1. INTRODUCTION
Heavy metal toxicity is the most common problem faced by agricultural soil and plants in the contemporary agricultural world. Perseverance of metal in nature, non-degradability in soil, and toxicity in biological cells and tissues cause metal toxicity. Heavy metals in soil can be absorbed by crops and accumulated in foods, eventually creating a negative collision on living beings. Heavy metals have an adverse impact on plant growth and human health is threatened through food chain [1]. Cadmium (Cd), a naturally occurring non-nutritive trace element in earth’s crust, is one of the major heavy metal contaminants in sewage sludge. Sewage sludge is an insoluble solid residue obtained after sewage treatment. It is a by-product of biological wastewater treatment process, which contains a wide range of non-essential heavy metals including Cd. Huge disposal of waste water and sewage sludge from industries and rainwater runoff enters into the drainage system and pollutes soil, water, and air [2]. The anthropogenic sources of Cd in urban areas of Coimbatore district, India, are likely to be derived from the gold making wastes that were channelized into sewage during disposal. Sewage water and solid waste are disposed for several years in nearby areas and the agricultural soil in such areas had a high cadmium content of 4.5–8.2 mg kg-1 [3]. Hence, this article presents an environmental containment to overcome the abovementioned menace. Washing agents, synthetic surfactants, and biosurfactants are used in separation methods that are utilized for removing heavy metals from the contaminated soil in most of the chemical and physical extractions. The process is expensive and the derivatives become secondary contaminants because of their toxicity and resistance to biodegradation [4]. Phytoremediation is the most cost-effective, environment-friendly, and practical approach in the remediation of heavy metals, which involves the removal, relocation, or reduction of metal contaminants using plants that hyperaccumulate these contaminants [5,6]. In the present study, maize is employed for phytoremediation of heavy metal pollution because of mixed pollutant removal ability of Zea mays L. [7] and high biomass production potential.
Arbuscular mycorrhiza (AM), which has a symbiotic relationship with most vascular plants, plays the most significant role of alleviating the toxic effects of heavy metals. The extended and highly branched mycelial network of AM fungi translocates the heavy metals along the hyphae to the intercellular structure and transfers them to the root at the symbiotic interface. The mechanisms involved are enhanced uptake by fungal transporters in mycorrhizal pathway, phytoextraction by increased biomass of mycorrhizal plants, heavy metal binding to the fungal cell wall, chelation by glomalin, heavy metal compartmentalization in the vacuoles, and activation of antioxidative defenses [8]. The fungal hyphae percolate in the cortical cells of plant roots and form arbuscules, vesicles, and hyphae [9]. The fungal hyphae bind with heavy metals and render them immobile at the cortical region of roots, prevent their translocation towards aerial parts of the plant, and prevent leaf tissues from damage [10]. AM fungi reduce heavy metal stress through heavy metal immobilization in fungal structure, precipitation, chelation in the rhizosphere, sequestration in vacuoles, and activation of antioxidant mechanisms in plants [11].
Therefore, phytoremediation strategies, possible responses of cadmium accumulation in plants, and AM symbiosis are ecofriendly and these approaches can be considered to improve soil health and protect plants against such abiotic stress. The present study was conducted to investigate the potential benefits of monoxenic or pure culture of AM fungi, Rhizophagus irregularis, developed through Agrobacterium rhizogenes transformed root organ culture in maize plant at different levels of artificially spiked Cd in the experimental soil.
2. MATERIALS AND METHODS
The experiments were conducted in the Department of Agricultural Microbiology, Tamil Nadu Agricultural University, Coimbatore (India), in 2019.
2.1. Steps in Establishment of Monoxenic Culture of R. irregularis
The monoxenic culture of arbuscular mycorrhizal fungi (AMF), R. irregularis strain MUCL 41833 obtained from the Department of Agricultural Microbiology, was multiplied for 4 months through root organ culture in MSR medium. When the spore size reached maturity, the spores were harvested using 10 mM sodium citrate (pH 6.0) and by allowing them to pass through 45 micron sieve. The AM spores were mixed in dextrin powder, a water-soluble carrier, under aseptic conditions and formulated. The population was maintained with a minimum of 5000 spores/50 g of inoculum (i.e., 100 spores g-1 inoculum) [12,13].
2.2. Seed coating technique of AM inoculum
Maize seeds (Fodder maize – African tall composite) were obtained from the Department of Forage Crops, TNAU, and were surface sterilized with 0.1% HgCl2 for 2 min, followed by 70% ethyl alcohol for 2 min, with intermittent washings using sterile distilled water. Seeds were coated with AM spores formulated in dextrin at the rate of 50 g Kg-1of seeds. It was air-dried at room temperature (37°C).
2.3. Pot Culture Study
The experimental soil was an alfisol, red sandy loam in texture, neutral in pH (7.2), and free from salinity (0.02 dSm-1). The mixture of soil and sand (2:1) was filled in earthen pots of 5 kg capacity (20 cm diameter and 20 cm height) and used for the experiment. The experimental soil was sterilized and spiked with hemi-pentahydrate (CdCl2.2.5.H2O) (Sigma-Aldrich CAS no. 202908) at different concentrations of 25, 50, and 75 ppm (mg kg-1) and incubated for about 24 h before sowing. The following treatments were imposed and seeds were sown in Cd-spiked soils to test the tolerance of varying levels of Cd in pot culture soils. Two maize plants were grown with and without inoculation of AM fungi (R. irregularis) at varying levels of Cd spiked in soil. The duration of study was 70 days. The treatments were: T1 – Uninoculated control, T2 – Inoculated with R. irregularis (without Cd), T3 – Uninoculated with R. irregularis (25 ppm Cd), T4 – Uninoculated (50 ppm Cd), T5 – Uninoculated (75 ppm Cd), T6 – R. irregularis + 25 ppm Cd, T7 – R. irregularis + 50 ppm Cd, and T8 – R. irregularis+ 75 ppm Cd.
2.4. Root and Shoot Dry Weight
The roots were removed from soil in the pots and the adhering soil was washed and cleaned with water. The roots were shade dried for 15 min to remove water. Plant samples were air-dried at sampling periods 45 and 70 DAS and then kept in an oven at 60–70°C until a constant weight was obtained. The weight of the dried root and shoot samples was recorded and expressed as g plant-1.
