri, Nannochloropsis gaditana, and Chlorella sp., algal species has been developed their higher biomass concentration as well as TAG (Triacylglycerol) lipids in the high light intensities (200 mmol m–2.s–1) [46,47]. C. reinhardtii, TLA1 gene has been engineered through the cell antenna size reduction process. The species has been shown higher biomass productivity during the modifications. Also N. gaditana, C. sorokiniana, species has been shown their higher biomass productivity throughout the DNA, RNA sequencing, and gene manipulation techniques [48]. C. protothecoide and C. kessleri have been inhibited their growth during various higher light intensities 200 mmol m–2.s–1 and 120 mmol m–2.s–1 respectively [49]. A study have been represented some algal species has been produces higher chlorophyll during the light condition, through the effects of organic carbon substrate such as sucrose, that is, C. globosa (9.56 mg L¹ chl-a and 3 mg L¹ chl-b) C. minutissima, (14.28 mg L¹ chl a and 4.4 mg L¹ chl b), and S. bijuga (17.05 mg L¹ chl a and 6.36 mg L¹ chl b) [50]. During the 45.5% light irradiation, Scenedesmus dimorphus have been reached 0.45 g.L–1 maximum biomass concentration. Tetraselmis sp. has been produces highest biomass 0.57 g.L–1 during the open pond cultivation systems with high light intensities [37]. A study has been shows that Dunaliella tertiolecta was cultivated under the various LED lights and high biomass growth; higher cellular pigments productivity was beheld during 7.5–8.5 pH range [51]. Phaeodactylum diatoms are being preferred blue lights for their maximum growth phase. P. tricornutum species has been triggered for the utmost biomass growth during the high light stress condition [52]. Many of algal species (e.g., Haematococcus pluvialis, Botryococcus braunii, Desmodesmus sp., and Chlorella sp. PCH90) have been grown in PBR (photobiorecator) during the high light severity and their biomass as well as lipid productivity was up to 40% rather than normal light and cultivation systems [53]. Haematococcus pluvialis have been raised astaxanthin production three-fold higher through the enhancement of light intensities. At higher light intensity (300 mmol photons m–2. s–1), lipid formation have been increased up to 35%. Furthermore, in this studies have been revealed that astaxanthin was reached more potency during blue light with low intensity 8 or 12 mmol photons m–2.s-¹ compared to red light with higher intensities (80–240 mmol photons m–2.s–1 [54]. When the light stress has been combined with the salt stress, nitrogen stress, temperature it was being more effective for the lipid accumulation and biomass growth for the algae. During the high light intensities 300 mmol photons m–2 s–1, 400 mmol photons m–2.s–1, P. tricornutum, I. galbana produces 31.4 mg.L–1.d–1, and 21.7 mg/L–1.d–1 TAG. Trentepohlia arborum contents 13.4% (3.0 mg L–1.d–1) DCW carotenoids with 26.3% (65.5 mg.L–1.d–1) of DCW lipid during the 150 mmol photons m–2.s–1 light stress conditions [55]. A study has been shown that (1000–2000 lx) light illumination was increased microalgal biomass accumulation [56]. During the 50, 250, and 400 mmol photons m–2.s–1 and 200, 500, 800, and 1500 mmol photons m–2.s–1 light intensities Scenedesmus sp., Tetradesmus obliquus has been reached 3.88 g.L–1 and 0.263 g.mol–1 photon highest biomass and fatty acid yield [45]. Scenedesmus obliquus has been increased 53–73% glucose of total carbohydrates during the 60–180 mmol m–2.s–1 photons light intensities [57].
4.3. Role of Bacterial Interactive Associations with Algae toward Improving Biomass Content
Some bacterial species were being used for algal growth cultivation and it has been exhibited most salient effects through several experimental analysis. Those bacterial species are eminently promotes the growth factors during the algal biomass cultivation systems. Hyphomonas sp. Rhizobium sp. Sphingomonas sp., bacterial species has been interacted with volatile compounds (e.g., Dimethyl disulphide, Tetranitromethane, Indole, and Dimethyl trisulfide) and works as nutrition supplements for algal (e.g., C. vulgaris) culture refinement. Furthermore, with the genetic modifications tnaA genes have been manipulated through knock out procedure with E. coli K12 mutant strain toward enhance the algal biomass generation [58]. Some zooplankton, bacteria, virus, and protozoa have also been used as an environmental key factors for algal (e.g., C. protothecoide and C. kessleri) biomass, lipid productivity. A study have been reported that Kinetin, gibberellic acid (GA3), indoleaceticacid (IAA), Jasmonic acid (JA), Salicylic acid (SA), Methyl jasmonate (MJ), Polycyclic aromatic hydrocarbons, Indomethacin (IM), Ethanolamine, 2,2_-azo-bis(2-amidinopropane)-dihydrochloride (AAPH), viologen (MV), 2-phenylacetic acid (PAA), Indole butyric acid (IBA), naphthalene acetic acid (NAA), N-6-benzylaminopurine, N-6-furfu-rylamineopurine, allantoin (AT), brefeldin A, anthranilic acid, and tryptamine, 2-phenylacetic acid (PAA) such as some chemicals have been metabolized with different types of interactions during the synthesis of various algal biomolecules from diverse microbial regimes (i.e., Chlorella vulgaris, Monorapbidium convolutum, Dunaliella salina, Spirulina platensis, Chlorella pyrenoidosa, Chlorococcum sp., Anabaena sp. PCC7120, Scenedesmus obliquus, Microcystis aeruginosa, Haematococcus pluvialis, Chlamydomonas reinhardtii, Selenastrum capricornutum, Nostoc muscorum, and Tolypothrix tenuis) including biomass generation [59]. A study has also been revealed that some algae-bacteria interactions have been synthetically induced for algal biomass growth augmentation. The phycosphere bacterial communities have been interacted with axenic C. vulgaris (ACV) algal species and significant algal biomass accretions (P < 0.05) were perceived [60]. During the Emiliania huxleyi algal cultivation, with Roseobacter species interaction has been widely studied. Azospirillum, Mesorhizobium species were provided the nutrients of the algal cells towards amelioration the biomass equality. Through the genetic modifications Roseobacterium sp. and Ruegeria pomeroyi DSS-3 have been interacted with Thalassiosira pseudonana CCMP1335 diatom strain. The bacterial consortium continuously has been provided the Vitamin B12 in to the algal culture towards raise the algal growth throughout the 2, 3 dihydroxypropane-1-sulphonate exhalation. The a-proteobacteria Roseobacter, g proteobacteria Marinobacter sp. were interacted with microalgal sp. and those bacterial species produces siderophore vibrioferrin compounds which binds with Fe (III) and gives nutrition in algal culture medium. Scenedesmus sp. BA032 and Neochloris oleoabundans algae have been fused with A. vinelendi species. This bacterium sp. is known as a higher supplier of nitrogen source for algal biomass cultivation. Chlorella vulgaris shows superior lipid production yield under the heterotrophic conditions throughout the faction of Azospirillum brasilense. It is a growth promoter bacterium species for higher plants. Moreover, Phaeobacter gallaeciensis is known as a growth enhancer and antibiotic producing Roseobacterium species for algal cells. A study has been proclaimed that under the suboptimal growth conditions E. huxleyi produces p-coumaric acid. P. gallaeciensis triggers the algaecide Roseobacticide during the biosynthesis. It has been lysed the microalgal cells at nanomolar concentrations and provided the nutrition and food during the cultivation in to the algal cells simultaneously [61].
