1. INTRODUCTION
The bidirectional reciprocal interaction between neuroendocrine and immune components known as the neuroendocrine-immune axis maintains the physiological homeostasis of animals. The hypothalamic–pituitary–adrenal (HPA) axis is a key component of this neuroendocrine–immune interaction, inducing an adaptive mechanism for animal survival. The HPA axis is a feedback mechanism system comprising hormones of the hypothalamus corticotropin-releasing factor (CRF), pituitary (ACTH), and adrenal gland (glucocorticoids) [1]. Stress can be defined as any physical, psychological, and immunological imbalance that disrupt the animal’s homeostasis, a responsive defense mechanism, and triggers the activation of the HPA axis [2]. The endotoxin lipopolysaccharide (LPS) is a cell wall component of Gram-negative bacteria (e.g., Escherichia coli) and is well established as an immune stressor [3]. The peripheral systemic administration of the LPS activates the HPA axis via cytokines stimulation [4,5]. The LPS administration releases a number of cytokines such as tumor necrosis factor-α (TNF-α), interleukine-1 (IL-1), and IL-6 from activated innate immune cells such as macrophages and subsequently increase CRF secretion in the hypothalamus [6]. The hypothalamic CRF is a crucial component of the HPA axis which induces ACTH secretion in the bloodstream from the anterior pituitary, which further stimulates the adrenal gland to release cortisol (corticosterone in rodents) [7]. The cortisol/corticosterone secretion inhibits the peripheral inflammation through their anti-inflammatory responses [8]. The glucocorticoids also send negative feedback signals to regulate the HPA axis. But in acute stress conditions, the inadequate response of the HPA axis to the cytokines might play a role in the commencement of neuropathology/psychopathology.
There are some neuropeptides (central and peripheral) that act as an atypical antipsychotic drug (AAPD), protect the neuroendocrine–immune axis impairment in LPS-exposed animals [9]. One of them is neurotensin (NT), a tridecapeptide found mainly in the brain and gut [10]. The NT functions as an endogenous antipsychotic agent that acts upon dopaminergic neurons to modulate behavioral impairment and psychiatric illness [11]. NT mediates their signaling through the NT receptor-1 (NTR-1), NTR-2 and G-protein-coupled receptors. Moreover, there are reports that the peripheral administration of NT reduces the inflammatory and oxidative stress response mechanism [12]. Although NT’s role in inhibiting peripheral inflammation is known, regulation of the neuroendocrine axis, particularly the HPA axis, is not well known. A few studies have been suggested the central administration of NT and HPA axis regulation. The peripheral NT injection cannot penetrate the blood–brain barrier (BBB) to reach the brain, which might be a reason for its limitation in regulating psychotic illness and HPA axis. Therefore, recently some NT analogs were developed to treat behavioral dysfunction, mental illness, and pain management [13]. PD 149163 is one of the NT agonists that acts upon NTR-1. The PD’s role in the psychosis and behavioral modulation in stress conditions is available, but its role in modulating the HPA axis impairment is unknown. Therefore, the present study hypothesized that PD modulates the HPA axis via an anti-inflammatory response. As the PD is an agonist of NT, this study also attempts to establish NT as an HPA axis modulator.
2. MATERIALS AND METHODS
2.1. Animal and Experimental Design
Swiss albino mice (female, 12 weeks) were procured from the Indian Institute of Toxicology Research, Lucknow, India, and were housed under controlled 12:12 hours dark–light cycle and temperature condition (25°C ± 10°C) and humidity (55% ± 5%) with free access feed and water. After acclimatizing for 2 weeks, mice were randomly divided into four groups (six mice/group): Group I (control) received saline (0.9% NaCl) from day 1 to 5; Group II (LPS) received LPS (1 mg/kg BW) from day 1 to 5 and was reared up to 21 days; Group III (LPS + PD) received LPS (1 mg/kg BW) from day 1 to 5, and after that treated with PD (300 µg/kg BW) for 21 days; and Group IV (PD) received saline (0.9% NaCl) from day 1 to 5 and after that treated with PD (300 µg/kg BW) for 21 days. All treatments were given intraperitoneal.