2.5. Total Chlorophyll Content
Total chlorophyll was measured by following the procedure of Sadasivam and Manickam [14]. One gram of fresh leaf sample was weighed and cut into fine pieces and the representative sample of 0.5 g was ground to a fine pulp in a chilled mortar with an addition of 20 ml of 80% acetone. The contents were centrifuged at 5000 rpm for 5 min. The supernatant was transferred to a 50 ml conical flask. The residue was ground again with 20 ml of 80% acetone and the supernatant was transferred to the same conical flask. The process was repeated until the residue turned colorless. The volume was made up to 50 ml with 80% acetone. Finally, absorbance of the solution was read at 645 and 663 nm against the solvent (80% acetone) in the spectrophotometer. The chlorophyll content was calculated at 45 and 60 DAS as per the formula given below.
 |
Where, A =Absorbance of specific wavelengths
V = Final volume of chlorophyll extract in 80% acetone
W = Fresh weight of tissue powdered
2.6. Assessment of AM Colonization
Root colonization was estimated during 30 and 45 DAS, as per the method described by Phillips and Hayman [15]. AM fungal spores were estimated from rhizosphere soil of maize (during 70 DAS) using wet sieving and decanting techniques [16].
2.7. Catalase and Peroxidase Activity
Catalase activities in the leaf and root samples were determined by quantitative measurement of interacted H2O2 with the titration method [17]. Five hundred milligrams of plant tissue were macerated with 10 ml of 0.1 M phosphate buffer and centrifuged at 3000 rpm for 10 min to collect the supernatant. Five beakers labeled 0–4 were taken with 5 ml of 1.5% sodium per borate, 1.5 ml of phosphate buffer, and 1 ml of supernatant. After 0th, 1st, 2nd, 3rd, and 4th min, 10 ml of 0.2 N H2SO4 was added and titrated against 0.5 N KMnO4. The reactive oxygen species (ROS) H2O2 was determined at 45 and 70 DAS in shoots and roots as per the formula given below. Lesser H2O2 production implies greater catalase activity and it is expressed as:
 |
Peroxidase is determined through spectrophotometric determination on measuring the rate of increase of absorbance at 430 nm, according to the protocol adopted by Sadasivam and Manickam [14]. Five hundred milligrams of plant tissues were macerated with 10 ml 0.1M phosphate buffer that was centrifuged at 3000 rpm for 10 min to collect the supernatant. A 3 ml pyrogallol was added to 0.1 ml of supernatant and adjusted to zero reading as blank. A 0.5 ml of H2O2 was added to the same solution and read at 430 nm at an interval of 30 s for a period of 3 min. The peroxidase activity was measured at 45 and 70 DAS in shoots and roots, and it was expressed as per the formula given below:
 |
2.8. Acid and Alkaline Phosphatase
The acid phosphatase enzyme activity was estimated as per the method followed by Tabatabai and Bremner [18]. It was expressed in mg of p-nitrophenol released g-1 of soil h-1 on dry weight basis at 35°C at pH 6.5.
2.9. Alkaline Phosphatase
The alkaline phosphatase enzyme activity was estimated as per the method followed by Tabatabai and Bremner [18]. It was expressed in mg of p-nitrophenol released per g of soil per h on dry weight basis at 35°C at pH 11.0
2.10. Soluble Proteins and Total Phenols
Soluble proteins were estimated in maize shoots and roots at 45 and 70 DAS and were expressed as mg protein g-1 plant tissue [19]. Total phenols were estimated by the Folin–Ciocalteu reagent method in maize shoots and roots at 45 and 70 DAS and were expressed as mg of catechol produced g-1 plant material [20].
2.11. Cd uptake in Shoot and Root Samples
Ten milliliters of concentrated nitric acid were added to 500 mg of plant samples and transferred to microwave digester. The temperature was maintained at 100°C for 1–2 h until it turned into a thick slurry form. It was cooled for another 15–30 min after which 2 ml of distilled water was added. The sample was filtered through Whatman No.1 filter paper and the volume was made up to 25 ml. Cd concentrations were determined using atomic absorption spectrophotometer (PerkinElmer) in the same conditions against Cd standards (5–20 ppm) [21].
2.12. Translocation Factor (TF)
TF for heavy metal Cd within a plant was calculated using the following formula (22).
 |
2.13. Tolerance Indices (Ti)
Ti of AM and non-AM plants to heavy metal (Cd) stress in soil was determined using the following formula [22].
 |
2.14. Statistical Analysis
The experimental design was completely a random one with three replications. All results were analyzed with ANOVA. The data were subjected to statistical analysis by variance (P = 0.05) with mean separation, as per the standardized methods detailed by Panse and Sukhatme [23]. The data were critically analyzed using computational tools available in R statistical programming to correlate and interpret various parameters through graphical representations.
3. RESULTS AND DISCUSSION
3.1. Monoxenic culture of R. irregularis in Root Organ Culture (ROC) and its Coating Efficiency
The ROCs grew tremendously with more number of lateral roots along with excessive branching of hyphae and profuse sporulation, which eventually filled the Petri plate in 4 weeks. The dynamic production of new spores after 3–4 months recorded nearly 5000–9000 spores per Petri plate [Plates 1 and 2]. The seed coating polymeric suspension was used to treat the dextrin-based carrier inoculum and it had the best coating efficiency with a maximum of 68% root colonizing potential after 25 days of treatment. Earlier conventional vermiculite-based method was used for obtaining AM inoculum from root segments of plants hosting AM fungi and/or the infected soil containing AM propagules [24]. However, this vermiculite-based AMF inoculum is highly voluminous and contaminated [25]. In the present study, a novel thick spore inoculum, extracted and prepared from in vitro root organ and cultured through a dual culture technique, was used for seed coating in maize. Mycorrhizal spores in the inoculum can be entrapped into cavities created inside the carrier matrix. Spore viability improved with the best coating efficiency of 83.2% [26]. Thus, it can be inferred that the mycorrhizal load in soil can be restored efficiently by applying the seed coating method that colonizes young roots early in a plant’s life, that is, immediately after the seed begins to sprout. Such a seed coating technique infects the emerging root system of inoculated plants.