4.4. Effects of pH on Algal Biomass Generation
The pH is another factor that directly or indirectly effects some algal growth. The pH used as a nutrient adsorbent for algal culture medium. The range of pH 10-11 or its above is not suitable for most of the algal biomass generation system. In the culture medium carbon source is being balanced by pH. And it plays very eminent role for algal biomass generation. Nannochloropsis sp.; Tetraselmis sp.; and Isochrysis sp. have grown in the 6.75–7.25 pH range. After 15 days, cultivation pH value has been reached 8.25. A recent study have been depicted that HCL, sodium bicarbonate can control the pH range during the cultivation of Tetraselmis suecica, Chlorella sp. carbonic acid have been formed by these compounds and used as a carbon source for algal culture medium toward more biomass production. During the 5–11 pH range Chlorella sp. produces 167.5 mg.L–1 highest biomass yields within 30 days cultivation. During 5.5–8 pH range Chlorella sp. produces 1600 mgl highest biomass yield during 50 days cultivation. During 6.5–8 pH value Tetraselmis suecica produces 950 mg/L highest biomass yield within 50 days cultivation. However, different algal species has been varies different pH ranges. Most of the algae have been grown within 7–9 pH range. Also some study has been revealed that Anabaena variabilis cultivation medium pH value was 8.2–8.4. At the 8–9 pH range, Nannochloropsis sp. has been generated their highest biomass. Thalassiosira pseudonana; Thalassiosira oceanica, and Skeletonema costatum are cultivated in >pH 8.8 and 6.5–9.4 pH range, respectively [46]. Pinguiococcus pyrenoidosus 2078 algal strain has been cultivated with 7 pH ranges culture medium. The highest PUFAs and EPA 38.75% and 23.13% of the total fatty acids yield has been observed [47]. Spirogyra and Oedogonium species has been observed highest growth rate during 7.5 pH condition. Chroococcus turgidus, Lyngbya confervoides, Nostoc commune, Chaetoceros calcitrans, and Skeletonema costatum species has been produced their highest biomass at maximum pH of 8.5 [62]. S. abundans produces 769 mg.L–1 and 179 mg.L–1 biomass and lipid yield during the 6–8 pH range. N. oleoabundans produces 1.04 g.L–1, and 19.1 mg.L–1.day–1 cell growth and lipid generation yield in pH 9.5. Ettlia sp. YC001 and S. obliquus have achieved 96.7 mg.L–1.d–1 biomass and lipid growth rate during the 8.5 and 7.0 pH conditions [55,63].
4.5. Influence of Nutrients, Salinity and Different Metal Ion Stress towards Improving Algal Biomass
Nutrients, salinity stress, and various metal ions are also plays a crucial role on algae cultivation and raise their biomass growth, respectively. A research experiment has been depicted that C. vulgaris and S. obliquus algal species were grown with Mg2+, Ca2+ and NaCl nutrients culture medium. It shows 1.6 g.L–1, 1.5 g.L–1, and 331 mg.L–1, 224 mg.L–1 lipid yields, respectively. Furthermore, ~40% (DCW) lipid yield has been observed in both cultures as well as in the NaCl supplemented medium [64]. Under the adequate nitrogen starvation estate algal regimes were increased rapidly 7–10 folds. During the nitrogen assimilation photosynthesis receptivity was developed and the TAG lipid production has been ameliorated. Prorocentrum donghaiense and Chlorella rathinasabapathi increase 2–3 fold lipid biomass yield under moderate nutrient stress and nitrogen deprivation conditions. Monallantus salina produces 72% lipid under nitrogen stress. Scenedesmus sp.; and Chlorella sorokiniana reached >40% lipid yield. During the iron concentration, Ankistrodesmus falcatus has been reached their highest biomass yield 6 mg L–1. Chlorella minutissima and Scenedesmus obliquus produces 29.19% and 18% to 40% dry weight biomass, respectively [46,47]. Furthermore, Dunaliella parva, Chlorella sp., Scenedesmus sp., Monodus subterraneus, Chaetoceros sp., Pavlova lutheri, Nanochloris atomus, and Sprirulina sp. several algal species can produce different quantities of biomass and lipid contents under phosphorus and trace metal stress (i.e., Fe3+, NaNO3, Mg2+, FeCl3, and ZnCl2 etc.). C. nivalis and Dunaliella sp. have been cultivated in extreme salt stress condition. In the higher NaCl concentration, those species has been produces their maximum biomass 70.13 g/L g.L–1 and 70% lipid (DCW) yield. Besides Chlorella minutissima and Nitzschia Laevis produces 40 and 20 g.L–1 lipid biomass yield in high salt concentration medium. An experimental study has been exposed that the usage of sodium ions during C. Mexicana and S. obliquus algae cultivation generated 0.8 g.L–1 and 0.65 g.L–1 higher biomass yield within 20 days cultivation period. Monodus subterraneus increase ~ 6 fold TGA lipid yield during the phosphorus limiting conditions [65]. Acutodesmus obliquus produces the highest biomass yield 160.71 mg.L–1.d–1 during the magnesium, calcium, and iron stress. A study have also been reported that Ankistrodesmus falcatus KJ671624 algal strain was cultivated and manipulated through the nitrogen, phosphorus and iron stress 750 mg.L–1, 0 mg.L–1, and 9 mg.L–1. It reaches to74.07 mg.L–1.d–1 lipid productivity yield [66,67]. 1.0–10% nitrogen content microalgal biomass. During this feasible ecological pressure on N deficiency lipid accumulation has been reached higher yield. Molybdenum, zinc, cobalt, copper, boron, and chloride are also elemental nutrients for algal growth. A study have been revealed that Monoraphidium sp. T4X algal strain has been cultivated in six different nitrate stress conditions and it has been seen that the highest biomass and lipid productivity yield emphasizing 0.36 g/l/day and 0.18 g/L/day simultaneously [53]. Throughout the sudden nitrogen stress conditions Chlorella vulgaris NIES-227, Nannochloropsis sp., Acutodesmus dimorphus produces 89% fatty acid methyl esters, 60% lipid, 75% neutral lipid of total dry algal biomass cells [55]. 35 g.L–1 glucose and 0.125 g.L–1 NaNO3 concentration Chlorella sorokiniana FACHB-275 strain content 320 mg.g–1 dry weight lipid yields. Scenedesmus sp. CCNM 1077 and Monoraphidium dybowskii LB50 strain produces 74.8% neutral lipids under several NaCl concentrations [45].
