1. INTRODUCTION
Mucuna pruriens (L.) DC., commonly known as Velvet bean, belongs to the family Fabaceae. This leguminous plant which grows in tropical and subtropical regions of Africa, America, Asia, and the Pacific Islands, is endemic to India. The pods of this plant are used as a vegetable, and the leaves are used as animal feed [1]. This underutilized legume has anti-inflammatory and antioxidant properties. Besides this, the plant is well known for its neuroprotective effect and is used to treat Parkinson’s disease [2]. The plant is also used to treat infertility in men. Levodopa (L-DOPA), a precursor of alkaloids, plays a key role in increasing the dopamine content abundantly found in M. pruriens. Hence, it can treat Parkinson’s disease [3].
During its growth in its natural habitat, the plant is exposed to a wide variety of biotic and abiotic stress. Exposure to heavy metals such as lead (Pb), cadmium (Cd), and mercury (Hg) in the soil is one such abiotic stress that impedes plant growth [4]. The soil gets accumulated with these heavy metals due to its contamination with industrial effluents, pesticides, and fertilizers [4,5]. While heavy metal Pb is toxic at higher concentrations, other heavy metals such as Hg and Cd are toxic even at lower concentrations. The Cd metal is known to be taken up more readily by the plant’s roots and retards the growth of shoots and roots in plants. It is also known to interfere with plants’ Ca, Mg, P, and K and water uptake [6]. The silver-white metal Hg enters the plants through the roots and alleviates oxidative stress [7]. Pb, at higher concentrations, induces detrimental effects in plants by affecting photosynthesis and transpiration, in turn, affecting the overall plant growth [8].
The stress induced by heavy metals may affect the efficacy and quality of the biosynthesis of secondary metabolites in medicinal plants. The mobile heavy metals are known to suppress the secondary metabolites production in some species by inactivating genes of enzymes involved in the biosynthetic pathway or enhancing the secondary metabolite production in some other species by activating the gene expression involved in the biosynthesis [9,10]. The present study investigates the effects of Cd, Hg, and Pb on the germination, growth, and biochemical characteristics of M. pruriens. Furthermore, the present work aspires to study the impact of the selected heavy metals on the L-DOPA content.
2. MATERIALS AND METHODS
2.1. Evaluation of lethal dose (LD50) for Heavy Metals on Seed Germination and its Growth
The authenticated seeds of Mucuna pruriens (L.) DC. (Arka Shubra) were collected from ICAR-Indian Institute of Horticultural Research, Hessaraghatta, Bengaluru, India. The seeds were surface sterilized with a soap solution (1–2 min) and sodium hypochlorite (4–5 min). The seeds were transferred to Petri dishes with filter paper moistened with 10 different concentrations of selected heavy metals ranging from 25–250 ppm of Cd, 25–250 ppm of Hg, and 200–2000 ppm of Pb and incubated at 25°C in a dark chamber for germination.
2.2. Measurement of Vegetative Growth Characteristics
The root and shoot length, fresh weight (FW), and dry weight (DW) of 21-day-old seedlings were measured.
2.3. Biochemical Characterization of the Seedlings
The proline content, total chlorophyll, protein, and carbohydrate content of the 21-day-old treated seedlings were estimated.
2.3.1. Estimation of proline
The proline content was estimated by following the method of Troll and Lindsley (1955) with some modifications [11]. 0.1 g of sample was homogenized with 5 mL of 3% sulfosalicylic acid, the sample was filtered, and 2 mL of this filtrate was mixed with 2 ml each of Ninhydrin reagent and glacial acetic acid. This mixture was then heated in a boiling water bath for an hour and cooled. A chromophore was formed by adding 4 mL toluene to this cooled solution. The absorbance was measured at 520 nm using the UV–VIS spectrophotometer (Shimadzu, UV-1900, Kyoto, Japan). Proline ranging from 0 to 10 µg mL−1 was used as standard, and a graph was plotted from which the proline content of the samples was estimated.
2.3.2. Estimation of chlorophyll
The total chlorophyll content was measured by following the modified method of Arnon (1949) [12]. 0.1 g of leaf sample was homogenized with 5 mL of 80% acetone and incubated overnight at room temperature. It was then centrifuged at 5000 rpm for 5 min. The supernatant was made up to 5 mL with 80% acetone and the optical densities were measured at 645 and 663 nm wavelengths using the UV–VIS spectrophotometer (Shimadzu, UV-1900, Kyoto, Japan).

Where, A is the absorbance at specific wavelength; V = final volume of chlorophyll extract (mL); and W = fresh weight(g).
2.3.3. Estimation of protein and carbohydrate
The protein content was estimated by following the method of Lowry (1951) [13]. The carbohydrate was estimated by phenol-sulfuric acid method as followed by Dubois et al., (1956) [14]. The absorbance of control and the treated samples was measured at 660 nm and 490 nm, respectively, using the microplate reader (BIO-RAD, iMARK™, Japan).
2.4. Preparation of Plant Methanolic Extract
The plant methanolic extract was prepared by soaking the dried plant samples (control and treated, 0.2 g each) in 5 mL methanol, covered with aluminum foil, and was incubated for 24 h with constant stirring. It was then filtered, and the filtrate was evaporated until the residue was obtained. The residue was redissolved in methanol to obtain a stock concentration of 10 mg mL−1. These methanolic extracts of control and treated plants were further used for the estimation of total phenol and flavonoid content and for antioxidant assays.
2.5. Estimation of Total Phenol Content (TPC)
The TPC was measured using Folin–Ciocalteu assay followed by Sembiring et al. (2018) [15]. 25 μL of the extract (500 μg mL−1) was used to which 25 μL of (1:1) Folin–Ciocalteu reagent and 100 μL of 7.5% sodium bicarbonate solution were added and incubated at room temperature for 2 h in dark condition. The absorbance was recorded at 765 nm using the microplate reader (BIO-RAD, iMARK™, Japan). Gallic acid ranging from 0 to 100 μg mL−1 was used as a standard to calculate the phenol content of the samples.
