1. INTRODUCTION
The butterfly pea (Clitoria ternatea L.), commonly known as the blue pea, is a plant species from the family Fabaceae. It has recently been of interest because of its potential for medical applications and as a source of natural food colorings and antioxidants that can be added to foods to increase the attractiveness of food products. This flower is one of the most interesting sources of polyacylated anthocyanins because it provides blue color in neutral and low acidic solutions [1,2]. However, the very short shelf life limits the commercial use of these flowers. The butterfly pea flower is a natural source of antioxidants with high concentrations of phenolic compounds, flavonoids, and anthocyanins in the extract [3], which play an important role in human health [4].
Extraction procedures include Soxhlet extraction and ultrasonic- and microwave-assisted extraction (MAE) [5]. MAE is one of the advanced extraction processes in the preparation of medicinal plants. This method is suitable for the extraction of flavonoids. The main advantage of MAE is its ability to rapidly heat sample solvent mixtures, leading to its wide applicability for the rapid extraction of analytes, including thermally unstable ones [5,6]. MAE has special advantages such as minimizing solvents and extraction time as well as increasing yield. The ultrasonic-assisted extraction process involves the use of sound energy at very high frequencies greater than 20 kHz to disrupt all plant cells and increase the drug surface area for the solvent to penetrate [5]. Ultrasonic-assisted extraction can be applied to small samples; extraction time was reduced, low amount of solvent used, and yield maximized [7]. The common solvents used in plant extracts are water, alcohol, acetone, and so forth. The effectiveness of solvents was varied in extracting bioactive compounds from plants, depending on the intended use. In this study, water was used as the solvent for the extraction, and the effectiveness of extraction methods was analyzed and compared. Multiple regression analysis was conducted for the most suitable extraction conditions to predict and get the optimal values.
2. MATERIAL AND METHODS
2.1. Preparation of Butterfly Pea Flowers
Butterfly pea flowers were grown at the College of Agriculture, Can Tho University, Vietnam. After harvesting, butterfly pea flowers were washed with running tap water and drained. The sample was arranged on trays (stainless steel) and dried at 70oC in a convectional air oven (SIBATA SD60, Japan) with an airflow speed of 1.0 m/s, parallel to the drying surface of the samples. The dried sample (5%–6% moisture) was packed in dark bags and kept at ambient temperature (28oC ± 2oC) for further use.
This study was conducted from August 2020 to May 2021 in the Department of Food Technology, Can Tho University, Vietnam.
2.2. Extraction Procedure
The ratio of raw materials (butterfly pea flower) to solvent (water) is 1:20 (w/v). The dried material (10 g) was used for each treatment.
2.2.1. Conventional extraction (CE)
Extraction was started by mixing 10 g of dried pea flowers (moisture content of 3%–5%) in 200 ml of water, using a 250 ml Erlenmeyer flask with different extraction temperatures (from 60oC to 80oC) and times (from 15 to 60 minutes). The samples were then centrifuged for 30 minutes (13,000 g, 25oC) (Z232K, Hermle, Gosheim, Germany).
2.2.2. Ultrasound-assisted extraction (UAE)
UAE was conducted in an ultrasonic bath (490 W, 42 kHz, USA). Samples were prepared similarly to the CE (as mentioned above). The process temperature was controlled at 60oC–80oC. Triplicate experiments were carried out.
2.2.3. Microwave-assisted extraction (MAE)
Samples were prepared in the same way as CE (as mentioned above). The extraction process was conducted in a microwave oven (LG MS2595DIS, Korea) with three energy levels (200, 400, and 600 W) for 3–6 minutes. Each experiment was performed three times. The extracts were centrifuged (Z232K, Hermle, Germany) for 30 minutes at 13,000 g at room temperature to obtain the supernatant. The clear liquid was obtained after being passed through the filter paper (Whatman no. 4, UK), which was stored in the refrigerator for further analysis.
2.3. Chemical Analysis
2.3.1. Determination of total anthocyanin content (TAC)
TAC was analyzed using the differential pH method described by Guisti and Wrolstad [8]. The pH-difference method is based on the reaction at pH 1 and 4.5, allowing accurate and rapid measurement of total anthocyanins. Briefly, 1 ml of the extract was reacted with potassium chloride buffer (0.0025 M, pH 1.0) and sodium acetate buffer (0.4 M, pH 4.5). The mixture was incubated for 15 minutes at ambient temperature. The absorbance of each dilution at 510 and 700 nm was measured on a blank cuvette filled with distilled water. The calculation of TAC is based on the equation described by Guisti and Wrolstad [8].
