1. INTRODUCTION
Nation’s economic growth largely depends on the rate of agricultural production and is the major source of food for both rural and urban populations [1]. Agrochemicals play a key role in enhancing agricultural production and reducing the effect of pests [2], but rigorous uses of agrochemicals throughout the world are familiar for all of us. There is a list of 30 varieties of fungicides, 108 classes of pesticides, 39 brands of weedicides, six types of rodenticides, and along five kinds of acaricides that have been used by the developing countries [3,4]. Food gets contaminated by pesticides through an adequate channel such as absorption or injection method. Intake pesticide-contaminated food is 5 times more unsafe to the exposure such as water and wind [5]. The World Health Organization argues that the most popular consumed food groups are fruits and vegetables, and around 30% of intake is rest on fruits and vegetables only. As fruits and vegetables are eaten in a row or semi-cooked form, there is a high chance of pesticide residue level compared to the other part of plants that prepare through cereal processing [6]. The application of insecticides and fungicides to protect vegetable crops is inevitable in agriculture. During rabi season, the requirement of crop protective agents is very high as the intensity of pests and diseases is very high on vegetable crops [7].
In okra (Abelmoschus esculentus L), fruit borer (Earias vitella, E. insulana), white fly (Bemisia tabaci), red spider mite (Tetranychus telarius), jassids (Amrasca bigutula), and aphids (Aphis gossypii) incidence are very common. Farmers are applying insecticides in higher doses than permissible limits and non-recommended pesticides for the crop. Farmers are using ethion for the control of fruit borer and sucking complex [8].
Estimation of ethion residues had done using gas chromatography. It had found that the average deposition of ethion at the initial stage on cucumbers was 4.97 and 2.40 mg kg−1 at double and single dosages, respectively. Degradation of ethion for half-life (T½) of ethion on cucumber was 1.8 and 2.1 at double and single doses, respectively. Intake of cucumber is recommended after 7 days [9]. The studies related to persistence of ethion and its residues in cowpea pods at the initial deposit which was 11.23 ppm, and the half-life (T½) value for the decay of ethion on cowpea pods was 2.90 days. The average initial deposit of ethion was 0.254 mg kg−1, which dissipated to 0.013 (94.88%) mg kg−1 on the 5th day [10,11]. Pesticide residues in maize exceeded the acceptable daily intake (ADI), and consumption was hazardous to humans in Ghana [12]. From the residual pesticide content, human health hazards can be estimated for both short-term and long-term exposure. The aim of the present study is an estimation of ethion dissipation in okra and its effect on human health using the hazardous index.
2. MATERIALS AND METHODS
Ethion has been exposed in okra through ingestion mode to observe its indulgence effect on the human being. Simultaneously, it is observed that an average adult of 60 kg can consume okra up to 150 gm in a day [13]. The recommended doses of ethion (468.75 g a.i./ha) were applied to okra (var. Arka Anamika) during the Rabi season of a farmer’s field, which field with alluvial soils (black cotton soils) located at Koonavarm (Godavari delta region) near Rajamahendravaram area. The field was made into many blocks, and ethion was applied in the selected three blocks. Mature vegetable samples were collected from the treated blocks on every alternate day from the initial day of applying the crop protective agent until 8th day, and a total five samples had collected (Day 0 [2 h after application], Day 2, Day 4, Day 6, and Day 8). The mature growth of okra (flower to fruit) is 7 days, and after 7 days, it is not palatable due to its high fiber content. After harvesting, samples were washed thoroughly with deionized water, sliced, oven-dried, grinded, and stored in 4°c cool storages before analyzing ethion content in it.
2.1. Residue Analysis
For detection of the residual level of pesticide, the sample should come through the examination of the maximum residual limit, which depends on the experiments of pesticide residue present on the food products [14]. The maximum amount of pesticide can be referring as maximum residue limit (MRL), and its presence can be notified at the time of its sale. Limits are denoted as mg/kg. In general, the national government confirms the pesticide limits for good agricultural practice followed by standardized international trade [15]. These limits consider as the margin line for health risks and provide the standard for food safety to the customers. ADI, which determines the health safety limits for human health. The formula for calculating human exposure to pesticides is rested on the average food intake of a person on a daily basis average adult weight with pesticide residue data [16].
