1. INTRODUCTION
Pesticide residues have been detected in many ecosystems of the environment, generating serious concerns about their uncontrolled use, which has outweighed the benefits gained [1,2] The scientific community is concerned about the possibility of pesticide management having a negative influence on numerous natural environment components [3,4]. Pesticides have been used in India alone in excess of 100,000 tons, mostly for agricultural pest control, because of their low cost and broad-spectrum toxicity [5–8]. Agricultural fertilizers and pesticides are applied worldwide in excess of 140 billion kilos per year, creating a major source of pollution through agricultural runoff [6]. Agricultural pollution refers to the biotic and abiotic waste products of agriculture that pollute, degrade, and/or harm humans, their economic interests, as well as the environment and ecosystems surrounding them [9,10]. Agrochemicals have the potencies to pollute food and water, putting human health at risk [11,12]. Due to their increasing toxicity, persistence, and potencies to accumulate in organisms, the use of such agrochemicals poses a significant risk to human health and has become a serious issue for the aquatic environment [13].
Human health has been posed with a huge risk when it comes to pesticides and their usage. However, initially, pesticides were synthesized to control pest population, but their usage has led to posed prospective risks to human health and nontarget environmental species [14–18]. Traditional toxicity testing highly depends on in-vivo single constituent studies, which have been thoroughly investigated at all levels of the system, including producer and consumer levels. However, in-vivo testing is time-consuming and expensive, and it necessitates a lot of upkeep and a large number of animals, which raises ethical concerns [19]. Thus, for economic, practical, and ethical reasons, in-vitro techniques have risen tremendously, and the use of cell lines as an alternative to in-vivo testing is being seriously examined [14,20,21].
In-vitro analysis of fish cells is gaining a promising alternative for mechanistic evaluation for toxicological assessment, with the potential to replace or reduce the usage of fish as a whole organism [22], which is also supported by the idea where maintenance of ?sh cells in culture conditions is easy and cost-effective. There have been a lot of studies carried out on hazardous substances to correlate the toxicity of xenobiotic in in-vitro and in-vivo experimentation, which has resulted in the usefulness of mitigating the usage of it [23,24]. Research is now intended toward assessing the toxicity of agrochemicals on different cell lines derived from fish organs. For instance, several cell lines have been developed from India, such as the Indian Catla catla Heart cell line from the heart of Catla catla; RE and Indian Catla catla Brain Cell line from the eye of L. rohita and brain of C. catla, respectively [25]; rohita eye cell line, rohita Fin cell line, and Cell line from L. rohita swim bladder from fin, heart and swim bladder of L. rohita, respectively [26]; from the fin tissue of Tor tor [27]; two cell lines from the fin and eye tissues of Tor chelynoides [28,29]; and heart and gill cell lines from C. catla [30,31].
Previous in-vivo studies have well established the toxic potential of all the classes of agrochemicals, viz. imidacloprid (IMI), curzate (CZ), micronutrients (MN), and pyrazosulfuron-ethyl (PE), which elucidated the alteration in hematological, histological, biochemical parameters, behavior, and neuroendocrine response as well [4,12,23,24,32–35]. However, there is a gap in our understanding with regards to the molecular mechanism. Thus to understand the mechanism of action, the present study was undertaken to unravel the alteration in cell cycle on exposure to agrochemicals (PE, CZ, MN and IMI) in fish cells Indian Catla catla gill cell line (ICG). More precisely, the loss of normal cell orchestration and cell proliferation was addressed by studying the cell cycle regulation and key proliferation markers.
Proliferating cell nuclear antigen (PCNA) is a vital component in replication in which it acts as a progression factor and DNA clamp for DNA polymerase δ, and additionally, it also plays a pivotal role in DNA repair, chromatin remodeling, and epigenetics. It is considered as a universal marker for cell proliferation [36,37]. The control of DNA replication is a key element in the proper functioning of a cell, and it influences genome stability [38]. Duplication of the genetic material that occurs in S phase of the cell cycle has to be coordinated with other cellular processes like mitosis. DNA replication is regulated mainly at the initiation step as a result of cooperation between different signaling pathways controlling the cell cycle [39,40].
In addition to PCNA, cyclin and cyclin-dependent kinases (CDKs) are yet other universal markers which are known to control cell cycle transitions. Several classes of cyclins have been described, of which cyclin E binds to G1 phase Cdk2, which is required for the transition from G1 to S phase of the cell cycle that determines initiation of DNA duplication [40]. During the S phase of the cell cycle, cyclin A is found in the nucleus and is involved in the initiation and completion of DNA replication [39,41]. Quantification of proliferative markers (PCNA and cyclin genes) can thus be crucial in understanding its role of xenobiotics in the cell cycle. The present inventory aims to understand the alterations in the expressions of the proliferative markers in fish cell line-ICG due to the exposure of different classes of agrochemicals (IMI, CZ, MN, and PE). The selection of the agrochemicals was based on the routine usage in the agricultural field and its in-vivo assessment [4,12,23,24]. Moreover, the gill cell line was taken as it is the first organ of the fish that is acquainted with any toxicant in the natural habitat.
