1. INTRODUCTION
Vitamin D, widely known as “sunshine vitamin,” is a vital component as it helps the human body in metabolizing calcium and phosphorus elements from food [1]. It is reported that 70% of the global population is deficient in Vitamin D level, whereas more than 95% of Indian grown-ups are having below <75 nmol [2]. Vitamin D deficiency in the human body causes health disorders related to bones such as rickets in teenagers and osteoporosis in grown-ups [3]. It is also reported that it can protect the human body from many types of cancers, diabetes, multiple sclerosis, inflammatory bowel diseases, muscular degeneration, mental illness, obesity, and heart diseases [3,4]. Vitamin D is also well recognized for its dynamic function against various respiratory diseases [5]. Recent research findings have shown noteworthy correlation between Vitamin D and currently persisting pandemic disease coronavirus disease 2019 (COVID-19), instigated by severe acute respiratory syndrome coronavirus 2 or novel coronavirus. It is reported that adequate levels of Vitamin D in humans are significantly helpful in reducing the mortality rate due to COVID-19 infection [6]. Earlier, it is reported that vitamin D deficiency was one of the major cause in serious COVID-19 patients showing higher virulence effect due to coronavirus [7].
Mushrooms are well acknowledged for non-animal origin food source having a significant concentration of provitamin-D2 (ergosterol) which gets transformed into Vitamin D2 through ultraviolet (UV) irradiation [8]. This process is analogous to the transformation of 7-de-hydrocholesterol present in human skin into cholecalciferol (Vitamin D3) through sun exposure. Vitamin D2 functions similarly to Vitamin D3 [9]. Thus, mushroom can be a potential source of Vitamin D2 preferably for vegans and people with less sunlight exposure.
Pleurotus eryngii (King Oyster) is a medicinal mushroom possessing various nutraceutical components including ergosterol (Vitamin D2 precursor), polysaccharides (i.e., beta-glucans), antioxidants (i.e., ergothioneine), terpenoids, and essential amino acids [10]. There are no previous studies available on enhancing Vitamin D2 in submerged cultivated P. eryngii mycelia using optimized UVB irradiated conditions. Accordingly, the present study was focused on optimizing the parameters for transforming ergosterol into Vitamin D2 in fresh P. eryngii mushroom mycelia obtained through submerged fermentation technique using response surface methodology (RSM) and thus enhancing the practical application of Vitamin D2 enriched mushroom mycelia in food processing and pharma industries.
2. MATERIALS AND METHODS
2.1. Chemicals
Standard of ergocalciferol (Vitamin D2) was obtained from Sigma-Aldrich (St. Louis, USA). Sodium hydroxide, potassium hydroxide, sodium L-ascorbate, ethanol, methanol, acetonitrile, and n-pentane used in this study were of high purity (>99%) chemicals purchased from Merck (Mumbai, India). The potato dextrose agar and broth were purchased from HiMedia (Mumbai, India).
P. eryngii Mycelia Cultivation
P. eryngii (18.K07) mycelial culture was obtained from Haryana Agro Research Centre, India, and maintained in potato dextrose agar plates. The seed culture was prepared in 100 ml potato dextrose broth medium (24 g/l) by transferring two 8 mm punches of mycelia taken from a full grown fresh agar plate using a cork borer, properly crushed using an Ultra-Turrax (IKA, Germany) homogenizer for 20 s at 12,000 rpm and incubated under aerobic conditions for 4 days at 130 rpm and 28°C. The production media composition was kept as described in Singh et al. 2020 [11]. About 4% homogenized inoculum was used to inoculate production media. The mycelia production was accomplished in 10 days with complete consumption of carbon source. The produced mycelia were collected, washed thrice using deionized water, and filtered (Whatman paper no.1, Sigma-Aldrich, USA).
2.3. UV Irradiation Set-up
The UV exposure set-up includes three 1.75 m long (100 W) UV-B tubes (UV Narrowband TL 100, Phillips) having a definite band of 290–315 nm (311 nm peak) (Wittig et al., 2013), was used for exposure of mycelia. The UV exposure experiments were performed using fresh homogenized mycelial biomass (50 ml) in glass Petri dishes (ID = 15 cm diameter). The sampling was done by removing the whole set of Petri plates (n = 3) for analysis. The samples were lyophilized after UV treatment and kept at −20°C till further analysis. The sample without UV treatment was set as a control. All the calculations were done on a dry cell weight basis.