Grinded fruit samples were analyzed using gas chromatography with mass spectrometer (GC-MS/MS). To estimate the pesticide residue level in okra fruit, determined samples (1 kg okra) were harvested from the identified three blocks after each treatment, along with the control sample. Initially, samples were analyzed using QuEChERS (quick, easy, cheap, effective, rugged, and safe) extraction method in the laboratory [17]. Logarithmic transformation was applied to pesticide residue values to correct skewness [18]. The half-life of the pesticide residue was calculated using the transformed values using the following equation.
The analyzed data (log pesticide concentration versus time elapsed) are then addressed to simple regression analysis, and the regression equation is calculated.
Thus,
log Rt = −kt + log R0
Where,
log Rt = log of pesticide concentration at any time t (in days)
log R0 = log of initial pesticide concentration
k = slope of the linear plot as shown by regression equation which is the rate of the degradation of pesticide with time t
t = time elapsed in days
Half-life is the time required to reduce the pesticide concentration to half of its initial concentration [19].
Thus,
log (1/2R0) = −kt + log R0
Consequently,
Half-life (t 1/2) = log 2/k
After completing the pesticide residue analysis, the data were log-transformed (for exponential relation) and tabulated and statistically analyzed, and presented.
From the pesticide residue data, the human hazard index (HI) was calculated [20]. To bring HI value, we need to estimate estimated daily intake (EDI), and the equation is:
EDI=C × F/D × W
Where,
EDI = Estimated daily intake
C = Mean of pesticide concentration possessed by an individual (mg kg−1)
F = Mean of fruit consumption per person per year (kg)
W = Mean weight of a person (60 kg)
D = Days per year (365 days)
To bring HI value, we need to divide the estimated EDI value to their equivalent known ADI value (mg kg−1 day−1).
HI = EDI/ADI
For consumption standards like MRL of ethion in okra, EU MRL was referred [21].
2.2. Estimation of Average Daily Intake (EADI)
EADI is grounded on the average body weight of an Indian adult. It is evaluated by multiplying the Food and Agriculture Organization per capita food intake rate for a specific kind of vegetable with the mean residual concentration (mg/kg) of individual pesticide residue in the vegetable and dividing the result by standard bodyweight, which is considered as 60 kg [12,22].
Thus
EADI = Cp × FCR/B
where
EADI – estimated average daily intake,
Cp – concentration of the pesticide,
FCR – the food consumption rate, and
BW – the average body weight
2.3. Risk Assessment
To manage and reduce the effect of pesticide residue in the community, regular risk assessment studies are in high demand. These types of studies can be helpful for managers and policymakers to make resolutions based on scientific proof with appropriate management selections [23].
The potential health risk index (HRI) can be obtained for the non-carcinogenic effect on the individual pesticide residue in the vegetables by dividing the EADI to their corresponding ADI values, as revealed in the equation below. If HRI value is more than 1, then it denotes that lifelong intake of vegetables contains a significant level of pesticide residues, which can cause health in the near future [22,24].
The HRI has been estimated by applying the equation below:
HRI = EADI/ADI
3. RESULTS AND DISCUSSION
The data derived from pesticides residue analysis has played a significant role in finding half-life and pre-harvest interval (minimum time in days) for pesticide application to fruit harvestmen, and calculation of ethion has resulted from HRI.