5. GENETICALLY ENGINEERED ALGAE FOR IMPROVING BIOMASS FOR SUSTAINABLE UTILIZATION
Genetically engineered algal regimes has been paid great attention than wild type background in term of gaining higher productivities and yields of desired value added products generations. There are several avenues which use to transfer exogenous genes for heterologous functionalization in sustainable algal host. However, any single genetic alteration is not fully satisfactory to solve aforementioned issues (lower productivity of value added products, and lower biomass productivities) in broad spectrum of algae considering diversity in algal cell wall composition and complicated structural assemblies. There are majorly three types of genetic material namely nuclear, chloroplast and mitochondrial genome which influence the level of gene expression and regulations in different compartments within algal cellular platform. Model eukaryotic algae Chlamydomonas reinhardtii is most effective algal host where lots of genetic manipulative approaches have been extensively applied by various researchers time to time [68-71]. However, the rate of genetic stability, biomass productivity, metabolic regulations, and heterologous expression levels are quiet inconsistent till date. However, few success rates have also been reported involving Volvox carteri, Chlorella sp. [72], Dunaliella salina [73], Haematococcus pluvialis [74] Euglena gracilis [75], Diatoms (Phaeodactylum tricornutum, Naviculasaprophila sp, Cyclotellacryptica sp. and Thalassiosira weissflogii), dinoflagellates (Amphidinium klebsii and Symbiodinium microadriaticum), and red algae Porphyridium sp. [76] and Cyanidioschyzon merolae). The genetically modified C. reinhardtii have generated through use of Agrobacterium tumefaciens mediated Ti plasmid based gene transfer approach [39]. Genetically modified C. reinhardtii transformants are being screened through using selective markers, that is, antibiotic dominant marker concerning genes which confers certain difficulties to grow. Therefore, certain specific kinds of selective marker has been designed and established for Chlamydomonas reinhardtii likely the R100.1 plasmid/bacteriophage T4/synthetic aminoglycoside adenyl transferase gene (aadA) confers resistance to spectinomycin and streptomycin [77], the Streptoalloteichus hindustanus selectable gene to zeomycin and phleomycin [78], the mutated Chlamydomonas reinhardtii protoporphyrinogen oxidase gene (ppx1) to the N-phenyl heterocyclic herbicide S-23142 [79]. An extended analysis has clearly been implemented to enhance algal biomass growth through the alternations and advancements of genetically modified tools. Light-harvesting complex stress-related (Lacer) proteins, orange carotenoids Protein (OCP), and over expression of RuBisCO tends to elevate the algal biomass yield. Two light harvesting complex proteins likely light-harvesting complex stress-related (LHCSR) and PSBS-photosystem II subunit-S are being used for expansion of the photosynthetic antenna size of C. reinhardtii with the 77K fluorescence spectrum. Super complex LHCSR3 proteins have fused with PSII-LHCII for fixation of photosynthetic stability [80,81]. To attain few algal cellular targets, that is, Arthrospira maxima and Synechocystis sp. have been fused with OCP-orange carotenoids protein. OCP is a photoactive protein with the carotenoids and 39-hydroxyechinenone main domains. However, slr1963 gene and OCP associated genes have been interacted in Synechocystis sp. toward acceleration of photosynthetic mechinaries. Synechocystis slr1964 gene has fused with fluorescence recovery protein (FRP) to accelerate the light independent binding efficacy. Synechocystis sp. PCC 6803 has encoded with flavodiiron proteins (FDPs) throughout the fusion of sll1521 (flv1), sll0219 (flv2), sll0550 (flv3), and sll0217 (flv4) multiple gene clusters for the induction of the photo-protection mechanism. During the dark period algal cultivation few promoters such as fcp and EF2 have been used for several downstream genetic expression in heterologous host, that is, P. tricornutum diatom for gene manipulation as well as light inducer [82,83]. Synechocystis sp. PCC6803 model algal species achieve 0.08 g dry cell weight per liter biomass with the addition of 1 g/L glucose through implementation of the proteome allocation system. In addition, during the downregulation of rbcS gene under the CO2 enriched conditions using of anti-sense RNA (35%) increase the photosynthesis rate 15%. This approach can only be applied for rice plant, since currently it should be a baronial approach for enhance algal biomass biosynthetic using the genetic modification [84]. CPC-operon deletion has been initiated in Synechocystis species (Dcpc) for elevate generation yield as well as photosynthesis efficiency. After the modifications of Planktothrix PCC 7821, Planktothrix PCC 7805 strains have shown that higher quantities of electron globules formations in the thylakoids membrane [85,86]. Rubisco enzyme system based metabolic activities has been applied to Haematococcus pluvialis species toward genetic alternation under mixotrophically cultivation state. Moreover, rbcL and rbcS genes are overexpressed to enhance algal biomass quantity. Subsequently, LhcsR1, LhcsR3.1, and LhcsR3.2 homologous genes in the C. reinhardtii algae are enciphered with LhcsR protein during the psbS1 and psbS2 gene fusion by psbS. Moreover, these immense genetic modification systems influence the variant signals such as light intensities (higher light efficiency, UV light, and red light consumption) during the gene expression [87,88]. In addition to dominant selectable marker are also established recessive markers which are required for auxotrophic mutants with mutations in the corresponding endogenous gene and the corresponding intact gene for complementation. Often selectable marker genes cannot be expressed under their own promoters, especially if they come from a heterologous source. There are several algal species have undergone genetic engineering approaches to improve the biomass generation, photosynthetic efficiencies, and carbon dioxide sequestration (Rubisco enzymatic activities) which are described in Table 2.