 | Plate 1: Growth of transformed roots and multiplication of Glomus intraradices spores. (a and b) Growth of transformed roots and (c) multiplication of Gi spores interlinked with extramycelial network. [Click here to view] |
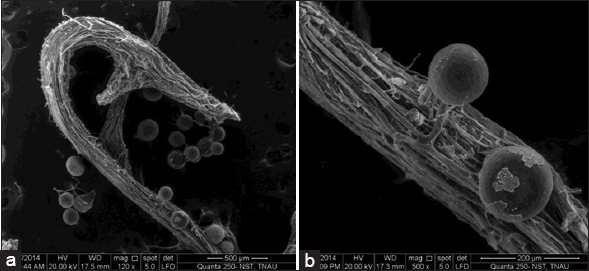 | Plate 2: Scanning electron microscopic view of Gi spores in root organ culture. (a) Bunch of spores in AM colonized root apexes and (b) globose-shaped spores with size ranging from 115 to 145 µm. [Click here to view] |
During 45 DAS, the root dry weight was significantly increased in T2 than in T1 and it was significantly reduced with increasing concentrations of Cd at 45 DAS. During 70 DAS, a decrease was noticed in root dry weight with respect to increasing concentrations in nonmycorrhizal plants, but the reduction was significant only in T5 (18.5%). However, T6 had a higher mean root dry weight (4.2 g-1 plant-1; interquartile range 1.18) than the other two mycorrhizal inoculated treatments (T7 and T8) during 45 DAS [Figure 1a]. Similarly, significant reduction of shoot dry weight was observed in both mycorrhizal and non-mycorrhizal plants at an increasing level of Cd at 45 and 70 DAS. T6 had a maximum mean shoot dry weight at 70 DAS (14 g-1 plant-1; interquartile range 1.65) [Figure 1b].
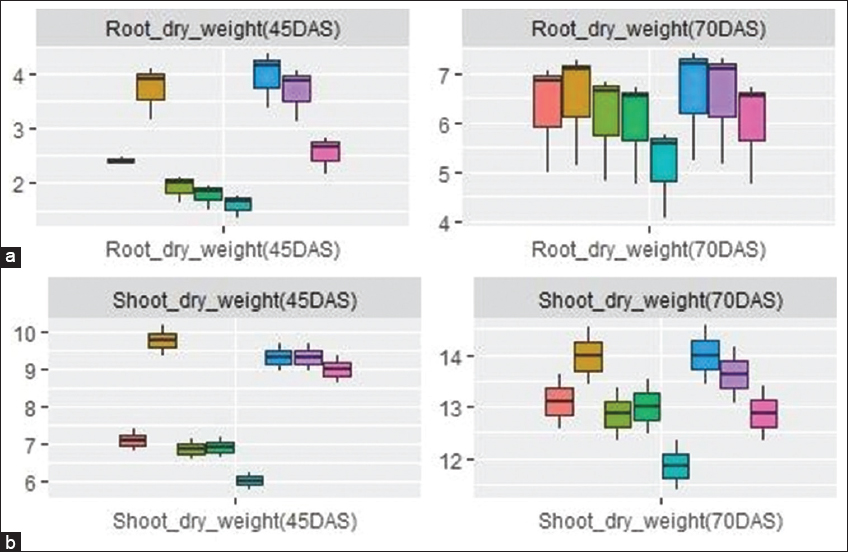 | Figure 1: Box and whisker plot showing root (a) and shoot (b) dry weight in maize in mycorrhiza inoculated and uninoculated treatments at different levels of Cd. Box and whisker plots represent minimum, lower quartile, median, upper quartile, and maximum values. [Click here to view] |
Furthermore, Figure 2 shows a higher mean plant height in mycorrhizal inoculated plants compared to uninoculated ones at both stages. The mean plant height was significantly reduced at 75 ppm Cd in AM uninoculated plants at both stages. Both T7 and T8 had a significant variation over T4 and T5 with the higher values in T7 at 70 DAS (82.2 cm; interquartile value 6.3 cm).
 | Figure 2: Box and whisker plot showing plant height in maize in mycorrhiza inoculated and uninoculated treatments at different levels of Cd. Box and whisker plots represent minimum, lower quartile, median, upper quartile, and maximum values. [Click here to view] |
Similarly, the root length was increased at 6.14–22.20% level over non-mycorrhizal Cd applied plants under mycorrhizal inoculation with different levels of Cd. During 40 and 70 DAS, the mean chlorophyll content was the highest in AM inoculated control, followed by mycorrhizal inoculated treatments T6, T7, and T8, with the possibility of reaching the maximum of the high mean level. All uninoculated treatments (T1, T3, T4, and T5) showed poor chlorophyll content with a possibility of reaching a much lower level than the uninoculated control [Figure 3]. The effect of a good AM infectivity potential on reducing the toxic effect of Cd in polluted soil needs to be essentially studied and the present study has served this purpose. Heavy metal stress affects plant growth, but the inoculation of AM fungi in Cd contaminated soil decreases the reduction of overall plant growth in the present investigation, showing an increase in root and shoot length as well as dry weight at both stages of observation, as presented in Figure 1. The reduction observed at all levels of artificial Cd contamination, irrespective of the comparison between mycorrhizal inoculation and the inoculated control (T2), is possibly due to the reduction in the profuse hyphal elongation and branching, which ultimately causes a decrease in prime nutrient uptake like phosphorus. The principal component analysis (PCA) biplot in Figure 4 depicts the positive Eigen vector values for alkaline and acid phosphatase and other parameters such as plant biomass production, root spread and root volume, root colonization potential, and chlorophyll content. This proves the massive sporulation and the growth of Gi in soil has a great impact on the growth of plant. This is well supported by a study on mycorrhiza-Cd interaction on plant growth, nutrients and Cd accumulation, and AMF root colonization [27]. The researchers posited that mycorrhiza promotes plant growth whereas Cd addition reduces plant biomass production. The PCA biplot [Figure 4] implies positive correlation among the variables, such as total chlorophyll content and root colonization, suggesting minimum variation among them. In addition, the correlogram [Figure 5] shows a highly significant positive correlation (0.97***) among the same variables.
