1. INTRODUCTION
Rice (Oryza sativa L.) is one of the most strategic crops worldwide, especially in developing countries, after wheat [1]. Due to its carbohydrate content and nutritional value, it is considered an important source of daily calories for more than 50% of the world’s population [2]. Rice as a semiaquatic plant is one of the most water-consuming crops; one hectare consumes more than 17,000 m3 of water, where continuous submergence is the most effective method of irrigation. It is, therefore, highly affected by drought stress [3]. Drought can be defined as shortage in soil moisture available for plant growth, development, and completion of its life cycle [4]. It is one of the most serious abiotic stresses affecting the biochemical, physiological, and morphological status of the plant [5]. Stomatal closure according to Vanisri et al. [6] is one of the earliest reactions to stress leading to a reduction in CO2 diffusion from the atmosphere to carboxylation sites inside chloroplast. Electrons that were supposed to be consumed in CO2 assimilation are accumulated and transferred to O2 forming reactive oxygen species (ROS) (oxidative stress) in chloroplast and mitochondria [7]. ROS such as superoxide (O2-), singlet oxygen (1/2 O2), hydrogen peroxide (H2O2), and hydroxyl radical (OH−) are highly active molecules that react and damage all biological molecules in the cell such as lipids, proteins, carbohydrates, nucleic acids, chlorophylls, and enzymes resulting in membrane lipid peroxidation, protein carbonylation, and chlorophyll loss and inactivating the –SH containing enzymes [8]. Thus, photosynthesis, growth, development, and yield are severely affected by drought [9].
Plant response to drought stress depends upon plant stage, stress duration, and plant species [10]. Plant uses any of these different strategies to cope with drought stress: (i) drought escape that will allow the plant to grow, develop faster, and complete its life cycle before severe water shortages, (ii) drought avoidance by decreasing the rate of transpiration (via reduction of stomatal conductance and leaf area) and increasing water uptake, (iii) drought tolerance by maintaining cell turgor by inducing osmolytes accumulation such as proline, glycine betaine, and certain types of sugars that allow plants to retain water, maintain growth, and restore plant function after drought stress [11]. Acclimation of plants to stress is accompanied by an increase in the plant’s antioxidant system, which has the ability to remove oxygen radicals, repair the damage, and keep the concentration of ROS at a relatively low concentration [7]. An antioxidant system includes enzymatic (such as superoxide dismutases and catalases) as well as nonenzymatic compounds (such as phenolic acids and flavonoids and other molecules) which are able to remove ROS and protect the cells from damage like proline. Gao et al. [12] reported that increasing antioxidant activity of plants under stress is considered characteristic of drought-resistant species. The more increasing the efficiency of ROS scavenging during stress, the more increasing the protection of photosynthetic apparatus, which leads to improving photoassimilates production. For instance, sugars (such as glucose, fructose, sucrose, maltose, and galactose) and sugar alcohols (which are also called polyols, such as mannitol, sorbitol, and xylose) are accumulated and act as typical osmoprotectants. Moreover, they play a crucial role in adapting rice plants to drought stress by maintaining osmotic adjustment to prevent intracellular dehydration [11], protecting macromolecules, and stabilizing cellular membranes, proteins, and enzymes as well as scavenging ROS molecules [4], in addition to the role of proline and organic acids in the adjustment of cellular osmotic stress and maintenance of intracellular water.
More efforts have now been made to increase rice crop productivity to more than 40% to meet population requirements [13]. It has become serious in the shade of increasing water scarcity and agricultural land that is expected to dry up all over the world.
The aim of the current work was to (i) study the changes in metabolite profile (including sugars, phenolic acids, flavonoids, organic acids, and phytohormones) in Egyptian rice in response to drought stress and (ii) highlight the differences in strategies that were exhibited by tolerance and sensitive cultivars.
2. MATERIAL AND METHODS
The present experiment was conducted at the botanical garden, Faculty of Science, Ain Shams University, during the summer season of 2018/2019. Two rice cultivars Giza 179 (Gz179) (a drought-tolerant cultivar) and Sakha 101 (Sk101) (a drought-sensitive one) were selected based on our previous study [3] where several physiological parameters were analyzed on six Egyptian rice cultivars to select the most tolerant and the most sensitive cultivars. In plastic pots (12 cm in diameter and 10 cm high), ten grains per pot were sown and watered to 80% of its water holding capacity and exposed to normal daylight and temperature (30°C ± 2°C, 16 hours light/8 hours dark cycle), each pot filled with one kilogram of soil (clay : washed and dried sand) (2:1 wt/wt). Ten-day-old seedlings were irrigated once weekly with a full-strength Hoagland nutrient solution [14]. The water holding capacity was maintained at 80% until 21-day-old seedlings and pots for each rice cultivar were divided into two groups. The first one was watered to 80% water holding capacity (act as control), and the second group was watered to 40% water holding capacity (act as drought-stressed plants). This water regime was maintained until symptoms of leaf rolling were detected as an indicator of drought stress. At the end of the experiment, 28-day-old plants were collected (Fig. 1). Metabolites such as proline, sugar content, phenolic acids, flavonoids, growth regulators, and organic acid contents were determined by high-performance liquid chromatography (HPLC).
2.1. Determination of Proline
Free proline was evaluated in fresh leaves as described by Bates et al. [15]. Values were expressed as µg/g fresh weight of the plant.