2.3.2. Determination of the ferric reducing antioxidant power (FRAP)
Antioxidant power was determined using the FRAP assay [9]. 0.1 ml of the butterfly pea flower extract was mixed with FRAP reagent in a volume ratio of 1:20. The mixture was incubated for the reaction in the dark at ambient temperature for 30 minutes. Based on the color products (iron tripyridyltriazine complex), its absorbance was measured at 593 nm using a UV/Vis spectrophotometer (Shimadzu, Kyoto, Japan). Calibration curves were established and the results expressed in μMFeSO4/100 ml.
2.4. Data Analysis
2.4.1. Identification of anthocyanin compounds
Anthocyanins and their derivatives were determined using LC-ESI-QQQ (6460 Triple Quadrupole System, Agilent, Santa Clara, CA) in combination with a UV detector (1260 Infinity, Agilent, Santa Clara, CA).
2.4.2. Measured data analysis
Statistically significant differences in the antioxidant properties of the butterfly pea flower extract were determined using software Statgraphics Centurion XV.I (Statistical Graphics Corp., USA). The multiple regression analysis was conducted with TAC and FRAP as the response. The reference equation was selected to fit the data, based on the R2 obtained from the multiple regression analysis. The selected reference equation should have a higher R2.
3. RESULTS AND DISCUSSION
3.1. Comparison of Different Extraction Techniques for Butterfly Pea Flowers
The changes of TAC under different extraction conditions and groups of factors affecting the extraction process are shown in Tables 1 and 2. The extracts were different in anthocyanin content corresponding to different extraction conditions. Currently, many new extraction methods have been introduced, including ultrasonic- and MAE [5,7,10]. These methods can reduce energy consumption and time and maintain high anthocyanin content in the extracts [11].
3.1.1. Conventional extraction (CE)
Anthocyanins were obtained differently when extracted in hot water under different conditions. When increasing the extraction time from 15 to 45 minutes, the obtained TAC gradually increased. However, when increasing the extraction time to 60 minutes, the TAC in the butterfly pea flower extract tended to decrease.
Furthermore, the extraction temperature also affects the TAC in the extract. Usually, by increasing the extraction temperature and time, the mass transfer from the plant material into the extract during extraction was easier due to the diffusion rate and solubility of the compounds that were increased [12]. In addition, under high temperatures, plant tissues softened and weak interactions affected cell membranes. As a result, the phenolic compounds can be easily extracted into the solvents [13]. However, a prolonged extraction time at 80oC reduced the extraction yield because the high temperature induced oxidation and decomposition of the desired compounds [14,15].
 | Table 1. CE, UAE, and microwave-assisted (MAE) extraction techniques for anthocyanin of butterfly pea flowers. [Click here to view] |
 | Table 2. Mean of anthocyanin in extract by treatment level under different extraction conditions. [Click here to view] |
The results showed that the obtained anthocyanins gradually increased when the heating temperature was from 60oC to 80oC, but, at the heating temperatures of 70oC and 80oC, the obtained solution did not have a significant difference in anthocyanin content. From the obtained results, it was found that a too short or too long time also could not obtain high TAC. Because of the short-time extraction, anthocyanin might not fully dissolve into the extraction solvent. On the other hand, anthocyanins are not temperature stable, so when extracted for a long time at high temperatures, TAC can be degraded [16].
3.1.2. Ultrasound-assisted extraction
Temperature has an effect on anthocyanin extract from butterfly pea flowers. TAC of the butterfly pea flower extract increased when the temperature increased from 60oC to 70oC (from 34.20 to 38.12 mg/l) and tended to decrease when the temperature reached 80oC (36.80 mg/l).
Besides avoiding exposure to high temperatures [17], UAE was used to reduce extraction time. The obtained results show that extraction time also affects anthocyanin content of the butterfly pea flower extract; this content increases with increasing time from 15 to 60 minutes, but there is no significant difference in TAC obtained from extract for 45 and 60 minutes.
These results are similar to those of Anthika et al. [18]. The extraction time of the butterfly pea flower was carried out in 45 minutes to obtain the highest anthocyanin content of 42.02 mg/l. Waves could cause tissue rupture for a long time of extraction, the solutes are removed and anthocyanins are produced more and more. This phenomenon occurs because the diffusion of the compound from the raw material into the solvent increases to a certain extent with the extraction time [17].