 | Figure 3: Box and whisker plot showing chlorophyll content in maize in mycorrhiza inoculated and uninoculated treatments at different levels of Cd. Box and whisker plots represent minimum, lower quartile, median, upper quartile, and maximum values. [Click here to view] |
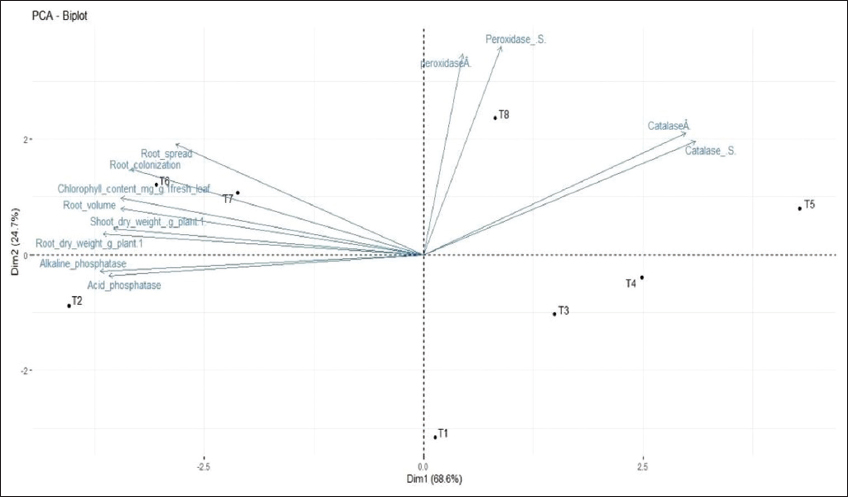 | Figure 4: PCA biplot representing correlation of growth parameters, root colonization, and antioxidative enzymes at different levels of Cd in maize. [Click here to view] |
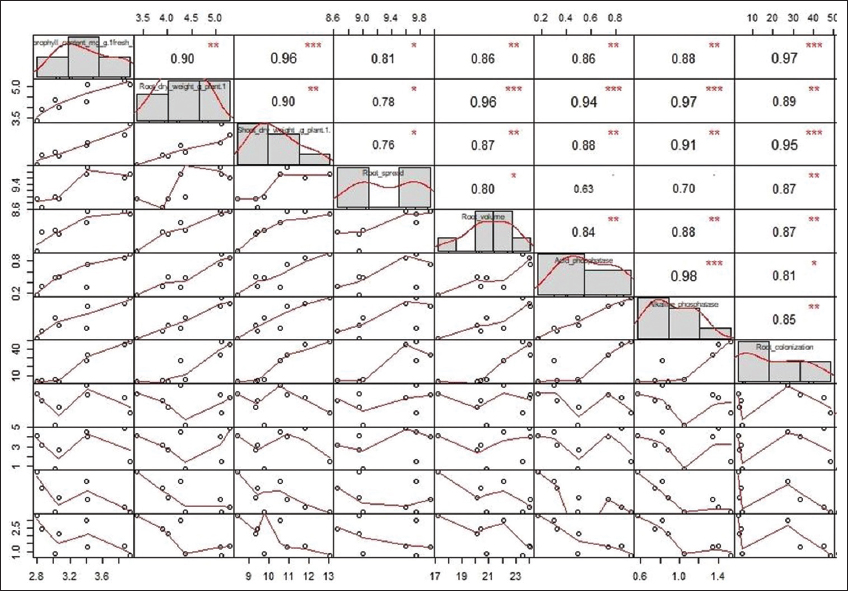 | Figure 5: Correlogram showing the summarized data of AM colonization, growth parameters, and enzyme studies at different levels of Cd in maize. [Click here to view] |
The correlation plot [Figure 5] depicts a significant correlation between the AM root colonization and different growth parameters such as chlorophyll content (0.97***), shoot dry weight (0.95***), root dry weight (0.89**), root spread (0.87**), volume (0.87**), and alkaline phosphatase (0.85**). The root volume was positively correlated with root colonization in PCA biplot [Figure 4] and it was found increased in AM (+) plants than AM (-) plants at all levels of Cd [Figure 6]. The overall mean values depicted that the decrease in the root volume was more in 75 ppm Cd level (T8). No significant changes were observed in root spread at all Cd levels during both stages. The mean percent root colonization in AM (+) and Cd treatments was high in T6 (48.0%; interquartile value 17.3), which had the highest probability to reach its maximum with inoculated control during 30 DAS. Increasing Cd level in T7 and T8 decreased the mean root colonization percent at 45 DAS. However, uninoculated treatments revealed poor infectivity percent at all spiked levels in both stages of observation [Figure 7].
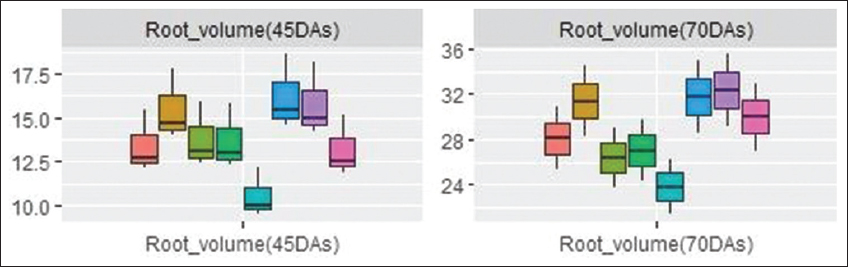 | Figure 6: Box and whisker plot showing maize root volume in mycorrhiza inoculated and uninoculated treatments at different levels of Cd. Box and whisker plots represent minimum, lower quartile, median, upper quartile, and maximum values. [Click here to view] |
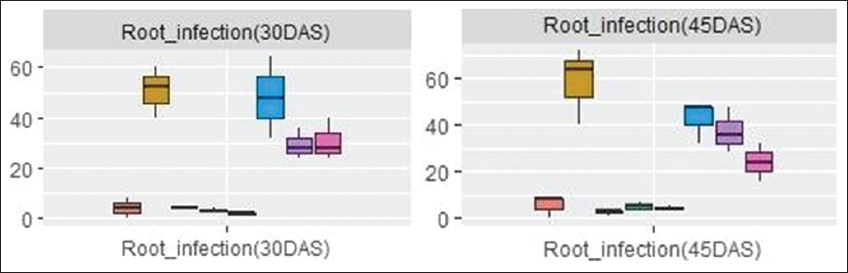 | Figure 7: Box and whisker plot showing AM root colonization percent in maize in mycorrhiza inoculated and uninoculated treatments at different levels of Cd. Box and whisker plots represent minimum, lower quartile, median, upper quartile, and maximum values. [Click here to view] |
Heavy metal contamination in soil is associated with iron deficiency. Low iron content results in chlorosis, since it inhibits both chloroplast development and chlorophyll biosynthesis [28]. Cd has a direct effect on the structure, composition, and functioning of photo system II domains in the thylakoid membrane of plants [29]. The results of the present study indicate an increase of 19.2–25.7% in the chlorophyll content in AM fungi inoculated plants over uninoculated plants at all Cd spiked levels. This finding was supported by Rivera-Becerril et al. [30] who reported an increase in plant photosynthetic activity by AM inoculation in Cd stress conditions. The increase in chlorophyll content is due to great phosphorous availability and other macro- and micronutrients imported to the plant by the extraordinary multiplication of mycorrhizal spores, high-speed elongation, and profuse branching of extracellular hyphal structures, seen in the in vitro pure inoculum of Gi. The box and whisker plot analysis [Figure 7] shows that mean root infection of Gi at 25 ppm Cd was significantly higher (48%) than at 50 and 75 ppm Cd (28%) during 30 DAS. However, the probability of further increase was not observed on continuous exposure of the same Cd level after 45 DAS. The results suggest that root infectivity varies in accordance with the period of exposure and concentrations of Cd were artificially contaminated in the experimental soil, as stated by Liao et al. [31]. Further, beyond a critical concentration, the plant system might be recalcitrant to heavy metal due to the activation of de nova metabolic pathways and triggering of antioxidant defense system.