2.6. Estimation of Total Flavonoid Content (TFC)
The TFC was evaluated using a modified aluminum chloride method by Sembiring et al. (2018) [15]. 50 μL extract (5 mg mL−1) was taken, to which 150 μL of 80% methanol, 10 μL aluminum chloride (10% [w/v]), and 10 μL 1M sodium acetate were added and incubated at room temperature for 45 min. The absorbance was recorded at 415 nm using the microplate reader (BIO-RAD, iMARK™, Japan). Quercetin ranging from 0 to 100 μg mL−1 was used as a standard to calculate the flavonoid content of the samples.
2.7. Antioxidant Activity
2.7.1. Radical scavenging activity using 2,2-diphenyl-1-picryl-hydrazyl (DPPH)
The antioxidant activity was assessed using a modified DPPH scavenging assay as described by Blois (1958) [16]. 30 μL (10 mg mL−1) of the plant extract was taken and made up to 3 mL with methanol. 1 mL of DPPH (0.004% [w/v]) was added and incubated for 30 min in the dark. The absorbance was recorded at 517 nm using the UV–VIS spectrophotometer (Shimadzu, UV-1900, Kyoto, Japan). Methanol (3 mL) served as the blank.
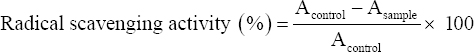
Where, Acontrol and Asample are the absorbances of control without plant extract and sample with plant extract, respectively.
2.7.2. Metal chelating activity
The method described by Chew et al. (2009) [17] was followed for analysis of metal chelating activity. 1 mL of plant extract (400 μg mL−1) was used to which 1 mL of 0.1 mM ferrous sulfate and 2 mL of 0.25 mM ferrozine were added. After incubating for 10 min at room temperature, the absorbance mixture was recorded at 562 nm using a UV–VIS spectrophotometer (Shimadzu, UV-1900, Kyoto, Japan).

Where, Acontrol and Asample are the absorbances of control without plant extract and sample with plant extract, respectively.
2.7.3. Reducing power assay
The FRAP assay was conducted following the method of Chung et al. (2014) [18]. 1 mL of plant extract (400 μg mL−1) was mixed with 2.5 mL of phosphate buffer (0.2 M) and potassium ferricyanide [K3Fe(CN6)](1%) each. The mixture was incubated at 50°C for 20 min. 2.5 mL of trichloroacetic acid (10%) was added to the mixture and centrifuged at 1000 rpm for 10 min. The upper layer of the solution (2.5 mL) was then mixed with 2.5 mL distilled water and ferric chloride (FeCl3) (0.5 mL, 0.1%). Absorbance was measured at 700 nm using a UV–VIS spectrophotometer (Shimadzu, UV-1900, Kyoto, Japan).
2.8. Analysis of Heavy Metal Accumulation in Seedlings of M. pruriens
The heavy metals were analyzed by the wet acid digestion method as described by Turek et al., (2019) [19]. The plant sample (control and treated) was oven dried at 60°C ± 2°C. 2 g of 24 h air-dried, powdered samples was digested using a mixture of 12 mL HNO3 and 4 mL HCl and was heated on hot plate maintained at 45°C ± 5°C in fume hood until the volume reduced to 10 mL. It was then made up to 25 mL with deionized water and filtered using Whatman No. 42 filter paper. The heavy metals were analyzed using atomic absorption spectrophotometer (Shimadzu, AA-6880, Japan).
2.9. Quantification of L-DOPA using HPLC
Quantification of L-DOPA from the heavy metal-treated seedlings was carried out following the method of Rakesh et al. (2021) [20]. 0.2 g of dried plant material (control and treated) was dissolved in 5 mL of 0.1M orthophosphoric acid and placed in an orbital shaker at 150 rpm at room temperature for 30 min. The mixture was centrifuged for 10 min at 10,000 rpm, and the supernatant was filtered using 0.45 μM nylon membrane and subjected to HPLC analysis. RP-HPLC system (Shimadzu) equipped with Sharpsil-U C-18, 250 (L) × 4.6 mm. The mobile phase consisted of a mixture of phosphate buffer (pH 2.5) and acetonitrile (80:20, v/v) as mobile phase at a flow rate of 0.5 mL min−1 and column temperature was maintained at 40°C and absorbance was set at 280 nm with isocratic elution and run time of 10 minutes. L-DOPA content ranging from 50 to 1000 μg mL−1 was used as a standard from which the L-DOPA content of the samples was estimated. The HPLC grade L-DOPA (purity – 99.3%) was procured from Natural Remedies Pvt. Ltd., Bengaluru, India.
2.10. Statistical Analysis
The assays were conducted in triplicates. The data obtained were statistically determined and presented as means ± SE. IBM SPSS Statistics software version 22.0 was used to analyze means, standard error, and run one-way ANOVA. The post hoc Duncan’s multiple range test at P ≤ 0.05 was carried out to analyze the significant differences among means of control and metal-treated groups using the same software.
3. RESULTS AND DISCUSSION
3.1. Estimation of LD50 Value on Seed Germination of M. pruriens
The effects of the concentrations of Cd, Hg, and Pb on seed germination of M. pruriens are presented in Figures 1 and 2. The seed germination was significantly reduced with an increase in the concentration of heavy metals. In the present study, the concentration of 150 ppm of Cd, 175 ppm of Hg, and 1200 ppm of Pb inhibited seed germination by 50%, respectively. The plants from the family Fabaceae have been subjected to concentrations ranging from 0 to 2000 ppm of Pb [21], 0 to 400 ppm of Cd [22], and 0 to 300 ppm of Hg [23]. The inhibition of seed germination percentage could be attributed to the heavy metal accumulation, which causes membrane damage, induces mineral leakage, impede food reserve mobilization, inhibit carbohydrate metabolizing enzymes such as amylases and invertase, and cause accumulation of lipid peroxidation products [24].