Table 2: Genetic and metabolic engineering toward algal biomass enhancement.
| Purpose of gene expression | Species | Gene | Mode of action | Biomass production and accessory observations | References |
|---|---|---|---|---|---|
| Photosynthetic CO2 assimilation (Calvin cycle) | S. elongatus | RuBisCO (rbcLS and rbcS) | Over-expression | 1–4 fold increase in Rubisco activity. | [90] |
| Synechocystis sp. | RuBisCO (rbcL and rbcS) | Over-expression | Increased growth rate and photosynthesis. | [91] | |
| Nannochloropsis oceanica | RuBisCoactivase (nNoRca-like) | Over-expression | Biomass and lipid productivity increased by 46 and 41%, respectively. | [92] | |
| Dunaliella bardawil, Chlamydomonas reinhardtii | Sedoheptulose 1,7 bis-phosphatase (CrSBPase) | Over-expression | Improved photosynthetic performance. | [93] | |
| Chlorella vulgaris | s-FBA | Over-expression | Increased photosynthesis by 1–2 fold and enhanced growth. | [94] | |
| Optimum light energy use efficiency | Chlamydomonas reinhardtii. | tla1 | Suppression | Higher photosynthetic productivity. | [22] |
| Chlorella vulgaris | CpSRP43 | Suppression | Higher photosynthetic yield | [95] | |
| Chlamydomonas reinhardtii | tla4 | Suppression (450 mmol photons m–2.s–1) | Higher photosynthetic quantum yield 0.154 ± 0.005 h–1 | [109] | |
| Chlamydomonas reinhardtii | TLA3-CpSRP43 | Suppression | Improved efficient solar energy conversion efficiency. | [104] | |
| Chlamydomonas reinhardtii | chlorophyllide a oxygenase (CAO) | Suppression | Two fold increase in photosynthetic rate | [103] | |
| Chlamydomonas reinhardtii | LHC1 | Suppression | Higher photosynthetic quantum yield increase photosynthetic efficiency levels 1–2% from any environment | [98] | |
| Chlamydomonas reinhardtii | Ad1/Pmp1, HLA3 | Over-expression | Maintain high CO2 concentration at the catalytic site of RuBisCO, induce photosynthetic Ci affinity | [97,105] | |
| Abiotic stress tolerance | Schizochytrium sp. | SOD | Over-expression | Enhanced PUFA production without compromising growth rate | [106] |
6. FUTURE OUTLOOK
In this current extensive literature review, there are several aspect of algal biomass based high value added product generation has been clearly depicted. There are several aspect of algal genetic modulatory approaches have also been included for accelerating algal biomass productivities and yields in different algal regimes for improving value added industrially feasible products generations. However, the current algal biomass aspired bio-platform or microbial cellular factory approach is still in its infancy toward large scale production or scaling up stage due to different biorefinery constraints. Moreover, algae derived biomolecules do contain several drawbacks which restrict algal biomass associated technology to fully implement in industrial scale production process to serve community as an economically alterative viable technology. These major demerits include higher production cost, CO2 generation during combustion, and algal toxins mediated human health and environmental issues [89]. To this end, these major disadvantages need to be addressed to make the algal biomass associated high value added products generations sustainable in near future.
7. AUTHORS’ CONTRIBUTIONS
Mr. Kamalendu De has initiated drafting, formatting and visualization of figures and table; Ms. Shrestha Debnath has continued reviewing the primary draft, formatting and grooming of figures and tables; Dr. Dipankar Ghosh has conceptualized the primary draft construction following final drafting, editing, reviewing, all correspondence, and supervision.
8. CONFLICT OF INTEREST
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this review article.
9. FUNDING
There is no funding to report.
10. DATA AVAILABILITY
Datasets are outcome of extensive literature survey and references are cited in the reference section.
11. Ethical approvals
This study does not involve experiments on animals or human subjects.
12. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Mostafa SS. Microalgal biotechnology:Prospects and applications. Plant Sci 2012;12:276-314.
2. Springmann M, Clark M, Mason-D'Croz D, Wiebe K, Bodirsky BL, Lassaletta L, et al. Options for keeping the food system within environmental limits. Nature 2018;562:519-25. [CrossRef]
3. Harvey M, Pilgrim S. The new competition for land:Food, energy, and climate change. Food Policy 2011;36:S40-51. [CrossRef]
4. Enzing C, Ploeg M, Barbosa M, Sijtsma L. Microalgae-based Products for the Food and Feed Sector:An Outlook for Europe. JRC Scientific and Policy Reports;2014. 19-37.