2.2. Peptide Preparation
The doses of PD were equivalent to NT and selected on the basis of previous studies with some modifications [14]. The stock solution of PD was prepared by first dissolving the amount of PD needed for the study in 1 ml of sterile water containing 0.1% (weight/volume) bovine serum albumin (BSA) diluted with normal saline containing 1% BSA. The PD solution was divided into equal aliquots of 0.1 ml and stored at −20°C. During the administration time, each aliquot was further diluted in sterile water to the required concentration (300 µg/kg BW) and given intraperitoneally as a bolus injection of 0.5 ml.
Animals were treated according to the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), MoEF&CC, India. The Institutional Animal Ethics Committee (IAEC) of the University of Allahabad approved the experimental protocols (approval number IAEC/AU/2017(1)/019).
2.3. Study of IL-6 and TNF-α
IL-6 and TNF-α blood plasma were assayed by a commercially available ELISA kit (Sigma-Aldrich, USA). Blood samples were immediately collected from the abdominal aorta of mice in 0.1% EDTA-treated vials, centrifuged at 2,500 rpm for 15 minutes, and plasma was collected and stored at −20°C until assay. During the assay, plasma samples (100 µl of each aliquot) were used in duplicate. The inter-assay and intra-assay coefficient variations for IL-6 and TNF-α were 12% and <10, respectively.
2.4. Immunohistochemistry of CRF
The immunohistochemical detection of CRF was conducted by the avidin–biotin complex (ABC) method [15]. Brain sections (10 µm thickness), including the hypothalamus, were stretched on a glass slide, deparaffinized in xylene, and rehydrated through graded alcohol to phosphate buffer saline (PBS). Sections were then incubated with primary antibody of anti-CRF (dilution, 1:800) (Sigma-Aldrich, USA) overnight at 4°C. After thorough washing in PBS, sections were subsequently incubated with the biotinylated antibody (Goat anti-Rabbit IgG-Biotin, 1:100) (Merck, India) and streptavidin–horseradish peroxidase (HRP) conjugate (Goat anti-Rabbit IgG, 1:100) (Merck, India), each for 1 hour. After that, the slides were incubated with a chromogen 3, 3′ DAB (diaminobenzidine, 10 mg in 0.1% H2O2) for 10–15 minutes for immunoreactive visualization.
The immunointensity of CRF was measured by mean optical density (OD) using ImageJ 1.32 software (NIH, Bethesda, MD) of different hypothalamus regions and corrected by subtraction of the background OD of the corresponding area.
2.5. Hormonal Assay of ACTH and Corticosterone
Plasma levels of ACTH and corticosterone were assayed according to the manufacturer’s protocols of the respective commercially ELISA kits purchased from Neogene Corporation (Lansing, MI) and Labor Diagnostika Nord (LDN) Diagnostics (Nordhorn, Germany). Blood was immediately collected from the aorta, centrifuged at 2,500 rpm (15 minutes), and kept at −20°C until assay. Samples of control and experimental groups were assayed in duplicate of 100 µl each aliquot. Both intra-assay and inter-assay coefficients of variations were <10% for both the tests.
2.6. Statistical Analysis
All data were represented in mean ± standard error mean (SEM) and analyzed using GraphPad Prism 5 software. One-way analysis of variance (ANOVA), followed by Tukey’s post-hoc test, was used for the study of significant level. p < 0.05 was considered significant.
3. RESULTS
3.1. IL-6 and TNF-α
The results of cytokines (IL-6 and TNF-α) are shown in Table 1. The plasma IL-6 and TNF-α level were significantly increased (p < 0.001 and p < 0.01, respectively) in LPS-challenged mice compared to control. The IL-6 level, not TNF-α, was also increased in only the PD group in comparison with control (p < 0.05); other groups showed no substantial changes. In Tukey’s post-hoc test, the mice of LPS + PD and PD groups showed significantly increased IL-6 (p < 0.01) and TNF-α (p < 0.001) compared to the LPS-exposed group. In LPS + PD versus PD group, only the IL-6 level was significantly increased (p < 0.05), not TNF-α (Table 1).
