1. INTRODUCTION
Food colorants play an important role in the food industry to make foods coloring and increase customer attractiveness and sensory acceptability [1]. Traditional foods are foods and dishes that are passed on through generations. They are traditional in nature and may have a historic precedent in a national dish, regional, or local cuisine. The Southern Folk Cake Festival is always held every year on certain occasions in the Mekong Delta, especially in Can Tho city. This festival introduces and promotes the traditional cake made from rice of the Mekong Delta. In addition, it also honors Southern culinary culture and traditional cake-making culture of this area. The festival is a great playground for not only Vietnamese people but also international tourists for cultural exchange.
In general, traditional/folk cakes are often prepared with the use of many different colors to create attractiveness, but some of them are not safe because a few enterprises still tend to favor the use of synthetic colorants instead of natural extracted pigments. Natural food colors consist of pigments such as anthocyanins, carotenoids, and chlorophyll that are extracted from plants [2]. The demand for food products with natural coloring agents has increased since consumption of synthetic colorings is believed to cause allergies, food intolerance, hyperactivity, irritability, and sleep disorders in children [3]. The new sources of natural blue color are extracted from Butterfly pea flowers (BPF) [4] and identified [5]. The next problem is necessary to control the stability of these pigments in different processing techniques. Moreover, when using blue color extracted from BPF in common food processing, anthocyanin will be added. Anthocyanins are known as non-toxic natural pigments. This bioactive compound has been shown to provide a number of health benefits such as antibacterial, antiviral, anti-allergic, anti-cancer, anti-inflammatory, anti-proliferative, and preventing various degenerative diseases [6]. Therefore, the use of anthocyanin as food colorants should be encouraged to provide these health benefits to consumers. Unfortunately, anthocyanin compounds are usually unstable, they are easily degraded by exposure to heat, light, pH, oxygen, and enzymes [7-9]. Thermal and pH degradation kinetics of anthocyanins in natural food colorant prepared from black rice bran have been studied by Loypimai et al. [10]. Jiang et al. [11] studied degradation of anthocyanin and polymeric color formation during heat treatment of purple sweet potato extract at different pH value. Degradation kinetics of anthocyanins in blackberry juice and concentrate have been investigated [12,13]. Degradation kinetics of anthocyanin from purple rice bran and effect of hydrocolloids on its stability were studied by Mulyawanti et al. [14]. Anthocyanin content decreased during processing and storage, but this concentration increased slightly during steaming and that the highest losses occurred during boiling. Recently, the thermal stability of anthocyanin in mixed raspberry-pomegranate-banana nectar in the presence of ascorbic acid and citric acid was studied [15]. However, data on the stability of anthocyanin in traditional Vietnamese foods during cooking at different temperatures and storage are still lacking. The use of natural colorants, of which blue is particularly rare, tends to be sensitive to processing and storage conditions. Considering the above, the aim of this work was to evaluate the anthocyanin remaining in traditional foods from different cooking methods. The kinetic parameters of anthocyanin degradation, including the rate constant and half-life values for cooking/storage time were determined.
2. MATERIALS AND METHODS
2.1. Preparation of BPF
Butterfly pea was grown at CanTho University (Vietnam). After harvesting, BPFs were washed with running tap water, drained and arranged on trays (stainless steel). It was dried at 70°C in an air convectional oven (SIBATA SD60, Japan) with an air flow speed of 1.0 m/s [16]. The moisture of the dried sample reached 5–6%, containing anthocyanin 1.50 ± 0.04 mg/g DW, and then it was packed in dark bags and kept at low temperature (6 ± 2°C) for further use.
2.2. BPF Extraction Procedure
The ratio of raw materials (BPF) and solvent (water) was 1:20 (w/v). The dried material (50 g) was used for each treatment. The extraction process was conducted in a microwave oven (LG MS2595DIS, Korea) with a medium power level (400 W) for 5 min. The obtained extracts were centrifuged (Z232K, Hermle, Germany) for 30 min at 12,000 rpm at room temperature to obtain the supernatant [4]. The clear liquid was passed through the filter paper and stored in the refrigerator for further use.