 | Figure 1: Lethal dose of Cd, Hg, and Pb on seed germination of Mucuna pruriens (L.) DC. Data represent mean values ± SE of three replicates; each experiment was repeated thrice. Means with common letters are not significantly different at P ≤ 0.05 according to Duncan’s multiple range test (DMRT). Cd: Cadmium, Hg: Mercury, Pb: Lead. [Click here to view] |
 | Figure 2: Germination of Mucuna pruriens (L.) DC seeds treated with different concentration of heavy metals (Cd, Hg and Pb). Cd: Cadmium, Hg: Mercury, Pb: Lead. [Click here to view] |
3.2. Effect of Different Concentrations of Cd, Hg, and Pb on Growth Parameters
The effects of the concentrations of Cd, Hg, and Pb on the growth parameters such as root and shoot length, and fresh and dry biomass of M. pruriens are presented in Table 1. The length of the root, shoot, and the fresh and dry mass of M. pruriens significantly reduced with increasing heavy metal concentration compared with untreated seeds (control). The highest root length of 5.9 ± 0.12 cm was observed in 25 ppm Hg and 200 ppm Pb, and the lowest of 2.07 ± 0.12 cm was observed in 250 ppm Hg-treated plants. The highest shoot length of 12.6±0.36 cm and the lowest shoot length of 1.5 ± 0.15 cm were recorded in 200 ppm Pb and plants treated with 250 ppm Cd, respectively. The least fresh and dry biomass of 1.32 ± 0.06 g and 0.34 ± 0 g was observed in plants treated with 250 ppm Hg, respectively; the highest fresh and dry biomass of 3.3 ± 0.02 g and 0.84 ± 0.03 g were achieved in plants treated with 200 ppm Pb. Studies have reported inhibition of early seedling growth in the presence of heavy metals [25,26]. The decrease in the root and shoot length could be due to change in the cell polarity, decreased mitotic activity, and inhibition of cell elongation caused by heavy metal accumulation, resulting in growth inhibition [27]. The decrease in the FW is mainly due to the loss of turgor pressure caused by the loss of water content affecting the cell wall expansion and overall growth [28].
Table 1: Effect of different concentrations of Cd, Hg and Pb on growth parameters of Mucuna pruriens.
| Cd conc. (ppm) | Control | 25 | 50 | 75 | 100 | 125 | 150 | 175 | 200 | 225 | 250 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Fresh biomass (g) | 2.99±0.08a | 2.91±0.03a | 2.56±0.06b | 2.35±0.01c | 2.26±0.06cd | 2.12±0.12de | 2.05±0.04ef | 2±0.09ef | 1.91±0.03f | 1.85±0.03f | 1.47±0.05g |
| Dry biomass (g) | 0.76±0.02a | 0.74±0.03a | 0.65±0.03b | 0.6±0.03bc | 0.58±0.04bcd | 0.54±0.05cde | 0.52±0.03cde | 0.51±0de | 0.49±0.02e | 0.47±0.01e | 0.38±0.01f |
| Shoot length (cm) | 11.7±0.21a | 11.43±0.35a | 10.39±0.28b | 9.73±0.27c | 9.8±0.17bc | 9.4±0.1c | 6.47±0.23d | 6.8±0.15d | 5.07±0.09e | 3.13±0.18f | 1.5±0.15g |
| Root length (cm) | 5.83±0.15a | 5.63±0.15a | 5.4±0.25a | 4.93±0.18b | 4.57±0.09b | 4.57±0.07b | 3.87±0.15c | 3.7±0.1c | 3.57±0.15c | 2.47±0.15d | 2.47±0.15d |
| Hg conc. (ppm) | Control | 25 | 50 | 75 | 100 | 125 | 150 | 175 | 200 | 225 | 250 |
| Fresh biomass (g) | 2.99±0.08bc | 2.78±0.06b | 2.61±0.01c | 2.56±0.03c | 2.26±0.05d | 2.16±0.08d | 1.98±0.05e | 1.92±0.06ef | 1.8±0.01f | 1.56±0.04g | 1.32±0.06h |
| Dry biomass (g) | 0.76±0.02a | 0.71±0.04ab | 0.67±0.03b | 0.65±0.03b | 0.58±0.01c | 0.55±0cd | 0.5±0.02de | 0.49±0.01de | 0.46±0.02ef | 0.4±0.01fg | 0.34±0g |
| Shoot length (cm) | 11.7±0.21a | 11.23±0.18a | 10.37±0.26ab | 9.13±0.12bc | 8.47±0.2bc | 7.63±0.15cd | 6.63±0.18de | 6.07±0.15e | 5.5±0.23f | 5.2±0.2g | 4.07±0.12h |
| Root length in cm | 5.83±0.15a | 5.9±0.12a | 5.57±0.19ab | 5.37±0.12b | 4.93±0.09c | 4.67±0.06c | 3.67±0.12d | 3.5±0.06d | 3.4±0.17d | 2.7±0.06e | 2.07±0.12f |
| Pb conc. (ppm) | Control | 200 | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 |
| Fresh biomass (g) | 2.99±0.08bc | 3.3±0.02a | 3.04±0.06bc | 2.83±0.08cd | 2.76±0.05d | 2.55±0.04e | 2.46±0.05ef | 2.31±0.06fg | 2.3±0.05fg | 2.24±0.04g | 2.04±0.04h |
| Dry biomass (g) | 0.76±0.02a | 0.84±0.03bcd | 0.78±0.04b | 0.72±0.02bc | 0.7±0.02cde | 0.65±0.02cde | 0.63±0.02def | 0.59±0.03ef | 0.59±0.03f | 0.57±0.01f | 0.52±0.03f |
| Shoot length (cm) | 11.7±0.21a | 12.6±0.36a | 11.97±0.29ab | 11.27±0.2abc | 10.17±0.23bcd | 9.33±0.26cde | 8.73±0.12def | 7.9±0.23defg | 6.97±0.18efg | 5.57±0.09g | 5.03±0.12fg |
| Root length (cm) | 5.83±0.15a | 5.9±0.1a | 5.87±0.07a | 5.63±0.07ab | 5.5±0.06ab | 5.23±0.09abc | 5.13±0.09abc | 4.8±0.06bc | 4.33±0.12cd | 3.5±0.06e | 3.07±0.12e |
Data represent mean values±SE of three replicates; each experiment was repeated thrice. Means with common letters are not significantly different at P≤0.05 according to Duncan’s multiple range test (DMRT). Cd: Cadmium, Hg: Mercury, Pb: Lead..