2. MATERIALS AND METHODS
2.1. Chemicals
Agrochemicals insecticide IMI (TATAMIDA), fungicide CZ (DuPontTM Curzate M8), herbicide PE (Saathi, UPL), and MN (LibrelTM , Ciba) were purchased from the local vendors and they were dissolved (individually) in water for the further experimentation.
2.2. Culturing of ICG Cells
The ICG gill cell line of C. catla was procured from the National Repository of Fish Cell Line (NRFC), Indian Council of Agricultural Research, National Bureau of Fish Genetic Resources (ICAR-NBFGR), Lucknow. The cell line was cultured in Leibovitz’s L-15 (AL0011A, HiMedia, India) supplemented with 10% FBS (RM9955, HiMedia, India) [11]. The flasks were incubated at 28°C in a biological incubator (LabTech) and the medium was changed every fourth day. Upon reaching 80%–85% confluence, the cells were subcultured in the ratio of 1:2 by using trypsin–EDTA solution (TC007, HiMedia, India).
2.3. MTT Assay
ICG cells were seeded in the density of 2 × 104 cells per well in 96-well tissue culture plates (TPC96, HiMedia, India) and were incubated overnight at 28°C. The medium was removed after incubation and the cells were treated with a medium containing agrochemicals (CZ, IMI, PE, and MN) for 96 hours. After a 96-hour exposure period, the test medium was replaced by 10 μl of 5 mg/ml 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) (TC191, HiMedia, India) in phosphate buffered saline (PBS) (M1866, pH 7.4, HiMedia, India). After incubation for 4 hours at room temperature in the dark, the solution was removed carefully and dimethyl sulfoxide (6644, SRL, India) was added per well to solubilize the purple formazan crystals produced. The absorbance of each well was measured at 570 nm and cell inhibitions were obtained using the following formula:
After obtaining the inhibition concentration (IC50), sublethal (1/20th, 1/10th, and 1/5th doses of IC50) concentrations were selected for further subacute studies as low dose (LD), medium dose (MD), and high dose (HD), respectively. Moreover, the ICG cells were exposed to all agrochemicals, i.e., IMI, CZ, MN, and PE, for 7 days and toxicity was analyzed with respect to untreated control (n = 3).
2.4. Cell Viability Assay
Trypan blue assay was used to understand the effect of agrochemicals on the viability of ICG cells. The cells were seeded at a density of 1 × 105 cells/ ml in a complete L-15 medium. Following 24 hrs of cell growth, different concentrations of agrochemicals (LD, MD, and HD) were added to the cells. After 7 days, cells were trypsinized, washed, and resuspended in PBS containing 0.4% trypan blue (TCL046, HiMedia, India). The number of viable cells was counted using hemocytometer (GW088, HiMedia, India) as per the standard protocol. Each experiment was carried out with three replicates (n = 3) for each group for statistical analysis.
2.5. Cell Morphology Analysis
Cells were plated into a 6-well culture plate (9.5 cm2, TPC6, HiMedia, India) at a density of 2 × 105 cells (in 2 ml complete medium). After overnight growth, the supernatants from the culture plates were aspirated and fresh aliquots of growth medium containing various concentrations (LD, MD, and HD) of agrochemicals were added. Upon incubation for 7 days, cells were washed with PBS (M1866, HiMedia, India, pH 7.4) and morphological changes were observed under an inverted phase-contrast microscope at 100× magnification.
2.6. Total RNA Isolation and cDNA Synthesis
Total RNA was extracted from ICG cells from control and treated cells for all agrochemicals using TRIzol reagent (15596-026, Invitrogen, USA) with standard protocol. The pellet was resuspended by adding 40 μl of diethyl pyrocarbonate in water (DBOS009, SRL, India), which was quantified spectrophotometrically using NanodropC and was stored at −20°C. The cDNA was synthesized from each sample using the standard kit protocol of Thermo Scientific Verso cDNA Synthesis Kit (AB-1453/A), for which 1 μg RNA was used as a template per reaction for single-strand cDNA synthesis using oligo dT primers.
2.7. Quantitative PCR Amplification
Quantitative RT-polymerase chain reaction (PCR) was carried out using the method where PowerUp SYBR Green Master Mix (A25741, Applied Biosystems, USA) was used and the amplification was carried out in Quant Studio 12K (Life technology) FAST real-time PCR machine with primers of PCNA, cyclin A, and cyclin E (Table 1). The melting curve of each sample was measured to ensure the specificity of the products. glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as an internal control to normalize the variability in the expression levels and data were analyzed using the 2??CT method [42].