2.3. Effect of Added Extract Concentration and Steaming Time on Anthocyanin Stability – A Case Study for Bloating Fern-Shaped Cake/Thin Cakes (Bánh Bèo)
Weigh the fixed cake dough of 170 g, add 100 g of coconut milk and 500 g of water. BPF extract (prepared) was added at different concentrations of 5, 10, and 15% (based on dough weight) for each treatment. Mix the dough until it was homogeneous and smooth. Perform the steaming process with the survey time of 5, 7, 9, and 11 min. After each arranged time, the cake (diameter: 5 cm, thickness: 0.2 cm) was taken out, cooled rapidly, photographed to monitor color change, and analyzed the remaining anthocyanin content (mg/g). The experiment was repeated 3 times.
2.4. Effect of Added Extract Concentration and Boiling Time on Anthocyanin Stability – A Case Study for Rice Ball Sweet Soup (Viên Chè ?)
Weigh the fixed weight of glutinous rice flour at 100 g for all treatments. Add 75 g of water to the dough. BPF extract was used with three different concentrations of 5, 10, and 15% (based on dough weight). Knead the dough until smooth and evenly colored. Shape into round dough balls with a weight of 5 g each. The glutinous rice balls were boiled for 3, 6, and 9 min. Products were taken out according to the assigned time. The following operations were similar to those in the previous experiment.
2.5. Effect of Added Extract Concentration and Frying Time on Anthocyanin Stability – Case Study for Meat Stuffed Rice Ball (Viên Th?t)
The mass of the powder was kept constant at 100 g (total dry matter). The weight ratio of glutinous rice flour: wheat flour (Bakers’ Choice 13, Vietnam) was 35:65. Use an additional amount of water of 75 g. Supplementation of BPF extract with three different concentrations of 5, 10, and 15% (based on dough weight). The dough was kneaded until smooth and uniform. Weigh each dough ball as 10 g and shaped it with the filling (the filling includes ground meat and spices). Perform frying in an electric frying pan (IESKIMOS, China) at a fixed temperature of 150°C with controlled frying times of 2, 4, and 6 min. The following operations were similar to those in the previous experiment.
2.6. Effect of Added Extract Concentration and Baking Temperature on Anthocyanin Stability – Case Study for Bread
The bread was made in the PETRUS automatic bread machine (China). The ingredients were weighed as follows: Flour 290 g, eggs 60 g, sugar 22 g, yeast 2 g, salt 3 g, 26 g butter, and 140 g non-sugar fresh milk. BPF extract was added with different concentrations [16, 18, and 20% (w/w)]. The bread was baked at 160, 165, and 170°C with a total baking time (including stuffing, incubation, and baking) of 3 h and 50 min. The following operations were similar to those in the previous experiment.
2.7. Effect of Added Extract Concentration and Sterilization Temperature on Anthocyanin Stability – Case Study for BPF Juice
BPF extract was added to water (water soluble used for drinking) at 3, 4, and 5%, and using sugar to made the total soluble solid content to 10° Brix. The juice was filled in glass bottles and sterilized in sterilizer (DG-S280C, China) at 125°C with a holding time of 1, 3, 5, and 7 min. After controlled times, the products were removed from the sterilizer and rapidly cooled to reach the ambient temperature. The following operations were similar to those in the previous experiment.
2.8. Effect of Storage Temperature on Anthocyanin Stability in BPF Drink
The experiment was designed with two storage temperatures: 28 ± 2°C and 6 ± 2°C. With the total time was 3 months, samples were checked and analyzed for anthocyanin content after every 1 week of storage.
2.9. Determination of Total Anthocyanin Content (TAC)
TAC was analyzed using the differential pH method described by Giusti and Wrolstad [17]. The pH-difference method was based on the reaction at pH 1.0 and 4.5, allowing accurate and rapid measurement of total anthocyanin. Briefly, 1 mL of the extract was reacted with potassium chloride buffer (pH 1.0) and sodium acetate buffer (pH 4.5). The mixture was incubated for 15 min. Measure the absorbance of each dilution at 510 and 700 nm. The calculation of TAC is based on the equation described by these authors.