3.2. Impact on Antioxidative Enzymes
The production of ROS was significantly less in all uninoculated and inoculated controls (T1 and T2) without Cd spiking. However, H2O2 production was high at 75 ppm Cd (T5) level in roots (6.80 µg H2O2 g-1 min-1) and shoots (6.377 µg H2O2 g-1 min-1) during 45 DAS and 70 DAS, respectively. However, treatments inoculated with mycorrhiza showed a relative decrease in ROS, thus confirming the increased catalase activity [Table 1]. A relative increase in the peroxidase activity with increasing Cd levels due to AM inoculation was observed in both roots and shoots with maximum in roots (6.12 change in OD min-1 g-1).
Table 1: Effect of Glomus intraradices inoculation on antioxidative enzymes, total phenols, and soluble proteins of maize grown in Cd-spiked soil.
| Treatments | Catalase (µg H2O2 g-1 min-1) | Peroxidase (change in OD min-1 g-1) | Total phenols (mg of catechol g-1) | Soluble proteins (mg g-1) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Shoot | Root | Shoot | Root | Shoot | Root | Shoot | Root | |||||||||
| 45 d | 70 d | 45 d | 70 d | 45 d | 70 d | 45 d | 70 d | 45 d | 70 d | 45 d | 70 d | 45 d | 70 d | 45 d | 70 d | |
| T1 | 1.27 | 2.12 | 0.65 | 0.85 | 0.09 | 0.31 | 0.61 | 0.81 | 1.38 | 1.74 | 1.40 | 1.80 | 6.81 | 10.51 | 0.56 | 1.17 |
| T2 | 0.43 | 0.70 | 0.87 | 0.72 | 1.25 | 1.44 | 1.89 | 1.10 | 1.59 | 1.80 | 1.69 | 1.83 | 9.53 | 13.68 | 0.66 | 1.54 |
| T3 | 2.12 | 2.97 | 2.55 | 2.13 | 1.73 | 2.15 | 2.19 | 3.12 | 1.79 | 2.02 | 2.25 | 2.67 | 6.62 | 10.34 | 0.45 | 1.16 |
| T4 | 2.97 | 3.40 | 4.25 | 2.45 | 3.05 | 2.21 | 3.45 | 2.89 | 1.81 | 2.14 | 2.70 | 2.80 | 5.60 | 8.01 | 0.36 | 1.08 |
| T5 | 4.25 | 6.37 | 6.80 | 3.31 | 2.88 | 3.63 | 4.87 | 3.34 | 2.40 | 2.94 | 2.95 | 2.97 | 6.01 | 8.27 | 0.28 | 0.83 |
| T6 | 1.70 | 2.12 | 1.27 | 1.34 | 2.79 | 2.97 | 6.12 | 3.67 | 1.90 | 2.40 | 3.10 | 2.80 | 10.51 | 12.39 | 0.64 | 1.88 |
| T7 | 2.40 | 2.55 | 2.97 | 1.27 | 4.19 | 2.11 | 4.15 | 4.02 | 2.45 | 3.10 | 3.46 | 3.28 | 10.82 | 13.00 | 0.56 | 1.54 |
| T8 | 3.53 | 3.82 | 5.10 | 2.97 | 4.29 | 3.88 | 4.68 | 4.31 | 2.34 | 3.00 | 2.71 | 2.92 | 5.79 | 6.81 | 0.50 | 1.71 |
| Mean | 2.37 | 3.00 | 3.05 | 1.88 | 2.53 | 2.33 | 3.50 | 2.91 | 1.96 | 2.39 | 2.53 | 2.63 | 7.71 | 10.38 | 0.50 | 1.36 |
| SEd | 0.22 | 0.28 | 0.30 | 0.17 | 0.42 | 0.37 | 0.64 | 0.51 | 0.19 | 0.23 | 0.09 | 0.26 | 0.51 | 0.87 | 0.04 | 0.12 |
| CD (0.05) | 0.46 | 0.59 | 0.64 | 0.36 | 0.89 | 0.80 | 1.35 | 1.08 | 0.40 | 0.48 | 0.20 | 0.56 | 1.08 | 1.84 | 0.09 | 0.25 |
Values represent mean of three replications, T1 – UIC, T2 – Gi seed coated, T3 – T1+25 ppm Cd, T4 – T1+50 ppm Cd, T5 – T1+75 ppm Cd, T6 – T2+25 ppm Cd, T7 – T2+50 ppm Cd, T8 – T2+75 ppm Cd. DAS – days after sowing, AMF - arbuscular mycorrhizal fungi, ROS – reactive oxygen species
PCA was performed using all attributes among different treatments. Out of the seven PCs, the first three axes with Eigen value of more than 1.0 evidenced that the identified attributes within the axes exhibited a significant influence among the treatments. The PCA biplot [Figure 4] showed T8 and T5 (75 ppm Cd with and without mycorrhizal inoculation) in the positive coordinates for antioxidants in both roots and shoots, whereas other parameters were not influenced significantly. PC1 and PC2 showed variances of 68.6% and 24.7%, respectively. Cd is an important abiotic stress element, which endorses the creation of ROS through the inhibition of electron transport, especially the primary events in Photo system-II [32]. It is a well-known fact that ROS would damage lipids, DNA, RNA, and proteins involved in the constructive metabolism of plant growth. Catalase is an ever-present tetrameric heme-containing enzyme that catalyzes the dismutation of two molecules of H2O2 into H2O and O2, and protects the cell against membrane lipid peroxidation through ROS [33,34].
