1. INTRODUCTION
Prolonged hyperglycemia is the early sign of diabetes. Considering the fact that about 25% of the world population are diabetic, its management is of great importance [1]. Furthermore, the various effects of hyperglycemia demand immediate treatment [2]. An optimal method of addressing diabetes comprises of lifestyle and medical interventions which reduce hyperglycemia thereby decreasing the likelihood of developing the associated diabetic complications [3-5].
Dietary carbohydrates are broken down to simple sugars by the action of α-glucosidase and α-amylase and these enzymes are the primary and most promising targets of medical intervention to reduce hyperglycemia [5,6]. However, in a more established disease condition, mere control of the blood glucose levels does not suffice and requires a treatment comprising the inhibition of several other enzymes that are involved in the development of complications. A significant amount of excess glucose enters the polyol pathway and involves aldose reductase (AR) enzyme for its conversion to sorbitol, which accumulates resulting in impairment in the membrane permeability and leads to the microvascular and cardiovascular complications [7]. The impact of hyperglycemia is also witnessed on the tissue proteins which undergo non-enzymatic glycation rendering them inactive. These proteins then hinder the normal signal transduction pathway thereby generating reactive oxygen species and lead to several oxidative stress-induced complications [8]. More recently, the close association of diabetes and cardiovascular complications are ascertained because of the effect of hyperglycemia on the blood lipid profiles. Evidences are increasing to suggest the abnormalities induced by hyperglycemia on the lipids and lipoprotein metabolism which in turn results in the triglyceride rich lipoproteins thereby raises the risks for coronary heart disorders [9].
In this context, our study aims at inhibition of hyperglycemia and encumbers the development of its associated complications by inhibiting a series of enzymatic and non-enzymatic reactions. Banana pseudostem, a secondary product of banana is a popular vegetable and also known for its medicinal properties in treatment of kidney stones. Indian traditional medicine Ayurveda also reports its benefits in management of diabetes [10]. With this background, in our previous study, we established the inhibitory potential of pseudostem ethanol extract (EE) on the key carbohydrate hydrolyzing enzymes α-glucosidase [11,12]. In this study, we aim at evaluating the in vitro α-glucosidase and α-amylase inhibitory activity and antioxidant potential of the compounds Stigmasterol (C1) and β-Sitosterol (C2) isolated from EE. Further, we aim to study the inhibition of extract and compounds on AR, advanced glycation end products (AGE) formation at every stage and also assess its in vivo antihyperlipidemic and antioxidant activities.
2. MATERIALS AND METHODS
2.1. Plant Material and Extraction
The collection and treatment of the sample (banana pseudostem) was carried out according to the previous study by the authors [11]. Treatment of pseudostem for the ethanol extraction using soxhlation apparatus, isolation of stigmasterol (C1) and β-Sitosterol (C2) using repeated silica gel column chromatography was conducted. Further, spectral measurements, mass spectrum evaluation, and IR spectra evaluation of the fraction containing a single compound in TLC were also done according to the previous study conducted by authors [11].
2.2. In Vitro Antioxidant Assays
The antioxidant ability of EE and the isolated compounds (C1 and C2) to scavenge radicals was tested using 1, 1-diphenyl-2-picrylhydrazyl (DPPH), 2, 2’-Azinobis-3-ethylbenzthiazoline-6-sulfonate (ABTS), and superoxide assays according to Razali et al. (2008) [13]. The EC50 values express the radical scavenging potential of the extracts, representing 50% of free (DPPH), cation (ABTS), and anion (superoxide) scavenging. The positive control used was butylated hydroxyl anisole (BHA). The antioxidant prospective was also projected for lipid peroxidation (LPO) inhibition in a liposome model and ferrous ion chelating activity according to Meghashri et al. (2010) [14]. Subsequently, determination of the reducing potential of iron ion for the extract and the compounds was performed according to Yen et al. (2002) [15]. All the samples were performed in three individual experiments. The following formula was used to calculate the absorbance of the antioxidant activity in percentage (%).
2.3. Antiglycemic Assays
The inhibitory activity of the isolated compounds against Type VI-B porcine pancreatic α-amylase and yeast type-1 α-glucosidase enzymes using starch and p-nitrophenyl-α-D-glucopyranoside (pNPG) as substrates, respectively, was carried out according to Ramu et al. 2014) [8]. The results were computed using the equation below, and are expressed as a percentage of α-amylase/α-glucosidase inhibition.