3.1. Ethion Dispersion and Half-life
Pesticide dissipation data are presented in Table 1. Pesticide residue of ethion in okra fruit samples were 8.02, 1.14, 0.14, 0.08, and 0.02 mg/kg from the samples collected on 0 (2 h after application), 2nd, 4th, 6th, and 8th days after application, respectively [Figure 1]. The correlation coefficient of ethion with respect concerning to the duration was significant (0.01 level) and recorded as 0.957, and the correlation of determination (r2) was 0.916. The dissipation percent is 0, 85.74, 98.25, 99.04, and 99.71 on 0th (2 h after application), 2nd, 4th, 6th, and 8th day, respectively, from the time, it had applied. Ethion dissipation percent is presented in Figure 1. From the residual data half-life of ethion in okra, fruit is 0.95 days. A similar trend was observed in okra by [11], wherein ethion was dissipated by 94.88% on the 5th day after application and recorded 1.27 days as the half-life of ethion in okra. From the study, he observed that after the 5th day, residues were below the detectable limit. A similar experimental study on cowpea resulted in 2.9 days half-life of ethion [10]. 2.4 and 4.9 mg/kg ethion residue was recorded in cucumber immediately after application of pesticide and with a half-life of 2.1 and 1.8 days, respectively, for a single and double dose of ethion [9,25] observed flubendiamide residue was dissipated to below detectable limit when tested samples were collected on the 7th day after application on okra.
Table 1: Ethion dissipation in okra fruit in relation to time.
| Ethion | Pesticide residue (mg/kg) | Standard deviation | TMRC (mg/person/day) | MPI (mg/person/day) | ADI (mg/kg bw) | EADI | HI |
|---|---|---|---|---|---|---|---|
| Before Spraying | 0.20 | ±0.02 | 0.0305 | 0.12 | 0.002 | 0.0005 | 0.254 |
| 0 Days after spraying | 8.02 | ±0.26 | 1.2025 | 0.12 | 0.002 | 0.0200 | 10.020 |
| 2 Days after spraying | 1.14 | ±0.11 | 0.1715 | 0.12 | 0.002 | 0.0029 | 1.429 |
| 4 Days after spraying | 0.14 | ±0.01 | 0.0210 | 0.12 | 0.002 | 0.0004 | 0.175 |
| 6 Days after spraying | 0.08 | ±0.01 | 0.0115 | 0.12 | 0.002 | 0.0002 | 0.096 |
| 8 Days after spraying | 0.02 | ±0.01 | 0.0035 | 0.12 | 0.002 | 0.0001 | 0.029 |
FCR: Food consumption rate, TMRC: Theoretical maximum residue concentration in mg/person/day = Residue × Average daily consumption (150 g), MPI: Maximum permissible intake in mg/person/day = ADI×Average body weight (60 kg), ADI: Acceptable Daily Intake mg/kg bw by PPDB, EADI: Estimated average daily intake = cp×FCR/bw, HI = EADI/ ADI
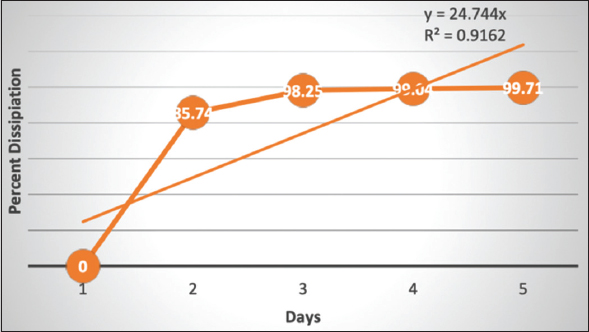 | Figure 1: Ethion dissipation percentage in relation to time. [Click here to view] |
3.2. Pre-harvested Interval
The ethion residue data [Tables 1 and 2] have found that harvesting of okra fruit on or before the second day from the day, it has applied, is not suggested as the theoretical maximum residue concentration of ethion residue is more than the maximum permissible intake (MPI) as per the guidelines of pesticide property database. MPI of treated okra was 0.12 mg/person/day, which is more than the ADI of ethion residue consumed by an adult of 60 kg. Consumption of 150 g an average daily intake of okra (other vegetables) is recommended by a nutritional [26].