The present study shows a significant reduction in H2O2 production in roots and shoots when Gi is inoculated at both stages (45 and 75 DAS) with increasing levels of Cd in soil. This steady increase of catalase activity in mycorrhizal roots under Cd stress is due to the decreased oxidative damage to rhizosphere, because AMF plants had consistently better plant biomass production [Figure 1]. A relative increase was observed in the peroxidase activity with increasing Cd levels due to the inoculation of Gi, and inoculum of root organ culture was observed in both roots and shoots. A positive correlation between the two enzymes prevented the negative effects of ROS during increased stress levels. PCA results evidenced the activation of both CAT and peroxidase activity in roots and shoots of AM primed maize seedlings. This study revealed that stress reaction is not perceived in mycorrhizal roots grown in artificial Cd polluted soil. Such less oxidative damage in AM seedlings might be due to the AM associations, conferring better defense mechanism and higher antioxidative defense systems [35]. Sensitivity of plants to heavy metals is determined by an interrelated network of physiological and molecular mechanisms [36] such as uptake and accumulation of metals through binding to extracellular exudates and cell wall, complexation of ions inside the cell by various substances (for example, organic acids, amino acids, ferritins, phytochelatins, and metallothioneins), general biochemical stress defense responses such as induction of antioxidative enzymes, activation, or modification of plant metabolism, and rapid repair of damaged cell structures.
3.3. Impact on Soluble Proteins and Total Phenols
The roots were generally observed with very low soluble proteins than shoots with an overall mean value ranging from 0.50 mg g-1 to 1.36 mg g-1 with the maximum in T6 (25 ppm Cd, AM inoculated), which was found on par with T2, T7, and T8 (AM inoculated). Mycorrhizal Cd applied plants registered higher protein concentration than non-mycorrhizal Cd plants to the tune of 42.2–78.6% on 45 DAS and 42.5–103% on 70 DAS. Obviously, the soluble protein content was decreased more profoundly in mycorrhizal plants at high level Cd. An increase in total phenol content was recorded in all plants exposed to the heavy metal Cd, irrespective of mycorrhizal inoculation. The total phenols were recorded at 45 and 70 DAS in maize shoots and roots and were expressed as mg of catechol produced g-1 plant tissue [Table 1]. The maximum value was recorded in T7 (shoot – 3.10; root – 3.28 mg of catechol g-1 plant tissue) during 70 DAS, where AM was inoculated at 50 ppm Cd level, which was significantly higher than T1 and T2. Furthermore, our study revealed a significant increase in soluble protein content in shoots and roots at both stages of observation when mycorrhiza (Gi) was treated in seeds in T6 and T7. However, when artificial level of contamination was increased at 75 ppm, a steep decrease in soluble protein was noticed in shoots even in mycorrhiza inoculated treatments, possibly due to the toxic negative impact of Cd on the hyphal proliferation and massive sporulation of Gi. In addition, Cd could induce DNA damage, such as single- and double-strand breaks, and modified bases that could lead to a decline in protein synthesis. Numerous studies have shown that genotoxicity of Cd is directly related to its effect on structure and function of DNA [37,38]. The total soluble protein increases as the mycorrhizal infectivity increases [27]. It is obvious to note a significant increase in total phenols at 50 ppm Cd in AM inoculated T7 in comparison to uninoculated T4. The phenolic compounds have the capacity of scavenging free radicals and donating hydrogen atoms. The electrons or chelate metal ions thus contribute to the antioxidant activity [39]. However, the increase in concentration of total phenols in Gi inoculated treatments might be caused by the increase in precursors of these compounds that are optimized through mycorrhizal inoculation [40]. In contrast, the profound decrease in soluble protein at high level of Cd (75 ppm) in mycorrhiza inoculated maize was due to severe reduction in intra/extra radical mycorrhizal colonization.
3.4. Cd Uptake in AM (+) and AM (−) Maize
In the present study, Cd was accumulated to a tune of 22.2–38.3% in mycorrhizal roots that were more than the accumulation in non-mycorrhizal roots [Table 2]. Moreover, Cd addition in mycorrhizal plants did not affect Cd accumulation in shoots, which was decreased significantly to a level of 28.0% in mycorrhizal plants amended with 25 and 50 ppm Cd. Therefore, the uptake in shoot increased even in mycorrhizal plants at higher concentrations of Cd, beyond a certain limit. TF is the ratio of metal concentration in shoot to root, which indicated the internal metal transportation [22]. TF between mycorrhizal and non-mycorrhizal treatments is apparent to show the reduced translocation of Cd in T5, T6, and T7 with the lowest factor (0.14) at 50 ppm Cd and the highest Ti of 100.59 and 98.34 in T6 and T7, respectively. The correlogram [Figure 8] represents the relationship among dry weights, Cd content, and its uptake in maize roots and shoots at different levels of artificial Cd spiking in soil. An increase in shoot or root cadmium content causes a decrease in shoot or root dry weight (negative correlation). Even though the correlation plot shows an overall negative correlation with the total biomass of the plant, tolerance index of Cd was 100.59 in T6. This represented that dry weight of the plant did not reduce due to artificial spiking of 25 ppm Cd when inoculated with AM fungi, R. irregularis, whereas it was positively correlated with Cd uptake in shoot, in spite of showing comparatively less values in T6 over T3 and in T7 over T4, respectively. Table 2 shows that the root dry weight was significantly increased in Gi inoculated Cd levels (T6 and T7) than in uninoculated Cd levels. Our study registered higher Ti in AM inoculated maize plants.
Table 2: Bioaccumulation of cadmium in maize.
| Treatments | Root | Shoot | Translocation factor (b/a) | Tolerance indices (%) | ||||
|---|---|---|---|---|---|---|---|---|
| Cadmium content (µg g-1) | Dry weight (g pot-1) | Cadmium uptake (mg pot-1) a | Cadmium content (µg g-1) | Dry weight (g pot-1) | Cadmium uptake (mg pot-1) b | |||
| T1 | 0 | 12.60 | 0 | 0 | 26.20 | 0 | - | - |
| T2 | 0 | 13.00 | 0 | 0 | 27.96 | 0 | - | - |
| T3 | 14.40 | 12.20 | 0.176 | 2.10 | 25.72 | 0.054 | 0.31 | 97.73 |
| T4 | 24.50 | 12.00 | 0.294 | 3.20 | 26.00 | 0.083 | 0.28 | 97.94 |
| T5 | 29.00 | 10.26 | 0.298 | 3.70 | 23.74 | 0.088 | 0.29 | 87.63 |
| T6 | 17.60 | 13.20 | 0.232 | 1.50 | 28.00 | 0.042 | 0.18 | 100.59 |
| T7 | 33.90 | 13.04 | 0.442 | 2.30 | 27.24 | 0.063 | 0.14 | 98.34 |
| T8 | 39.00 | 12.60 | 0.491 | 3.90 | 25.74 | 0.100 | 0.19 | 93.60 |
| Mean | 28.1 | 12.37 | 0.346 | 2.78 | 26.34 | 0.071 | 0.23 | 95.97 |
| SEd | 4.91 | 0.50 | 0.076 | 0.12 | 1.08 | 0.008 | ||
| CD (0.05) | 10.69 | 1.07 | 0.165 | 0.26 | 2.29 | 0.019 | ||
Values represent mean of three replications,
dry weight of two plants per pot, T1 – UIC, T2 – Gi seed coated, T3 – T1+25 ppm Cd, T4 – T1+50 ppm Cd, T5 – T1+75 ppm Cd,
T6 – T2+25 ppm Cd, T7 – T2+50 ppm Cd, T8 – T2+75 ppm Cd.