3.2. IHC of CRF
The IHC and IHC of CRF are summarized in Figures 1 and 2, respectively. The CRF-immunoreactive cells were observed mainly in the median eminence (ME) and third ventricle (3V) of the hypothalamus. The control group mice exhibited fewer homogenously distributed CRF-immunoreactive cells than the experimental group (Fig. 1A). In LPS-induced mice, CRF immunoreactive cells’ expression was increased and concentrated in patches (Fig. 1B). The ME showed high reactivity of CRF than 3V. After PD treatment of LPS-challenged mice, the CRF expression was downgraded to control (Fig. 1C). This result shows the counteraction effect of PD on LPS. The only PD-exposed mice also showed moderately increased CRF expression to control, but much less than LPS exposure.
The immunointensity of CRF was significantly increased in only LPS-challenged mice (Fig. 2) compared to control. Other groups revealed no substantial increase/decrease in CRF immunoreactivity. However, only PD-exposed mice showed some increased IHC in comparison with control, but not significantly. In post-hoc analysis, LPS and LPS + PD versus other groups showed no substantial CRF IHC changes (Fig. 2).
3.3. ACTH and Corticosterone
The result of ACTH and corticosterone level is summarized in Table 1. The plasma ACTH and corticosterone levels were significantly increased (p < 0.01) in LPS-challenged mice compared to control. In Tukey’s multiple tests, the LPS + PD and PD groups revealed substantially increased ACTH (p < 0.05 for both) and corticosterone (p < 0.05 for both) compared to the LPS-exposed group. Compared to control, LPS and LPS + PD, other groups showed no significant change in ACTH/corticosterone level.
4. DISCUSSION
The present study evaluates the psychopharmacological effect of PD 14163 on the HPA axis. Fine-tuning between the neuroendocrine–immune axes is a requisite for homeostasis and animal survival; studying this aspect is an emerging research area. The HPA axis is a crucial component of the endocrine–immune axis. As the immune activation leads to the HPA activation, peripheral immune activation modulation might regulate the HPA axis. So, we hypothesized that the PD protects the HPA axis impairment through its immune modulation activity. The systemic infection of LPS impaired the HPA axis directly or indirectly through cytokine secretion [16]. In the peripheral system, the LPS directly stimulates several cytokines such as IL-1, IL-6, TNF-α, and chemokines from innate immune cells. The LPS binds with the toll-like receptor, particularly Toll like receptor-4 (TLR-4) , present on innate immune cells such as macrophages and triggers cytokine release signaling [17]. These cytokines cross the BBB to regulate the HPA axis activity through multiple cytokine receptors [18]. Thus, the HPA axis’s hyperactivation is a sign of depression associated with the proinflammatory cytokine such as IL-6, IL-1β, and TNF-α stimulated CRF release [19,20]. In our study, the LPS-exposed (1 mg/kg BW) mice showed the substantially increased proinflammatory cytokines IL-6 and TNF-α, which was associated with the increased immunoreactivity of CRF in the hypothalamus. The exact mechanism through which LPS and cytokines activate the HPA axis is not well known. Although some studies have been suggested the TLR-4 present in the hypothalamic region, LPS penetration through BBB is not proved yet. Therefore, we cannot surely say that the LPS reaches the brain to bind with TLR-4 to activate the HPA axis. On the other hand, there are reports that the LPS through local production of IL-6 and IL-1 play a vital role in the hypothalamus in the stimulation of the HPA axis. Hence, the present study correlated the enhancement of IL-6 and TNF-α and secretion of hypothalamic CRF. Furthermore, there is evidence in rodents that cytokines stimulate the hypothalamic CRF in vitro and in vivo [21,22]. The CRF plays a significant role in regulating the HPA axis, induced by proinflammatory cytokines such as IL-1β, IL-6, IL-10, and TNF-α upon LPS exposure. The increased numbers of CRF-expressing neurons and elevated CRF mRNA expression were found in the Paraventricular nucleus (PVN) of the hypothalamus of patients with depression [23]. The dysregulation of CRF caused extensive adverse effects on the body, such as reducing appetite, stress-induced analgesia, sleep disturbances, and anxiety. CRF is transported by the local vascular system, binds with the CRF receptor-1 in the anterior pituitary, and stimulates the pro-opiomelanocortin transcription and ACTH release [24, 25]. ACTH via blood circulation reaches the adrenal cortex and binds with its receptor in zona fasciculata to stimulate the synthesis and release of glucocorticoids (cortisol and corticosterone), which have broad biological effects on the body. We also found that both ACTH and corticosterone were significantly increased after LPS injection for 5 days (Table 1). The glucocorticoids are the main end effectors of the HPA axis activation and send negative feedback to the hypothalamus and the pituitary gland to inhibit Corticotropin-releasing hormone (CRH) and ACTH production. In depression, excess glucocorticoid levels caused by hyperactivity of the HPA axis result in tissue damage and immune disturbances.