2.8. Statistical Analysis
Experiments were carried out in triplicate (n = 3) for each exposure concentration. Data were analyzed with GraphPad Prism 9 (GraphPad Software) and one-way analysis of variance (p ≤ 0.05) was carried out. The post-hoc test was carried out by Dunnett’s multiple comparison test to further understand the level of significance (p ≤ 0.05; p ≤ 0.01).
3. RESULTS
Table 2 and Figures 1–4 show the IC50 values and sublethal concentrations of different classes of agrochemicals. IMI was determined to be the most harmful of all the agrochemicals, followed by CZ and MN, with PE being the least toxic. ICG cells were treated with sublethal concentrations [LD (1/20th), MD (1/10th), and HD (1/5th)] of all agrochemicals (IMI, CZ, MN, and PE) for 7 days. Cell viability assay carried out by Trypan blue (Table 3) showed that cell proliferation was significantly (p < 0.05) affected upon treatment with agrochemicals in a dose-dependent manner. At all exposures, cell viability was found to be highest in PE and MN, low in CZ, and lowest in IMI. Among all the groups, HD of IMI, CZ, MN, and PE showed a significant decrease in viability compared to control.
 | Table 1: Real-time PCR primer sequences. [Click here to view] |
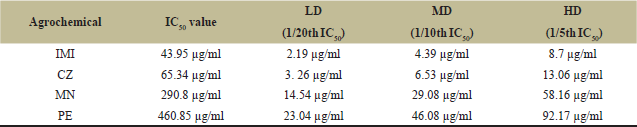 | Table 2: IC50 values and their sublethal doses for IMI, CZ, MN, and PE for ICG cell line. [Click here to view] |
 | Figure 1: ICG cell mortality against different concentrations of IMI. [Click here to view] |
 | Figure 2: ICG cell mortality against different concentrations of CZ. [Click here to view] |
 | Figure 3: ICG cell mortality against different concentrations of MN. [Click here to view] |
Morphological alterations were also observed, such as loss of integrity of membrane, membrane blebbing, detachment of cells, and formation of apoptotic bodies compared to the control cells which showed healthy cell morphology. Dose-dependent morphological changes were observed in cells exposed to agrochemicals, where MN- and PE-treated cells exhibited fewer alterations, whereas IMI- and CZ-treated cells exhibited the highest alterations in comparison to control. The observed alterations in morphology of ICG cells are shown in Figure 5.
 | Figure 4: ICG cell mortality against different concentrations of PE. [Click here to view] |
Subacute exposure of agrochemicals for 7 days resulted in differential expressions of the proliferative markers. Expression of the proliferative marker genes, such as PCNA and cyclin A, showed different expressions. A significant dose-dependent decrease (p < 0.01) was seen in PCNA expression (Fig. 6) in all the treated groups for all the doses compared to control, while cyclin A was found to be significantly decreasing only at MD and HD of IMI (p < 0.05; p < 0.01), CZ (p < 0.01), and MN (p < 0.01) exposure compared to the control. PE exposure resulted in a significant (p < 0.01) decrease only at HD (Fig. 7). Cyclin E expression resulted in a dose-dependent significant (p < 0.01) decrease in exposure to IMI, CZ, and PE. However, MN exposure was found to be significantly decreased (p < 0.01) only at HD compared to the control (Fig. 8).
4. DISCUSSION
Under the Green Revolution, agrochemicals and chemical fertilizers were widely employed to protect crops from pests and increase yield, resulting in increased productivity and economic benefit of agricultural output to satisfy the rising demand for food due to the fast-growing population [43]. Runoff and groundwater leaching from a range of chemicals used in agricultural activities have a significant potential of contaminating aquatic habitats that flow through the agricultural regions. Fish is the most economically important nontarget species that is adversely affected by severe agrochemical pollution [43–45].
To evaluate the toxic potential of agrochemicals many scientists have worked on the toxic effect on fish in in-vivo and in-vitro systems. ICG cells have been found to be good candidates for assessing in-vitro acute cytotoxicity of hazardous compounds and heavy metals [29]. We employed ICG cells to assess the in-vitro toxicity of agrochemicals such as IMI, CZ, MN, and PE. The half-maximal inhibitory concentration (IC50) is a measure of the potency of a chemical in inhibiting a specific biological or biochemical function [45–47].
 | Figure 5: Alterations in the cell morphology of ICG cells exposured to agrochemicals. [Click here to view] |
 | Figure 6: The level of PCNA (in folds) in ICG cells treated with sublethal doses of IMI, CZ, MN, and PE. Each value represents the mean ± SEM (n = 3). The significant level is indicated by * p < 0.05 and ** p < 0.01. [Click here to view] |
The effect of agrochemicals on the ICG cells of C. catla was assessed by the uptake of MTT and its following reduction in the mitochondria of living cells to MTT formazan [48]. This is the first time in-vitro studies are reported in which we found the IC50 of four different agrochemicals in fish gill cells ICG. According to previous studies, IMI proved to be toxic to many nontarget organisms [19,49–51]. Earlier studies have reported that LC50 values have proved that neonicotinoids IMI is the most toxic to the nontarget organisms in in-vivo conditions, followed by CZ, MN, and PE [4,12,23,24,32,35]. Furthermore, in-vitro studies have also suggested that the neonicotinoids are more toxic compared to other agrochemicals. The IC50 of IMI is 0.023 mM, which was reported previously in the prostate epithelial WPM-Y.1 cell line [52]. The IC50 values of neutral red, MTT, and total cell protein were 41.86, 38.46, and 39.08 g/ml, respectively, in an in-vitro study of the pesticide IMI in the gill cell line of Flounder (FG) [53].