Table 2: Ethion residue standards for consumption.
| Ethion | mg/kg bw | MPI (mg/person/day) |
|---|---|---|
| Acceptable daily intake of Ethion | 0.002 | 0.12 |
PPDB: Pesticide property database, MPI: Maximum permissible intake
3.3. HI
Too much application of pesticide in a crop field is disturbing human health directly and indelicately as the chemical proportion of pesticide is not support the life system and causes cancer [14]. Properties of pesticides such as lethality, lipophilicity, bioaccumulation, persistent characteristics have a negative impact on human health, which is a concerning factor for all. Intake the pesticide-contaminated food can cause a hazardous and reproductive effects. The consequence of pesticides can be analyzed by considering the period of contact and lethality of a specific pesticide. From the residue data, ADI and EADI were calculated, and data are presented in Table 1. From the ADI and EADI, the HI was computed. Consumption of okra as vegetables having HI more than unit (One) is hazardous to human health [12,20]. Consumption of okra fruits from the 4th day after application of ethion is non-hazardous to human health.
4. CONCLUSION
Ethion residues identified in the vegetable samples were 8.02, 1.14, 0.14, 0.08, and 0.02 mg/kg for 0th, 2th, 4th, 6th, and 8th day from the day it has applied, respectively. The correlation coefficient of ethion residue in relation to duration was highly significant (<0.01 level) and r value recorded as 0.957. Ethion dissipation percentages were 85.74, 98.25, 99.04, and 99.71 from samples collected on 2nd, 4th, 6th, and 8th days, respectively, when compared with residue immediately after two hours of application of ethion. During the study, the half-life of ethion was 0.95 days. A pre-harvest interval of more than 2 days is needed from the day after ethion application. The human HI of ethion was 10.02, 1.429, 0.175, 0.096, and 0.029 for samples collected on 0, 2, 4, 6, and 8th day, respectively, when consumed 150 g/day/person. Consumption of vegetables with a HI of more than one is not recommended. From the experiment, it is clear that the consumption of okra from the 4th day after the application of ethion is non-hazardous to human health.
5. ACKNOWLEDGMENT
The authors are thankful to the Head, Research Department, ITC, ABD, Rajamahendravaram, for providing the necessary research facilities to carry out these studies.
6. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
7. FUNDING
There is no funding to report.
8. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
9. ETHICAL APPROVALS
The article does not contain any studies conducted on human or animal subject.
10. DATA AVAILABILITY
All data generated and analysed are included within this research article.
11. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Khan N, Yaqub G, Hafeez T, Tariq M. Assessment of health risk due to pesticide residues in fruits, vegetables, soil, and water. J Chem 2020;2020:5497952. [CrossRef]
2. Endalew M, Gebrehiwot M, Dessie A. Pesticide use knowledge, attitude, practices and practices associated factors among floriculture workers in Bahirdar city, North West, Ethiopia, 2020. Environ Health Insights 2022;16:117?2221076250. [CrossRef]
3. Caratelli V, Fegatelli G, Moscone D, Arduini F. A paper-based electrochemical device for the detection of pesticides in aerosol phase inspired by nature:A flower-like origami biosensor for precision agriculture. Biosens Bioelectron 2022;205:114119. [CrossRef]
4. Moreno JF, Liébanas FA, Frenich AG, Vidal JM. Evaluation of different sample treatments for determining pesticide residues in fat vegetable matrices like avocado by low-pressure gas chromatography-tandem mass spectrometry. J Chromatogr A 2006;1111:97-105. [CrossRef]
5. Claeys WL, Schmit JF, Bragard C, Maghuin-Rogister G, Pussemier L, Schiffers B. Exposure of several Belgian consumer groups to pesticide residues through fresh fruit and vegetable consumption. Food Control 2011;22:508-16. [CrossRef]
6. Bempah CK, Buah-Kwofie A, Denutsui D, Asomaning J, Tutu AO. Monitoring of pesticide residues in fruits and vegetables and related health risk assessment in Kumasi Metropolis, Ghana. Res J Environ Earth Sci 2011;3:761-71.