3.3. Effect of Different Concentrations of Cd, Hg, and Pb on Proline and Chlorophyll Content
Proline is a multifunctional amino acid that serves as a non-enzymatic antioxidant when plants are exposed to stress [29]. A wide range of studies report proline accumulation in response to stress [30]. M. pruriens exhibited high proline content on exposure to Cd, Hg, and Pb stress. The highest proline accumulation of 11.55 ± 0.28 μ mol g−1 FW was observed in plants treated with 225 ppm Hg and the least in control showing 6.52 ± 0.05 μ mol g−1 FW [Table 2]. On exposure to heavy metals, proline accumulation could be due to protein hydrolysis, reduced proline dehydrogenase activity, and decreased usage of proline [31].
Table 2: Effect of different heavy metal concentration on biochemical parameters of Mucuna pruriens.
| Cd conc. (ppm) | Control | 25 | 50 | 75 | 100 | 125 | 150 | 175 | 200 | 225 | 250 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Proline (µ mol g-1 FW) | 6.52±0.05de | 6.53±0.1de | 6.35±0.09e | 6.72±0.11de | 7.06±0.06c | 7.81±0.09b | 7.99±0.04b | 8.73±0.04a | 8.87±0.08a | 8.93±0.22a | 8.98±0.11a |
| Chlorophyll (mg g-1 FW) | 12.9±0.09a | 12.82±0.05a | 11.24±0.06b | 10.12±0.05c | 9.89±0.14cd | 9.61±0.05d | 9.59±0.09d | 8.08±0.26e | 7.01±0.05f | 7.1±0.04f | 6.01±0.06g |
| Protein (mg g-1 FW) | 65.63±0.73a | 61.47±0.69b | 63.13±0.93c | 48.57±0.03d | 44.1±0e | 40.77±0.61f | 37.53±0.52g | 38.8±0.36g | 30.63±0.67h | 31.57±0.03h | 25.57±0.38i |
| Carbohydrate (mg g-1 FW) | 85.49±0.42a | 81.32±0.5b | 79.44±0.54b | 76.11±0.42c | 75.42±1.34cd | 73.96±0.48d | 70.76±0.49e | 68.54±0.36f | 65.97±0.7g | 65.69±0.57g | 63.19±0.85h |
| Phenol content (mg g-1 GAE) | 119.16±0.24g | 107.89±0.64i | 111.63±0.42h | 121.09±0.48f | 132.03±0.4d | 169.09±0.25a | 160.49±0.19b | 141.63±0.16c | 126.63±0.49e | 118.03±0.36g | 111.49±0.64h |
| Flavonoid (mg g-1 QE) | 3.84±0.11de | 3.28±0.1e | 3.97±0.11cde | 4.3±0.22cd | 5.81±0.4b | 7.42±0.21a | 5.6±0.4b | 4.72±0.11c | 4.08±0.34cde | 3.3±0.23de | 1.99±0.34f |
| Hg conc. (ppm) | Control | 25 | 50 | 75 | 100 | 125 | 150 | 175 | 200 | 225 | 250 |
| Proline (µ mol g-1 FW) | 6.52±0.05e | 6.53±0.05e | 8.79±0.07d | 8.83±0.16d | 9.58±0.14c | 10.03±0.1c | 10.83±0.21b | 11.04±0.13ab | 11.37±0.51ab | 11.55±0.28a | 11.23±0.07ab |
| Chlorophyll (mg g-1FW) | 12.9±0.09a | 12.99±0.1a | 11.25±0.1b | 11.09±0.08b | 11.07±0.06b | 10.39±0.18c | 9.71±0.04d | 9.41±0.16d | 8.98±0.03e | 8.65±0.06e | 8.06±0.01f |
| Protein (mg g-1 FW) | 65.63±0.73a | 58.57±0.37b | 50.3±0.61c | 45.13±0.93d | 44.6±0.4d | 44.2±0.89d | 42.13±0.86e | 40.77±0.03e | 40.43±0.54e | 36.03±0.43f | 34.3±0.64f |
| Carbohydrate (mg g-1 FW) | 85.49±0.42a | 79.31±0.66b | 80±0.52b | 77.64±0.49c | 77.15±0.49c | 76.32±0.61c | 76.25±0.43c | 74.1±0.3d | 71.39±0.49e | 70.56±0.3e | 63.75±0.55f |
| Phenol content (mg g-1 GAE) | 119.16±0.24h | 117.69±0.23i | 131.96±0.18f | 134.83±0.08e | 139.43±0.87c | 141.49±0.17b | 143.16±0.52a | 138.03±0.49d | 129.49±0.26g | 100.09±0.02j | 100.03±0.12j |
| Flavonoid (mg g-1 QE) | 3.84±0.11d | 3.47±0.34d | 4.36±0.25d | 5.16±0.41d | 5.61±0.ab | 6.06±0.57ab | 6.52±0.15a | 6.64±0.41a | 5.67±0.64ab | 4.26±0.22cd | 4.36±0.06cd |
| Pb conc. (ppm) | Control | 200 | 400 | 600 | 800 | 1000 | 1200 | 1400 | 1600 | 1800 | 2000 |
| Proline (µ mol g-1 FW) | 6.52±0.05ef | 6.48±0.03ef | 6.38±0.03f | 6.53±0.04ef | 6.63±0.06ef | 6.64±0.04ef | 6.6±0.18ef | 7.57±0.11d | 8.56±0.06c | 8.78±0.05b | 10.4±0.05a |
| Chlorophyll (mg g-1 FW) | 12.9±0.09a | 13.43±0.18a | 12.5±0.04ab | 12.14±0.07ab | 11.65±0.04ab | 11.27±0.04abc | 10.55±0.09abc | 9.51±0.02bc | 9.23±0.12bc | 8.02±0.06c | 6.38±0.68c |
| Protein (mg g-1 FW) | 65.63±0.73a | 64.8±0.45a | 61.1±1.54b | 58.43±0.64c | 57.8±0.36c | 53.33±0.78d | 45.07±0.42e | 41.33±0.56f | 42.07±0.7f | 40.03±0.32f | 41.4±0.35f |
| Carbohydrate (mg g-1 FW) | 85.49±0.42a | 82.71±0.55b | 80.28±0.46c | 78.75±0.36d | 68.06±0.54e | 66.74±0.5ef | 65.42±0.84f | 63.75±0.36g | 63.82±0.14g | 63.75±0.55g | 58.68±0.5h |
| Phenol content (mg g-1 GAE) | 119.16±0.24f | 122.36±0.24e | 125.76±0.35d | 136.36±0.85c | 144.89±0.51b | 145.43±0.46b | 150.29±0.43a | 104.23±0.06 | 103.09±0.17g | 98.29±0.18h | 86.63±0.06i |
| Flavonoid (mg g-1 QE) | 3.84±0.11fg | 3.89±0.09g | 4.78±0.21e | 5.33±0.43d | 5.87±0.41d | 7.27±0.14a | 7.66±0.2- | 6.39±0.33b | 5.42±0.25cd | 4.42±0.32ef | 3.36±0.39g |
Data represent mean values±SE of three replicates; each experiment was repeated thrice. Means with common letters are not significantly different at P≤0.05 according to Duncan’s multiple range test (DMRT).