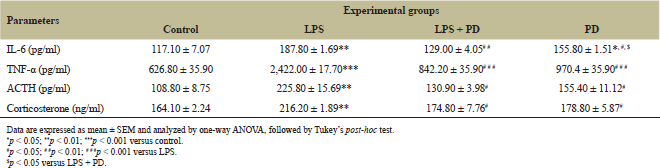 | Table 1: Effect of LPS and PD on cytokines and hormonal level. [Click here to view] |
 | Figure 1: IHC of hypothalamic CRF: Control (A), LPS (B), LPS + PD (C), and PD (D). The arrow shows the CRF-immunoreactive cells (A–D) in the ME and third ventricle (3V) of the hypothalamus. Magnification: 40×, bar = 50 µm. [Click here to view] |
 | Figure 2: The graph showing CRF immunointensity. The data are represented as mean ± SEM and analyzed by one-way ANOVA, followed by Tukey’s post-hoc test. *p < 0.05 compared to respective control. [Click here to view] |
The role of AAPD in the prevention of stress-induced HPA axis was reported previously [26,27]. The NT acts as an endogenous neuroleptic in the central nervous system and an anti-inflammatory agent in the peripheral system. The Central nervous system (CNS) co-exists with the dopaminergic system and regulates the dopamine’s function, resulting in psychosis management [28]. In the peripheral, NT is bound mainly upon NTR-1 present on immune cells and enteroendocrine cells to alleviate the proinflammatory cytokines’ secretion. There are reports that the NT, acting through NTR-1, modulates the immune impairment by down-regulating proinflammatory such as IL-6 and TNF-α, as well as accelerating anti-inflammatory cytokines (IL-10) and growth factors such as Epidermal growth factor (EGF), Vascular endothelial growth factor (VEGF), and PDGF [12,29]. As the activation of the immune system is suppressed, it further regulates the hyperactivity of the HPA axis. Therefore, we used the NT agonist PD, which also acts upon the NTR-1 to evaluate the efficacy as an immune and HPA axis modulator. The PD can cross the BBB in the brain and act upon the dopaminergic system, like NT, which plays a significant role in stress. In the current study, the PD treatment counteracted the LPS-induced impairment at different HPA axis levels: hypothalamic CRF expression, the plasma level of pituitary hormone ACTH, and adrenal corticosterone. The PD treatment may regulate the HPA axis by anti-depression and anti-inflammatory mechanisms. A recent study found that systemic administration of the highly selective NTR-1 receptor agonist PD149163 exhibited an antidepressant drug-like effect comparable to the tricyclic antidepressant drug imipramine and the AAPD risperidone [30]. The PD’s psychopharmacological property had been tested in behavioral dysregulation by open field tests and forced swimming tests [31]. Despite this, the PD’s role in the HPA axis regulation and immune modulation has not been tested yet. As the interaction between NT and NTR-1 modulates the immune disruption, PD might also regulate the immune impairment and, further, HPA axis activation. Like NT, PD modulates dopamine release by acting upon NTR-1. By normalizing the CRF expression, a critical coordinator of the HPA axis regulation, PD down-regulates the plasma level of ACTH and corticosterone. After PD treatment, the inhibition of the HPA axis could be linked to the suppression of immune activation.
5. CONCLUSION
The neuroendocrine and immune communication is disrupted by the LPS, an immune stressor. The systemic LPS exposure disturbs the peripheral innate immune system and releases the proinflammatory cytokines IL-6 and TNF, which subsequently activate the stress axis (i.e., HPA). The increased CRF expression and ACTH and corticosterone level is a marker of HPA axis activation. After the PD 149163 treatment, the HPA axis modulated through inhibition of cytokines IL-6 and TNF-α. The antipsychotic-like activity of PD also balanced the neuroendocrine–immune axis. Thus, the present study will help for more exploration of mechanisms in the future.
6. ACKNOWLEDGMENT
The authors would like to thank Dr. B. Mohanty, Department of Zoology, University of Allahabad, for her guidance and support.