5. Milledge JJ. Commercial application of microalgae other than as biofuels:A brief review. Rev Environ Sci Biotechnol 2011;10:31-41. [CrossRef]
6. Spolaore P, Joannis-Cassan C, Duran E, Isambert A. Commercial applications of microalgae. J Biosci Bioeng 2006;101:87-96. [CrossRef]
7. Wilkie DS, Bennett EL, Peres CA, Cunningham AA. The empty forest revisited. Ann N Y Acad Sci 2011;1223:120-8. [CrossRef]
8. Scott D, Becken S. Adapting to climate change and climate policy:Progress, problems and potentials. J Sustain Tour 2010;18:283-95. [CrossRef]
9. Renita A, Kumar PS. Valorization of waste algal boom for value-added products. In:Bioprocess Engineering for Bioremediation. Berlin, Cham:Springer;2020. 129-37. [CrossRef]
10. Yadav G, Sen R. Microalgal green refinery concept for biosequestration of carbon-dioxide vis-à-vis wastewater remediation and bioenergy production:Recent technological advances in climate research. J CO2 Util 2017;17:188-206. [CrossRef]
11. Chisti Y. Biodiesel from microalgae. Biotechnol Adv 2007;25:294-306. [CrossRef]
12. Raja R, Hemaiswarya S, Kumar NA, Sridhar S, Rengasamy R. A perspective on the biotechnological potential of microalgae. Crit Rev Microbiol 2008;34:77-88. [CrossRef]
13. Del Campo JA, García-González M, Guerrero MG. Outdoor cultivation of microalgae for carotenoid production:Current state and perspectives. Appl Microbiol Biotechnol 2007;74:1163-74. [CrossRef]
14. Ghosh D, Hallenbeck PC. Cyanobacteria and photosynthetic bacteria:Metabolic engineering of hydrogen production. U S Air Force Acad 2015;8:112-24. [CrossRef]
15. Ghosh D, Hallenbeck PC. Metabolic engineering of hydrogen production by green algae. Adv Enzym Convers Biomass Biofuels 2015;1:96-110. [CrossRef]
16. Sheehan J, Dunahay T, Benemann J, Roessler P. A look back at the US department of Energy's aquatic species program:Biodiesel from algae. Natl Renew Energy Lab 1998;328:1-294. [CrossRef]
17. Khoeyi ZA, Seyfabadi J, Ramezanpour Z. Effect of light intensity and photoperiod on biomass and fatty acid composition of the microalgae, Chlorella vulgaris. Aquac Int 2012;20:41-9. [CrossRef]
18. De Clerck O, Guiry MD, Leliaert F, Samyn Y, Verbruggen H. Algal taxonomy:A road to nowhere?J Phycol 2013;49:215-25. [CrossRef]
19. Ciferri O. Spirulina, the edible microorganism. Microbiol Rev 1983;47:551-78. [CrossRef]
20. Holm-Hansen O, Gerloff GC, Skoog F. Cobalt as an essential element for blue-green algae. Physiol Plant 1954;7:665-75. [CrossRef]
21. Polle JE, Kanakagiri SD, Melis A. Tla1, a DNA insertional transformant of the green alga Chlamydomonas reinhardtii with a truncated light-harvesting chlorophyll antenna size. Planta 2003;217:49-59. [CrossRef]
22. Adedayo MR, Ajiboye EA, Akintunde JK, Odaibo A. Single cell proteins:As nutritional enhancer. Adv Appl Sci Res 2011;2:396-409.
23. De K, Ghosh D. Microbial single cell protein generation:A comparative existing state of art. In:De D, Roy DS, Bera GC, editors. Biotechnology and Nature. Midnapore Sadar:Kabitika;2018. 94-8.
24. Suman G, Nupur M, Anuradha S, Pradeep B. Single cell protein production:A review. Int J Curr Microbiol Appl Sci 2015;4:251-62.
25. Heidarpour A, Fourouz AD, Eghbalsaied S. Effects of Spirulina platensis on performance, digestibility and serum biochemical parameters of Holstein calves. Afr J Agric Res 2011;6:5061-5.
26. Juneja A, Ceballos RM, Murthy GS. Effects of environmental factors and nutrient availability on the biochemical composition of algae for biofuels production:A review. Energies 2013;6:4607-38. [CrossRef]
27. Khan MI, Shin JH, Kim JD. The promising future of microalgae:Current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb Cell Fact 2018;17:1-21. [CrossRef]
28. Godman JE, Molnár A, Baulcombe DC, Balk J. RNA silencing of hydrogenase (-like) genes and investigation of their physiological roles in the green alga Chlamydomonas reinhardtii. Biochem J 2010;431:345-52. [CrossRef]
29. Pinto TS, Malcata FX, Arrabaça JD, Silva JM, Spreitzer RJ, Esquível MG. Rubisco mutants of Chlamydomonas reinhardtii enhance photosynthetic hydrogen production. Appl Microbiol Biotechnol 2013;97:5635-43. [CrossRef]
30. Usher PK, Ross AB, Camargo-Valero MA, Tomlin AS, Gale WF. An overview of the potential environmental impacts of large-scale microalgae cultivation. Biofuels 2014;5:331-49. [CrossRef]
31. De Morais MG, Vaz BD, De Morais EG, Costa JA. Biologically active metabolites synthesized by microalgae. BioMed Res Int 2015;2015:835761. [CrossRef]
32. Michalak I, Chojnacka K. Algae as production systems of bioactive compounds. Eng Life Sci 2015;15:160-76. [CrossRef]
33. Santhosh S, Dhandapani R, Hemalatha N. Bioactive compounds from microalgae and its different applications - a review. Adv Appl Sci Res 2016;7:153-8.