2.2. Extraction, Separation, and Identification of Soluble Sugars using HPLC
2.2.1. Extraction of the soluble sugars
Fresh leaf samples (0.5 g) were frozen using liquid nitrogen and extracted for 10 minutes in boiling water. Then samples were incubated for 20 minutes at 70°C and centrifuged (K280R, Centurion Scientific, Chichester, UK) for 10 minutes at 4,000 g, the liquid phase was discarded, and the residue was reextracted 2 times at 70°C.
2.2.2. Quantification of the soluble sugars
Leaf extracts were diluted 1:10 (v/v) and quantified using a chromatographic system coupled to the refractive index detector (HPLC-RI) which was equipped with a degasser, quaternary pump (Waters 2695 Alliance, Milford, MA), autoinjector, and Waters RI 2414 refractive index detector (Milford, MA) [16].
2.3. Extraction, Separation, and Identification of Phenolic Compounds using HPLC
2.3.1. Extraction of the phenolic compounds
Fresh leaves of rice (0.5 g) were grounded in liquid nitrogen into a fine powder, and 10 ml methanol HPLC grade was added and slightly stirring. Tubes were then centrifugated at 1,500 g for 10 minutes. The liquid phase was collected for quantification by HPLC [17,18].
 | Figure 1: 28-days old rice seedlings: Gz179 (tolerant) and Sk101 (sensitive). C: 80% WHC (control), T: 40% WHC (drought-stressed). [Click here to view] |
2.3.2. Identification of the phenolic compounds
Phenolic compounds were measured using HPLC consisting of a solvent delivery module LG-980-02, detector UV/Vis UV-970, and pump PU-980. The column was Phenomenex, UK, an RP-C18 Luna column, 4.60 mm i.d. × 250 mm and particle size = 5 μm. Detection was carried out at 280 and 340 nm and the compounds were identified by comparing their retention time values with those of the standards [19].
2.4. Extraction, Separation, and Identification of Flavonoid Compounds using HPLC
2.4.1. Extraction of the flavonoid compounds
Flavonoids compounds were extracted from dried leaf tissues according to Hertog et al. [20].
2.4.2. Quantification of the flavonoid compounds
The analytical HPLC system used consisted of a Hewlett-Packard Gas Chromatography high-performance liquid chromatograph equipped with an HP1090 Series II diode array and an eight-channel electrochemical coulometric array detector (EC; Esa Inc., Chelmsford, MA). HP 3D Chem Station computer program was used to calculate the data. Standards’ calibration curves were performed by diluting stock standards to 2–20 μg/ml using methanol [21].
2.5. Extraction, Separation, and Identification of Growth Regulators
2.5.1. Extraction of the plant growth regulators
Phytohormones were determined in fresh leaf tissues. 10 ml of acidified acetonitrile (1% acetic acid in acetonitrile) was added to one-gram fresh leaves. After homogenization, 4 g of MgSO4 (magnesium sulfate) and 1.5 g of NaOAc (sodium acetate) were added to the mixture and then vortexed and centrifuged for 3 minutes at 4,000 g. 2 ml of the liquid phase was loaded into a clean 2 ml vial containing 25 mg C18 sorbent and 150 mg anhydrous MgSO4, then vortexed for 1 minute, and centrifuged at 16,000 g for 5 minutes.
2.5.2. Quantification of the growth regulators
Growth regulators were quantified using Agilent1260 infinity HPLC Series (Agilent@, Santa Clara, CA), provided with quaternary pump, a Zorbex Eclipse Plus C18 column 100 × 4.6 mm run at 35°C. Gradient elution with first 5 mM ammonium-acetate/0.05% formic acid in water and then acetonitrile (beginning with 95% solution A and 5% solution B) was used for separation. The injected volume was 20 μl. For detection, Variable Wavelength Detector was set at 254 nm [22].
2.6. Extraction, Separation, and Identification of Organic Acids using HPLC
2.6.1. Extraction of organic acids
Rice leaf samples (0.5 g) were maintained by adding 96% ethanol. Samples had a rather complex matrix; proteins, enzymes, and sugars were present in addition to organic acid. In order to prevent the deterioration of the analytical column resulting from highly retained substances, the sample was washed using solid-phase extraction prior to the examination.
2.6.2. Quantification of the organic acids
1 mm3 sample was injected into the chromatographic system [HP 1090 Hewlett-Packard liquid chromatograph with a Cl8 guard column (25 × 4.6 mm ID)] and a Nucleosil ODS 100-5 analytical column. The column was operated at 55°C. Precipitation method (standard industrial method for the determination of citric acid) was performed as described by Wodecki et al. [23]. Organic acid contents were represented as µg/g f.wt.
2.7. Statistical Analysis
With the Statistical Program for Social Sciences (SPSS) v 20.0 software (SPSS Inc., Chicago, IL), the experimental findings were analyzed, and analysis of variance was used for statistical analysis. Means were tested by Duncan’s test [24] to determine the level of significance (p < 0.05).
3. RESULTS
3.1. Proline
Data presented in Figure 2 showed the intense accumulation of proline in drought-stressed Gz179 (205% over the control value) compared with 29% increase in case of Sk101.
3.2. Soluble Sugars
The highest accumulation of sugar was recorded by stressed Gz179 (6.075 μg/g f.wt), 6-fold increase compared with its control, while drought-stressed Sk101 had doubled sugar accumulation compared to its control. Interestingly, Gz179 accumulated stachyose (20-fold), sucrose (12-fold), galacturonic (11.5-fold), rhamnose (10.5-fold), galactose (10.4-fold), and sorbitol (10-fold) compared with its control, while Sk101 accumulated stachyose (5.9-fold) and galacturonic (5.2-fold) as compared to its control (Table 1).