The light-absorbing pigment chlorophyll plays a crucial role in photosynthesis [32]. In the present study, the chlorophyll content decreased significantly with the increasing metal concentration with the highest chlorophyll content of 13.43 ± 0.18 mg g−1 FW in the 200 ppm Pb-treated plants and the least of 6.01 ± 0.06 mg g−1 FW in plants treated with 250 ppm of Cd [Table 2]. The reduction in chlorophyll may be due to the accumulation of metal ions that displaces and impairment of magnesium ions and denatures the chlorophyll [33]. The heavy metals also disturb the chlorophyll biosynthetic pathways by interfering with enzymes involved in the pathway [34].
3.4. Effect of Different Concentrations of Cd, Hg, and Pb on Protein and Carbohydrate Content
Proteins are polymers of amino acids that play various structural, enzymatic, and functional roles such as biosynthesis, photosynthesis, storage, transport, and overall plant growth [35]. M. pruriens subjected to Cd, Hg, and Pb stress showed a sharp decline in the protein content with the highest protein content of 65.63 ± 0.73 mg g−1 FW in control plants and the least protein content of 25.57 ± 0.38 mg g−1 FW in plants treated with 250 ppm Cd [Table 2]. Heavy metal stress studies conducted on germination in Vigna radiata showed a reduction in the protein content on exposure to heavy metals [36]. Heavy metals alter the native conformation of proteins and destruct their biological activity. The metals either suppress or inhibit the de novo synthesis of proteins or degrade into amino acids [37].
Carbohydrates are the first formed organic molecules resulting from photosynthesis that serve as the primary source of energy and take part in defense against plant pathogens and wounds. The carbohydrate content decreased in the metal-treated plants with increasing metal concentrations. The highest carbohydrate content of 85.49 ± 0.42 mg g−1 FW and the least of 58.68 ± 0.5 mg g−1 FW were reported in untreated control plants and plants treated with 2000 ppm Pb, respectively [Table 2]. In response to heavy metal stress, the carbohydrate content has decreased in some plants such as Cajanus cajan [38]; however, it has also been found to increase in some plants such as Glycine max at lower heavy metal concentrations [39]. The loss of carbohydrates in the metal-treated plant can be attributed to the defective photosynthetic machinery caused by metal accumulation. In turn, the suppression in carbohydrate metabolism results in stunted growth [40].
3.5. Effect of Different Concentrations of Cd, Hg, and Pb on Total Phenol and Flavonoid Content
Phenols and flavonoids are non-enzymatic antioxidants that get stimulated in response to heavy metal stress and serve as stress tolerance bioindicators. These secondary metabolites scavenge the molecular species of active oxygen and serve as metal chelators [41]. In the present study, the phenol content increased up to 125 ppm of Cd, 150 ppm of Hg, and 1200 ppm of Pb and further decreased. The highest phenol content of 169.09 ± 0.25 mg g−1 gallic acid equivalent (GAE) and the least of 86.63 ± 0.06 mg g−1 GAE were observed at 125 ppm Cd and 2000 ppm of Pb, respectively [Table 2]. The flavonoid content increased in a dose-dependent manner up to 125 ppm of Cd, 175 ppm of Hg, and 1200 ppm of Pb and further decreased with increasing concentrations. The highest flavonoid content of 7.66 ± 0.21 mg g−1 quercetin equivalent (QE) and lowest of 1.99 ± 0.34 mg g−1 QE were recorded at 1200 ppm Pb and 250 ppm of Cd, respectively [Table 2]. Studies have reported increasing phenol and flavonoid content with increasing metal concentrations [27]. The phenols and flavonoids increased initially but decreased further due to inhibition of peroxide formation by products of secondary oxidation further formed at higher concentrations [42].