Microscopic observation also revealed the presence of many abnormal cells; some cells had lost their normal cell morphology: loss of cell shape and sphericity and increase in cell granularity. Moreover, the cells were seen to get detached, float, and die. The morphological alterations were observed to be in the proportion of concentrations of the agrochemicals. Our studies are in agreement with previous reports on the assessment of cytotoxicity of the organophosphorus pesticide parathion on FG-9307 cells in-vitro system. They concluded that with the increase in the parathion concentration, the degree of damage to the cellular structures was more serious [54]. Moreover, morphological changes were observed in two fish cell lines, RTG-2 cells and PLHC-1 cells, on exposure to sodium fluoroacetate during previous cytotoxic studies [55]. Cytotoxic effects of benzonitrile herbicides using two human cell lines, Hep G2 and HEK293T, were studied where they have reported the alteration in morphology in a dose-dependent and time-dependent manner [56]. Apart from these assessment studies on cytotoxicity of imidazolium in the ovarian fish cell line CCO, the human cell line HeLa also revealed the same results [57]. Our results support the previously reported changes in cell shape, granularity, and alter morphology observed on exposure to toxicants. Of all the agrochemicals, the morphological changes in the IMI and CZ-treated groups were much more significant. The observed toxicity in ICG cells could be ranked in the following decreasing order: IMI > CZ > MN > PE on exposure to agrochemicals.
 | Figure 7: The level of cyclin A (in folds) in ICG cells treated with sublethal doses of IMI, CZ, MN, and PE. Each value represents the mean ± SEM (n = 3). The significant level is indicated by * p < 0.05 and ** p < 0.01. [Click here to view] |
PCNA is a well-conserved protein present in all eukaryotic species, as well as Archaea. PCNA was initially discovered to function as a processivity factor for DNA polymerase, which plays a role in DNA replication [57]. Moreover, PCNA activities are involved with other critical cellular processes such as chromatin remodeling, DNA repair, sister-chromatid cohesion, and cell cycle control [37,57]. Because cells spend more time in the G1 to S phase transition, PCNA expression is considered an indicator of cell proliferation. Furthermore, as part of the DNA replication and repair mechanism, this plays an important function in nucleic acid metabolism [58].
PCNA has been found in a variety of cell types in mammalian tissues, as well as in a variety of fish organs [21]. The effect of Mirex pesticide on the expression of PCNA levels has been reported [59]. It has been stated that organophosphate insecticides cause a substantial decrease in cell proliferation in liver cells [60]. Our results are in agreement with earlier reported studies. There was a significant dose-dependent decrease observed on the exposure of all the agrochemicals, suggesting that the decrease in the PCNA mRNA has probably lead to an impaired repair mechanism leading to a decreased replication process in the S-phase of the cells. Furthermore, the results also indicate that cells may have undergone stress conditions leading them to cell death [61].
The control of cell cycle progression is central to not only maintaining homeostasis but its alteration may also lead to imbalances in proliferation; cell death is governed by cyclins and CDKs. Normal cell proliferation is regulated by checkpoints that are situated at different stages of the cell cycle. Deregulation of these checkpoint events and the chemicals linked to them may cause cell cycle progression to halt. Cyclin D and E govern the transition from G1 to S phase; cyclin A regulates the development from G2 to M phase; and cyclin B regulates the transition from G2 to M phase [39]. By connecting with and activating its catalytic partner Cdk2, cyclin E is required for advancement through the G1 phase of the cell cycle and activation of DNA replication. The targets of cyclin E/Cdk2 phosphorylation are Rb, which is the critical component of cell proliferation, and Cdc6 and nucleophosmin, which are important for DNA replication [62]. The results of the present study show a decrease in the dose-dependent manner in the expression of cyclin A on exposure to IMI, CZ, and MN; however, with reference to PE, the pattern was not the same and a significant decrease was noted only at a high dose.
 | Figure 8: The level of cyclin E (in folds) in ICG cells treated with sublethal doses of IMI, CZ, MN, and PE. Each value represents the mean ± SEM (n = 3). The significant level is indicated by * p < 0.05 and ** p < 0.01. [Click here to view] |
 | Table 3: Cell viability at sublethal doses for IMI, CZ, MN, and PE for ICG cell line. [Click here to view] |
There was a dose-dependent significant reduction observed in cyclin e expression in cells exposed to IMI, CZ, and PE, whereas cells exposed to MN showed decreased expression in HD only. A decrease in cyclin A and E is suggestive of a decrease in the transition from G1 to S phase and an arrest happening at S phase through which the cell cycle regulation is getting hampered. Most likely, pesticide exposure changed this process by preventing cell cycle progression from G1 to DNA synthetic S phase, where certain endogenous anti-mitogenic signals might have been working through CDK inhibitors to decrease the cyclin–CDK complex activity and impede G1/S transition [63–65].