2.4. Analysis of Vitamin D2 and Mycelial Nutritional Quality
Vitamin D2 (present in mycelia) was extracted and analyzed as described by Singh et al., 2020. Precisely, to 0.5 g powdered (freeze-dried) mycelia, 4 ml of sodium ascorbate solution (17.5% w/v in 1 M NaOH), 50 ml ethanol, and 10 ml of 50% (w/v) KOH were added in a 250 ml round bottom flask. This mixture was refluxed at 80°C for 1 h and then cooled to 25°C. The refluxed mixture was extracted in a separating funnel with distilled water (50 ml), ethanol (15 ml), and n-pentane (50 ml, 50 ml, and 20 ml), respectively. The clear organic layer obtained was washed thrice with 50 ml of 3% (w/v) KOH in 5% (v/v) ethanol and, thereafter, neutralized with the deionized water. The organic layer was evaporated at 40°C to dryness in a rata vapor. The residue left was redissolved in 10 ml HPLC-grade methanol (ultrasonicate for 5 min) and filtered through a 0.45 μm filter. Vitamin D2 concentration was determined using a reverse phase C18 column (Shimadzu) under a UV detector (264 nm) in an Agilent-1260 HPLC system. Acetonitrile/methanol mixture (75:25, v/v) was used as mobile solvent at a flow rate of 1.0 mL/min.
The β-glucan content in the freeze-dried mycelia was quantified using a yeast-mushroom β-glucan kit (Megazyme, Ireland). Reducing power was calculated using a method reported by Reis et al., 2012 [12]. Crude protein and crude fat were analyzed [13]. Micro-Kjeldahl method (AOAC-925.10) was used for calculating crude protein with a conversion factor of 6.25 [14], crude fat (AOAC-2003.05) was analyzed through Soxhlet extraction with hexane. All the calculations were made on a dry weight basis.
2.5. Selection of Irradiation Conditions
Preliminary experiments were performed to select an appropriate exposure time, UV-B intensity, and ambient temperature. First, a range of temperature (20–50°C) selected keeping exposure time and UV-B intensity constant at 20 min and 0.6 W/m2 , respectively. Second, UV-B intensity varied from 0.3 to 1.2 W/m2 , and the optimal temperature was taken as 30°C from the first step, keeping exposure time constant at 20 min. Finally, using optimal temperature and intensity obtained from the previous two steps, exposure time varied from 10 to 40 min. On the basis of these results, the upper, middle, and lower levels of the independent factors were selected for RSM.
2.6. Design of Experiments and Statistical Optimization using RSM
The RSM was used for the statistical optimization of process parameters – experimental temperature (A, °C), duration of exposure time (B, min), and intensity of UV-B irradiation (C, W/m2 ) for ergosterol to Vitamin D2 conversion. A central composite design was applied for three variables having replicates at the midpoint. A total of 20 experimental trials were designed and the Vitamin D2 concentration (μg/g, dry biomass) was set as a response variable with a mean of triplicate values. For experimental design and validation of a model for the experimental conditions – Design Expert (12th version; Stat-Ease Inc., USA) was employed in the present study.
3. RESULTS AND DISCUSSION
3.1. Selection of Parameter for Vitamin D2 Synthesis
The effect of the type of UV radiation (A, B, and C) has been studied by many authors [15-17] and it is evident from these studies that the maximum Vitamin D2 formation from ergosterol present on fungal mushrooms is obtained by UV-B irradiation. Hence, UV-B irradiation was selected for Vitamin D2 synthesis in P. eryngii mycelia.
Preliminary experiments have been performed to estimate the upper, middle, and lower levels of each factor and the outcomes are shown in Table 1. From the results, it was clear that the Vitamin D2 content increased until the surrounding temperature reached to 30°C. The study conducted by Jasinghe and Perera, 2006, on mushrooms fruiting bodies reported that the optimum temperature for Vitamin D2 was 35°C while Lee and Aan, 2016, conducted a study on Agaricus bisporus mushroom powder and found a temperature of 26.3°C for maximum Vitamin D2 synthesis. It has also been observed that as the temperature increases, mycelial surface turns brown which could also negatively affect the Vitamin D2 synthesis process [18].
Thus, for experimental design, 30°C was taken as temperature midpoint and lower and upper values were set at 20 and 40°C, respectively. To study the effect of exposure time, the temperature and intensity of UV irradiation were fixed at 30°C and 0.6 W/m2 with exposure time varying from 10 to 40 min. The results showed [Table 1] that Vitamin D2 content enhanced as the exposure time increased and accordingly the three levels of exposure time were chosen for RSM as 10, 20, and 30 min, respectively. The analysis showed that with increasing exposure time, Vitamin D2 content increases initially up to 20 min but later on, no significant conversion occurred possibly because of pigmentation on the mycelial surface. A rapid increase in Vitamin D2 was noticed with an increase of irradiation intensity [Table 1]. The preliminary experiments [Table 1] have shown that after an optimum intensity, the rate of conversion begins to decline as higher doses of intensity could possibly induce photodegradation reactions [19], causing a reduction in Vitamin D2 content. Furthermore, it can negatively affect the mycelial moisture content, color, and nutritional quality [20]. Hence, irradiation intensities for RSM were taken as 0.6, 0.9, and 1.2 W/m2 .