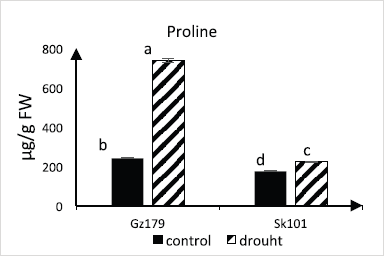 | Figure 2: Changes in proline in response to water deficit in two rice cultivars (Gz179 and Sk101) differing in drought tolerance. [Click here to view] |
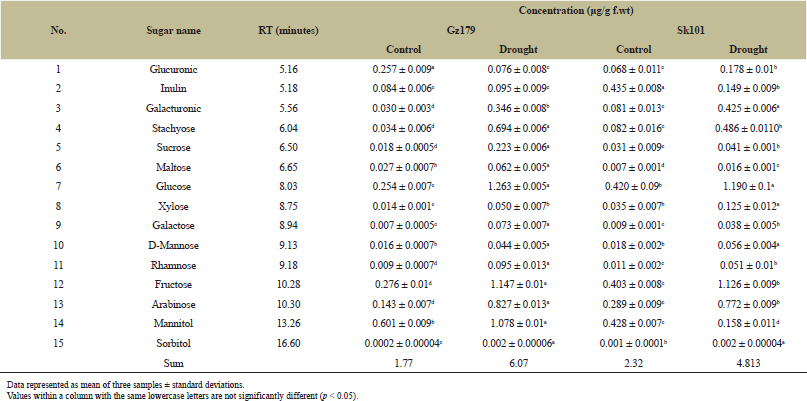 | Table 1: Identification and quantification of sugars in Egyptian rice cultivars Gz179 and Sk101 as determined by HPLC. [Click here to view] |
3.3. Phenolic Compounds
Table 2 represents the concentrations of different phenolic compounds present in rice shoot system and their response to drought stress. Total analyzed phenols in plant seedlings showed that stressed Gz179 contained 2,012.66 μg/g f.wt, approximately 1.5-fold of its control which recorded 1,365.66 μg/g f.wt, while treated Sk101 recorded 1,130.92 μg/g f.wt, 1.03-fold of its control which recorded 1,089.76 μg/g f.wt. This means that untreated Gz179 accumulated phenolic compounds more than treated Sk101. Moreover, treated Gz179 recorded an increase in isoferulic (5.5-fold), pyrogallol (5-fold), and cinnamic (4.3-fold) compared with control, while Sk101 grown under deficit condition recorded an increase in ferulic (2.8-fold) and ellagic (2.2-fold) compared with control. Gallic and alpha-coumaric were decreased in treated Gz179 and increased in Sk101. In contrast, catechin and catechol were decreased in stressed Sk101 and increased in stressed Gz179. Protocatechuic was decreased and benzoic disappeared from both cultivars under drought stress.
3.4. Flavonoid Compounds
The results of flavonoid compounds analyzed in the two rice cultivars under normal and drought stress conditions are presented in Table 3. Drought-stressed Gz179 exhibited an approximately double increase in flavonoids concentration compared with control, where stressed seedling was recorded (4,979.9 μg/g f.wt) and control seedling was recorded (2,693 μg/g f.wt), while stressed Sk101 recorded 1,592.4 μg/g f.wt compared with its control which recorded 1,513.69 μg/g f.wt, and at another time, the concentration of flavonoid compounds in untreated Gz179 exceeds treated Sk101. Treated Gz179 recorded an increase in kaempferol (5.3-fold), quercetrin (2.7-fold), kaempferol 3-(2-p-comaroyl) glucose (2.5-fold), and each apigenin 6-arabinose, apigenin 7-glucose, apigenin 7-O-neohespiroside, and kaempferol 3-7-diramoside (recording double increase), while luteolin 7-glucose recorded a significant decrease in treated Gz179 and apigenin 6-rhamnose 8-glucose and acacetin 7-neohesperidin that are characteristics of sensitive cultivar (Sk101).
3.5. Plant Growth Regulators
Plant growth promoters [indole acetic acid (IAA), indole butyric acid (IBA), and gibberellic acid (GA3)] as well as inhibitors [abscisic acid (ABA) and salicylic acid (SA)] have been quantified in controlled and drought-stressed rice cultivars (Fig. 3). The results showed that growth promoters were reduced in both cultivars in response to drought compared to control, where IAA, IBA, and GA3 were decreased by percent 20%, 38%, and 50%, respectively, in tolerant cultivar (Gz179) under stress, while in sensitive one (Sk101), they were decreased by 29%, 53%, and 50%, respectively. In contrast, ABA and SA markedly were increased to 2-fold in Gz179 and approximately 3-fold in Sk101.
3.6. Organic Acids
Results in Figure 4 showed that the accumulation of oxalic acid in both rice cultivars in normal conditions was not changed due to drought stress. Tartaric acid was increased 10-fold in treated Gz179 and two-fold in treated Sk101 compared to control. Succinic acid has been detected in control plants and has shown a slight increase in both cultivars under stress. Moreover, glutaric acid displayed an increase in Gz179 and decrease in Sk101 in response to water deficit. In contrast, the accumulation of citric acid was decreased significantly during the drought period.