5. CONCLUSION
According to the results of the study on alterations in proliferation in ICG cells exposed to pesticides, IMI is the most toxic of all the agrochemicals studied, followed by CZ, MN, and PE. The study also suggests that dose-dependent morphological alterations were observed in ICG cells exposed to all agrochemicals compared to the control which showed healthy cell morphology. There was a significant decrease in proliferation markers like PCNA and cyclin genes in ICG cells when exposed to all agrochemicals. The study suggests that agrochemicals possess multimodal actions, i.e., it does not alter a single gene, instead works on multiple pathways.
6. ACKNOWLEDGMENT
The authors are thankful to the NRFC, ICAR-NBFGR, Lucknow, for providing the fish cell line. They are also thankful to the head of the Department of Zoology, The M.S. University of Baroda, Vadodara, for giving facilities to carry out effective work.
7. AUTHORS’ CONTRIBUTION
All authors contributed significantly to the conception, design, data analysis, and interpretation; participated in the drafting of the manuscript and critically revised it for its content; and have approved the final draft submitted for publication to the current journal. All the writers are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
8. CONFLICTS OF INTEREST
The authors report no conflicts of interest in this work.
9. FUNDING
The authors received no direct funding for this research.
10. ETHICAL APPROVAL
This study does not involve experiments on animals or human subjects.
PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Ali U, Syed JH, Malik RN, Katsoyiannis A, Li J, Zhang G, et al. Organochlorine pesticides (OCPs) in South Asian region: a review. Sci Total Environ 2014;476–7; http://doi.org/10.1016/j.scitotenv.2013.12.107
2. Anbarkeh FR, Nikravesh MR, Jalali M, Sadeghnia HR, Sargazi Z. The effect of diazinon on cell proliferation and apoptosis in testicular tissue of rats and the protective effect of vitamin E. Int J Fertil Steril 2019;13(2):154–60; http://doi.org/10.22074/ijfs.2019.5612
3. Ansoar-Rodríguez Y, Christofoletti CA, Marcato AC, Correia JE, Bueno OC, Malaspina O, et al. Genotoxic potential of the insecticide imidacloprid in a non-target organism (Oreochromis niloticus–Pisces). J Environ Prot 2019;6:1360–7.
4. Pandya P, Parikh P, Ambegaonkar A. Evaluating the toxic potential of agrochemicals on the hypothalamic-pituitary-thyroid axis in tilapia (Oreochromis mossambicus). J Appl Ichthyol 2020;36(2):203–11.
5. Alewu B, Nosiri C. Pesticides and human health. In: Stoytcheva M (ed.). Pesticides in the modern world—effects of pesticides exposure. InTech, Rijeka, Croatia pp 231–50, 2011.
6. Arora VK, Nath JC, Singh CB. Analyzing potato response to irrigation and nitrogen regimes in a sub-tropical environment using SUBSTOR-Potato model. Agricult Water Manage 2013;124:69–76.
7. Nicolopoulou-Stamati P, Maipas S, Kotampasi C, Stamatis P, Hens L. Chemical pesticides and human health: the urgent need for a new concept in agriculture. Front Public Health 2016;1–8; http://doi.org/10.3389/fpubh.2016.00148
8. Caron-Beaudoin É, Viau R, Sanderson JT. Effects of neonicotinoid pesticides on promoter-specific aromatase (CYP19) expression in Hs578t breast cancer cells and the role of the VEGF pathway. Environ Health Perspect 2018;126(4):47014; http://doi.org/10.1289/EHP2698
9. Saha NC, Giri SK, Chatterjee N, Biswas SJ, Bej S. Evaluation of acute toxicity of dichlorvos (Nuvan) to freshwater fish, Oreochromis mossambicus (W. K. H. Peters, 1852) and their ethological changes. Int Educ Sci Res J 2016;2(7):47–9.
10. Sarkar C, Bej S, Saha NC. A study on the acute toxicity of Triazophos to Branchiura sowerbyi (Beddard, 1982) and their behavioural changes. Int J Sci Res 2016;5(5):730–2.