With appropriate blanks in between, all the experiments were performed in triplicates. As a positive control, acarbose was used. Under the given test circumstances, the inhibitory concentration of the extract and compounds to inhibit the 50% of α-amylase/α-glucosidase activity is known as IC50. The kinetic analysis of enzyme inhibition was conducted according to Ramu et al. (2014) [6] using Lineweaver-Burk plot method [16]. The activity of human recombinant AR was assessed using the absorbance method (decrease in NADPH absorption) at 340 nm using quercetin as positive control. All the tests were performed in triplicates and IC50 was calculated as described in the above section [8]. The antiglycation potential of extract and its components was assessed at various stages adopting the BSA/fructose method, as described by Ramu et al. (2014) [6]. The effects of EE, C1, and C2 on multistage glycation markers such as fructosamines (early phase), protein carbonyls (middle phase), and AGE (antiglycation end products, late phase), as well as thiol measurements, were studied after 3 weeks of incubation.
2.4. Animals and Experimental Design
Wistar rats of either sex weighing 220–10 g were used for the in vivo studies according to the protocol designed by Friedewald et al. (1972) [17]. The Institutional Animal Ethics Committee had approved the trials and the same were carried out according to the guidelines of CPCSEA, Chennai, India. Rats fed with basal diets were used as controls (group 1). Diabetes was induced using alloxan monohydrate (120 mg/kg) to group 2–7. EE was administered to diabetic rats in groups 3 and 4 at dosages of 100 and 200 mg/kg, respectively. C1 and C2 were given at 100 mg/kg of body weight to diabetic rats in groups 5 and 6. Metformin was given to diabetic rats in group 7 at a dose of 250 mg/kg of body weight.
Fasting blood glucose, total cholesterol (TC), triacylglycerol (TG), very low-density lipoprotein-C (VLDL-C), low-density lipoprotein-cholesterol (LDL-C), high-density lipoprotein-cholesterol (HDL-C), and atherogenic index (AI) were calculated according to the protocol given by Friedewald et al. (1972) [17]. Further, antioxidant enzyme activity evaluation was carried out according to the protocols by Marklund and Marklund (1974) [18], Rotruck et al. (1973) [19], and Sinha (1972) [20] were used to evaluate the activity of antioxidant enzymes (catalase, glutathione peroxidase, superoxide dismutase). Glutathione levels were determined using the Ellman (1959) method [21]. Ascorbic acid and α-tocopherol, the non-enzymatic antioxidants were evaluated according to Baker et al. (1980) [22] and Omaye et al. (1979) [23]. Furthermore, a colorimetric method for assessing LPO utilizing the thiobarbituric acid reaction with malondialdehyde described by Ohkawa et al. (1979) was used for the same [24].
2.5. Statistical Analysis
All the outcomes of this study have been shown as Mean ± SD. Comparison of statistical data between the treated and normal was performed using Duncan’s Multiple Range Test (version 21.0, Chicago, USA) and SPSS Software’s one-way analysis of variance (ANOVA). Only Results with only “P” ≤ 0.05 were considered as statistically significant.
3. RESULTS
3.1. Antiglycemic and In Vitro Antioxidant Activity
The inhibitory potential of the isolated compounds (C1 andC2) on the α-amylase/α-glucosidase, the carbohydrate metabolizing enzymes, was assessed using acarbose as positive control. Results described in terms of IC50 values [Table 1] illustrated that the α-amylase inhibitory effect of C1 (IC50: 42.58 ± 2.14 μg/ml) and C2 (IC50: 43.43 ± 1.03 μg/ml) was comparatively lower than acarbose (IC50: 29.71 ± 1.51 μg/ml). In contrast, C1 and C2 inhibited α-glucosidase with IC50 values of 7.31 ± 0.12 and 7.80 ± 0.93 μg/ml, respectively, and possessed a strong inhibition compared to acarbose (9.68 ± 0.48 μg/ml). In vitro inhibitory studies indicated that compounds had effective α-glucosidase inhibitory potential compared to α-amylase inhibition.
During inhibitory kinetic assays, the presence of C1 and C2, a comparative increase in Km values and a decrease in Vmax values ensures the typical reversible mixed-type plots. Diverse quantities of C1 [Figure a1] and C2 [Figure b1] intersected in the second quadrant, revealing a pattern of mixed-type inhibition, as shown by the plots. Mixed-type inhibition straddles the line between competitive and non-competitive inhibition. Using the Lineweaver-Burk method using secondary plots of the slope and Y-intercept versus compound (inhibitor) concentration, the dissociation constants of the K1 (inhibitor-bound enzyme complex) and the K2 (inhibitor-bound enzyme substrate complex) were calculated. C1 (0.55 ± 0.05 g, Figure a2) and C2 (0.43 ± 0.09 g, Figure b2) were found to have lower K1 values than C1 (4.92 ± 0.10 g, Figure a3) and C2 (2.38 ± 0.07 g, Figure b3), showing that chemicals bind more to α-glucosidase enzyme than to α-glucosidase-pNPG substrate complex. While, in terms of IC50 values, C1 had stronger inhibition toward α-glucosidase than C2 was also affirmed by smaller values of inhibitory constants exhibited by K1 than K2.