34. Basily HS, Nassar MM, El Diwani GI, El-Enin SA. Exploration of using the algal bioactive compounds for cosmeceuticals and pharmaceutical applications. Egypt Pharm J 2018;17:109-20. [CrossRef]
35. Balasubramaniam V, Gunasegavan RD, Mustar S, Lee JC, Mohd Noh MF. Isolation of industrial important bioactive compounds from microalgae. Molecules 2021;26:943. [CrossRef]
36. Del Pozo JC, Lopez?Matas MA, Ramirez?Parra E, Gutierrez C. Hormonal control of the plant cell cycle. Physiol Plant 2005;123:173-83. [CrossRef]
37. Salama ES, Hwang JH, El-Dalatony MM, Kurade MB, Kabra AN, Abou-Shanab RA, et al. Enhancement of microalgal growth and biocomponent-based transformations for improved biofuel recovery:A review. Bioresour Technol 2018;258:365-75. [CrossRef]
38. Davies PJ. Regulatory factors in hormone action:Level, location and signal transduction. In:Plant Hormones. Netherlands, Dordrecht:Springer;2010. 16-35. [CrossRef]
39. Kumar SV, Misquitta RW, Reddy VS, Rao BJ, Rajam MV. Genetic transformation of the green alga-Chlamydomonas reinhardtii by Agrobacterium tumefaciens. Plant Sci 2004;166:731-8. [CrossRef]
40. Bajguz A, Piotrowska-Niczyporuk A. Interactive effect of brassinosteroids and cytokinins on growth, chlorophyll, monosaccharide and protein content in the green alga Chlorella vulgaris (Trebouxiophyceae). Plant Physiol Biochem 2014;80:176-83. [CrossRef]
41. Kokkiligadda S, Pandey B, Ronda SR. Effect of plant growth regulators on production of alpha-linolenic acid from microalgae Chlorella pyrenoidosa. S?dhan?2017;42:1821-4. [CrossRef]
42. Piotrowska-Niczyporuk A, Bajguz A, Kotowska U, Zambrzycka-Szelewa E, Sienkiewicz A. Auxins and cytokinins regulate phytohormone homeostasis and thiol-mediated detoxification in the green alga Acutodesmus obliquus exposed to lead stress. Sci Rep 2020;10:1-4. [CrossRef]
43. Bajguz A. Effect of brassinosteroids on nucleic acids and protein content in cultured cells of Chlorella vulgaris. Plant Physiol Biochem 2000;38:209-15. [CrossRef]
44. Correa-Aguado HC, Cerrillo-Rojas GV, Rocha-Uribe A, Soria-Guerra RE, Morales-Domínguez JF. Benzyl Amino purine and gibberellic acid coupled to nitrogen-limited stress induce fatty acids, biomass accumulation, and gene expression in Scenedesmus obliquus. Phyton 2021;90:515. [CrossRef]
45. Chu WL. Strategies to enhance production of microalgal biomass and lipids for biofuel feedstock. Europ J Phycol 2017;52:419-37. [CrossRef]
46. Sajjadi B, Chen WY, Raman AA, Ibrahim S. Microalgae lipid and biomass for biofuel production:A comprehensive review on lipid enhancement strategies and their effects on fatty acid composition. Renew Sustain Energy Rev 2018;97:200-32. [CrossRef]
47. Paliwal C, Mitra M, Bhayani K, Bharadwaj SV, Ghosh T, Dubey S, et al. Abiotic stresses as tools for metabolites in microalgae. Bioresour Technol 2017;244:1216-26. [CrossRef]
48. Dall'Osto L, Cazzaniga S, Guardini Z, Barera S, Benedetti M, Mannino G, et al. Combined resistance to oxidative stress and reduced antenna size enhance light-to-biomass conversion efficiency in Chlorella vulgaris cultures. Biotechnol Biofuels 2019;12:1-7. [CrossRef]
49. Zhou W, Chen P, Min M, Ma X, Wang J, Griffith R, et al. Environment-enhancing algal biofuel production using wastewaters. Renew Sustain Energy Rev 2014;36:256-69. [CrossRef]
50. Bhatnagar A, Chinnasamy S, Singh M, Das KC. Renewable biomass production by mixotrophic algae in the presence of various carbon sources and wastewaters. Appl Energy 2011;88:3425-31. [CrossRef]
51. Kumar SS, Saramma AV. Effect of plant growth regulators on growth and pigment composition of microalga, Nannochloropsis salina DJ Hibberd. Appl Biol Res 2018;20:228-33. [CrossRef]
52. Fu W, Wichuk K, Brynjólfsson S. Developing diatoms for value-added products:Challenges and opportunities. N Biotechnol 2015;32:547-51. [CrossRef]
53. Moreno-Garcia L, AdjalléK, BarnabéS, Raghavan GS. Microalgae biomass production for a biorefinery system:Recent advances and the way towards sustainability. Renew Sustain Energy Rev 2017;76:493-506. [CrossRef]
54. Markou G, Nerantzis E. Microalgae for high-value compounds and biofuels production:A review with focus on cultivation under stress conditions. Biotechnol Adv 2013;31:1532-42. [CrossRef]
55. Chen B, Wan C, Mehmood MA, Chang JS, Bai F, Zhao X. Manipulating environmental stresses and stress tolerance of microalgae for enhanced production of lipids and value-added products-a review. Bioresour Technol 2017;244:1198-206. [CrossRef]
56. Jaiswal KK, Banerjee I, Singh D, Sajwan P, Chhetri V. Ecological stress stimulus to improve microalgae biofuel generation:A review. Octa J Biosci 2020;8:48-54.