3.6. Antioxidant Activity in Response to Heavy Metal Stress
Plants have adopted a defensive antioxidant system to combat heavy metal stress and alleviate cellular damage by scavenging free radicals that would otherwise cause cellular damage. Plants have exhibited antioxidant activity in response to heavy metal stress [43]. DPPH radical scavenging activity, reducing power assay, and metal chelating activity were performed to determine the antioxidant potential of M. pruriens under heavy metal stress. The effects of Cd, Hg, and Pb concentrations on DPPH radical scavenging, metal chelating activity, and reducing potential in M. pruriens are presented in Figure 3 and Table 3, respectively. The DPPH activity and metal chelating activity increased up to 125 ppm Cd, 150 ppm Hg, and 1200 ppm Pb whereas the reducing power increased up to 125 ppm Cd, 125 ppm Hg, and 1000 ppm Pb. The highest DPPH activity of 81.76% and the lowest of 65.57% were achieved at 150 ppm of Hg and 2000 ppm of Pb, respectively. The highest metal chelating activity of 73.23% and the lowest of 58.14% were achieved at 1200 and 2000 ppm of Pb, respectively. The highest reducing power activity of 0.71 OD was observed at 125 ppm Cd and 1000 ppm Pb, and the lowest activity of 0.46 OD was observed at 200 ppm Pb. The initial increase and further decrease in antioxidant activity can be attributed to the increase and further decrease in the phenolic and flavonoid content.
 | Figure 3: DPPH radical scavenging activity and metal chelating activity of heavy metal (Cd, Hg, and Pb) treated seedlings of Mucuna pruriens (L.) DC. Data represent mean values ± SE of three replicates; each experiment was repeated thrice. Means with common letters are not significantly different at P ≤ 0.05 according to Duncan’s multiple range test (DMRT). Cd: Cadmium, Hg: Mercury, Pb: Lead. [Click here to view] |
Table 3: Reducing power of Mucuna pruriens subjected to heavy metal stress.
| Cd conc. (ppm) | Reducing power | Hg conc. (ppm) | Reducing power | Pb conc. (ppm) | Reducing power |
|---|---|---|---|---|---|
| Control | 0.48±0.02d | Control | 0.48±0.01f | Control | 0.48±0.02f |
| 25 | 0.57±0.03bc | 25 | 0.52±0.01de | 200 | 0.46±0.01f |
| 50 | 0.59±0bc | 50 | 0.58±0.01b | 400 | 0.55±0e |
| 75 | 0.65±0ab | 75 | 0.6±0.02b | 600 | 0.59±0d |
| 100 | 0.69±0.01a | 100 | 0.67±0.01a | 800 | 0.69±0.01b |
| 125 | 0.71±0.01a | 125 | 0.69±0.01a | 1000 | 0.71±0.01a |
| 150 | 0.69±0.05a | 150 | 0.59±0.01b | 1200 | 0.7±0a |
| 175 | 0.68±0ab | 175 | 0.58±0b | 1400 | 0.7±0.01a |
| 200 | 0.65±0.05ab | 200 | 0.57±0.01bc | 1600 | 0.67±0.01bc |
| 225 | 0.64±0ab | 225 | 0.54±0.01cd | 1800 | 0.66±0.01c |
| 250 | 0.52±0.05d | 250 | 0.5±0.01ef | 2000 | 0.53±0.01e |
Data represent mean values±SE of three replicates; each experiment was repeated thrice. Means with common letters are not significantly different at P≤0.05 according to Duncan’s multiple range test (DMRT). Cd: Cadmium, Hg: Mercury, Pb: Lead.
3.7. Accumulation of Heavy Metals in Seedlings of M. pruriens
The bioaccumulation analysis of three heavy metals, Cd, Hg, and Pb, in M. pruriens exposed to 10 different concentrations, represents the pattern of metal accumulation. It also helps understand the effect of these accumulated metals on plant growth and its biochemical characteristics. The effects of the concentrations of Cd, Hg, and Pb on metal accumulation in M. pruriens are presented in Figure 4. The highest metal accumulation of 940.32 ± 0.48 mg kg−1 DW of Pb, 143.49 ± 1.12 mg kg−1 DW of Hg, and 7.19 ± 0 mg kg−1 DW of Cd was observed in plants treated with 2000 ppm of Pb, 250 ppm Hg, and 250 ppm Cd, respectively. Studies conducted in medicinal plants grown in heavy metal (Cd, Cr, Hg, and Pb) contaminated soil have reported that the plants have been well adapted to the contaminated soil and are not suitable for their use in herbal formulations [44]. In the present study, the heavy metals Cd, Hg, and Pb accumulated in a dose-dependent manner. This indicates that M. pruriens could uptake, hyperaccumulate, and get well adapted to Cd, Hg, and Pb toxicity.
 | Figure 4: Accumulation of Cd, Hg, and Pb in seedlings of Mucuna pruriens (L.) DC. Data represent mean values ± SE of three replicates; each experiment was repeated thrice. Means with common letters are not significantly different at P ≤ 0.05 according to Duncan’s multiple range test (DMRT). Cd: Cadmium, Hg: Mercury, Pb: Lead. [Click here to view] |
3.8. Effect of Heavy Metals on L-DOPA Content in Seedlings of M. pruriens
The effect of Cd, Hg, and Pb on L-DOPA content in the seedlings of M. pruriens are presented in Figure 5 and Table 4. In the present study, the L-DOPA content increased up to 125 ppm Hg, 150 ppm Cd, and 1000 ppm Pb. It decreased further with increasing metal concentration. The highest L-DOPA accumulation of 105.25 ± 0.09 mg g−1 DW was observed in 1000 ppm Pb, and the least of 19.97 ± 0.11 mg g−1 DW was observed in the control plants. The effect of drought stress on L-DOPA production in Vicia faba L. has been reported by Etemadi et al. (2018) [45]. It has been concluded that the drought stress enhanced the L-DOPA accumulation. The oxidative stress due to heavy metal treatment could trigger the signaling pathway of secondary plant metabolites [9]. The initial increase in L-DOPA with metal concentration was due to the induction of biosynthesis and accumulation of L-DOPA; however, the plants fail to tolerate the stress induced by a further increase in metal concentration, leading to reduction in plant growth and inhibition of biosynthetic activity.
 | Figure 5: HPLC chromatogram for (a) Standard, (b) Control, (c and d) - 25 ppm and 125 ppm Cd treated seedlings of Mucuna pruriens (L.) DC. Cd: Cadmium. [Click here to view] |
Table 4: L-DOPA content in Mucuna pruriens subjected to heavy metal stress.