In this study, the compounds C1 (IC50: 1.80 ± 0.36 μg/ml) and C2 (IC50: 1.93 ± 0.37 μg/ml) isolated from banana pseudostem EE (IC50: 2.67 ± 0.27 μg/ml) were found to inhibit AR at far better levels than the EE [Table 1]. In AR inhibition tests, a phenolic compound known as quercetin (IC50: 3.420.14 g/ml) was used as a positive control. In general, EE, C1 and C2 exhibited stronger inhibition (P < 0.05) toward AR than quercetin.
 | Table 1: Inhibitory potential of EE, C1 and C2 against α–amylase, α–glucosidase, and aldose reductase enzymes. [Click here to view] |
EE and its derivatives showed a similar trend in radical scavenging activity testing. Table 2 summarises the estimated half effective concentration, or EC50 (g of testing per ml), of EE and associated components. In all three antioxidant experiments examined, stigmasterol had considerable free radical scavenging activities, but C2 was relatively lesser (in terms of EC50 values) compared to C1 (P < 0.05). In all in vitro antioxidant studies, EE and compounds extracted from them excelled BHA, and the activity ascended in the order C1>C2>EE>BHA.
In a liposome model, EE and its components effectively prevented the peroxidation of egg yolk homogenate compared to their EC50 values. When compared to the normal BHA, the ethanolic extract had 1.5-fold more metal (ferrous ion) chelating activity, while the isolated components had 2-fold higher metal (ferrous ion) chelating activity [Table 2]. The capacity of EE and its constituents to reduce ferric chloride and potassium ferricyanide complex was also used to measure their reducing power. Figure 2a shows a dose-dependent increase in absorbance (700 nm), which indicates that EE and its components are losing their effectiveness.
The antiglycation properties of EE and its components were assessed using a BSA/fructose glycation model. Test samples were examined for inhibition of multistage glycation markers such as fructosamines, protein carbonyls, and AGE, along with thiol protection, after a 3-week incubation period, as shown in Figure 2b-e, respectively. Adding EE and its components to the BSA-fructose model for 21 days resulted in a significant concentration-dependent decrease in AGE formation. At a concentration of 100 g/ml, EE inhibited the AGE formation by 8%, whereas the C1 and C2 inhibited the same by 83%, and 74%, respectively. The aminoguanidine inhibited the AGE formation by 70%, and was much lower in comparison with the extract and the isolated compounds [Figure 2b].
Figure 3a shows the fructosamine content of the Amadori product. Fructosamine level in the BSA/fructose system increased steadily as the experiment went, compared to non-glycated BSA. Fructosamine production was progressively diminished after treatment with EE and its components at a concentration of 100 g/ml. Fructosamine synthesis was inhibited in a dose-dependent way toward the end of the experiment, with stronger inhibition than aminoguanidine at various dosages [Figure 2c].
In comparison to non-glycated BSA, high doses of fructose increased the amount of protein carbonyl by 5-fold [Figure 3c]. The results demonstrated that adding 100 g/ml of EE, C1, and C2 to the BSA/fructose system decreased protein carbonyl levels considerably. The chemicals (100 g/ml) and EE together lowered protein carbonyl levels by 80–90% at the end of the trial. At a concentration of 100 g/ml, aminoguanidine lowered protein carbonyl concentration and inhibited by 77% [Figure 2d].
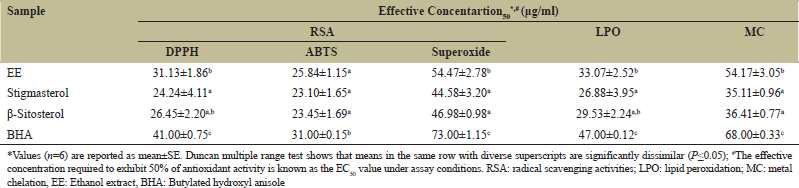 | Table 2: In vitro antioxidant activity of EE and its isolated compounds. [Click here to view] |
 | Figure 1: Lineweaver-Burk plot (LBp) on inhibition of α-glucosidase activity by Stigmasterol (a1) and β-Sitosterol (b1) at diverse concentrations for the estimation of K1 and K2. (a2, a3 and b2, b3) curve represents K1 and K2 calculation for the pNPG substrate, respectively. [Click here to view] |
Fructose-mediated glycation of BSA reduced the thiol groups in BSA over time in the same way that non-glycated BSA did. The extract and its constituents, on the other hand, prevented thiol group loss in a concentration-dependent manner [Figure 2e]. Thiol protection levels ranged from 65% to 85% in the presence of EE, C1 and C2, with significant denaturation shielding evident as thiol concentrations were nearly recovered to native BSA levels [Figure 3b].