2.10. Degradation Kinetics of Anthocyanin
The previous studies showed that thermal degradation of anthocyanin followed a first-order reaction kinetics [15,18,19]. Storage temperature also impacts on anthocyanin degradation [20]. During the long-term storage, total and individual anthocyanin degraded following first-order reaction kinetics at the rate strongly dependent on temperature. The first-order reaction rate constants (k in min−1), the time needed for 50% degradation in the visual color and anthocyanin, or half-life (t1/2 in min/h/week) was calculated using the following equations 1 and 2.
ln (Ct/Co) = kt (1)
t1/2 = -ln 0.5/k (2)
Where Co is initial anthocyanin content; Ct is anthocyanin content after t (min/h) heating/storage at a given temperature and k is the degradation rate constant. When the natural logarithm of the residual concentration is plotted as a function of the treatment time, the degradation rate constant (k) can be estimated by linear regression analysis and the k value is derived from the slope of the regression line.
3. RESULTS AND DISCUSSION
3.1. Effect of Added Extract Concentration and Steaming Time on Anthocyanin Stability in Bloating Fern-Shaped Cake/Thin Cakes (Bánh Bèo)
The effects of temperature on anthocyanin stability in Bloating fern-shaped cake were investigated at a steaming temperature of 100°C with different concentrations of added extract. When the logarithm of the concentration was plotted as a function of time, thermal degradation of anthocyanin could be described by a first-order model with a high regression coefficient (≥0.97) [Figure 1].After 11 min of the treatments, the anthocyanin degradation was ranging from 14.33%, 16.50%, and 20.00% when extract used 15%, 10%, and 5%, respectively. The degradation rate constant (k) and half-life (t1/2) values of anthocyanins in cakes steamed at 100oC with different added extract ratios are shown in Table 1. A decrease in anthocyanin degradation rate constant (k) was observed when the percent of extract used increased, which corresponded to an increase in the value of t1/2 when the extract ratios increased from 5 to 15%.
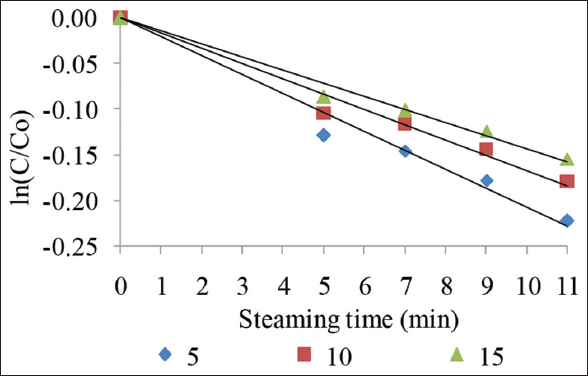 | Figure 1: The remaining anthocyanin in cake at steaming time using different % of extract. [Click here to view] |
Table 1: The rate constant and half-life values of anthocyanin degradation at 100°C of steaming using different extract ratios.
| Percentage of BPF extract (%) | The rate constant [k (min−1)] | R2 | t1/2 (min) |
|---|---|---|---|
| 5 | 0.0196±0.0017 | 0.983 | 35.36 |
| 10 | 0.0159±0.0014 | 0.982 | 43.59 |
| 15 | 0.0137±0.001 | 0.984 | 50.59 |
Standard error of regression. BPF: Butterfly pea flower.
It is well known that anthocyanin pigments make up a good source of natural food colorants. However, they are unstable, where temperature is an important factor influencing the stability of this pigment. Larief and Dirpan [21] reported that TAC is lost mainly due to destruction by heat and during processing. In this case, it has been shown that higher utilization ratios of extracts have better retention of anthocyanin with the same heating time.
Surh and Koh [22] showed that the steaming process reduced anthocyanin content in raw materials, after 1 h of steaming of black rice, anthocyanin content was reduced by 88%, this loss was mainly due to thermal degradation. The color of the cake added with BPF extract after the end of the steaming process has been changed. The color of the product gradually faded as the steaming time was prolonged, but this change was not significantly different [Figure 2]. The product still retained its bright color and high sensory value after 11 min of steaming. Combined with sensory evaluation, the product preference was highest at the percentage of BPF extract used 10% and the steaming time was 7 min.