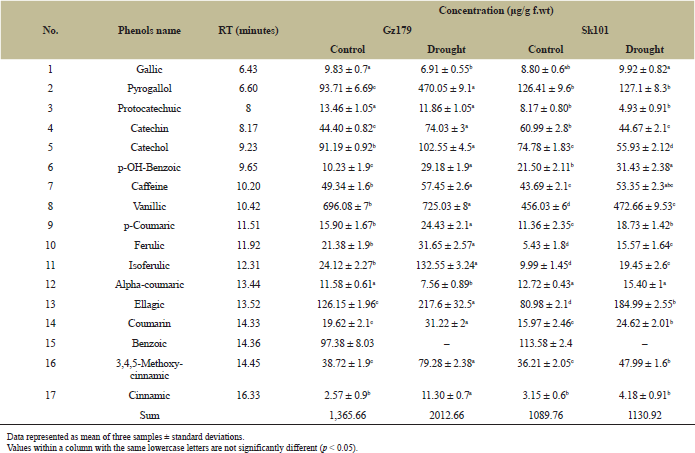 | Table 2: Identification and quantification of phenols in Egyptian rice cultivars Gz179 and Sk101 as determined by HPLC. [Click here to view] |
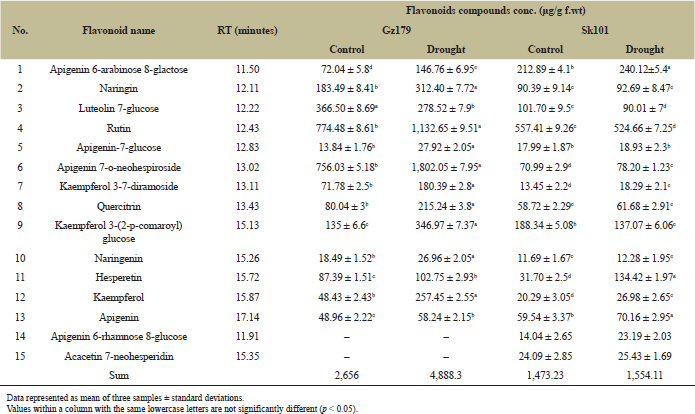 | Table 3: Identification and quantification of flavonoids in Egyptian rice cultivars Gz179 and Sk101 as determined by HPLC. [Click here to view] |
4. DISCUSSION
Rice growth and grain development are severely affected by drought [5]. In order to meet growing global demand, a thorough understanding of the impact of drought stress on rice growth and production is therefore crucial to increasing biomass yields [25]. In our previous work [3], we have shown that the sensitive cultivar Sk101 is more affected by oxidative damage caused by water deficiency than tolerant cultivar Gz179 as indicated by increased level of lipid peroxidation (detected by Malondialdehyde (MDA) content as a decomposition product of polyunsaturated fatty acids), increased permeability (as indicated by electrolyte leakage increase), and loss of cellular integrity (decrease in membrane stability index). Consequently, morphological criteria of plants (especially plant length, leaf area, and leaf number per plant) as well as photosynthetic pigment content were significantly affected in Sk101 compared to Gz179 [3].
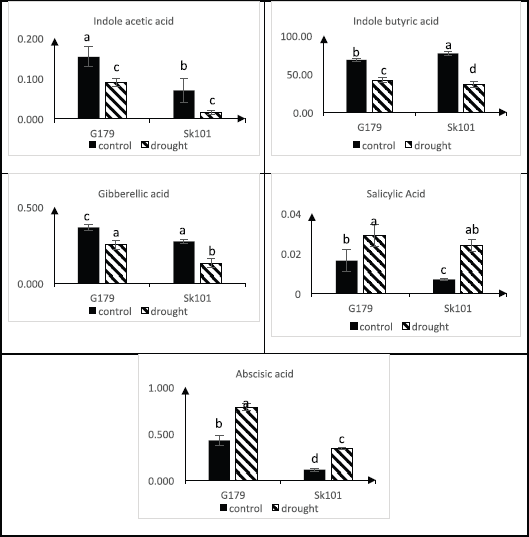 | Figure 3: Changes in plant growth regulators in response to water deficit in two Egyptian rice cultivars (Gz179 and Sk101) differing in their drought tolerance as determined by HPLC. [Click here to view] |
In the current work, the physiological and metabolic responses of these two cultivars were compared: drought tolerant (Gz179) and the drought sensitive (Sk101) for better understanding of drought-tolerant strategies in Egyptian rice.
The development of enzymatic and nonenzymatic antioxidants that scavenge ROS and turn it into harmless molecules is one of the most effective mechanisms for preventing oxidative stress-induced damage in tolerant plants [26]. In this study, we focused on nonenzymatic molecules (such as phenolic acids and flavonoids). Exposing the plant to drought stress improves the secondary metabolites biosynthesis as a part of the nonenzymatic mechanisms. Phenolic compounds are one of the major groups of secondary metabolites. Phenolic compounds show an increase in their accumulation under drought stress [27]. This is clearly evident from our results (Table 2), where tolerant cultivar showed higher accumulation in phenols than sensitive cultivar under the same stress conditions. Tolerant cultivar is characterized by high levels of isoferulic, cinnamic, and pyrogallol. Plant may tolerate the negative effect of drought stress on membranes by increasing ferulic acid that acts as a precursor for oryzanol (one of phytosterol in plant cell), which increases the stabilizing property and strengthens the cell membrane as a result of its polarity and 3D interaction with lipid bilayer membrane [28]. Ferulic acid is also known as the precursor to lignin that protects cell wall from microbial degradation and imparts rigidity to cell wall [29].