 | Figure 8: Correlation plot showing the summarized data of cadmium uptake in shoot and roots. [Click here to view] |
The present study primarily focused on the role of mycorrhiza in overcoming Cd stress in contaminated soil, which is possible only by its symbiotic association with the selected plant species. The bizarre bountiful extracellular hyphal branching of the root organ culture of R. irregularis improved the accumulation of Cd tied in AM structures surrounded by the root system and restricted its movement toward the shoot or economical part of the plant. The results warrant that AM fungi might have some other mechanisms that act as a barrier for the movement of Cd in the plant system through xylem and phloem vessels. AM produces a type of insoluble protein called glycoprotein, which binds heavy metals beyond the plant rhizosphere [41]. Passive adsorption of this protein to the hyphae leads to a binding of up to 0.5 mg Cd g-1 dry biomass [42].
The capacity of plants to tolerate different heavy metals is indicated by the Ti [43] which is important for sinking the toxic stratum of heavy metal from the polluted soil. Our results show that mycorrhiza reduced the transport of Cd from root to shoot and it can reduce metal accumulation in the above ground parts of the plant, thus increasing the tolerance level. Mycorrhizal roots act as a blockade against Cd transport, tumbling its transfer from roots to shoots. Even if the plant tolerance to abiotic heavy metal stress is improved by its mutualistic relationship with AM fungi, the overall performance in heavy polluted soils highly depends on the efficient fungal isolate, nature of plant selected, and the type of heavy metal involved [8]. The distinct strategies involved in molecular mechanisms of plant cells include sequestration of non-essential heavy metals such as cadmium, mercury, and arsenic in root vacuoles. It involves vesicular trafficking in heavy metal detoxification [44].
4. CONCLUSION
The present study on Cd tolerance of R. irregularis MUCL 41833 revealed mycorrhizal plants through seed treatment could be exploited in polluted soils. The seed coating technique is focused to evaluate the in vitro inoculum of R. irregularis developed through root organ culture. It plays a significant ecological role in the phytostabilization of Cd polluted soils through metal sequestration and translocation. Mycorrhizal plants can adapt well to Cd stress conditions at 25 ppm and tolerate up to 50 ppm reducing translocation of heavy metal from roots to shoots. This novel technique can be exploited for alleviation of other heavy metals using other hyperaccumulators.
5. ACKNOWLEDGMENT
The authors are thankful to the Department of Environmental Sciences, Tamil Nadu Agricultural University, Coimbatore, India, for the atomic absorption spectrophotometric analysis of cadmium.
6. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
7. FUNDING
There is no financial assistance for executing this research.
8. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
9 ETHICAL APPROVALS
This study did not involve any animal studies.
10. DATA AVAILABILITY
All data are available in manuscript.
11. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Liu G, Yu Z, Liu X, Xue W, Dong L, Liu Y. Aging process of cadmium, copper and lead under different temperatures and water contents in two typical soils of China. J Chem Volume 2020, Article ID 2583819:1-10. [doi:10.1155/2020/2583819]. [CrossRef]
2. Zhang X, Wang X, Wong D. Immobilization of heavy metals in sewage sludge during land application process in China:A review. Sustainability 2017;9:1-19. [CrossRef]
3. VinuRadha R, Marimuthu P, Kumutha K. Assessment of Cadmium contamination of soils of sewage disposal areas of Coimbatore District, Tamil Nadu, India. Current World Environ 2014;9:379-86. [CrossRef]
4. Qi X, Xu X, Zhong C, Jiang T, Wei W, Song X. Removal of cadmium and lead from contaminated soils using sophorolipids from fermentation culture of Starmerella bombicola CGMCC 1576 fermentation. Int J Environ Res Public Health 2018;15:E2334. [CrossRef]
5. Parmar S, Singh V. Phytoremediation approaches for heavy metal pollution:A review. J Plant Sci Res 2015;2:1-8.
6. Wuana RA, OkieimenFE. Heavy metals in contaminated soils:A review of sources, chemistry, risks and best available strategies for remediation - A review article. ISRN Ecol Volume 2011, Article ID 402647:1-20. [CrossRef]
7. Hechmi N, Ben Aissa N, Abdennaceur H, Jedidi N. Phytoremediation potential of maize (Zea mays L.) in co-contaminated soils with pentachlorophenol and cadmium. Int J Phytoremediation 2013;15:703-13. [CrossRef]
8. Ferrol N, Tamayo E, Vargas P. The heavy metal paradox in arbuscular mycorrhizas:From mechanisms to biotechnological applications. J Exp Bot 2016;67:6253-65. [CrossRef]
9. Verma R, Tapwal A, Kumar D, Parkash V, Puri S. Vesicular arbuscular mycorrhizal diversity in some important ethno medicinal plants of Western Himalaya. Med Plants 2019;11:279-85. [CrossRef]
10. Dhalaria R, Kumar D, Kumar H, Nepovimora E, Kuca K, Islam MT, et al. Arbuscular mycorrhizal fungi as potential agents in ameliorating heavy metal stress in plants. Agronomy 2020;10:815. [CrossRef]
11. Mishra A, Bhattacharya A, Mishra N. Mycorrhizal symbiosis:An effective tool for metal bioremediation. In:Singh JS, editor. New and Future Developments in Microbial Biotechnology and Bioengineering. Amsterdam, The Netherlands:Elsevier;2019. 113-28. [CrossRef]
12. Declerck S, Strullu DG, Plenchette C. Monoxenic culture of the intraradical forms of Glomus sp. isolated from a tropical ecosystem:A proposed methodology for germplasm collection. Mycologia 1998;90:579. [CrossRef]
13. Cranenbrouck S, Voets L, Bivort C, Renard L, Strullu DG, Declerck S. Methodologies for in vitro cultivation of Arbuscular Mycorrhizal fungi with root organs. In:Declerck S, Strullu DG, Fortin JA, editors. In Vitro Culture of Mycorrhizas. Heidelberg:Springer Publications;2005. 341. [CrossRef]
14. Sadasivam S, Manickam A. Biochemical Methods. New Delhi:New Age International (P) Limited Publishers;1992. 256.
15. Phillips JM, Hayman DS. Improved procedures for clearing and staining parasitic and vesicular arbuscular mycorrhizal fungi for rapid assessment of infection. Trans Br Mycol Soc 1970;13:31-2.