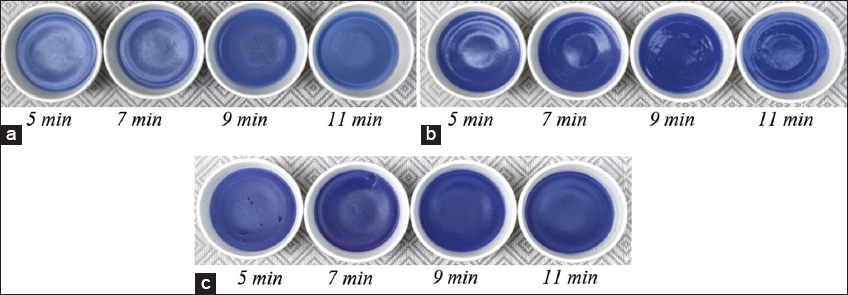 | Figure 2: (a) Butterfly pea flowers extract used: 5%, (b) Butterfly pea flowers extract used: 10%, and (c) Butterfly pea flowers extract used: 15%, Product color change according to the concentration of extract used and steaming time. [Click here to view] |
3.2. Effect of Added Extract Concentration and Boiling Time on Anthocyanin Stability in Rice Ball Sweet Soup (Chè ?)
The anthocyanin content was well maintained in this product over time of boiling. With the longest boiling time of 9 min, anthocyanin losses were recorded as 7.41, 9.33, and 10.26% with the product supplemented with 15, 10, and 5% extract, respectively (data not shown). In the case of boiling, the destruction of TAC clearly followed the first-order reaction kinetic model with high regression coefficient (≥0.95), the rate constant k and half-life values are given in Table 2. Compared with the steaming method, the total anthocyanin in the boiled product was better maintained, partly due to the shorter cooking time. Less destruction of anthocyanin was found in this cooking method and the half-life values have been calculated for higher values than steaming. The study of Liao et al. [23] showed that boiling and steaming did not significantly affect the TAC. Almost all monoacylated anthocyanins increased significantly after boiling. However, Burgos et al. [24] reported that boiling significantly reduced the TAC but increased the total phenol content. Hong and Koh [25] studied on effects of various cooking methods on anthocyanin and also confirmed that boiling and steaming reduced anthocyanin content. The color of rice-ball sweet soup at different ratios of BPF extract and boiling time is shown in Figure 3.
Table 2: The rate constant and half-life values of anthocyanin degradation at boiling temperature (100°C) using different extract ratios.
| Percentage of BPF extract (%) | The rate constant [k (min-1)] | R2 | t1/2 (min) |
|---|---|---|---|
| 5 | 0.0117±0.0017 | 0.959 | 59.24 |
| 10 | 0.0112±0.0017 | 0.956 | 61.88 |
| 15 | 0.0090±0.0014 | 0.954 | 77.02 |
Standard error of regression. BPF: Butterfly pea flower.
 | Figure 3: (a) Butterfly pea flowers extract used: 5%, (b) Butterfly pea flowers extract used: 10%, and (c) Butterfly pea flowers extract used: 15%, Product color change according to the extract concentration used and boiling time. [Click here to view] |
The color of the rice ball sweet soup supplemented with BPF extract was changed after boiling. The higher the percentage of BPF extract, the darker color was observed in product. The longer the boiling time, the lighter the product, but this change did not make a big difference. The product still retained its bright color and high sensory value after 9 min of boiling. The time of 6 min made the glutinous rice ball completely gelatinized, the product was edible at this time.
3.3. Effect of Added Extract Concentration and Frying Time on Anthocyanin Stability in Fried Meat Stuffed Rice Ball (Viên Th?t)
The results obtained for fried meat stuffed rice ball for 2, 4, and 6 min at 150°C and the addition of different concentration of extract is presented in Table 3.
Table 3: The rate constant and half-life values of anthocyanins degradation at frying temperature (150°C) using different extract ratios.
| Percentage of BPF extract (%) | The rate constant [k (min−1)] | R2 | t1/2 (min) |
|---|---|---|---|
| 5 | 0.0598±0.003 | 0.994 | 11.59 |
| 10 | 0.0597±0.003 | 0.994 | 11.61 |
| 15 | 0.0581±0.002 | 0.996 | 11.93 |
Standard error of regression. BPF: Butterfly pea flower.