 | Figure 2: At varying doses of ethanol extract of pseudostem and its separated components, inhibitory effects on Reducing power (a); AGE formation (b); Fructosamine (c); Protein carbonyls (d); Protein thiols protection (e). The mean and standard deviation (n=3) are used to calculate the values. [Click here to view] |
3.2. In Vivo Antihyperlipidemia and Antioxidant Activity
In each group, TC, triglycerides (TG), HDL-C, VLDL-C, LDL-C, AI, LDL-C/HDL-C ratio, HTR (percent), free fatty acids (FFA), and phospholipids (PL) levels are shown in Table 3. When compared to the normal control groups, alloxan induction resulted in a substantial (P < 0.05) increase in TC, TG, LDL-C, VLDL-C, AI, PL, FFA, and HTR (percent), as well as a drop in HDL-C levels. After 28 days of oral therapy with EE, C1, C2, and metformin in diabetic rats, all of the parameters were returned to near-normal values. The amounts of TC, TG, PL, and FFA found in the livers of diabetic and non-diabetic rats are shown in Table 4. In diabetic rats’ tissues, lipid indices were significantly higher (P < 0.05) than in normal rats’ tissues. The lipid profile in liver tissues was dramatically lowered following 28 days of administration of EE and the drugs.
The concentrations of enzymatic and non-enzymatic antioxidants in serum and liver, respectively, are shown in Tables 5 and 6. When compared to the normal control group, the serum and liver of untreated diabetic rats (group 2) had a substantial increase in lipid peroxide concentration. For 28 days, diabetic rats were given two doses of EE, C1, C2, and metformin (group 3–7) and their serum/liver lipid peroxide concentration was significantly reduced. The activities of enzyme antioxidants in serum and liver were reduced in diabetic control rats (P < 0.05). When diabetic rats were administered with EE, C1, C2, and metformin, their enzyme activity was significantly higher (P < 0.05) than diabetic control rats. Furthermore, when diabetic untreated rats were compared to normal control rats, non-enzymatic antioxidants like alpha-tocopherol, GSH, and ascorbic acid activities in the blood and liver were considerably reduced (P < 0.05). The results reverted to near normal after 28 days of oral treatment of extract (100 and 200 mg/kg) and compounds (100 mg/kg), similar to metformin (250 mg/kg).
4. DISCUSSION
The major symptom of diabetes is hyperglycemia, which is defined as consistently elevated levels of plasma glucose due to impairments in body metabolism, as well as the absence or insufficient insulin production [25]. Hyperglycemia elicits a complex of several reactions, both enzymatically and non-enzymatically, resulting in the formation of diabetic complications and such a condition require treatment that not only targets hyperglycemia but also various other pathways in order to achieve optimal management [26]. In the current scenario, antihyperglycemic effect is achieved by the use of enzyme blockers (α-glucosidase and α-amylase) that restrict the breakdown of dietary carbohydrates thereby preventing excess glucose entering the blood [6]. However, this further leads to undesirable breakdown of stored form of glucose (glycogen) or breakdown of proteins into individual amino acids which undergo gluconeogenic cycle to replenish glucose into the plasma both of which leads to loss of body weight that is witnessed in diabetic patient. Furthermore, thus generated excessive glucose enters various pathways leading to vascular complications which collectively prove fatal to the patients. Despite control of hyperglycemia being the pre-requisite in control of diabetes, addressing the complications associated with them also is equally important and this is achieved by the use of an adjunct that inhibits hyperglycemia (α-glucosidase and α-amylase inhibitors) along with inhibition of AR (the key enzyme of the polyol pathway), glycation of proteins (at various stages to form AGE), oxidative stress which are the major contributors of microvascular problems and inhibitor of hyperlipidemia which is the primary cause of macrovascular complication [8,27-29]. Hyperglycemia being the prime source of all the complications of diabetes, the first target of the present study was inhibition of the two key carbohydrates hydrolyzing enzymes viz., α-glucosidases and α-amylase which are located in the brush borders of the intestine and secreted by the pancreatic cells, respectively. Standard inhibitors namely acarbose, voglibose, and miglitol are antihyperglycemic but they also lead to undesirable problems due to which use of such synthetic inhibitors are less advocated [30]. Previous studies on banana plant also indicate the antioxidant and antidiabetic activity. Bioactive compounds from banana plant responsible for these pharmacological actions have been depicted in Tables 7 and 8, respectively. Considering this, a huge attention is driven towards identification of safer formulations derived from natural sources and on these lines, in our previous study; we evaluated the inhibitory potential of EE on α-glucosidases from yeast and mammalian origin [8,36,37]. EE effectively inhibited α-glucosidases paving way for its use as antihyperglycemic sources. However, the antihyperglycemic effects of the compounds extracted from EE were investigated to further support the findings. The inhibition of α-glucosidase by the compounds C1 (7.31 g/ml) and C2 (7.80 g/ml) was clearly higher than that of the standard medication acarbose (9.68 g/ml). Kinetic investigations (for α-glucosidase inhibition) revealed a mixed type of inhibition by both drugs, with two inhibitory constants, one representing the inhibitor-bound enzyme complex (K1) and the other representing the inhibitor-bound enzyme substrate complex (K2). The compounds’ lower K1 values of C1 (0.55 ± 0.05 μg) and C2 (0.43 ± 0.09 μg) in comparison to their K2 values of (C1: 4.92 ± 0.10 μg and C2: 2.38 ± 0.07 μg) in the study suggest that they work by binding to the enzyme rather than the enzyme-substrate complex. We also looked at the -amylase inhibitory potential of C1 and C2, which revealed lesser inhibition than the conventional medicine acarbose, which is consistent with prior findings that plant-derived inhibitors are weak α-amylase inhibitors but robust α-glucosidase inhibitors.