7. FUNDING
This work was funded by UGC-CSIR [Award No. 09/001(0404)/2017-EMR-I], Govt. of India.
REFERENCES
1. Joseph DN, Whirledge S. Stress and the HPA axis: balancing homeostasis and fertility. Int J Mol Sci 2017;18:2224; doi:10.3390/ijms18102224 CrossRef
2. Smith SM. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin Neurosci 2006;8(4):383–395. CrossRef
3. Beishuizen A, Thijs LG. Endotoxin and hypothalamo-pituitary-adrenal (HPA) axis. J Endotoxin Res 2003;9(1):3–24; doi:10.1179/096805103125001298 CrossRef
4. Ménard C, Pfau ML, Hodes GE, Russo SJ. Immune and neuroendocrine mechanisms of stress vulnerability and resilience. Neuropsychopharmacol Rev 2017;42:62–80; doi:10.1038/npp.2016.90 CrossRef
5. Kanczkowski W, Alexaki V, Tran N, Großklaus S, Zacharowskic K, Martinezd A, et al. Hypothalamo-pituitary and immune-dependent adrenal regulation during systemic inflammation. Proc Natl Acad Sci U S A 2013;110:14801–6. CrossRef
6. Pham GS, Mathis KW. Lipopolysaccharide challenge reveals hypothalamic-pituitary-adrenal axis dysfunction in murine systemic lupus erythematosus. Brain Sci 2018;8:184; doi:10.3390/brainsci8100184 CrossRef
7. Jiang Y, Peng T, Gaur U, Silva M, Little P, Chen Z, et al. Role of corticotropin releasing factor in the neuroimmune mechanisms of depression: examination of current pharmaceutical and herbal therapies. Front Cell Neurosci 2019;13:290. CrossRef
8. Bellavance M, Rivest S. The HPA – immune axis and the immunomodulatory actions of glucocorticoids in the brain. Front Immunol 2014;5:136; doi:10.3389/fimmu.2014.00136 CrossRef
9. Meyer U, Schwarz MJ, Müller N. Inflammatory processes in schizophrenia: a promising neuroimmunological target for the treatment of negative/cognitive symptoms and beyond. Pharmacol Ther 2011;132:96–110. CrossRef
10. Caceda R, Kinkead B, Nemeroff BC. Neurotensin: role in psychiatric and neurological diseases. Peptides 2006;27:2385–404; doi:10.1016/j.peptides.2006.04.024. CrossRef
11. Boules MM, Fredrickson P, Muehlmann AM, Richelson E. Elucidating the role of neurotensin in the pathophysiology and management of major mental disorders. Behav Sci 2014;4:125–53; doi:10.3390/bs4020125 CrossRef
12. da Silva L, Neves BM, Moura L, Cruz MT, Carvalho E. Neurotensin downregulates the pro-inflammatory properties of skin dendritic cells and increases epidermal growth factor expression. Biochim Biophys Acta 2011;1813:1863–71; doi:10.1016/j.bbamcr.2011.06.018 CrossRef
13. Boules M, Li Z, Smith K, Paul Fredrickson P, Richelson E. Diverse roles of neurotensin agonists in the central nervous system. Front Endocrinol 2013;4:36; doi:10.3389/fendo.2013.00036 CrossRef
14. Akcan A, Muhtaroglu S, Akgun H, Akyildiz H, Kucuk C, Sozuer E, et al. Ameliorative effects of bombesin and neurotensin on trinitrobenzene sulphonic acid-induced colitis, oxidative damage and apoptosis in rats. World J Gastroenterol 2008;14(8):1222–30; doi:10.3748/wjg.14.1222 CrossRef
15. Jensen K, Krusenstjerna-Hafstrøm R, Lohse J, Petersen KH, Derand H. A novel quantitative immunohistochemistry method for precise protein measurements directly in formalin-fixed, paraffin-embedded specimens: analytical performance measuring HER2. Mod Pathol 2017;30:180–93; doi:10.1038/modpathol.2016.176 CrossRef
16. Turnbull AV, Rivier CL. Regulation of the hypothalamic-pituitary-adrenal axis by cytokines: actions and mechanisms of action. Physiol Rev 1999;79(1):1–71. CrossRef