11. Taju G, Abdul Majeed S, Nambi KSN, Sahul Hameed AS. Application of fish cell lines for evaluating the chromium induced cytotoxicity, genotoxicity and oxidative stress. Chemosphere 2017;184:1–12; http://doi.org/10.1016/j.chemosphere.2017.05.151
12. Upadhyay A, Pandya P, Parikh P. Acute exposure of pyrazosulfuron ethyl induced haematological and blood biochemical changes in the freshwater teleost fish Oreochromis mossambicus. Int J Adv Res Biol Sci 2014;1(2):179–86.
13. Joseph B, Raj SJ. Impact of pesticide toxicity on selected biomarkers in fishes. Int J Zoo Res 2011;(7):212–22; http://doi.org/10.3923/ijzr.2011.212.222
14. Roux F. Fish cell lines and their potential uses in ecotoxicology: from cytotoxicity studies and mixture assessment to a co-culture model and mechanistic analyses. Department of Biological and Environmental Sciences, University of Gothenburg, Gothenburg, Sweden, 2015.
15. Sarkar C, Bej S, Saha NC. Acute toxicity of triazophos to common carp (Cyprinus Carpio) fry and their behavioural changes. Paripex Indian J Res 2016;5(6):19–21.
16. Saha NC, Giri SK, Chatterjee N, Biswas SJ, Bej S. Acute toxicity of dichlorvos to Branchiura sowerbyi (Beddard, 1982). Glob J Res Analys 2016;5(5):138–9.
17. Saha NC, Giri SK, Chatterjee N, Biswas SJ, Bej S. Acute toxic effects of Mancozeb to fish Oreochromis mossambicus (W. K. H. Peters, 1852) and their behaviour. Int J Adv Res Biol Sci 2016;3(6):40–4.
18. Bej S, Ghosh K, Chatterjee A, Saha NC. Assessment of biochemical, hematological and behavioral biomarkers of Cyprinus carpio on exposure to a type-II pyrethroid insecticide Alpha-cypermethrin. Environ Toxicol Pharmacol 2021;87:103717; http://doi.org/10.1016/j.etap.2021.103717
19. Bej S, Chatterjee HN, Giri L, Mukhopadhyay KS, Ganguly P, Mukherjee S. Modulation of blood profile of Cyprinus carpio exposed to Imidacloprid. Int J Life Sci 2017;5(4):627–30.
20. Kasi Elumalai L. Synergistic effect of seaweed manure and Bacillus sp. on growth and biochemical constituents of Vigna radiata L. J Biofertil Biopest 2012;03(03); http://doi.org/10.4172/2155-6202.1000121
21. Dubey A, Goswami M, Yadav K, Mishra A, Kumar A. Establishment of a novel muscle cell line from Wallago attu for in -vitro study of pesticide toxicity. Gene Cell Tissue 2015;2(1):1–7; http://doi.org/10.17795/gct-25568
22. Bols NC, Dayeh VR. Use of fish cell lines in the toxicology and ecotoxicology of fish. Piscine cell lines in environmental toxicology. Biochem Mol Biol Fishes 2005;6:43–84.
23. Patel B, Pandya P, Parikh P. Effects of agrochemicals on antioxidant enzymes and lipid peroxidation in Oreochromis mossambicus and Labeo rohita. Int J Zoo Appl Biosci 2016;1(3):163–72.
24. Patel B, Upadhyay A, Parikh PH. Histological changes in the tissues of Oreochromis mossambicus and Labeo rohita on exposure to imidacloprid and curzate. Int J Res Appl Nat Soc Sci 2016;4(5):149–60.
25. Ahmed VPI, Chandra V, Sudhakaran R, Kumar SR, Sarathi M, Babu VS, et al. Development and characterization of cell lines derived from rohu, Labeo rohita (Hamilton), and catla, Catla catla (Hamilton). J Fish Dis 2009;32(3):211–8; http://doi.org/10.1111/j.1365-2761.2008.00966.x
26. Lakra WS, Swaminathan TR, Joy KP. Development, characterization, conservation and storage of fish cell lines?: a review. 2011;1–20; http://doi.org/10.1007/s10695-010-9411-x
27. Yadav K, Lakra WS, Sharma J, Goswami M, Singh A. Development and characterization of a cell line TTCF from endangered mahseer Tor tor (Ham.). Fish Physiol Biochem 2011;38:1035–45; http://doi.org/10.1007/s10695-011-9588-7
28. Goswami M, Sharma BS, Tripathi AK, Yadav K, Bahuguna SN, Nagpure NS, et al. Development and characterization of cell culture systems from Puntius Tor chelynoides (McClelland). Gene 2012;500(1):140–7; http://doi.org/10.1016/j.gene.2012.03.016
29. Goswami M, Nagpure NS, Jena JK. Fish cell line repository: an enduring effort for conservation. Curr Sci 2014;107(5):738–9; http://doi.org/10.18520/cs/v107/i5/738-739
30. Taju G, Majeed SA, Nambi KS, Hameed AS. In-vitro assay for the toxicity of silver nanoparticles using heart and gill cell lines of Catla catla and gill cell line of Labeo rohita. Comp Biochem Physiol C Toxicol Pharmacol 2014;161(1):41–52; http://doi.org/10.1016/j.cbpc.2014.01.007
31. Nagpure NS, Mishra AK, Ninawe AS, Rasal A, Dubey A, Kumar A, et al. Molecular and cytogenetic characterization of fish cell lines and its application in aquatic research. Natl Acad Sci Lett 2016;39(1):11–6; http://doi.org/10.1007/s40009-015-0365-5
32. Sadekarpawar S, Desai B, Parikh P. Acute toxicity and behavioural responses of Oreochromis mossmbicus (Peters, 1852) to insecticide, fungicide and plant nutrient. Biohelica 2010;1(2):16–21.