 | Figure 3: After 3 weeks of incubation, the effects of ethanol extract of pseudostem (EE), Stigmasterol (C1), and β-Sitosterol (C2) on Fructosamine content (a), Protein carbonyl content (b), and Thiols group content (c) in the BSA/Fructose system. [Click here to view] |
 | Table 3: After 28 days of treatment, the effect of EE, C1 and C2 on the serum lipid profile of experimental normal and diabetic rats. [Click here to view] |
 | Table 4: After 28 days of treatment, the effect of EE, C1 and C2 on the liver lipid profile of experimental normal and diabetic rats. [Click here to view] |
 | Table 5: After 28 days of treatment, the effect of EE, C1 and C2 on lipid peroxidation and non.enzymatic/enzymatic antioxidants levels in the serum of experimental normal and diabetic rats. [Click here to view] |
 | Table 6: After 28 days of treatment, the effect of EE, C1 and C2 on lipid peroxidation and non.enzymatic/enzymatic antioxidants levels in the liver of experimental normal and diabetic rats. [Click here to view] |
 | Table 7: Antioxidant activities of the previously reported phytocompounds from banana plant. [Click here to view] |
 | Table 8: Antidiabetic enzyme inhibition activities of the previously reported phytocompounds from banana plant [Click here to view] |
The subsequent target of therapy in hyperglycemic state is to prohibit the excessive glucose present in circulation from entering various pathways that lead to the vascular complications. A significant amount of glucose enters an intermediary pathway known as polyol pathway that converts glucose to sorbitol [38]. Despite the exact mechanism not being known, the excessive amount of sorbitol accumulation is linked to cataract development and in this regard, inhibition of AR, the key enzyme of this pathway is proven to reverse the effect [7,8]. Thus, in the present study, the effect of C1 and C2 was tested on the activity of AR in comparison to the standard phenolic drug quercetin. The results exhibited better inhibition of the enzyme by C1 and C2 as against quercetin thus ascertaining their use in diabetic management.
Another phenomenon that co-exists with hyperglycemia is the free radical mediated oxidative stress which also augments to the vascular damage leading the complications reported during diabetes [39]. Hence, EE and the compounds C1 and C2 were tested for their antioxidant ability by employing radical scavenger’s viz., DPPH, ABTS and superoxide. Another important marker of oxidative stress is lipid peroxides and hence, the ability of EE and compounds were tested for their inhibition using egg yolk homogenate in liposome model [14]. In contrast to BHA, C1 and C2 showed better antioxidant activities with C1 faring better than C2 without differing significantly. It was also witnessed that the pure compounds fared better than EE suggesting them to be more potent than the extract itself. Further, a momentous inhibition was seen by EE and compounds against the LPO suggesting its protective role during myocardial infractions. Despite the varying principles of antioxidant roles in the above study, an overall reducing potential of the extract needs to be elucidated and for this purpose, reduction of ferric chloride to potassium ferricyanide was estimated. There was a dose-dependent increase in the absorbance at 700 nm suggesting a proportional increase in the reducing power with respect to its concentration. Perhaps, free radicals generated during physicochemical reactions are not the only contributors of oxidative stress. Some of the metal ions present in circulation like the iron can undergo reduction to form reactive hydroxyl radicals augmenting to oxidative stress [40]. To assess the protective role of EE and compounds on this phenomenon, their metal chelating ability for Fe2+ was carried out. Apparently, in comparison to a standard BHA, EE exhibited a 1.5 fold while the compounds showed a 2-fold increase in the metal chelating ability proposing their interference in the formation of the highly reactive hydroxyl radicals.