3.2. RSM Model Fitting
A central composite rotatable design was used to determine the effect of physical parameters, that is, irradiation intensity, ambient temperature, and exposure time for conversion of ergosterol present on the cell membrane of P. eryngii mycelia into Vitamin D2. The design of experiments and obtained outcomes are given in Table 2. A multiple regression analysis was conducted, and the concentration of Vitamin D2 in P. eryngii mycelia was assessed with the help of a second-order polynomial equation stated below:
Y = 324.41 - 24.61A + 19.67B + 28.46C - 12.68AB - 7.92AC - 6.83BC - 28.11A2 –26.46B2 – 20.51C2 (1)
 | Table 1: Effect of different parameters on Vitamin D2 formation in Pleurotus eryngii mycelia. [Click here to view] |
The significance of fitting of the second-order polynomial equation was determined using the analysis of variance (ANOVA) for the assessment of experimental values, as depicted in Table 3. The “F-value” of the model is 6.42 and P = 0.0077 indicates the significance of the model. In this model, values for individual variables and their interactions are significant, as shown in Table 3. It is clearly visible from Table 2 that irradiation intensity (C) is the most significant factor affecting the ergosterol conversion into Vitamin D2 in P. eryngii mushroom mycelia. The determination coefficient (R2 ) of the model is 0.8785 which indicates that the variability of this model can be 87%. The “lack of fit F-value” of 5.34 represents not a significant lack of fit compared to the pure error. Thus, the projected model fits satisfactorily to the data obtained through experiments. The “Adeq. Precision” is utilized to determine the ratio between response and deviation, and a ratio beyond 4 indicates an acceptable signal. In the present investigation, the ratio was 7.072 indicating that the signal was adequate and thus the above model could be further utilized to direct the design space.
Figure 1a shows the interaction effects of temperature (20–40°C) and exposure time (10–30 min) along with a set UV-B intensity of 0.9 W/m2 . It indicates the quadratic effect of two variables on the Vitamin D2 synthesis.
Figure 1b indicates that under lower values of temperature, the irradiation intensity resulted in a linear rise in the Vitamin D2 synthesis while exposure time was fixed at 20 min. Furthermore, the ambient temperature shows a quadratic effect on the outcome. Figure 1c indicates the effects of the interactions between exposure time and UV-B intensity with a similar tendency.
 | Table 2: Experimental design and central composite design results. [Click here to view] |
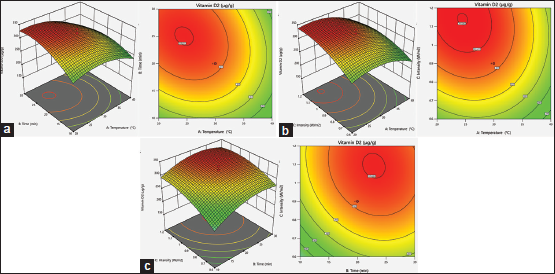 | Figure 1: Vitamin D2 contour plots for synthesis in Pleurotus eryngii mycelia. A: Impacts of temperature and exposure time on Vitamin D2 production in P. eryngii mycelia at a fix intensity of 0.9 W/m2 ; B: Impacts of temperature and intensity on Vitamin D2 production in P. eryngii mycelia at a fix time of 20 min; C: Impacts of exposure time and intensity on Vitamin D2 production in P. eryngii mycelia at a fix temperature of 30°C. [Click here to view] |
The optimum operating conditions for Vitamin D2 synthesis are 24.29°C, 23.88 min, and 1.099 W/m2 for ambient temperature, exposure time, and UV-B intensity, respectively, and the predicted Vitamin D2 synthesis using these optimized parameters was 346.84 μg/g, with a confidence level of 95%.
Ergosterol, when exposed to UV irradiation first, converts into unstable pre-Vitamin D after the opening of its “B-ring” and then gets rearranged into Vitamin D2 along with various conformational isomeric forms of photoproducts such as tachysterol, lumisterol, Vitamin D4, suprasterol, and toxysterol [21]. The rate of formation of Vitamin D2 is a function of temperature, type of UV, and the intensity of UV, thus, it is imperative to expose fungal mycelia with the optimum doses of the same. Ergosterol to Vitamin D2 conversion is also limited due to its further conversion into hydroxylated forms possibly by monooxygenases or through oxidative reactions [22]. The ergosterol to Vitamin D conversion efficiency increases when mushrooms are homogenized before UV exposure possibly because of increases in the surface area exposed [18].