33. Sadekarpawar S, Pandya P, Upadhyay A, Parikh PA comparative assessment of trace metal accumulation in Oreochromis mossambicus and Labeo rohita exposed to plant nutrient librel TM. Int J Curr Adv Res 2015;4(10):441–9.
34. Pandya P, Parikh P. Agrochemicals induced gene expression alterations in O. mossambicus. Int J Adv Res 2016;4(6):830–40; http://doi.org/10.21474/IJAR01
35. Pandya P, Upadhyay A, Thakkar B, Parikh P. Evaluating the toxicological effects of agrochemicals on glucocorticoid receptor and serum cortisol level in Mozambique tilapia. Cogent Biol 2018;4(1):1480338 2017.
36. Kubben FJ, Peeters-Haesevoets A, Engels LG, Baeten CG, Schutte B, Arends JW, et al. Proliferating cell nuclear antigen (PCNA): a new marker to study human colonic cell proliferation. Gut 1994;35(4):530–5; http://doi.org/10.1136/gut.35.4.530
37. Leung AYH, Leung JCK, Chan LYY, Ma ESK, Kwan TTF, Lai KN, et al. Proliferating cell nuclear antigen (PCNA) as a proliferative marker during embryonic and adult zebrafish hematopoiesis. Histochem Cell Biol 2005;124(2):105–11; http://doi.org/10.1007/s00418-005-0003-2
38. Strzalka W, Ziemienowicz A. Proliferating cell nuclear antigen (PCNA): a key factor in DNA replication and cell cycle regulation. Ann Bot 2011;107(7):1127–40; http://doi.org/10.1093/aob/mcq243
39. Ekholm-Reed S, Mendez J, Tedesco D, Zetterberg A, Stillman B, Reed SI. Deregulation of cyclin E in human cells interferes with prereplication complex assembly. J Cell Biol 2004;165:789–800; http://doi.org/10.1083/jcb.200404092
40. Duffy KT, McAleer MF, Davidson WR, Kari L, Kari C, Liu CG, et al. Coordinate control of cell cycle regulatory genes in zebrafish development tested by cyclin D1 knockdown with morpholino phosphorodiamidates and hydroxyprolyl-phosphono peptide nucleic acids. Nucleic Acids Res 2005;33(15):4914–21; http://doi.org/10.1093/nar/gki799
41. Bertoli C, Skotheim JM, De Bruin RA. Control of cell cycle transcription during G1 and S phases. Nat Rev Mol Cell Biol 2013;14(8):518–28.
42. Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 2001;25(4):402–8.
43. Gill RJ, Raine NE. Chronic impairment of bumblebee natural foraging behaviour induced by sublethal pesticide exposure. Funct Ecol 2014;28(6):1459–71; http://doi.org/10.1111/1365-2435.12292
44. Pandey S, Kumar R, Sharma S, Nagpure NS, Srivastava SK, Verma MS. Acute toxicity bioassays of mercuric chloride and malathion on air-breathing fish Channa punctatus (Bloch). Ecotoxicol Environ Saf 2005;61(1):114–20; http://doi.org/10.1016/j.ecoenv.2004.08.004
45. Jacquin L, Petitjean Q, Côte J, Laffaille P, Jean S. Effects of pollution on fish behavior, personality, and cognition: some research perspectives. Front Ecol Evol 2020;8:86; http://doi.org/10.3389/fevo.2020.00086
46. Yilmaz AD, Coban T, Suzen S. Synthesis and antioxidant activity evaluations of melatonin-based analogue indole-hydrazide/hydrazone derivatives. J Enzyme Inhib Med Chem 2012;27(3):428–36; http://doi.org/10.3109/14756366.2011.594048
47. Vellonen KS, Honkakoski P, Urtti A. Substrates and inhibitors of efflux proteins interfere with the MTT assay in cells and may lead to underestimation of drug toxicity. Eur J Pharm Sci 2004;23(2):181–8; http://doi.org/10.1016/j.ejps.2004.07.006
48. Rai Y, Pathak R, Kumari N, Sah DK, Pandey S, Kalra N, et al. Mitochondrial biogenesis and metabolic hyperactivation limits the application of MTT assay in the estimation of radiation induced growth inhibition. Sci Rep 2018;8(1):1–15; http://doi.org/10.1038/s41598-018-19930-w