16. Gerdemann JW, Nicolson TH. Spores of Mycorrhizal Endogonespecies extracted from soil by wet sieving and decanting. Trans Br Mycol Soc 1963;46:235-44. [CrossRef]
17. Euler HV, Josephson K. Uber katalase. Eur J Org Chem 1927;452:158-81. [CrossRef]
18. Tabatabai MA, Bremner JM. Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol Biochem 1969;1:301-7. [CrossRef]
19. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem 1951;193:265-75. [CrossRef]
20. Mallick CP, Singh MB. Plant Enzymology and Histo Enzymology. New Delhi:Kalyani Publishers;1980. 286.
21. Micheal JS. Examination of water for pollution control. A reference handbook:Physical, chemical and radiological examination. Publisher Oxford;New York:Pergamon, 1982;2:202-10.
22. Stoltz E, Greger M. Accumulation properties of As, Cd, Cu, Pb and Zn by four wetland plant species growing on submerged mine tailings. Environ Exp Bot 2002;47:271-80. [CrossRef]
23. Panse VG, Shukatme PV. Statistical methods for Agricultural Workers. New Delhi:Indian Council of Agricultural Research;1978. 327.
24. Douds DD Jr., Nagahashi G, Hepperly PR. On-farm production of inoculum of indigenous arbuscular mycorrhizal fungi and assessment of diluents of compost for inoculum production. Bioresour Technol 2010;101:2326-30. [CrossRef]
25. Gaur A, Adholeya A. Response of threevegetablecrops to VAM fungal inoculation in nutrientdeficientsoilsamendedwithorganicmatter. Symbiosis 2000;29:19-31.
26. VinuRadha R, Marimuthu P, Kumutha K. Improved performance of seed coating of AM spore inoculum developed through root organ culture. Asian J Microbiol Biotechnol Environ Sci 2019;21:183-91.
27. Andrade SA, Silviera AP. Mycorrhiza influence on maizedevelopmentunder Cd stress and P supply. Braz J Plant Physiol 2008;20:39-50. [CrossRef]
28. Imsande J. Iron, sulfur and chlorophyll deficiencies:A need for an integrative approach in plant physiology. Plant Physiol 1998;103:139-44. [CrossRef]
29. Becerril JM, Munoz-Rueda A, Aparico-Tejo P, Gonzalez-Murua C. The effects of cadmium and lead on photosynthetic electron transport in clover and Lucerne. Plant Physiol Biochem 1998;26:357-63.
30. Rivera-Becerril F, Calantzis C, Turnau K, Caussane JP, Belimov A, Gianianazzi S, et al. Cadmium accumulation and buffering of cadmium induced stress by arbuscular mycorrhizal in three Pisumsativum. L. genotypes. J Exp Bot 2002;53:1177-85. [CrossRef]
31. Liao JP, Lin XG, Cao ZH, Shi YQ, Wong MH. Interactions between arbuscular mycorrhizae and heavy metals under sand culture experiment. Chemosphere 2003;50:847-53. [CrossRef]
32. Sandalio LM, Dalurzo HC, Gómez M, Romero-Puertas MC, Río LA. Cadmium-induces changes in the growth and oxidative metabolism of pea plants. J Exp Bot 2001;52:2115-26. [CrossRef]
33. Sharma P, Jha AB, Dubey RS, Pessarakli M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stress conditions. J Bot 2012;2:1-26. [CrossRef]
34. Kurutas EB. The importance of antioxidants which play the role in cellular response against oxidative/nitrosative stress:Current state. Nutr J 2016;15:71. [CrossRef]
35. Wu QS, Srivatsava AK, Zou YN. AMF induced tolerance to drought stress in citrus. A review. Sci Hortic 2013;164:77-87. [CrossRef]
36. Upadhyaya H, Panda SK, Bhattacharjee MK, Dutta S. Role of arbuscular mycorrhiza in heavy metal tolerance in plants:Prospects for phytoremediation. J Phytol 2010;2:16-27.
37. Liu W, Sun L, Zhong M, Zhou Q, Gong Z, Li P, et al. Cadmium-induced DNA damage and mutations in Arabidopsis plantlet shoots identified by DNA fingerprinting. Chemosphere 2012;89:1048-55. [CrossRef]
38. Cambier S, Gonzalez P, Durrieu G, Bourdineaud JP. Cadmium-induced genotoxicity in zebrafish at environmentally relevant doses. Ecotoxicol Environ Saf 2010;73:312-9. [CrossRef]
39. Santos-Sanchez NF, Coronado RS, Canongo CV, Carlos BH. Antioxidant compounds and their antioxidant mechanism. In:Shalaby E, editor. Antioxidants. UK:IntechOpen;2019. [doi:10.5772/intechopen.85270]. [CrossRef]
40. Silva FA, Ferreira MR, Soares LA, Sampaio EV, Silva FS, Maia LC. Arbuscular mycorrhizal fungi increase gallic acid production in leaves of field grown Libidibia ferrea (Mart. ex Tul.) L. P. Queiroz J Med Plants Res 2014;8:1110-5. [CrossRef]
41. Kazemalilou S, Delangiz N, Lajayer BA, Ghorbanpour M. Insight into plant bacteria fungi interactions to improve plant performance via remediation of heavy metals:An overview. In:Molecular Aspects of Plant Beneficial Microbes in Agriculture. 1st ed., Vol. 9. Netherlands:Elsevier Inc.;2020. [CrossRef]
42. Gonzalez Chavez MC, Carrillo R, Wright SF, Nichols KA. The role of glomalin, a protein produced by arbuscular mycorrhizal fungi, in sequestering potential toxic element. Environ Pollut 2004;130:317-23. [CrossRef]
43. Chen L, Long XH, Zhang ZH, Zheng XT, Rengel Z, Liu ZP. Cadmium accumulation and translocation in two Jerusalem artichoke (Helianthus tuberosus L.) cultivars. Pedosphere 2011;21:573-80. [CrossRef]
44. Sharma SS, Dietz KJ, Mimura T. Vacuolar compartmentalization as indispensable component of heavy metal detoxification in plants. Plant Cell Environ 2016;39:1112-26. [CrossRef]