3.3. Validation of the Model
The experiments for validation were conducted at the predicted conditions obtained through RSM analysis. The Vitamin D2 concentration in P. eryngii mushroom mycelia was 329.43±5.32 μg/g with a confidence level of 95% for the projected maximum concentration (346.84 μg/g). The satisfactory correlation in the experimental and predicted values validates the effectiveness and applicability of the model suggested in the present study.
3.4. Impact of UV-B Irradiation on the Nutritional Quality of P. eryngii Mycelia
Contents of β-glucans and reducing power were determined in model optimized UV-B treated mushroom mycelia of P. eryngii and the corresponding values were compared with the untreated (control) mycelia [Table 4]. β-glucan in UV-B treated mycelia 29.43±0.78% was found to be increased in comparison to untreated mycelia 26.02±0.42%, which indicates the elicitation effect of ultraviolet irradiation on the contents of β-glucan. The cell wall of mycelia contains glucans as a major component and the UV irradiation has a stimulating effect on enhancing β-glucan content in mushrooms [23,24].
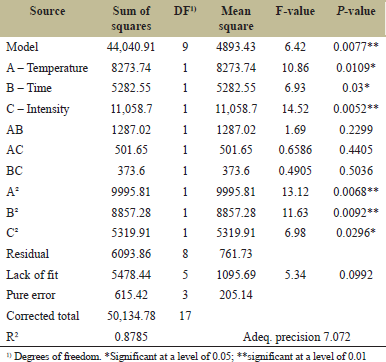 | Table 3: ANOVA results for the response surface quadratic model. [Click here to view] |
 | Table 4: Effect of ultraviolet exposure on nutraceutical content of Pleurotus eryngii mycelia. [Click here to view] |
Reducing power of UV irradiated mycelia (0.98±0.22 mg/ml) was slightly improved than non-irradiated control samples (1.18±0.18 mg/ml). No significant changes were found in the contents of protein, fat, and carbohydrate after mycelial UV treatment [Table 4]. Similar observations in UV irradiated oyster mycelia were also reported by Huang et al. (2015) and Simon et al. (2011). Our results in this regard are corroborated by their findings. A detailed study is still to be done for effects of UV-B irradiation on other bioactive compounds in the submerged cultivated mushroom mycelia.
4. CONCLUSION
The study concluded that the conversion of ergosterol present on the cell wall of P. eryngii mushroom mycelia into Vitamin D2 was significantly affected by optimized parameters (temperature, exposure time, and UV-B irradiation intensity) achieved through RSM (CCD). The optimized conditions of UV-B irradiations also improved other nutraceutical attributes in the mycelia of P. eryngii.
It is evident from this study that submerged cultivated mushroom mycelia can be a good alternative as a resource of Vitamin D2 in comparison to fruiting bodies of mushrooms and hence can be utilized in food processing and pharma industry for the manufacturing of Vitamin D2 enriched food products and health supplements, respectively. Further, the study unlocks the opportunities for enhancing Vitamin D2 as well as improving inherent important nutraceutical properties of many other kinds of mushroom mycelia through this tested statistical tool for health benefits.
5. CONFLICTS OF INTEREST
Authors declare that no conflicts of interest exist.
6. FUNDING
This work was funded by the Ministry of Food Processing Industries, GOI (Sanctioned number F. No. Q-11/9/2018-R&D dt. December 17, 2018).
7. AUTHOR CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work. All the authors are eligible to be an author as per the international committee of medical journal editors (ICMJE) requirements/guidelines.
8. ETHICAL APPROVALS
Not applicable.
9. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Ko JA, Lee BH, Lee JS, Park HJ. Effect of UV-B exposure on the concentration of Vitamin D2 in sliced shiitake mushroom (Lentinula edodes) and white button mushroom (Agaricus bisporus). J Agric Food Chem 2008;56:3671-4. CrossRef
2. Palacios C, Gonzalez L. Is Vitamin D deficiency a major global public health problem? J Steroid Biochem Mol Biol 2014;144 Pt A:138-45. CrossRef
3. Holick MF. Medical progress: Vitamin D deficiency. N Engl J Med 2007;357:266-81. CrossRef
4. Kalaras MD, Beelman RB, Elias RJ. Effects of postharvest pulsed uv light treatment of white button mushrooms (Agaricus bisporus) on Vitamin D2 content and quality attributes. J Agric Food Chem 2012;60:220-5. CrossRef
5. Taofiq O, Fernandes Â, Barros L, Barreiro MF, Ferreira IC. UV-irradiated mushrooms as a source of Vitamin D2: A review. Trends Food Sci Technol 2017;70:82-94. CrossRef
6. Ilie PC, Stefanescu S, Smith L. The role of Vitamin D in the prevention of coronavirus disease 2019 infection and mortality. Aging Clin Exp Res 2020;6:1-4. CrossRef
7. Munshi R, Hussein MH, Toraih EA, Elshazli RM, Jardak C, Sultana N, et al. Vitamin D insufficiency as a potential culprit in critical COVID-19 patients. J Med Virol 2020;93:733-40. CrossRef
8. Roberts JS, Teichert A, McHugh TH. Vitamin D2 formation from post-harvest UV-B treatment of mushrooms (Agaricus bisporus) and retention during storage. J Agric Food Chem 2008;56:4541-4. CrossRef
9. Koyyalamudi SR, Jeong SC, Pang G, Teal A, Biggs T. Concentration of Vitamin D2 in white button mushrooms (Agaricus bisporus) exposed to pulsed UV light. J Food Compos Anal 2011;24:976-9. CrossRef
10. Rathore H, Prasad S, Sharma S. Mushroom nutraceuticals for improved nutrition and better human health: A review. PharmaNutrition. 2017;5:35-46. CrossRef
11. Singh U, Gautam A, Singha TK, Tiwari A, Tiwari P, Sahai V, et al. Mass production of Pleurotus eryngii mycelia under submerged culture conditions with improved minerals and Vitamin D2. Lwt 2020;131:109665. CrossRef
12. Reis FS, Martins A, Barros L, Ferreira IC. Antioxidant properties and phenolic profile of the most widely appreciated cultivated mushrooms: A comparative study between in vivo and in vitro samples. Food Chem Toxicol 2012;50:1201-7. CrossRef
13. Association of Official Analysis Chemists International. Official Methods of Analysis of AOAC International. Rockville, Maryland: Association of Official Analysis Chemists International; 2005.
14. Lowry OH, Randall RJ. Protein measurement byt the folin reagent. J Biol Chem 1951;193:265-75. CrossRef
15. Jasinghe VJ, Perera CO. Ultraviolet irradiation: The generator of Vitamin D2 in edible mushrooms. Food Chem 2006;95:638-43. CrossRef
16. Abillon E, Mermet-Bouvier R. Effect of wavelength on production of previtamin D2. J Pharm Sci 1973;62:1688-91. CrossRef
17. Wu WJ, Ahn BY. Statistical optimization of ultraviolet irradiate conditions for Vitamin D2 synthesis in oyster mushrooms (Pleurotus ostreatus) using response surface methodology. PLoS One 2014;9:e95359. CrossRef
18. Ahlborn J, Calzolari N, Spielmeyer A, Avci SS, Zimmer M, Rühl M. Enrichment of Vitamin D2 in mycelium from submerged cultures of the agaric mushroom Pleurotus sapidus. J Food Sci Technol 2018;55:3833-9. CrossRef
19. Jasinghe VJ, Perera CO, Sablani SS. Kinetics of the conversion of ergosterol in edible mushrooms. J Food Eng 2007;79:864-9. CrossRef
20. Huang SJ, Lin CP, Tsai SY. Vitamin D2 content and antioxidant properties of fruit body and mycelia of edible mushrooms by UV-B irradiation. J Food Compos Anal 2015;42:38-45. CrossRef
21. Keegan RJ, Lu Z, Bogusz JM, Williams JE, Holick MF. Photobiology of Vitamin D in mushrooms and its bioavailability in humans. Dermatoendocrinol 2013;5:165-76. CrossRef
22. Jasinghe VJ, Perera CO. Distribution of ergosterol in different tissues of mushrooms and its effect on the conversion of ergosterol to Vitamin D2 by UV irradiation. Food Chem 2005;92:541-6. CrossRef
23. Rathore H, Prasad S, Sehwag S, Sharma S. Vitamin D2 fortification of Calocybe indica mushroom by natural and artificial UVB radiations and their potential effects on nutraceutical properties. 3 Biotech 2020;10:41. CrossRef
24. Ryoo R, Sou HD, Ka KH, Park H. Elicitor-induced β-glucan contents in fruit body of cauliflower mushroom (Sparassis latifolia). Forest Sci Technol 2018;14:119-25. CrossRef