3.1.3. Microwave-assisted extraction (MAE)
After extraction at 3 and 5 minutes, anthocyanin content of the butterfly pea flower extract increased with extraction time from 32.86 to 39.60 mg/l, respectively. However, when the extraction time was over 5 minutes, anthocyanin content in the butterfly pea flower extract tended to decrease. This is probably because extraction time affects the dielectric properties of the solvent. Therefore, prolonged exposure to microwaves can cause degradation of target compounds due to substances in the system overheating [19].
A higher temperature helps induce intracellular pressure which ruptures the cell wall and leads to higher anthocyanin solubility as well as reduced viscosity of the extract [20]. Anthocyanin was easily affected by heat. As the temperature increases too high, the anthocyanin content in the extract may decrease. Anthocyanin content of the butterfly pea flower extract decreased from 37.29 to 31.56 mg/l when the energy levels were set at 200 and 600 W, respectively.
3.1.4. Comparison of selection conditions by different techniques for butterfly pea flowers
The obtained results from our study showed that both the UAE and MAE methods clearly improved the TAC in the extract, with the highest concentration found by UAE (Table 3). At the same temperature and extraction time, anthocyanin content of the butterfly pea flower extract from the UAE and MAE techniques increased to 15.01% and 14.11%, respectively. UAE can effectively improve the recovery of bioactive compounds, and ultrasonic radiation can disrupt cell walls allowing better penetration of solvents into the material’s interior, thereby improving mass transfer and cell disruption [7,21]. MAE is also an efficient extraction technique that converts microwave energy to heat quickly [22], with reduced solvent usage, shorter time, and higher yield recovery [23]. In this study, it was clearly observed that using UAE for extracting the anthocyanin in butterfly pea flowers was more effective than MAE. Ultrasound creates holes, disrupting the cell walls, allowing solvents to transfer mass, thereby increasing extraction efficiency [24,25].
3.2. Multiple regression analysis for antioxidant properties for butterfly pea flower extract by UAE
Multiple regression is allowed to determine the overall fit of the model and the relative contribution of each predictor to the total explained variance. Statgraphics Centurion XV.I was used to analyze the actual data (Table 4). The quadratic models for anthocyanin and iron reducing antioxidant power (FRAP) of butterfly pea flower extracts affected by extraction time and temperature using the UAE technique are presented in (1) and (2).
The high values of R2 for each response indicated that the multiple regression analysis fitted well into the model that was developed. These results confirmed the predictability of the models in determining the optimum conditions needed to obtain the highest TAC and antioxidant activity of the butterfly pea flowers extract.
 | Table 3. Comparison of anthocyanin contents of extract from different extraction methods. [Click here to view] |
 | Table 4. The change of antioxidant properties under different extraction conditions by the UAE technique. [Click here to view] |
TPCUAE = −92.72 + 3.59A − 0.008B − 0.0258A2 + 0.004AB − 0.0026B2 (1)
R2 = 91.07%, R2 (adj.) = 89.58%, SEE = 0.78,
FRAPUAE = −384.824 + 14.48A + 0.058B − 0.104A2 + 0.012AB − 0.008B2 (2)
R2 = 90.16%, R2 (adj.) = 88.52%, SEE = 1.10,
where A is extraction temperature (oC) and B is extraction time (minute).
Analysis of variance showed that the p values of the models and effects were less than 0.05, indicating that they were significant at the 95.0% confidence level (Table 5 and Fig. 1). From Figures 2 and 3 confirmed with previous results, extraction temperature and time strongly influenced anthocyanin content and antioxidant capacity of the extract. Under suitable conditions, the highest concentration in the extract could be observed. However, it should be considered that there are other affecting factors to avoid the loss of TAC and antioxidant activity.
Figure 4 shows the combination of desired functional maximization factor levels (TAC and FRAP maximization) on the indicated region. It also showed the combination of factors at which the optimal level is reached. The optimal values of TAC and FRAP obtained were 39.90 mg/l and 146.56 μMFeSO4/100 ml when the butterfly pea flower was extracted at approximately 74oC for 56.88 minutes.