The anthocyanin content of samples after frying at three extract concentrations was retained quite high, 69.23, 69.33, and 70.37%, corresponding to the concentration of extract added 5, 10, and 15%, probably due to the short frying time. The destruction of anthocyanin content during frying also followed a first-order kinetics model with a high regression coefficient (R2>0.99). It was observed that the higher k values, corresponding to a lower half-life values, were obtained compared with the steaming and boiling methods. In this case, high temperatures rapidly destroy anthocyanin. As indicated, TAC of the fried products was lost about 30.77, 30.67, and 29.63% with additional extraction ratios of 5, 10, and 15%, respectively. Anthocyanin loss in this study was shown to be lower than in other studies performed previously. Kita et al. [26] have studied on frying of red- and purple-fleshed potatoes (Solanum tuberosum L.) that are a good source of anthocyanin and reported that frying process caused anthocyanin degradation of 38–70%. Rytel et al. [27] found that at a temperature of 150°C, chips maintained 35% anthocyanin, while at higher temperature (170°C), the product maintained lower content (only about 16% original anthocyanin content in raw materials). The effect of frying on anthocyanin content in Hwajeon with the change ratio of black glutinous rice flour was studied by Jeonghee and Eunmi [28]. They showed that Hwajeon maintained up to 60% of anthocyanins in the dough after frying, that is, a loss of about 40% after frying. The color of cakes with different extraction rates (5–15%) and fried at different times (2–6 min) is shown in Figure 4. Although the high temperature of frying reduced TAC, the product still maintained a nice blue color after 4 min of frying and extract concentration ≥10% was used. In a pilot plant testing (20 kg/batch), we also found similar results [Figure 5].
 | Figure 4: (a) Butterfly pea flowers extract used: 5%, (b) Butterfly pea flowers extract used: 10%, and (c) Butterfly pea flowers extract used: 15%, Product color change according to the concentration of extract used and frying time. [Click here to view] |
 | Figure 5: Color of product in pilot plan testing (added 10% extract and fried for 4 min at 150°C). [Click here to view] |
3.4. Effect of BPF Extract Ratio and Baking Temperature on Anthocyanin Content and Color of Bread
The analysis results of anthocyanin remaining in the bread baked at different temperatures (160–170°C) with the same time and extract added (16–20%) are presented in Table 4.
Table 4: The remaining anthocyanin content (%) in bread supplemented with added extract percentage and baking at different temperatures.
| Extract (%) – Baking temperature (°C) | The remaining anthocyanin (%) | Extract (%) – Baking temperature (°C) | The remaining anthocyanin (%) | Extract (%) – Baking temperature (°C) | The remaining anthocyanin (%) |
|---|---|---|---|---|---|
| 16–0 | 100 | 18–0 | 100 | 20–0 | 100 |
| 16–160 | 48.98 | 18–160 | 50.51 | 20–160 | 52.28 |
| 16–165 | 44.67 | 18–165 | 46.06 | 20–165 | 48.27 |
| 16–170 | 40.14 | 18–170 | 42.02 | 20–170 | 44.08 |
The color of bread with added BPF extract and baked at different temperatures is shown in Figure 6.
 | Figure 6: (a) Baking temperature 160°C, (b) Baking temperature 165°C, and (c) Baking temperature 170°C, Color of bread with added extract (16–20%) and baking temperature (160–170°C). [Click here to view] |
The study results showed that the higher the percentage of BPF extract, the higher the anthocyanin content maintained in the products. TAC in bread supplemented with thre concentrations of extract was significantly lost after baking (from 47.72% to 59.86%). Anthocyanins are destroyed by high temperature. Liao et al. [23] reported that baking can be reduced the TAC about 11%.
3.5. Effect of Sterilization Conditions on Anthocyanin Content of BPF (Contained in Glass Bottles)
To ensure stable food quality during storage, heat treatment was selected. Liquid foods that undergo sterilization will not be damaged or harmful to the health of consumers under normal storage conditions. To ensure sterility of low-acid bottled/canned food (pH >4.6), it should be processed at 121°C for at least 2.4 min [29]. The optimum heat treatment time and temperature combination are measured in F value; for the reduction of spores of Clostridium botulinum, the most heat-resistant pathogen, the minimum F value was 2.52 [30]. Our obtained results showed that TAC of BPF juice decreased with time of sterilization (at different extract concentrations used). The linear regression confirmed the destruction of anthocyanin in juice followed by a first-order reaction [Figure 7]. The values of the first-order model and the half-life values at the same temperature are calculated and given in Table 5. Research results have shown that anthocyanin losses were about 28.48, 23.22, and 16.61% after 9 min of sterilization at 125°C, corresponding to the extract used 3, 4, and 5%. Combined with color observation, the product maintained its beautiful color after 3 min of sterilization. From 5 min on, the color decreased significantly. As discussed, 3 min was suitable for the sterilization process of this juice, anthocyanin persisted to 82.28; 90.04; and 91.51% with extracts using 3, 4, and 5%, respectively. Nie et al. [31] studied the effect of sterilization on anthocyanin in blueberry; it was observed that TAC was decreased 46% by sterilization.