Excess glucose present in circulation also leads to glycation of various proteins thereby inactivating them. More recently, fructose has been identified in glycation directly or by activating the polyol pathway to increase the levels of fructose formation from glucose. According to studies, fructose participates in glycation at a faster rate than glucose, implying greater damage [41]. The nucleophilic addition reaction between the free amino groups of proteins and the carbonyl group of the reducing sugar initiates the creation of a Schiff’s base, which results in irreversible Amadori products such fructosamine. Fructosamines in Amadori products are transformed to glyoxal, methylglyoxal, and deoxyglucosones in the second step of glycation. Thus, generated carbonyl compounds form carbonyl proteins leading to loss of protein thiols which constitute promising protein oxidation indicators [42]. As the glycation process advances by formation of insoluble fluorescent products frequently referred to as AGE, they combine with glycated proteins and accumulate in the cells. Within the cells, they impede normal protein functioning while in the extracellular matrix, they lead to irregular cross-linking of the matrix thus hindering their normal function. Apart from vascular complications that AGEs bring about, they also lead to generation of ROS which is associated to most of the complications observed in diabetes [43].
With this under concern, we aimed to examine if EE, C1, and C2 could prevent each stage of glycation and the production of AGE. The first stage of protein glycation was created in vitro by exposing BSA to high fructose levels which was expected to glycate BSA. As anticipated, there was elevated fructosamine levels as against the non-glycated BSA levels and treatment of EE, C1 and C2, significantly reduced this and proved better than the standard inhibitor aminoguanidine. Further, the elevated protein carbonyl groups that were produced after exposure of BSA to high fructose load and inversely low levels of protein thiols presented the second stage of protein glycation. This was ameliorated by treatment of EE, and compounds to about 80–90% signifying its protective role. Finally, the AGE formation was evaluated by the ability of the compounds to form fluorescent products and the results suggested a marked decrease in the formation of AGEs. Hence the potential role of EE and the compounds in preventing each stage of protein glycation in turn suggesting their promising use to control various complications associated with it is asserted.
The positive aspects of EE and its compounds (C1 and C2) as proposed by the in vitro studies were further assessed by in vivo studies, by feeding the purified compounds to alloxan-induced diabetic rats. It is well known that alloxan is preferentially toxic to beta cells as well as lead to ROS formation [44]. High plasma glucose levels, together with ROS increase the risks for cardiovascular disorders (CVDs) in diabetic patients. In a preliminary study, EE and compounds were evaluated for their in vivo antihyperglycemic properties and hence, in the present study we assessed its protective role in the development of macrovascular complications. Since hyperlipidemia is the major and direct cause for CVDs, EE as well as the compounds were tested for their antihyperlipidemic properties. Elevated plasma levels of TG, TC and imbalanced HDL: LDL ratio were characteristic to diabetic patients [45]. Post administration of EE and compounds, a significant improvement in the blood lipid profile was evident in alloxan-induced diabetic rats in comparison to diabetic control rats.
ROS by far have been the major contributors for most of the complications that exists during diabetes because of the hyper-physiological challenges posed by the free-radicals [46] and hence EE and compounds were also tested for their ability to induce the expression of antioxidant enzymes. In this regard, we evaluated the serum and liver antioxidant enzyme levels which were identified to be lowered during diabetes. The levels of antioxidant enzymes viz., SOD, GPx and CAT as well as non-enzymatic antioxidants viz., GSH, alpha-tocopherol and ascorbic acid tested after administration of EE and the compounds, exhibited a momentous improvement as purported by the in vitro studies.
5. CONCLUSION
The outcomes of this study suggest the tremendous potential of EE, C1, and C2 in ameliorating the damages induced by diabetic condition thereby advocating their use in antidiabetic therapy. Despite continued progress in understanding the phytochemical and pharmacological potential of Musa spp. biomasses such as fruit peels (for example), more research is needed to produce a phytochemical therapeutic agent from Musa spp. biomasses. In both preclinical and clinical studies, relevant elements of Musa spp. extract use will be addressed, with a special focus on overall bioavailability. Finally, given Musa spp.’s diversity and tolerance for a varied climatic condition, ethnopharmacological and rational oriented research should provide sufficient justification for Musa spp. secondary metabolites’ therapeutic use in modern medicine.
6. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
7. FUNDING
Nil.
8. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
9. ETHICAL APPROVALS
The experiments were approved by Institutional Animal Ethics Committee (JSSCP/IAEC/Ph.D./PH.COLOGY/01/2013-14) and conducted as per the guidelines of CPCSEA, Chennai, India.