57. Lage S, Gojkovic Z, Funk C, Gentili FG. Algal biomass from wastewater and flue gases as a source of bioenergy. Energies 2018;11:664. [CrossRef]
58. Cho K, Heo J, Cho DH, Tran QG, Yun JH, Lee SM, et al. Enhancing algal biomass and lipid production by phycospheric bacterial volatiles and possible growth enhancing factor. Algal Res 2019;37:186-94. [CrossRef]
59. Yu X, Chen L, Zhang W. Chemicals to enhance microalgal growth and accumulation of high-value bioproducts. Front Microbiol 2015;6:56. [CrossRef]
60. Cho DH, Ramanan R, Heo J, Lee J, Kim BH, Oh HM, et al. Enhancing microalgal biomass productivity by engineering a microalgal–bacterial community. Bioresour Technol 2015;175:578-85. [CrossRef]
61. Fuentes JL, Garbayo I, Cuaresma M, Montero Z, González-del-Valle M, Vílchez C. Impact of microalgae-bacteria interactions on the production of algal biomass and associated compounds. Mar Drugs 2016;14:100. [CrossRef]
62. Sharma J, Kumar SS, Bishnoi NR, Pugazhendhi A. Enhancement of lipid production from algal biomass through various growth parameters. J Mol Liq 2018;269:712-20. [CrossRef]
63. Shahid A, Malik S, Zhu H, Xu J, Nawaz MZ, Nawaz S, et al. Cultivating microalgae in wastewater for biomass production, pollutant removal, and atmospheric carbon mitigation;a review. Sci Total Environ 2020;704:135303. [CrossRef]
64. Gorain PC, Bagchi SK, Mallick N. Effects of calcium, magnesium and sodium chloride in enhancing lipid accumulation in two green microalgae. Environ Technol 2013;34:1887-94. [CrossRef]
65. Salama ES, Kim HC, Abou-Shanab RA, Ji MK, Oh YK, Kim SH, et al. Biomass, lipid content, and fatty acid composition of freshwater Chlamydomonas mexicana and Scenedesmus obliquus grown under salt stress. Bioprocess Biosys Eng 2013;36:827-33. [CrossRef]
66. Singh P, Guldhe A, Kumari S, Rawat I, Bux F. Combined metals and EDTA control:An integrated and scalable lipid enhancement strategy to alleviate biomass constraints in microalgae under nitrogen limited conditions. Energy Convers Manag 2016;114:100-9. [CrossRef]
67. Singh P, Guldhe A, Kumari S, Rawat I, Bux F. Investigation of combined effect of nitrogen, phosphorus and iron on lipid productivity of microalgae Ankistrodesmus falcatus KJ671624 using response surface methodology. Biochem Eng J 2015;94:22-9. [CrossRef]
68. Fuhrmann M. Expanding the molecular toolkit for Chlamydomonas reinhardtii-from history to new frontiers. Protist 2002;153:357. [CrossRef]
69. Franklin SE, Mayfield SP. Prospects for molecular farming in the green alga Chlamydomonas reinhardtii. Curr Opin Plant Biol 2004;7:159-65. [CrossRef]
70. Coll JM. Methodologies for transferring DNA into eukaryotic microalgae:A review. Span J Agric Res 2006;4:316-30. [CrossRef]
71. Tam LW, Lefebvre P. Cloning of flagellar genes in Chlamydomonas reinhardtii by DNA insertional mutagenesis. Genetics 1993;135:375-84. [CrossRef]
72. Sugimoto I, Hiramatsu S, Murakami D, Fujie M, Usami S, Yamada T. Algal-lytic activities encoded by Chlorella virus CVK2. Virology 2000;277:119-26. [CrossRef]
73. Jin E, Polle JE, Melis A. Involvement of zeaxanthin and of the Cbr protein in the repair of photosystem II from photoinhibition in the green alga Dunaliella salina. Biochim Biophys Acta 2001;1506:244-59. [CrossRef]
74. Teng C, Qin S, Liu J, Yu D, Liang C, Tseng C. Transient expression of lacZ in bombarded unicellular green alga Haematococcus pluvialis. J Appl Phycol 2002;14:497-500. [CrossRef]
75. Doetsch NA, Favreau MR, Kuscuoglu N, Thompson MD, Hallick RB. Chloroplast transformation in Euglena gracilis:Splicing of a group III twintron transcribed from a transgenic psbK operon. Curr Genet 2001;39:49-60. [CrossRef]
76. Lapidot M, Raveh D, Sivan A, Arad SM, Shapira M. Stable chloroplast transformation of the unicellular red alga Porphyridium species. Plant Physiol 2002;129:7-12. [CrossRef]
77. Cerutti H, Johnson AM, Gillham NW, Boynton JE. A eubacterial gene conferring spectinomycin resistance on Chlamydomonas reinhardtii:Integration into the nuclear genome and gene expression. Genetics 1997;145:97-110. [CrossRef]
78. Stevens DR, Purton S, Rochaix JD. The bacterial phleomycin resistance geneble as a dominant selectable marker in Chlamydomonas. Mol Gen Genet 1996;251:23-30. [CrossRef]
79. Randolph-Anderson BL, Sato R, Johnson AM, Harris EH, Hauser CR, Oeda K, et al. Isolation and characterization of a mutant protoporphyrinogen oxidase gene from Chlamydomonas reinhardtii conferring resistance to porphyric herbicides. Plant Mol Biol 1998;38:839-59. [CrossRef]
80. Pinnola A, Cazzaniga S, Alboresi A, Nevo R, Levin-Zaidman S, Reich Z, et al. Light-harvesting complex stress-related proteins catalyze excess energy dissipation in both photosystems of Physcomitrella patens. Plant Cell 2015;27:3213-27. [CrossRef]
81. Gomaa MA, Al-Haj L, Abed RM. Metabolic engineering of cyanobacteria and microalgae for enhanced production of biofuels and high-value products. J Appl Microbiol 2016;121:919-31. [CrossRef]
82. Pathak J, Ahmed H, Singh PR, Singh SP, Häder DP, Sinha RP. Mechanisms of photoprotection in cyanobacteria. In:Cyanobacteria:From Basic Science to Applications. New York:Elsevier;2019. 145-71. [CrossRef]
83. Seo S, Jeon H, Hwang S, Jin E, Chang KS. Development of a new constitutive expression system for the transformation of the diatom Phaeodactylum tricornutum. Algal Res 2015;11:50-4. [CrossRef]