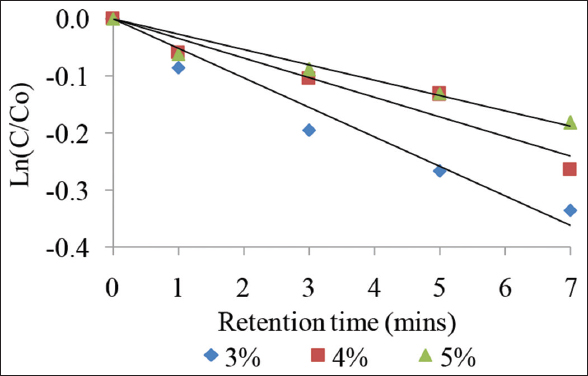 | Figure 7: Degradation of anthocyanin in Butterfly pea flower flowers juice during sterilization at 125°C. [Click here to view] |
Table 5: The rate constant and half-life values of anthocyanin degradation in juice at sterilization temperature of 125°C using different extract ratios.
| Percentage of butterfly pea flowers extract (%) | The rate constant (k) | R2 | t1/2 (min) |
|---|---|---|---|
| 5 | 0.047±0.0046 | 0.973 | 14.91 |
| 10 | 0.033±0.0052 | 0.935 | 20.82 |
| 15 | 0.024±0.0027 | 0.968 | 29.495 |
Standard error of regression.
3.6. Storage Capacity of BPF Drink at Different Storage Temperatures
As expected, anthocyanin contents in juice decrease in time during storage at different temperatures. Linear regression confirmed that the degradation of anthocyanin in BPF juice was also confirmed that the kinetics reaction followed first-order [Figure 8]. The rate of degradation of anthocyanin (k) and time of half-life of anthocyanins (t1/2) in the juice after storage was given in Table 6. After 12 weeks of storage, anthocyanin content was still maintained with relatively high amounts, 80% and 84%, corresponding to the losses of 20% and 16% at two storage temperatures 28 ± 2°C and 6 ± 2°C were determined, respectively. Beaulieu et al. [32] studied on quality and sensory changes in rabbit eye blueberry and reported that total anthocyanin decreased of 84.5% and 85.5% after 4 months storage in non-clarified juice and clarified juice, respectively. Brownmiller et al. [33] studied on blueberry juice and reported more than 50% total anthocyanin loss after 6 months of storage.
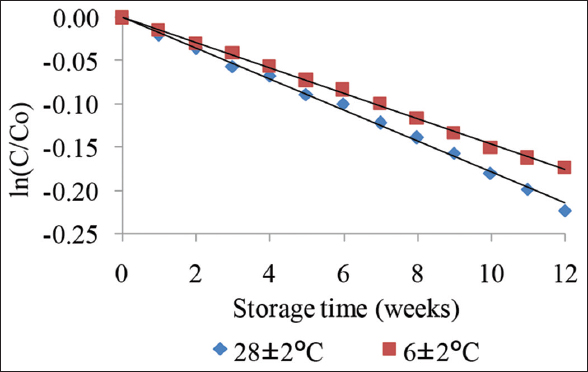 | Figure 8: Degradation of anthocyanin in Butterfly pea flower juice (4% added extract) during storage at different temperature. [Click here to view] |
Table 6: The rate constant and half-life values of anthocyanin degradation in juice at different storage temperatures.
| Storage temperatures (°C) | The rate constant (k) | R2 | t1/2 (weeks) |
|---|---|---|---|
| 28±2 | 0.0181±0.0003 | 0.991 | 38.29 |
| 6±2 | 0.0148±0.0001 | 0.994 | 46.83 |
4. CONCLUSION
Research on the stability of anthocyanin helps to suggest suitable ways to add BPF extract and control its content in different cooking and preservation techniques of some traditional Vietnamese food products. The degradation of anthocyanins depends on the cooking and storage conditions, the half-life of anthocyanin depends on the cooking method. Research results can have direct implications for food businesses to help use safe natural colorants instead of synthetic colorants for traditional/popular food products, increasing the attractiveness of their products and controlling the color degradation, corresponding to the loss of anthocyanin content during processing and storage. These results also showed that cooking conditions need to be carefully applied to maintain the product’s natural color. In addition, further bioavailability studies are needed to evaluate the possible benefits of these food products on human health.
5. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
6. FUNDING
This research received no external funding.
7. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
8. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
9. DATA AVAILABILITY
All data generated and analyzed are included within this research article.
10. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Lin WS, He PH, Chau CF, Liou BK, Li S, Pan MH. The feasibility study of natural pigments as food colorants and seasonings pigments safety on dried tofu coloring. Food Sci Human Wellness 2018;7:220-8. [CrossRef]
2. Sen T, Barrow CJ, Deshmukh SK. Microbial pigments in the food industry-challenges and the way forward. Front Nutr 2019;6:7. [CrossRef]
3. Feketea G, Tsabouri S. Common food colorants and allergic reactions in children:Myth or reality?Food Chem 2017;230:578-88. [CrossRef]
4. Thuy NM, Ben TC, Minh VQ, Tai NV. Effect of extraction techniques on anthocyanin from butterfly pea flowers (Clitoria ternatea L.) cultivated in Vietnam. J Appl Biol Biotech 2021;9:173-80. [CrossRef]
5. Thuy NM, Minh VQ, Ben TC, Thi Nguyen MT, Ha HT, Tai NV. Identification of anthocyanin compounds in butterfly pea flowers (Clitoria ternatea L.) by ultra performance liquid chromatography/Ultraviolet coupled to mass spectrometry. Molecules 2021;26:4539. [CrossRef]
6. Liu J, Zhou H, Song L, Yang Z, Qiu M, Wang J, et al. Anthocyanins:Promising natural products with diverse pharmacological activities. Molecules 2021;26:3807. [CrossRef]
7. Sadilova E, Stintzing FC, Kammerer DR, Carle R. Matrix dependent impact of sugar and ascorbic acid addition on color and anthocyanin stability of black carrot, elderberry and strawberry single strength and from concentrate juices upon thermal treatment. Food Res Inter 2009;42:1023-33. [CrossRef]
8. Patras A, Brunton N, O'Donnell C, Tiwari B. Effect of thermal processing on anthocyanin stability in foods;mechanisms and kinetics of degradation. Trends Food Scie Tech 2010;21:3-11. [CrossRef]
9. Cisse M, Vaillant F, Kane A, Ndiaye O, Dornier M. Impact of the extraction procedure on the kinetics of anthocyanin and colour degradation of roselle extracts during storage. J Sci Food Agric 2012;92:1214-21. [CrossRef]
10. Loypimai P, Moongngarm A, Chottanom P. Thermal and pH degradation kinetics of anthocyanins in natural food colorant prepared from black rice bran. J Food Sci Technol 2016;53:461-70. [CrossRef]
11. Jiang T, Mao Y, Sui L, Yang N, Li S, Zhu Z, et al. Degradation of anthocyanins and polymeric color formation during heat treatment of purple sweet potato extract at different pH. Food Chem 2019;274:460-70. [CrossRef]
12. Wang W, Xu S. Degradation kinetics of anthocyanins in blackberry juice and concentrate. J Food Eng 2007;82:271-5. [CrossRef]
13. Das AB, Goud VV, Das C. Degradation kinetics of anthocyanins from purple rice bran and effect of hydrocolloids on its stability. J Food Process Eng 2020;49:e13360. [CrossRef]
14. Mulyawanti I, Budijanto S, Yasni S. Stability of anthocyanin during processing, storage and simulated digestion of purple sweet potato pasta. Indonesian J Agric Sci 2018;19:1-8. [CrossRef]
15. Thuy NM, Han LN, Tai NV. Thermal stability of anthocyanin in mixed raspberry-pomegranate- banana nectar in the presence of ascorbic acid and citric acid. J Appl Biol Biotech 2022;10:189-95.