10. DATA AVAILABILITY
Data sharing is not applicable to this article as no new data were created or analyzed in this study.
11. ACKNOWLEDGMENT
Ramith Ramu thanks TEQIP, New Delhi, India for awarding the Research fellowship. All the authors thank JSS Academy of Higher Education and Research, Mysuru-570015, Karnataka, India for their constant support and encouragement
12. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Atlas D. International diabetes federation. In: IDF Diabetes Atlas. 7th ed. Brussels, Belgium: International Diabetes Federation; 2015.
2. Maiti R, Jana D, Das UK, Ghosh D. Antidiabetic effect of aqueous extract of seed of Tamarindus indica in streptozotocin-induced diabetic rats. J Ethnopharmacol 2004;92:85-91. CrossRef
3. Fowler MJ. Microvascular and macrovascular complications of diabetes. Clin Diabetes 2008;26:77-82. CrossRef
4. Patil SM, Shirahatti PS, Ramu R. Azadirachta indica A. Juss (neem) against diabetes mellitus: A critical review on its phytochemistry, pharmacology, and toxicology. J Pharm Pharmacol 2021;40:rgab098. CrossRef
5. Ramu R. Investigation of Banana (Musa sp. Var. Nanjangud Rasabale) Flower and Pseudostem in the Management of Hyperglycemia, Doctoral Thesis. Belagavi, India: Visvesvaraya Technological University; 2017.
6. Ramu R, Shirahatti PS, Zameer F, Prasad MN. Investigation of antihyperglycaemic activity of banana (Musa sp. var. Nanjangud rasa bale) pseudostem in normal and diabetic rats. J Sci Food Agric 2015;95:165-73. CrossRef
7. Jaiswal N, Srivastava SP, Bhatia V, Mishra A, Sonkar AK, Narender T, et al. Inhibition of alpha-glucosidase by Acacia nilotica prevents hyperglycemia along with improvement of diabetic complications via aldose reductase inhibition. J Diabetes Metab 2012;6:1-7. CrossRef
8. Ramu R, Shirahatti PS, Zameer F, Ranganatha LV, Prasad MN. Inhibitory effect of banana (Musa sp. var. Nanjangud rasa bale) flower extract and its constituents umbelliferone and lupeol on α-glucosidase, aldose reductase and glycation at multiple stages. South Afr J Bot 2014;95:54-63. CrossRef
9. Kusunoki J, Aragane K, Kitamine T, Kozono H, Kano K, Fujinami K, et al. Postprandial hyperlipidemia in streptozotocin-induced diabetic rats is due to abnormal increase in intestinal acyl coenzyme A: Cholesterol acyltransferase activity. Arterioscler Thromb Vasc Biol 2000;20:171-8. CrossRef
10. Joshi S. Eugenia jambolana, Musa paradisiaca L. Med Plants 2000;286:294.
11. Ramu R, Shirahatti PS, Nayakavadi S, Vadivelan R, Zameer F, Dhananjaya BL, et al. The effect of a plant extract enriched in stigmasterol and β-sitosterol on glycaemic status and glucose metabolism in alloxan-induced diabetic rats. Food Funct 2016;7:3999-4011. CrossRef
12. Patil SM, Martiz RM, Ramu R, Shirahatti PS, Prakash A, Kumar BP, et al. Evaluation of flavonoids from banana pseudostem and flower (quercetin and catechin) as potent inhibitors of α-glucosidase: An in silico perspective. J Biomol Struct Dyn 2021;40:1-5. CrossRef
13. Razali N, Razab R, Junit SM, Aziz AA. Radical scavenging and reducing properties of extracts of cashew shoots (Anacardium occidentale). Food Chem 2008;111:38-44. CrossRef
14. Meghashri S, Kumar HV, Gopal S. Antioxidant properties of a novel flavonoid from leaves of Leucas aspera. Food Chem 2010;122:105-10. CrossRef
15. Yen GC, Duh PD, Tsai HL. Antioxidant and pro-oxidant properties of ascorbic acid and gallic acid. Food Chem 2002;79:307-13. CrossRef
16. Lineweaver H, Burk D. The determination of enzyme dissociation constants. J Am Chem Soc 1934;56:658-66. CrossRef
17. Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972;18:499-502. CrossRef
18. Marklund S, Marklund G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 1974;47:469-74. CrossRef
19. Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra W. Selenium: Biochemical role as a component of glutathione peroxidase. Science 1973;179:588-90. CrossRef
20. Sinha AK. Colorimetric assay of catalase. Analytbiochem 1972;47:389-94. CrossRef
21. Ellman GL. Tissue sulfhydryl groups. Arch Biochem Biophys 1959;82:70-7. CrossRef
22. Baker H, Frank O, DeAngelis B, Feingold S. Plasma tocopherol in man at various times after ingesting free or acetylated tocopherol. Nutr Rep Int 1980;21:531-6.