Flavonoids are low molecular weight polyphenolic compounds, which play an important role in the growth, development, reproduction, and defense of plants under stress. The current study showed an increase in total flavonoid content following treatment with drought, which is also consistent with the results obtained by Yuan et al. [30] and Liu et al. [31]. As shown from results (Table 3), tolerant cultivar had a high content of flavonoids under normal conditions and has the ability to accumulate more flavonoids under stress compared to sensitive cultivar that has low flavonoid content under control conditions and recorded no marked increase under drought treatment. The most drought-induced flavonoids in tolerant cultivar were kaempferol and quercetrin which indicated that their accumulation was associated with drought tolerance in rice. Similar results were obtained in tomato [32]. The protective role of flavonoids in plant defense against drought stress was achieved through their strong activity in ROS scavenging. Their role achieved by either preventing the ROS generation [33,34] or rapid scavenging of ROS [35] resulted in the justification of oxidative stress under drought condition [36]. The key cause of this accumulation of phenolic compounds is phenylpropanoid biosynthetic pathway modulation. Many important genes that encode master enzymes in the phenylpropanoid pathway, which induce the development of phenolic compounds, are regulated by drought [27].
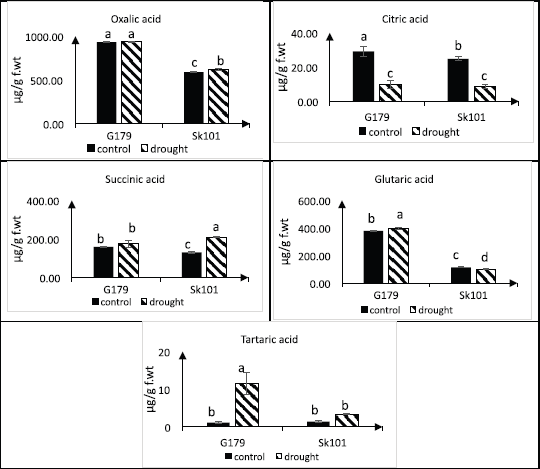 | Figure 4: Changes in organic acids in response to water deficit in two Egyptian rice cultivars (Gz179 and Sk101) differing in their drought tolerance as determined by HPLC. [Click here to view] |
The adaptive role of antioxidants in tolerant cultivar conflict over the efficacy of chlorophyll, photosynthetic rate, and photoassimilate production (such as proline, sugars, sugar alcohols, and organic acid), which accumulated as osmoprotectants. Osmoprotectants are low molecular weight molecules that are highly water-soluble, uncharged, and nontoxic at molar concentrations [37]. They help plants to survive under severe osmotic stress, through osmotic adjustment, which ensures continuous water uptake under drought, maintaining its turgidity and growth. This is another important strategy used by rice plants to cope with reduced water availability under drought stress [38]. Proline highly accumulated in the drought-tolerate cultivar Gz179 (3-fold of control) as one of the metabolic responses; the plants use it under stress. It has several functions as a free radical scavenger, a protective agent for enzymes, and stabilizing the cell membrane and protein structure in addition to its role in osmotic adjustment maintenance [39]. Many researchers have noted a significant increase in proline concentration due to drought stress in many plants such as wheat [40], peanut [41], soybean [42], and rice [43].
Sugars play a vital role in normal plant growth and development, and their contents are greatly increased under drought stress (Table 2), acting as osmoprotective agents to adjust osmotic pressure and overcome water shortages, in addition to their role in secondary metabolites biosynthesis (glycosylation process), by which active sugar hexoses such as glucose, galactose, and rhamnose and pentoses such as arabinose and xylose have been transferred to other molecules such as flavonoid in flavonoid modification process to increase the stability and solubility of hydrophobic flavonoids. In addition, its reaction with lipid to form galactolipid, which was considered to be the major lipid in plant cell membranes and the most abundant in chloroplast membrane, increased its content under drought stress to increase the stability of the thylakoid membranes, thus maintaining the efficiency of photosynthesis [44]. Tolerant rice cultivar is characterized by a significant increase in mannitol accumulation in response to drought that was not shown in sensitive cultivar. In addition, the increase in stachyose, sucrose, and galacturonic in tolerant cultivar was much higher than that detected in sensitive one in response to water deficit. Similar results were obtained by Gundaraniya et al. [45] who noted the accumulation of sugar alcohols such as myo-inositol and D-mannitol in the leaf sample of drought-tolerant peanut (Arachis hypogaea L.) cultivar in response to water stress. Sugar alcohols are osmoprotectants as well as potent ROS quenchers and accumulated in different concentrations under water stress. The hydroxyl group of sugar alcohol can substitute the hydroxyl group of water during interaction with membrane lipids and proteins, maintaining their structure and properties under drought conditions.