| Cd conc. (ppm) | L-DOPA (mg g-1) DW | Hg conc. (ppm) | L-DOPA (mg g-1) DW | Pb conc. (ppm) | L-DOPA (mg g-1) DW |
|---|---|---|---|---|---|
| Control | 19.97±0.11d | Control | 19.97±0.11i | Control | 19.97±0.11h |
| 25 | 19.73±0.2d | 25 | 30.83±0.15h | 200 | 31.07±0.07g |
| 50 | 30.73±0.16cd | 50 | 44.1±0.12g | 400 | 47.97±0.07f |
| 75 | 39.44±0.14bcd | 75 | 44.26±0.46g | 600 | 56.52±0.11e |
| 100 | 54.45±0.04abc | 100 | 58.95±0.15d | 800 | 67.77±0.09d |
| 125 | 63.29±0.06ab | 125 | 77.13±0.15a | 1000 | 105.25±0.09a |
| 150 | 68.18±0.07a | 150 | 71.85±0.1b | 1200 | 102.91±0.08ab |
| 175 | 66.84±0.08a | 175 | 68.76±0.11c | 1400 | 99.06±6.86ab |
| 200 | 47.89±0.01abc | 200 | 56.43±0.1e | 1600 | 98.58±1.28ab |
| 225 | 49.36±0.1abc | 225 | 47.37±0.1f | 1800 | 97.74±0.16b |
| 250 | 40.22±0.1bcd | 250 | 68.33±0.14c | 2000 | 84.04±0.21c |
Data represent mean values±SE of three replicates; each experiment was repeated thrice. Means with common letters are not significantly different at P≤0.05 according to Duncan’s multiple range test (DMRT).
4. CONCLUSION
The present research enables an understanding of the effect of Cd, Hg, and Pb on the pattern of metal accumulation, the vegetative growth, and biochemical and physiological responses in M. pruriens. The study provides insight into the metal stress tolerance ability of the plant. Despite the negative impact of the heavy metals on plant growth, it has been observed that L-DOPA content has increased significantly up to specific heavy metal concentrations. This concept can be implemented to promote L-DOPA production of M. pruriens. by growing them in heavy metal-contaminated soil. As a future perspective, studies on molecular and proteomic approaches to elucidate and identify the target genes or proteins triggered by the heavy metals and enhance the secondary metabolite production have to be studied.
5. ACKNOWLEDGMENT
Akshatha Banadka would like to thank Karnataka Science and Technology Promotion Society (KSTePS), Government of Karnataka, for providing the DST scholarship (LIF.10:2020-21). The authors are indebted to Mr. Jyothis Devasia, for the HPLC analysis and to the administration and Dept. of Life Sciences, CHRIST (Deemed to be University), for providing us the infrastructure.
6. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
7. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
8. ETHICAL APPROVALS
This study does not involve experiments on animals and/or humans.
9. DATA AVAILABILITY
All data generated and analyzed are included in the form of tables and figures within this research article.
10. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Lampariello LR, Cortelazzo A, Guerranti R, Sticozzi C, Valacchi G. The magic velvet bean of Mucuna pruriens. JTradit Complement Med 2012;2:331-9. [CrossRef]
2. Rai SN, Birla H, Singh SS, Zahra W, Patil RR, Jadhav JP, et al. Mucuna pruriens protects against MPTP intoxicated neuroinflammation in Parkinson's disease through NF-κB/pAKT signaling pathways. Front Aging Neurosci 2017;9:421. [CrossRef]
3. Katzenschlager R. Mucuna pruriens in Parkinson's disease:A double blind clinical and pharmacological study. J Neurol Neurosurg Psychiatry 2004;75:1672-7. [CrossRef]
4. Kalaivanan D, Ganeshamurthy AN. Mechanisms of heavy metal toxicity in plants. In:Rao N, Shivashankara K, Laxman R, editors. Abiotic Stress Physiology of Horticultural Crops. New Delhi:Springer;2016. [CrossRef]
5. Briffa J, Sinagra E, Blundell R. Heavy metal pollution in the environment and their toxicological effects on humans. Heliyon 2020;6:e04691. [CrossRef]
6. Das K, Dang R, Shivananda TN, Sur P. Interaction between phosphorus and zinc on the Biomass yield and yield attributes of the medicinal plant stevia (Stevia rebaudiana). Sci World J 2005;5:390-5. [CrossRef]
7. Bhattacharya S. Medicinal plants and natural products can play a significant role in mitigation of mercury toxicity. InterdiscipToxicol 2018;11:247-54. [CrossRef]
8. Nas FS, Ali M. The effect of lead on plants in terms of growing and biochemical parameters:A review. MOJ Ecol Environ Sci 2018;3:265-8. [CrossRef]
9. Nasim SA, Dhir B. Heavy metals alter the potency of medicinal plants. Rev Environ Contam Toxicol 2010;203:139-49. [CrossRef]
10. Lajayer BA, Ghorbanpour M, Nikabadi S. Heavy metals in contaminated environment:Destiny of secondary metabolite biosynthesis, oxidative status and phytoextraction in medicinal plants. Ecotoxicol Environ Saf 2017;145:377-90. [CrossRef]
11. Troll W, Lindsley J. A photometric method for the determination of proline. J Biol Chem 1955;215:655-60. [CrossRef]
12. Arnon DI. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 1949;24:1-15. [CrossRef]
13. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J BiolChem 1951;193:265-75. [CrossRef]
14. DuBois M, Gilles KA, Hamilton JK, Rebers PA, Smith F. Colorimetric method for determination of sugars and related substances. Anal Chem 1956;28:350-6. [CrossRef]
15. Sembiring EN, Elya B, Sauriasari R. Phytochemical screening, total flavonoid and total phenolic content and antioxidant activity of different parts of Caesalpinia bonduc (L.) roxb. Pharmacogn J 2017;10:123-7. [CrossRef]
16. Blois MS. Antioxidant determinations by the use of a stable free radical. Nature 1958;181:1199-200. [CrossRef]
17. Chew YL, Goh JK, Lim YY. Assessment of in vitro antioxidant capacity and polyphenolic composition of selected medicinal herbs from Leguminosae family in Peninsular Malaysia. Food Chem 2009;116:13-8. [CrossRef]
18. Chung IM, Ali M, Praveen N, Yu BR, Kim SH, Ahmad A. New polyglucopyranosyl and polyarabinopyranosyl of fatty acid derivatives from the fruits of Lycium chinense and its antioxidant activity. Food Chem 2014;151:435-43. [CrossRef]
19. Turek A, Wieczorek K, Wolf WM. Digestion procedure and determination of heavy metals in sewage sludge--an analytical problem. Sustain Sci Pract Policy 2019;11:1753. [CrossRef]
20. Rakesh B, HimaBindu K, Praveen N. Variations in the L-DOPA content, phytochemical constituents and antioxidant activity of different germlines of Mucuna pruriens (L.) DC. Asian J Chem 2021;33:1881-90. [CrossRef]
21. Amin H, Arain BA, Jahangir TM, Abbasi MS, Amin F. Accumulation and distribution of lead (Pb) in plant tissues of guar (Cyamopsis tetragonoloba L.) and sesame (Sesamum indicum L.):Profitable phytoremediation with biofuel crops. Geol Ecol Landsc 2018;2:51-60. [CrossRef]