49. Majeed SA, Nambi KSN, Taju G, Vimal S, Venkatesan C, Hameed ASS. Cytotoxicity, genotoxicity and oxidative stress of malachite green on the kidney and gill cell lines of freshwater air breathing fish Channa striata. Environ Sci Pollut Res 2014;21(23):13539–50; http://doi.org/10.1007/s11356-014-3279-8
50. Dezfuli BS, Giari L, Lui A, Squerzanti S, Castaldelli G, Shinn AP, et al. Proliferative cell nuclear antigen (PCNA) expression in the intestine of Salmo trutta trutta naturally infected with an acanthocephalan. Parasites Vectors 2012;5(1):1; http://doi.org/10.1186/1756-3305-5-198
51. Crayton SM, Wood PB, Brown DJ, Millikin AR, McManus TJ, Simpson TJ, et al. Bioaccumulation of the pesticide imidacloprid in stream organisms and sublethal effects on salamanders. Glob Ecol Conserv 2020;24:e01292; http://doi.org/10.1016/j.gecco.2020.e01292
52. Abdel-Halim KY, Osman SR. Cytotoxicity and oxidative stress responses of imidacloprid and glyphosate in human prostate epithelial WPM-Y.1 cell line. J Toxicol 2020; http://doi.org/10.1155/2020/4364650
53. Su F, Zhang S, Li H, Guo H. In vitro acute cytotoxicity of neonicotinoid insecticide imidacloprid to gill cell line of flounder Paralichthy olivaceus. Chin J Oceanol Limnol 2007;25(2):209–14; http://doi.org/10.1007/s00343-007-0209-3
54. Ilboudo S, Fouche E, Rizzati V, Toé AM, Gamet-Payrastre L, Guissou PI. In vitro impact of five pesticides alone or in combination on human intestinal cell line Caco-2. Toxicol Rep 2014;1:474–89.
55. Zurita JL, Jos Á, Cameán AM, Salguero M, López-Artíguez M, Repetto G. Ecotoxicological evaluation of sodium fluoroacetate on aquatic organisms and investigation of the effects on two fish cell lines. Chemosphere 2007;67(1):1–12; http://doi.org/10.1016/j.chemosphere.2006.10.027
56. Lovecka P, Thimova M, Grznarova P, Lipov J, Knejzlik Z, Stiborova H, et al. Study of cytotoxic effects of benzonitrile pesticides. BioMed Res Int 2015; http://doi.org/10.1155/2015/381264
57. Cvjetko M, Radoševi? K, Tomica A, Slivac I, Vorkapi?-Fura? J, Gaurina Sr?ek V. Cytotoxic effects of imidazolium ionic liquids on fish and human cell lines. Arhiv Za Higijenu Rada i Toksikologiju 2012;63(1):15–20; http://doi.org/10.2478/10004-1254-63-2012-2132
58. Maga G, Hubscher U. Proliferating cell nuclear antigen (PCNA): a dancer with many partners. J Cell Sci 2003;116(15):3051–60; http://doi.org/10.1242/jcs.00653
59. de Oliveira MG, Lauxen I da S, Chaves ACM, Rados PV, Sant’Ana Filho M. Immunohistochemical analysis of the patterns of p53 and PCNA expression in odontogenic cystic lesions. Med Oral Patol Oral Cir Bucal 2008;13(5):E275–80.
60. El-Bayomy AA, Smoak IW, Branch S. Embryotoxicity of the pesticide mirex in vitro. Teratog Carcinog Mutag 2002;22(4):239–49; http://doi.org/10.1002/tcm.10016
61. Hreljac I, Zajc I, Lah T, Filipic M. Effects of model organophosphorous pesticides on DNA damage and proliferation of HepG2 cells. Environ Mol Mutag 2008;49(5):360–7; http://doi.org/10.1002/em.20392
62. Sanden M, Olsvik PA. Intestinal cellular localization of PCNA protein and CYP1A mRNA in Atlantic salmon Salmo salar L. exposed to a model toxicant. BMC Physiol 2009;9(1):1–11; http://doi.org/10.1186/1472-6793-9-3
63. Mazumder S, DuPree EL, Almasan A. A dual role of cyclin E in cell proliferation and apoptosis may provide a target for cancer therapy. Curr Cancer Drug Targets 2004;4(1):65–75; http://doi.org/10.2174/1568009043481669
64. Cunningham JJ, Roussel MF. Cyclin-dependent kinase inhibitors in the development of the central nervous system. Cell Growth Differentiation. 2001 12(8):387–96.
65. Burke K, Cheng Y, Li B, Petrov A, Joshi P, Berman RF, et al. Methylmercury elicits rapid inhibition of cell proliferation in the developing brain and decreases cell cycle regulator, cyclin E. NeuroToxicol 2006;27(6):970–81.