Complex biochemical and physiological strategies have been established by rice plants to allow them to respond to sudden changes in climate [46]. Most of these adaptations are regulated by plant hormones that respond to changing environmental conditions by altering signal transduction to control and organize both growth and/or stress tolerance in order to promote survival or escape from environmental stress [47]. Du et al. [48] found that the equilibrium between auxin and ABA biosynthesis played a key part in the plant’s response to stress. Auxin biosynthesis is known to be monitored through the downregulation of YUCCAs (IAA biosynthesis genes), as well as the upregulation of IAA-conjugated genes (GH3s) [49]. The current results proved that auxins (IAA and IBA) content was significantly decreased, whereas ABA contents were markedly elevated in response to water deficit (Fig. 2); the changes were much more pronounced in sensitive cultivar. The observed changes in phenolic concentration such as kaempferol and quercetrin (increases with 5.3- and 2.7-fold, respectively, in tolerant cultivar) as shown in Table 2 acting as auxin transport inhibitor that influences the cycling of auxin efflux carriers and alters the apoplastic pH affecting the intake and distribution of auxin [50]. Meanwhile, the associated increment in ABA level under drought stress was attributed to upregulation of nine-cis-epoxycarotenoid dioxygenase 3 (NCED3), which acts as a key enzyme in ABA biosynthesis in Arabidopsis [51] and in rice [52] and to stimulate ABA-glucose deconjugation [53]. This increase in ABA concentration led to an increase in potassium ion efflux, a loss of turgor in guard cells, and, consequently, stomatal closure [54]. ABA signaling pathway can be considered important to the drought resistance of rice since higher levels of ABA were observed in drought-tolerant cultivar under both normal and stress conditions in the present study. Similar results were obtained by You et al. [55].
SA plays multiple roles in cell metabolism and also plays a key role in modulating the response of plants to abiotic stress by enhancing antioxidant enzymes and reducing ROS molecules [56]. SA, like ABA, is involved in stomatal regulation through Ca2+-dependent protein kinases located downstream of the peroxidase-mediated ROS signaling pathway in Arabidopsis guard cells [57]. In the current study, a further increase (as a percentage of control) in SA was detected in sensitive cultivar Sk101 that could be a coping strategy to overcome the impact of oxidative damage caused by drought.
Organic acids present in normal cells as intermediates to primary and secondary metabolite pathways and increase under drought stress to act as osmoprotectants. According to the results obtained (Fig. 4), tartaric acid showed a significant increase in the tolerant cultivar compared to that was detected in the sensitive one. Furthermore, the tolerant rice cultivar Gz179 was characterized by an increase in glutaric acid that was not detected in Sk101. Guo et al. [58] worked on wheat and stated that, in order to avoid drought stress, tartaric acid accumulates to sustain intracellular ion equilibrium and nutrient uptake.
5. CONCLUSION
Developing genetically modified rice plants with improved resistance and yield under adverse conditions remains a challenge. One major task is to define the metabolomic and transcriptomic changes that are responsible for the tolerance of abiotic stress in rice. The current study investigated the metabolomic differences between tolerant and sensitive Egyptian rice cultivars in response to drought stress. We found that the tolerant cultivar is characterized by a vast array of metabolites that have not been detected or less accumulated in the sensitive cultivar and appears to have a key role in ROS scavenging, osmotic adjustment, strengthening cell cytoskeleton, and stabilizing cell membrane properties, and hence drought tolerance. These metabolomes include both primary and secondary metabolites, namely, proline, sugars (mannitol, stachyose, sucrose, and galacturonic), organic acids (tartaric and glutaric), phenols (isoferulic, cinnamic, and pyrogallol), and flavonoids (kaempferol and quercetrin). Induction of these metabolites is responsible, at least in part, for drought stress tolerance in Egyptian rice cultivar Gz179 and can be used as efficient biomarkers.
7. FUNDING
The work was supported by Ain Shams University.
8. AUTHOR CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work. All the authors are eligible to be an author as per the international committee of medical journal editors (ICMJE) requirements/guidelines.
9. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
10. ETHICAL APPROVALS
Not applicable.
11. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Dien DC. Proline and carbohydrate metabolism in rice varieties (Oryza sativa L.) under various drought and recovery conditions. Plant Physiol Rep 2019;24(3):376–87. CrossRef
2. Mishra BK, Chaturvedi GS, Hindu B. Flowering stage drought stress resistance in upland rice in relation to physiological, biochemical traits and yield. Int J Curr Microbiol Appl Sci 2018;7(2):71–82. CrossRef
3. Ibrahim EK, Hashem HA, Mahmoud RA, Hassanein AA. Comparative physiological study on six egyptian rice cultivars differing in their drought stress tolerance. Acta Sci Agric 2019;3(3):44–52.
4. Farooq M, Hussain M, Siddique KHM. Drought stress in wheat during flowering and grain-filling periods. Crit Rev Plant Sci 2014;33:331–49. CrossRef
5. Larkunthod P, Nounjan N, Jonaliza L, Toojinda T, Sanitchon J, Theerakulpisut P. Physiological responses under drought stress of improved drought-tolerant rice lines and their parents. Not Bot Hortic Agrobotanici 2018;46(2):679–87. CrossRef
6. Vanisri S, Sreedhar M, Jeevan L, Pavani A, Chaturvedi A. Evaluation of rice genotypes for chlorophyll content and scavenging enzyme activity under the influence of mannitol stress towards drought tolerance. Int J Curr Microbiol Appl Sci 2017;6(12):2907–17. CrossRef
7. Dat J, Vandenabeele S, Vranová E, Montagu MV, Inz,é D, Breusegem FV. Dual action of the active oxygen species during plant stress responses. Cell Mol Life Sci 2000;57(5):779–95. CrossRef
8. Helena M, Carvalho CD. Drought stress and reactive oxygen species. Plant Signal Behav 2008;3(3):156–65. CrossRef
9. Mattos LM, Moretti CL. Oxidative stress in plants under drought conditions and the role of different enzymes. Enzyme Eng 2015;5(1): 1–6. CrossRef
10. Fahad S, Bajwa AA, Nazir U, Anjum SA, Farooq A. Crop production under drought and heat stress: plant responses and management options. Front Plant Sci 2017;8:1–16. CrossRef
11. Pereira A, Basu S, Ramegowda V, Kumar A. Plant adaptation to drought stress. F1000Res 2016;5:1–10. CrossRef
12. Gao D, Gao Q, Xu HY, Ma F, Zhao CM, Liu Q. Physiological responses to gradual drought stress in the diploid hybrid Pinus densata and its two parental species. Trees 2009;23:717–28. CrossRef
13. Wapna SS, Hylaraj KSS. Screening for osmotic stress responses in rice varieties under drought condition. Rice Sci 2017;24(5):253–63. CrossRef