7. Peshin R, Kranthi KR, Sharma R. Pesticide use and experiences with integrated pest management programs and Bt cotton in India. In:Integrated Pest Management. Dordrecht:Springer;2014. 269-306. [CrossRef]
8. Pandey SA. Relative bio-efficacy of newer insecticide molecules against okra shoot and fruit borer and their dissipation:A review. J Pharmacogn Phytochem 2018;7:93-104.
9. Hassanzadeh N, Bahramifar N, Abbas ES, Saberfar F. Dissipation of ethion applied on greenhouse cucumbers by duration of a pre-harvest interval (PHI). Int J Agric Enviorn Biotechnol 2010;3:179-83.
10. Soliman MM. Persistence of new insecticides and their efficacy against insect pests of cowpea. Aust J Basic Appl Sci 2011;5:82-9.
11. Parmar KD, Korat DM, Shah PG, Singh S. Dissipation and decontamination of some pesticides in/on okra. Pestic Res J 2012;24:42-6.
12. Akoto O, Andoh H, Darko G, Eshun K, Osei-Fosu P. Health risk assessment of pesticides residue in maize and cowpea from Ejura, Ghana. Chemosphere 2013;92:67-73. [CrossRef]
13. Rao BN. Fruits, vegetables, milk, and animal foods in balanced Indian diets-a critical appraisal. NFI Bull 2013;34:1-5.
14. Akhtar S, Yaqub G, Hamid A, Afzal Z, Asghar S. Determination of pesticide residues in selected vegetables and fruits from a local market of Lahore, Pakistan. Curr World Environ 2018;13:242-50. [CrossRef]
15. Chowdhury AZ, Hasan M, Karim N, Fakhruddin AN, Hossain S, Chowdhury AA, et al. Contamination and health risk assessment of pesticide residues in vegetables from agricultural fields of Gazipur District, Bangladesh. Sigma 2014;2:421-7.
16. Seo YH, Cho TH, Hong CK, Kim MS, Cho SJ, Park WH, et al. Monitoring and risk assessment of pesticide residues in commercially dried vegetables. Prev Nutr Food Sci 2013;18:145. [CrossRef]
17. Anastassiades M, Lehotay SJ, Štajnbaher D, Schenck FJ. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid-phase extraction“for the determination of pesticide residues in produce. J AOAC Int 2003;86:412-31. [CrossRef]
18. Rangaswamy R. A Text Book of Agricultural Statistics. New Delhi:New Age International (P) Limited, Publishers;2000.
19. Fantke P, Gillespie BW, Juraske R, Jolliet O. Estimating half-lives for pesticide dissipation from plants. Environ Sci Technol 2014;48:8588-602. [CrossRef]
20. Chen C, Qian Y, Chen Q, Tao C, Li C, Li Y. Evaluation of pesticide residues in fruits and vegetables from Xiamen, China. Food Control 2011;22:1114-20. [CrossRef]
21. European Food Safety Authority (EFSA), Cabrera LC, Pastor PM. The 2019 European Union report on pesticide residues in food. EFSA J 2021;19:e06491. [CrossRef]
22. Wang C, Wu J, Zhang Y, Wang K, Zhang H. Field dissipation of trifloxystrobin and its metabolite trifloxystrobin acid in soil and apples. Environ Monit Assess 2015;187:1-7. [CrossRef]
23. Kiwango AP, Kassim N, Kimanya EM. Pesticide residues in vegetables:Practical interventions to minimize the risk of human exposure in Tanzania. Curr J Appl Sci Technol 2018;26:1-18. [CrossRef]
24. Darko G, Akoto O. Dietary intake of organophosphorus pesticide residues through vegetables from Kumasi, Ghana. Food Chem Toxicol 2008;46:3703-6. [CrossRef]
25. Das SK, Mukherjee I, Das SK. Dissipation of flubendiamide in/on okra [Abelmoschus esculenta (L.) Moench] fruits. Bull Environ Contam Toxicol 2012;88:381-4. [CrossRef]
26. World Health Organization. The WHO Recommended Classification of Pesticides by Hazard and Guidelines to Classification. Geneva:World Health Organization;2010.