By UPLC/UV/MS analysis, five anthocyanin compounds, delphinidin-hex, cyanidin-hexose-deoxyHex, delphinidin-deoxyhex-hex, cyanidin-hexose-2 deoxyHex, and delphinidin-hexose-2 deoxyHex, were identified (Fig. 5).
 | Table 5. Analysis of variance of models. [Click here to view] |
 | Figure 1: Standardized pareto chart for TAC (a) and FRAP (b) in extract. [Click here to view] |
 | Figure 2: Main effects plot for TAC (a) and FRAP (b) in extract. [Click here to view] |
 | Figure 3: Estimated response surface plot for TAC (a) and FRAP (b) in extract. [Click here to view] |
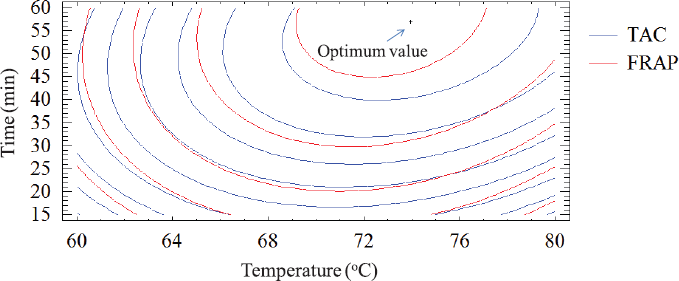 | Figure 4: The overlay plot for TAC and FRAP in extract and optimum value. [Click here to view] |
 | Figure 5: Anthocyanin compounds in butterfly pea flower by UPLC/UV/MS. [Click here to view] |
4. CONCLUSION
Ultrasonic- and MAE processes can be successfully applied to extract bioactive compounds from butterfly pea flowers with high bioactivity. However, using the UAE technique has many advantages over other techniques. With this extraction method, the anthocyanin content and antioxidant capacity of the extract were obtained at 39.90 mg/l and 146.56 μMFeSO4/100 ml, respectively. The obtained extract could be effectively used in processing traditional cakes and popular dishes in Vietnam instead of using synthetic colorants.
5. AUTHOR CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work. All the authors are eligible to be an author as per the international committee of medical journal editors (ICMJE) requirements/guidelines.
6. FUNDING
There is no funding to report.
7. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
8. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
9. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Mohamad MF, Nasir SNS, Sarmidi MR. Degradation kinetics and colour of anthocyanins in aqueous extracts of butterfly pea. Asian J Food Ag-Ind 2011;4(05):306–15.
2. Marpaung AM, Andarwulan N, Prangdimurti E. Optimization of anthocyanin pigment extraction from butterfly pea (Clitoria ternatea L.) petal using response surface methodology. Acta Hortic 2013;1011:205–11; doi:10.17660/ActaHortic.2013.1011.24
3. Jaafar NF, Ramli ME, Salleh RM. Optimum extraction condition of Clitorea ternatea flower on antioxidant activities, total phenolic, total flavonoid and total anthocyanin contents. Trop Life Sci Res 2020;31(2):1–17; doi:10.21315/tlsr2020.31.2.1
4. Pandey KB, Rizvi SI. Plant polyphenols as dietary antioxidants in human health and disease. Oxid Med Cell Longev 2009;2(5): 270–8; doi:10.4161/oxim.2.5.9498
5. Abubakar AR, Haque M. Preparation of medicinal plants: basic extraction and fractionation procedures for experimental purposes. J Pharm Bioallied Sci 2020;12:1–10; doi:10.4103/jpbs.JPBS_175_19
6. Kataoka H. Microwave assisted extraction. In: Townshend A, Poole CF, Miró M, Worsfold P (eds.). Encyclopedia of analytical science. 3rd edition, Elsevier, Amsterdam, Netherlands, 2019.