14. Hoagland DR, Arnon DI. The water-culture method for growing plants without soil. Calif Agric 1950;347:1–32.
15. Bates L, Waldren RP, Teare ID. Rapid determination of free proline for water-stress studies. Plant Soil 1973;39(1):205–7. CrossRef
16. Chinnici F, Spinabelli U, Riponi C, Amati A. Optimization of the determination of organic acids and sugars in fruit juices by ion-exclusion liquid chromatography. J Food Compost Anal 2005;18:121–30. CrossRef
17. JakopiÄ J, VeberiÄ R, Štampar F. Extraction of phenolic compounds from green walnut fruits in different solvents. Acta Agric Slov 2009;93(1):11–5. CrossRef
18. Marja PK, Anu IH, Heikki JV, Jussi-Pekka R, Kalevi P, Tytti SK, et al. Antioxidant activity of plant extracts containing phenolic compounds. J Agric Food Chem 1999;47(10):3954–62. CrossRef
19. Schieber A, Keller P, Carle R. Determination of phenolic acids and flavonoids of apple and pear by high-performance liquid chromatography. J Chromatogr A 2001;910:265–73. CrossRef
20. Hertog MGL, Hollman JPCH, Martijn BK. Content of potentially anticarcinogenic flavonoids of 28 vegetables and 9 fruits commonly consumed in the netherlands. J Agric Food Chem 1992;40:2379–83. CrossRef
21. Mattila P, Astola J, Kumpulainen J. Determination of flavonoids in plant material by HPLC with diode-array and electro-array detections. J Agric Food Chem 2000;48:5834–41. CrossRef
22. Ma L, Zhang H, Xu W, He, X. Simultaneous determination of 15 plant growth regulators in bean sprout and tomato with liquid chromatography – triple quadrupole tandem mass spectrometry. Food Anal Methods 2013;6:941–51. CrossRef
23. Wodecki ZJ, Torlop B, Slebioda M. Chromatographic determination of citric acid for monitoring the mould process. J Chromatogr A 1991;558:302–5. CrossRef
24. Kirk-Patrick LA, Feeney BC. A simple guide to IBM SPSS statistics for version 20.0. student Ed. Wadsworth, Cengage Learning, Belmont, CA, 2013.
25. Zhang J, Zhang S, Cheng M, Jiang H, Zhang X, Peng C, et al. Effect of drought on agronomic traits of rice and wheat: a meta-analysis. Int J Environ Res Public Health 2018;15(5):839–53. CrossRef
26. Khan A, Numan M, Khan AL, Lee I, Imran M, Asaf S, et al. Melatonin : awakening the defense mechanisms during plant oxidative stress. Plants 2020;9(4):1–22. CrossRef
27. Sharma A, Shahzad B, Rehman A, Bhardwaj R, Landi M. Response of phenylpropanoid pathway and the role of polyphenols in plants under abiotic stress. Molecules 2019;24:1–22; doi:10.3390/molecules24132452 CrossRef
28. Khan F, Upreti P, Singh R, Shukla PK. Physiological performance of two contrasting rice varieties under water stress. Physiol Mol Biol Plants 2017;23(1):85–97. CrossRef
29. Vanholme R, Demedts B, Morreel K, Ralph J, Boerjan W. Lignin biosynthesis and structure. Plant Physiol 2010;153(3):895–905. CrossRef
30. Yuan Y, Liu Y, Wu C, Chen S, Wang Z, Yang Z, et al. Water deficit affected flavonoid accumulation by regulating hormone metabolism in scutellaria baicalensis georgi roots. PLoS One 2012;7(10):1–10. CrossRef
31. Liu M, Li X, Liu Y, Cao B. Regulation of flavanone 3-hydroxylase gene involved in the flavonoid biosynthesis pathway in response to UV-B radiation and drought stress in the desert plant, Reaumuria soongorica. Plant Physiol Biochem 2013;73:161–7. CrossRef
32. Sanchez-Rodriguez E, Moreno DA, Ferreres F, Rubio WM, Ruiz JM. Differential responses of five cherry tomato varieties to water stress: changes on phenolic metabolites and related enzymes. Phytochemistry 2011;72:723–9. CrossRef
33. Melidou M, Riganakos K, Galaris D. Protection against nuclear DNA damage offered by flavonoids in cells exposed to hydrogen peroxide: the role of iron chelation. Free Radic Biol Med 2005;39(12):1591–600. CrossRef
34. Agati G, Tattini M. Multiple functional roles of flavonoids in photoprotection. New Phytol 2010;186(4):786–93. CrossRef
35. Jaakola L, Hohtola A. Effect of latitude on flavonoid biosynthesis in plants. Plant Cell Environ 2010;33(8):1239–47. CrossRef
36. Nakabayashi R, Mori T, Saito K. Alternation of flavonoid accumulation under drought stress in arabidopsis thaliana. Plant Signal Behav 2014;9(8):1–3. CrossRef
37. Kumar R. Role of naturally occurring osmolytes in protein folding and stability. Arch Biochem Biophys 2009;491:1–6. CrossRef
38. Szabados L, Savouré A. Proline: a multifunctional amino acid. Trends Plant Sci 2010;15(2):89–97. CrossRef
39. Liang X, Zhang L, Natarajan SK, Becker DF. Proline mechanisms of stress survival. Antioxid Redox Signal 2013;19(9):998–1011. CrossRef
40. Itam M, Mega R, Tadano S, Abdelrahman M, Matsunaga S, Yamasaki Y, et al. Metabolic and physiological responses to progressive drought stress in bread wheat. Sci Rep 2020;10:17189; doi:10.1038/s41598-020-74303-6 CrossRef
41. Solanki J. Effect of drought stress on epicuticular wax load in peanut genotypes. J Appl Biol Biotechnol 2015;2(10):301–9.