17. Vakharia K, Hinson JP. Lipopolysaccharide directly stimulates cortisol secretion by human adrenal cells by a cyclooxygenase-dependent mechanism. Endocrinology 2005;146(3):1398–402; doi:10.1210/en.2004-0882 CrossRef
18. Utsuyama M, Hirokawa K. Differential expression of various cytokines receptors in the brain after stimulation with LPS in young and old mice. Exp Gerontol 2001;37(2–3):411–20; doi:10.1016/s0531-5565(01)00208-x CrossRef
19. Dentino AN, Pieper CF, Rao KMK, Currie MS, Harris T, Blazer DG, et al. Association of Interleukin-6 and other biologic variables with depression in older people living in the community. J Am Geriatr SOC 1999;47(1):6–11. CrossRef
20. Kariagina A, Romanenko D, Ren S, Chesnokova V. Hypothalamic-pituitary cytokine network. Endocrinology 2004;145(1):104–12; doi:10.1210/en.2003-0669 CrossRef
21. Knapp DJ, Whitman BA, Wills TA, Angel RA, Overstreet DH, Criswell HE, et al. Cytokine involvement in stress may depend on corticotrophin releasing factor to sensitize ethanol withdrawal anxiety. Brain Behav Immun 2011;25:S146–54; doi:10.1016/j.bbi.2011.02.018 CrossRef
22. Vasconcelos M, Stein DJ, Gallas-Lopes M, Landau L, de Almeida RMM. Corticotropin-releasing factor receptor signaling and modulation: implications for stress response and resilience. Trends Psychiatry Psychother 2020;42(2):195–206. CrossRef
23. Raadsheer FC, Hoogendijk WJ, Stam FC, Tilders FJ, Swaab DF. Increased numbers of corticotropin-releasing hormone expressing neurons in the hypothalamic paraventricular nucleus of depressed patients. Neuroendocrinology 1994;60(4):436–44; doi:10.1159/000126778 CrossRef
24. Lightman SL. The euroendocrinology of stress: a never ending story. J Neuroendocrinol 2008;20:880–84; doi:10.1111/j.1365-2826.2008.01711.x CrossRef
25. Slominski AT, Zmijewski MA, Zbytek B, Tobin DJ, Theoharides TC, Rivier J. Key role of CRF in the skin stress response system. Endocr Rev 2013;34(6):827–84; doi:10.1210/er.2012-1092 CrossRef
26. Park SW, Choi SM, Lee JG, Lee CH, Lee SJ, Kim NR, et al. Differential effects of ziprasidone and haloperidol on immobilization-stress-induced CRF mRNA expression in the hypothalamic paraventricular nucleus of rats. Neuropsychobiology 2011;63:29–34; doi:10.1159/000322288 CrossRef
27. Cohrs S, Röher C, Jordan W, Meier A, Huether G, Wuttke W, et al. The atypical antipsychotics olanzapine and quetiapine, but not haloperidol, reduce ACTH and cortisol secretion in healthy subjects. Psychopharmacology 2006;185:11–8; doi:10.1007/s00213-005-0279-x CrossRef
28. Cusack B, Boules M, Tyler BM, Fauq A, McCormick DJ, Richelson E. Effects of a novel neurotensin peptide analog given extracranially on CNS behaviors mediated by apomorphine and haloperidol. Brain Res 2000;856:48–54. CrossRef
29. da Silva LP, Neves BM, Moura L, Cruz MT, Carvalho E. Neurotensin decreases the proinflammatory status of human skin fibroblasts and increases epidermal growth factor expression. Int J Inflam 2014;2014:248240. http://dx.doi.org/10.1155/2014/248240. CrossRef
30. Hillhouse TM, Shankland Z, Matazel KS, Keiser AA, Prus AJ. The quetiapine active metabolite N-Desalkylquetiapine and the neurotensin NTS1 receptor agonist PD149163 exhibit antidepressant-like effects on operant responding in male rats. Exp Clin Psychopharmacol 2014;22:548–56. CrossRef
31. Carey LM, Rice RJ, Prus AJ. The neurotensin NTS1 receptor agonist PD149163 produces antidepressant-like effects in the forced swim test: further support for neurotensin as a novel pharmacologic strategy for antidepressant drugs. Drug Dev Res 2017;78:196–202. CrossRef