16. Thuy NM, Minh VQ, Ben TC, Ha HT, Tai NV. Impact of different thin layer drying temperatures on the drying time and quality of butterfly pea flowers. Food Res 2021;5:197-203. [CrossRef]
17. Giusti MM, Wrolstad RE. Characterization and measurement with UVvisible spectroscopy. In:King S, Gates M, Scalettar L, editors. Current Protocols in Food Analytical Chemistry. New York:Wiley;2001. p. F1.2.1-F1.2.13. [CrossRef]
18. Nayak B, Berrios Jde J, Powers JR, Tang J. Thermal degradation of anthocyanins from purple potato (cv. Purple Majesty) and impact on antioxidant capacity. J Agric Food Chem 2011;59:11040-9. [CrossRef]
19. Hou Z, Qin P, Zhang Y, Cui S, Ren G. Identification of anthocyanins isolated from black rice (Oryza sativa L.) and their degradation kinetics. Food Res Inter 2013;50:691-7. [CrossRef]
20. Muche BM, Speers RA, Rupasinghe HPV. Storage temperature impacts on anthocyanins degradation, color changes and haze development in juice of “Merlot“and “Ruby“Grapes (Vitis vinifera). Front Nutr 2018;5:100. [CrossRef]
21. Larief R, Dirpan A. Purple yam flour (Dioscorea alata L.) processing effect on anthocyanin and antioxidant capacity in traditional cake “Bolu Cukke“making. IOP Conf Ser Earth Environ Sci 2018;207:012043. [CrossRef]
22. Surh J, Koh E. Effects of four different cooking methods on anthocyanins, total phenolics and antioxidant activity of black rice. J Scie Food Agric 2014;94:3296-304. [CrossRef]
23. Liao M, Zou B, Chen J, Yao Z, Huang L, Luo Z, et al. Effect of domestic cooking methods on the anthocyanins and antioxidant activity of deeply purple-fleshed sweetpotato GZ9. Heliyon 2019;5:e01515. [CrossRef]
24. Burgos G, Amoros W, Munõa L, Sosa P, Cayhualla E, Sanchez C. Total phenolic, total anthocyanin and phenolic acid concentrations and antioxidant activity of purple-fleshed potatoes as affected by boiling. J Food Compos Anal 2013;30:6-12. [CrossRef]
25. Hong KH, Koh E. Effects of cooking methods on anthocyanins and total phenolics in purple-fleshed sweet potato. J Food Proces Preser 2016;40:1054-63. [CrossRef]
26. Kita A, Bakowska-Barczak A, Hamouz K, Ku?akowska K, Lisi?ska G. The effect of frying on anthocyanin stability and antioxidant activity of crisps from red- and purple-fleshed potatoes (Solanum tuberosum L.). J Food Compos Anal 2013;32:169-75. [CrossRef]
27. Rytel E, Tajner-Czopek A, Kita A, Tkaczy?ska A, Kucharska AZ, Sokó?-??towska A. The influence of the production process on the anthocyanin content and composition in dried potato cubes, chips, and french fries made from red-fleshed potatoes. Appl Sci 2021;11:1104. [CrossRef]
28. Jeonghee S, Eunmi K. Effect of pan-frying on anthocyanin content in hwajeon with varying proportion of glutinous black rice flour. Korean J Food Cookery Sci 2014;30:378-84. [CrossRef]
29. Peck MW, van Vliet AH. Impact of Clostridium botulinum genomic diversity on food safety. Curr Opin Food Sci 2016;10:52-9. [CrossRef]
30. Heinz G, Hautzinger P. Meat Processing Technology for Small-To Medium-Scale Producers. Thailand, Bangkok:Food and Agriculture Organization of the United Nations;2007. Avaialble from:http://www.fao.org/docrep/010/ai407e/AI407E22.htm#fn11 [Last accessed on 2021 Jun 28].
31. Nie Q, Feng L, Hu J, Wang S, Chen H, Huang X, et al. Effect of fermentation and sterilization on anthocyanins in blueberry. J Sci Food Agric 2017;97:1459-66. [CrossRef]
32. Beaulieu JC, Stein-Chisholm RE, Lloyd SW, Bett-Garber KL, Grimm CC, Watson MA, et al. Volatile, anthocyanidin, quality and sensory changes in rabbiteye blueberry from whole fruit through pilot plant juice processing. J Sci Food Agric 2017;97:469-78. [CrossRef]
33. Brownmiller C, Howard LR, Prior RL. Processing and storage effects on monomeric anthocyanins, percent polymeric color, and antioxidant capacity of processed blueberry products. J Food Sci 2008;73:H72-9. [CrossRef]