84. Jahn M, Vialas V, Karlsen J, Maddalo G, Edfors F, Forsström B, et al. Growth of cyanobacteria is constrained by the abundance of light and carbon assimilation proteins. Cell Rep 2018;25:478-86. [CrossRef]
85. Kirst H, Formighieri C, Melis A. Maximizing photosynthetic efficiency and culture productivity in cyanobacteria upon minimizing the phycobilisome light-harvesting antenna size. Biochim Biophys Acta 2014;1837:1653-64. [CrossRef]
86. Djediat C, Feilke K, Brochard A, Caramelle L, Tiam SK, Sétif P, et al. Light stress in green and red Planktothrix strains:The orange carotenoid protein and its related photoprotective mechanism. Biochim Biophy Acta 2020;1861:148037. [CrossRef]
87. Pang N, Xie Y, Oung HM, Sonawane BV, Fu X, Kirchhoff H, et al. Regulation and stimulation of photosynthesis of mixotrophically cultured Haematococcus pluvialis by ribose. Algal Res 2019;39:101443. [CrossRef]
88. Vecchi V, Barera S, Bassi R, Dall'Osto L. Potential and challenges of improving photosynthesis in algae. Plants 2020;9:67. [CrossRef]
89. Sharma P, Sharma N. Industrial and biotechnological applications of algae:A review. J Adv Plant Biol 2017;1:1-25. [CrossRef]
90. Atsumi S, Higashide W, Liao JC. Direct photosynthetic recycling of carbon dioxide to isobutyraldehyde. Nat Biotechnol 2009;27:1177-80. [CrossRef]
91. Liang F, Lindblad P. Synechocystis PCC 6803 overexpressing RuBisCO grow faster with increased photosynthesis. Metab Eng Commun 2017;4:29-36. [CrossRef]
92. Wei L, Wang Q, Xin Y, Lu Y, Xu J. Enhancing photosynthetic biomass productivity of industrial oleaginous microalgae by overexpression of RuBisCO activase. Algal Res 2017;27:366-75. [CrossRef]
93. Fang L, Lin HX, Low CS, Wu MH, Chow Y, Lee YK. Expression of the Chlamydomonas reinhardtii Sedoheptulose-1, 7-bisphosphatase in Dunaliella bardawil leads to enhanced photosynthesis and increased glycerol production. Plant Biotechnol J 2012;10:1129-35. [CrossRef]
94. Yang B, Liu J, Ma X, Guo B, Liu B, Wu T, et al. Genetic engineering of the Calvin cycle toward enhanced photosynthetic CO2 fixation in microalgae. Biotechnol Biofuels 2017;10:1-3. [CrossRef]
95. Shin WS, Lee B, Kang NK, Kim YU, Jeong WJ, Kwon JH, et al. Complementation of a mutation in CpSRP43 causing partial truncation of light-harvesting chlorophyll antenna in Chlorella vulgaris. Scie Rep 2017;7:17929. [CrossRef]
96. De Bhowmick G, Koduru L, Sen R. Metabolic pathway engineering towards enhancing microalgal lipid biosynthesis for biofuel application-a review. Renew Sustain Energy Rev 2015;50:1239-53. [CrossRef]
97. Duanmu D, Miller AR, Horken KM, Weeks DP, Spalding MH. Knockdown of limiting-CO2-induced gene HLA3 decreases HCO3-transport and photosynthetic Ci affinity in Chlamydomonas reinhardtii. Proc Nat Acad Sci 2009;106:5990-5. [CrossRef]
98. Mussgnug JH, Thomas-Hall S, Rupprecht J, Foo A, Klassen V, McDowall A, et al. Engineering photosynthetic light capture:Impacts on improved solar energy to biomass conversion. Plant Biotechnol J 2007;5:802-14. [CrossRef]
99. Fatemeh L, Mohsen D. Effects of environmental factors on the growth, optical density and biomass of the green algae Chlorella vulgaris in outdoor conditions. J Appl Sci Environ Manag 2016;20:133-9. [CrossRef]
100. Xia L, Song S, He Q, Yang H, Hu C. Selection of microalgae for biodiesel production in a scalable outdoor photobioreactor in north China. Bioresour Technol 2014;174:274-80. [CrossRef]
101. Praveenkumar R, Kim B, Choi E, Lee K, Park JY, Lee JS, et al. Improved biomass and lipid production in a mixotrophic culture of Chlorella sp. KR-1 with addition of coal-fired flue-gas. Bioresour Technol 2014;171:500-5. [CrossRef]
102. Abdelaziz AE, Ghosh D, Hallenbeck PC. Characterization of growth and lipid production by Chlorella sp. PCH90, a microalga native to Quebec. Bioresour Technol 2014;156:20-8. [CrossRef]
103. Perrine Z, Negi S, Sayre RT. Optimization of photosynthetic light energy utilization by microalgae. Algal Res 2012;1:134-42. [CrossRef]
104. Kirst H, Garcia-Cerdan JG, Zurbriggen A, Ruehle T, Melis A. Truncated photosystem chlorophyll antenna size in the green microalga Chlamydomonas reinhardtii upon deletion of the TLA3-CpSRP43 gene. Plant Physiol 2012;160:2251-60. [CrossRef]
105. Wang Y, Spalding MH. An inorganic carbon transport system responsible for acclimation specific to air levels of CO2 in Chlamydomonas reinhardtii. Proc Natl Acad Sci 2006;103:10110-5. [CrossRef]
106. Zhang S, He Y, Sen B, Chen X, Xie Y, Keasling JD, et al. Alleviation of reactive oxygen species enhances PUFA accumulation in Schizochytrium sp. through regulating genes involved in lipid metabolism. Metab Eng Commun 2018;6:39-48. [CrossRef]
107. Fan J, Ning K, Zeng X, Luo Y, Wang D, Hu J, et al. Genomic foundation of starch-to-lipid switch in oleaginous Chlorella spp. Plant Physiol 2015;169:2444-61. [CrossRef]
108. Hu X, Zhou J, Liu G, Gui B. Selection of microalgae for high CO2 fixation efficiency and lipid accumulation from ten Chlorella strains using municipal wastewater. J Environ Sci 2016;46:83-91. [CrossRef]
109. Jeong J, Baek K, Kirst H, Melis A, Jin E. Loss of CpSRP54 function leads to a truncated light-harvesting antenna size in Chlamydomonas reinhardtii. Biochim Biophys Acta 2017;1858:45-55. [CrossRef]
110. Bhattacharjee ME. Pharmaceutically valuable bioactive compounds of algae. Asian J Pharm Clin Res 2016;9:43-7. [CrossRef]