42. Mwenye OJ, Rensburg L, Biljon A, Merwe R. The role of proline and root traits on selection for drought-stress tolerance in soybeans: a review. S Afr J Plant Soil 2016;33(4):245–56. CrossRef
43. Dien DC, Mochizuki T, Yamakawa T. Effect of various drought stresses and subsequent recovery on proline, total soluble sugar and starch metabolisms in rice (Oryza sativa L) varieties. Plant Prod Sci 2019;22(4):530–45. CrossRef
44. Shimojima M, Ohta H. Critical regulation of galactolipid synthesis controls membrane differentiation and remodeling in distinct plant organs and following environmental changes. Prog Lipid Res 2011;50(3):258–66. CrossRef
45. Gundaraniya SA, Ambalam PS, Tomar RS. Metabolomic profiling of drought-tolerant and susceptible peanut (Arachis hypogaea L.) genotypes in response to drought stress. ACS Omega 2020;5(48):31209–19; doi:10.1021/acsomega.0c04601 CrossRef
46. Krasensky J, Jonak C. Drought, salt, and temperature stress-induced metabolic rearrangements and regulatory. J Exp Bot 2015;63(4):1593–608. CrossRef
47. Du H, Wu N, Fu J, Wang S, Li X, Xiao J, et al. A GH3 family member, OsGH3-2, modulates auxin and abscisic acid levels and differentially affects drought and cold tolerance in rice. J Exp Bot 2012;63(2):695–709. CrossRef
48. Du H, Wu N, Chang Y, Li X. Carotenoid deficiency impairs ABA and IAA biosynthesis and differentially affects drought and cold tolerance in rice. Plant Mol Biol 2013a;83:475–88. CrossRef
49. Du H, Liu H, Xiong L. Endogenous auxin and jasmonic acid levels are differentially modulated by abiotic stresses in rice. Front Plant Sci 2013b;4:1–10. CrossRef
50. Potters G, Pasternak TP, Guisez Y, Jansen MAK, Simrs R. Different stresses, similar morphogenic responses: integrating a plethora of pathways. Plant Cell Environ 2009;32:158–69. CrossRef
51. Cheng WH, Endo A, Zhou L, Penney J, Chen HC, Arroyo A, et al. A unique short-chain dehydrogenase/reductase in Arabidopsis glucose signaling and abscisic acid biosynthesis and functions. Plant Cell 2002;14(11):2723–43. CrossRef
52. Ye N, Zhu G, Liu Y, Li Y, Zhang J. ABA controls H2O2 accumulation through the induction of OsCATB in rice leaves under water stress. Plant Cell Physiol 2011;52(4):689–98. CrossRef
53. Seiler C, Harshavardhan VT, Rajesh K, Reddy PS, Strickert M, Rolletschek H, et al. ABA biosynthesis and degradation contributing to ABA homeostasis during barley seed development under control and terminal drought-stress conditions. J Exp Bot 2011;62(8):2615–32. CrossRef
54. Hubbard KE, Nishimura N, Hitomi K, Getzoff ED, Schroeder JI. Early abscisic acid signal transduction mechanisms: newly discovered components and newly emerging questions. Genes Dev 2010;24(16):1695–708. CrossRef
55. You J, Zhang Y, Liu A, Li D, Wang X, Dossa K, et al. Transcriptomic and metabolomic profiling of drought-tolerant and susceptible sesame genotypes in response to drought stress. BMC Plant Biol 2019;19:267. CrossRef
56. Ganesan V, Thomas G. Salicylic acid response in rice: influence of salicylic acid on H2O2 accumulation and oxidative stress. Plant Sci 2001;160(6):1095–106. CrossRef
57. Prodhan MY, Munemasa S, Nahar MN, Nakamura Y, Murata Y. Guard cell salicylic acid signaling is integrated into abscisic acid signaling via the Ca2+/CPK-dependent pathway. Plant Physiol 2018;178:441–50. CrossRef
58. Guo R, Shi L, Jiao Y, Li M, Zhong X, Gu F, et al. Metabolic responses to drought stress in the tissues of drought-tolerant and drought-sensitive wheat genotype seedlings. AoB Plants 2018;10(2):1–13. CrossRef