22. Swapna B. Impact of cadmium on germination and early seedling growth of Cajanus cajan L. Int J Curr 2016;8:27.
23. Madhan M, Mahesh K, Rao SS. Effect of 24-epibrassinolide on aluminium stress induced inhibition of seed germination and seedling growth of Cajanus cajan (L.) Millsp. Int J Multidiscip Curr Res 2014;2:286-90.
24. Sethy SK, Ghosh S. Effect of heavy metals on germination of seeds. J Nat Sci Biol Med 2013;4:272-5. [CrossRef]
25. Gang A, Vyas A, Vyas H. Toxic effect of heavy metals on germination and seedling growth of wheat. J Environ Res Dev 2013;8:206-13.
26. Acharya S, Sharma DK. Study on the effects of heavy metals on seed germination and plant growth on Jatropha curcas. Int J Agric Sci Res 2014;3:31-4.
27. Chandana C. Heavy metal accumulation and the biochemical responses in selected medicinal plants [Dissertation]. Kerala:Mahatma Gandhi University;2018.
28. Varshney DS. Effect of some Heavy metals on the growth and development of some medicinally important plants [Dissertation]. Uttar Pradesh:Aligarh Muslim University;2014.
29. Hayat S, Hayat Q, Alyemeni MN, Wani AS, Pichtel J, Ahmad A. Role of proline under changing environments:A review. Plant Signal Behav 2012;7:1456-66. [CrossRef]
30. Dey SK, Dey J, Patra S, Pothal D. Changes in the antioxidative enzyme activities and lipid peroxidation in wheat seedlings exposed to cadmium and lead stress. Braz J Plant Physiol 2007;19:53-60. [CrossRef]
31. Lin YC, Kao CH. Proline accumulation induced by excess nickel in detached rice leaves. Biol Plant 2007;51:351-4. [CrossRef]
32. Li Y, He N, Hou J, Xu L, Liu C, Zhang J, et al. Factors influencing leaf chlorophyll content in natural forests at the biome scale. Front Ecol Evol. 2018;6:1-10. [CrossRef]
33. Parekh D, Puranik RM, Srivastava HS. Inhibition of chlorophyll biosynthesis by cadmium in greening maize leaf segments. Biochem Physiol Pflanz 1990;186:239-42. [CrossRef]
34. Pant PP, Tripathi AK, Dwivedi V. Effect of heavy metals on some biochemical parameters of Sal (Shorea robusta) seedling at nursery level, Doon Valley, India. J Agric Sci 2011;2:45-51. [CrossRef]
35. Rasheed F, Markgren J, Hedenqvist M, Johansson E. Modeling to understand plant protein structure-function relationships-implications for seed storage proteins. Molecules 2020;25:873. [CrossRef]
36. Nair SR, Rajani V. Effects of heavy metals on seed germination and protein content of Vigna radiate (L.) Wilczek. Int J Adv Res 2015;3:1306-17.
37. Tripathi AK, Tripathi S. Changes in some physiological and biochemical characters in Albizia lebbek as bio-indicators of heavy metal toxicity. J Environ Biol 1999;20:93-8.
38. Kumar H, Dubey RC, Maheshwari DK. Effect of plant growth promoting rhizobia on seed germination, growth promotion and suppression of Fusarium wilt of fenugreek (Trigonellafoenum graecum L.). Crop Prot 2011;30:1396-403. [CrossRef]
39. Duan Y, Sangani CB, Muddassir M, Soni KV. Copper, chromium and nickel heavy metal effects on total sugar and protein content in Glycine max. Res Sq 2020;1:1-20. [CrossRef]
40. Trivedi LD. A study on interactive effects of heavy metal on growth and biochemical changes in medicinal plants [Dissertation]. Ahmedabad:Gujarat University;2015.
41. Michalak A. Phenolic compounds and their antioxidant activity in plants growing under heavy metal stress. Pol J Environ Stud 2006;15:523-30.
42. Aryal S, Baniya MK, Danekhu K, Kunwar P, Gurung R, Koirala N. Total phenolic content, flavonoid content and antioxidant potential of wild vegetables from Western Nepal. Plants 2019;8:96. [CrossRef]
43. Antony A, Nagella P. Effect of heavy metals on the andrographolide content, phytochemicals and antioxidant activity of Andrographis paniculata. Asian J Chem 2020;32:2748-52. [CrossRef]
44. Abdussalam AK. Physiological and biochemical studies on heavy metal toxicity in selected medicinal plants [Dissertation]. Kerala:University of Calicut;2010. 259.
45. Etemadi F, Hashemi M, Randhir R, ZandVakili O, Ebadi A. Accumulation of L-DOPA in various organs of faba bean and influence of drought, nitrogen stress, and processing methods on L-DOPA yield. Crop J 2018;6:426-34. [CrossRef]