7. Chemat F, Rombaut N, Anne-GaëlleS, Meullemiestre A, Fabiano-Tixier A, Abert-Vian M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason Sonochem 2017;34:540–60; doi:10.1016/j.ultsonch.2016.06.035
8. Guisti MM, Wrolstad RE. Characterization and measurement of anthocyanins by UV-Visible spectroscopy. In: Wrolstad RE, editor. Current Protocols in Food Analytical Chemistry (F1.21-F1.2.13). John Wiley & Sons, New York, NY, 2001; doi:10.1002/0471142913.faf0102s00. Available via https://currentprotocols.onlinelibrary.wiley.com/ doi/10.1002/0471142913.faf0102s00
9. Adedapo AA, Jimoh FO, Afolayan AJ, Masika PJ. Antioxidant properties of the methanol extracts of the leaves and stems of Celtis africana. Rec Nat Prod 2009;3(1):23–31. Available via https://www.acgpubs.org/doc/201808051331252-RNP%200808-43.pdf
10. Backes E, Pereira C, Barros L, Prieto MA, Genena AK, Barreiro MF, et al. Recovery of bioactive anthocyanin pigments from Ficus carica L. peel by heat, microwave, and ultrasound based extraction techniques. Food Res Int 2018;113:197–209; doi:10.1016/j.foodres.2018.07.016
11. Piyapanrungrueang W, Chantrapornchai W, Haruthaithanasan V, Sukatta U, Aekatasanawan C. Comparison of anthocyanin extraction methods from high anthocyanin purple corn cob hybrid: KPSC 901, and quality of the extract powder. J Food Process Pres 2016;40(5):1125–33; doi:10.1111/jfpp.12693
12. Oancea S, Stoia M, Coman D. Effects of extraction conditions on bioactive anthocyanin content of Vaccinium corymbosum in the perspective of food applications. Procedia Eng 2012;42:489–95; doi:10.1016/j.proeng.2012.07.440
13. Shi J, Yu J, Pohorly J, Young JC. Optimization of the extraction of polyphenols from grape seed meal by aqueous ethanol solution. J Food Agric Environ 2003;1:42–7. Available via https://agris.fao.org/agris-search/search.do?recordID=FI2016100307
14. Naczk M, Shahidi F. Extraction and analysis of phenolics in food. J Chromatogr A 2004;1054:95–111; doi:10.1016/S0021-9673(04)01409-8
15. Silva EM, Souza JNS, Rogez H, Rees JF Larondelle Y. Antioxidant activities and polyphenolic contents of fifteen selected plant species from the Amazonian region. Food Chem 2007; 101:1012–8; doi:10.1016/j.foodchem.2006.02.055
16. Hou Z, Qin P, Zhang Y, Cui S, Ren G. Identification of anthocyanins isolated from black rice (Oryza sativa L.) and their degradation kinetics. Food Res Int 2013;50(2):691–7; doi:10.1016/j.foodres.2011.07.037
17. Syafa’atullah AQ, Amira A, Hidayati S, Mahfud M. Anthocyanin from butterfly pea flowers (Clitoria ternatea) by ultrasonic-assisted extraction. In AIP Conference Proceedings. AIP Publishing LLC, Melville, NY, 2020, vol 2237(1), p 020069; doi:10.1063/5.0005289
18. Anthika B, Kusumocahyo SP, Sutanto, H. Ultrasonic approach in Clitoria ternatea (butterfly pea) extraction in water and extract sterilization by ultrafiltration for eye drop active ingredient. Procedia Chem 2015;16:237–44; doi:10.1016/j.proche.2015.12.046
19. Orsat V, Routray W. Microwave-assisted extraction of flavonoids. In Munoz MJG, Dominguez H (eds.). Water extraction of bioactive compounds. Elsevier, Amsterdam, Netherlands, pp 221–244, 2017; doi:10.1016/B978-0-12-809380-1.00008-5
20. Chan CH, Lim JJ, Yusoff R, NgohGC. A generalized energy-based kinetic model for microwave-assisted extraction of bioactive compounds from plants. Sep Purif Technol 2015;143:152–60; doi:10.1016/j.seppur.2015.01.041
21. Bonfigli M, Godoy E, Reinheimer MA, Scenna NJ. Comparison between conventional and ultrasound-assisted techniques for extraction of anthocyanins from grape pomace. Experimental results and mathematical modeling. J Food Eng 2017;207:56–72; doi:10.1016/j.jfoodeng.2017.03.011
22. Jain NN, Ohal CC, Shroff SK, Bhutada RH, Somani RS, Kasture VS, et al. Clitoria ternatea and the CNS. Pharmacol Biochem Behav 2003;75:529–36; doi:10.1016/S0091-3057(03)00130-8
23. Armenta S, Garrigues S, De la Guardia M. Green analytical chemistry. Trend Anal Chem 2008;27:497–511; doi:10.1016/j.trac.2008.05.003
24. Chemat F, Rombaut N, Sicaire AG, Meullemiestre A, Fabiano-Tixier AS, Abert-Vian M. Ultrasound assisted extraction of food and natural products. Mechanisms, techniques, combinations, protocols and applications. A review. Ultrason Sonochem 2017;34:540–60; doi:10.1016/j.ultsonch.2016.06.035
25. Thuy NM, Tuyen NTM, Cuong NP, Huyen LTN, Phuong NP, Nguyen LTT, et al. Identification and extraction method of quercetin from flesh and skin of shallot (Allium ascalonicum) cultivated in Soc Trang Province, Vietnam. Food Res 2020;4(2):358–65; doi:10.26656/fr.2017.4(2).306