23. Omaye ST, Turnbull JD, Sauberlich HE. Selected methods for the determination of ascorbic acid in animal cells, tissues, and fluids. Meth Enzymol 1979;62:3-11. CrossRef
24. Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 1979;95:351-8. CrossRef
25. Brealey D, Singer M. Hyperglycemia in critical illness: A review. J Diabetes Sci Technol 2009;3:1250-60. CrossRef
26. Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001;414:813-20. CrossRef
27. Negre-Salvayre A, Salvayre R, Auge N, Pamplona R, Portero-Otin M. Hyperglycemia and glycation in diabetic complications. Antioxid Redox Signal 2009;11:3071-109. CrossRef
28. Ceriello A. Postprandial hyperglycemia and diabetes complications: Is it time to treat. Diabetes 2005;54:1-7. CrossRef
29. Sheetz MJ, King GL. Molecular understanding of hyperglycemia’s adverse effects for diabetic complications. JAMA 2002;288:2579-88. CrossRef
30. Madar Z. The effect of acarbose and miglitol (BAY-M-1099) on postprandial glucose levels following ingestion of various sources of starch by nondiabetic and streptozotocin-induced diabetic rats. J Nutr 1989;119:2023-9. CrossRef
31. Pereira A, Maraschin M. Banana (Musa spp) from peel to pulp: Ethnopharmacology, source of bioactive compounds and its relevance for human health. J Ethnopharmacol 2015;160:149-63. CrossRef
32. Sheng Z, Dai H, Pan S, Wang H, Hu Y, Ma W. Isolation and characterization of an α-glucosidase inhibitor from Musa spp.(Baxijiao) flowers. Molecules 2014;19:10563-73. CrossRef
33. Nguyen D, Zachariasova A, Spurna K, Hricko J, Phung H, Viktorova J, et al. Antidiabetic compounds in stem juice from banana. Czech J Food Sci 2017;35:407-13. CrossRef
34. Chiang SH, Yang KM, Lai YC, Chen CW. Evaluation of the in vitro biological activities of Banana flower and bract extracts and their bioactive compounds. Int J Food Prop 2021;24:1-6. CrossRef
35. Veeresham C, Rama Rao A, Asres K. Aldose reductase inhibitors of plant origin. Phytother Res 2014;28:317-33. CrossRef
36. Patil SM, Shirahatti PS, Ramu R, Prasad N. Azadirachta indica A. Juss (neem) as a contraceptive: An evidence-based review on its pharmacological efficiency. Phytomedicine 2021;88:153596. CrossRef
37. Patil SM, Ramu R, Shirahatti PS, Shivamallu C, Amachawadi RG. A systematic review on ethnopharmacology, phytochemistry and pharmacological aspects of Thymus vulgaris Linn. Heliyon 2021;7:e07054. CrossRef
38. Frank RN. The aldose reductase controversy. Diabetes 1994;43:169-72. CrossRef
39. Giacco F, Brownlee M. Oxidative stress and diabetic complications. Circ Res 2010;107:1058-70. CrossRef
40. Uttara B, Singh AV, Zamboni P, Mahajan RT. Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant therapeutic options. Curr Neuropharm 2009;7:65-74. CrossRef
41. Tupe RS, Sankhe NM, Shaikh SA, Phatak DV, Parikh JU, Khaire AA, et al. Aqueous extract of some indigenous medicinal plants inhibits glycation at multiple stages and protects erythrocytes from oxidative damage-an in vitro study. J Food Sci Technol 2015;52:1911-23. CrossRef
42. Goh SY, Cooper ME. The role of advanced glycation end products in progression and complications of diabetes. J Clin Endocrinol Metab 2008;93:1143-52. CrossRef
43. Ahmed N. Advanced glycation endproducts-role in pathology of diabetic complications. Diabetes Res Clin Pract 2005;67:3-21. CrossRef
44. Rohilla A, Ali S. Alloxan induced diabetes: Mechanisms and effects. Int J Res Pharm Biomed Sci 2012;3:819-23.
45. Ramachandran V, Saravanan R, Senthilraja P. Antidiabetic and antihyperlipidemic activity of asiatic acid in diabetic rats, role of HMG CoA: In vivo and in silico approaches. Phytomedicine 2014;21:225-32. CrossRef
46. Kavishankar GB, Moree SS, Lakshmidevi N. Hepatoprotective and antioxidant activity of N-trisaccharide in different experimental rats. Phytomedicine 2014;21:1026-31. CrossRef