1. INTRODUCTION
Synthetic chemicals are widely used as medicines in the treatment of diseases encompassing various side effects. Different plants were explored as a source of bioactive agents for the treatment of ailments like cancer. The plant possesses immense biological properties due to the presence of different chemical substances which perform several important physiological functions. Among 4,22,000 flowering plants reported from the whole world, more than 50,000 plants are reported to have medicinal and pharmacological uses. A rich diversity of medicinal plants is found in India [1]. Utilization of plants for medicinal usage has been documented long back in ancient works of literature. Such documentation of prime traditional knowledge on medicinal plants provided many important drugs of the modern era. Nyctanthes arbor-tristis L. (Oleaceae) is an important medicinal plant having a lot of medicinal uses since ancient times. Different parts of this plant have been used as local and traditional medicines. Nyctanthes arbor-tristis is utilized in Ayurveda, Siddha-Ayurveda, and Yunani systems of medicines as a laxative, diuretic, as anti-venoms, digestives, mild bitter tonic, and expectorant [2]. Nyctanthes arbor-tristis Linn. (Division: Magnoliophyta; Class: Magnoliopsida; Order: Lamiales; Family: Oleaceae), commonly known as Harshingar and Night jasmine [3], is a well-documented plant. It is a shrub or small tree growing to 10 m tall terrestrial woody perennial plant having 5–20 years of lifespan. It is a well-known tree having a wide range of pharmacological activities and is widely cultivated in tropical and subtropical regions all over the world [4,5]. In India, it is distributed in the outer Himalayas and tracts of Jammu and Kashmir, Nepal to the east of Assam, Bengal, Tripura extended through the central region up to the Godavari in the South [6]. It usually grows in red and black soils, having pH of 5.6–7.5 and prefers arid and semi-arid climatic conditions [7]. Previously, the systemic position of the Nyctanthes arbor-tristis was not clear. It was placed in the Oleaceae by Bentham and Hooker. An investigation was taken to assign the best matching family by Vaishampayan and Sharma, 1983. It is being used in folk medicine for antibilious, gynecological troubles, and hepatoprotective activity since the ancient time [8]. In central India, various parts of this plant are used by the tribal people to relieve cough, hiccup, dysentery, snakebite, and sores. In addition, N. arbor-tristis has screened by researchers for antihistaminic, CNS, analgesic and anti-inflammatory, antipyretic, amoebicidal, anthelmintic, antidepressant, antiviral, antioxidant, and anticancer activities [6]. Present review reports a comprehensive collection of studies based on pharmacological, anticancer, and antioxidant activities of different plant parts of N. arbor from 1987 to till date. Reports of some important pure bioactive compounds extracted from different parts of this plant have also documented which can be investigated for their possible use as therapeutic agents in the treatment of cancer. N. arbor-tristis is wonder tree and hub of medicinal properties. The present study may provide information to different research groups to validate its ethnomedicinal uses using advance technologies and to explore the proper mechanism of action of biological compounds found in this plant. This review is prepared to document almost all pharmacological studies performed on this plant in vitro and in vivo till date and second, a detailed survey of antioxidant and anticancer activities including information regarding effective doses of crude extracts as well as isolated pure compounds in Tables 1 and 2, respectively. Isolated pure compounds like 4-hydroxy hexahydrobenzofuran-7-one which is Benzofuran derivative [9], Naringenin (5, 7, 4′-trihydroxy flavonone), a bioflavonone isolated from stem [10], Arbortristoside A and 6-β Hydroxyloganin (Iridoid glycosides) isolated from seeds [11] are reported to possess significant anticancer activities tested on different cell lines. Isolated compounds were chemically transformed to obtained derivatives, which enhance their anticancer potential. The plant is also a rich source of antioxidants which can serve as a source for agents used in cancer therapy. Highly branched polysaccharides which are present in leaves reported for dose-dependent antioxidant potential [12].
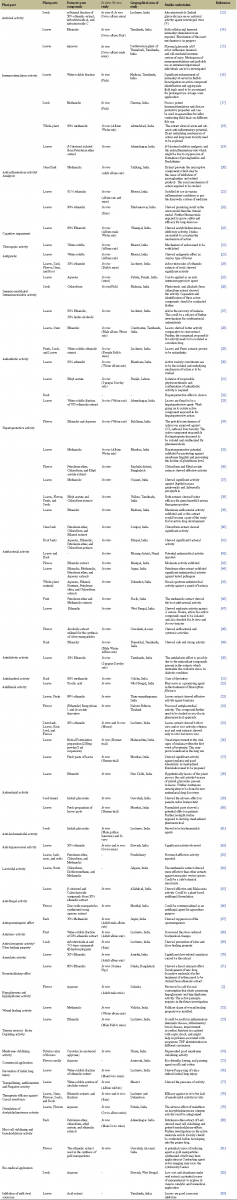 | Table 1: A detailed survey of pharmacological activities of different extracts/compounds from different parts of Nyctanthes arbor-tristis. [Click here to view] |
2. CONCLUSION AND FUTURE PROSPECTS
Indeed, N. arbor is among the wonder of medicinal plants as it has a wide variety of medicinal properties. All the pharmacological studies done so far are of a preliminary kind. These studies need identification and characterization of the bioactive compound and their molecular mechanism elucidation. Studies related to immunostimulatory, immunomodulatory, antipyretic, analgesic, anti-arthritic, hepatoprotective, antistress, anxiolytic, antiulcerogenic, hypoglycemic, and hypolipidemic activities should be checked for its toxicity level and finally for human intervention. Studies like antimalarial, antitrypanosomal, antifilarial, antileishmanial, and larvicidal activities are generally cured effectively with some herbal formulation or paste preparations and crude extracts. One should go for combinatorial formulation with some other similar medicinal property containing plant for better results. Overall, a significant number of in vitro antioxidant studies provide substantial evidence that N. arbor and its ethanolic fraction of acetone (NEA), butanolic extract, and dried extracts of plant parts are effective against the carcinogenic process. Phenolic acid and flavonoids, Naringenin, a bioflavone, should be studied In vitro and in vivo for its anticancer activity and further steps for molecular chemistry and molecular docking analysis to gain an insight into its molecular interactions with cellular component would give the path for designing drug and then finally human trials against human cancers. Only a few references were found explaining the molecular mechanism of isolated compounds. Hence, rest of the study on the antioxidant and anticancer activity should be undertaken for research to address responsible phytoconstituents and its mechanism of actions which would support the said therapeutic action and then in vivo study will give the lead for drug development. Further research should focus on how genetic variability and environmental factors influence on its anticancer property attributed to N. arbor. Moreover, molecular marker-assisted selection study to improve desired traits; use of plant tissue culture technique and recombinant technology can be undertaken to increase the production of bioactive molecules.
 | Table 2: Different extracts from various parts of Nyctanthes arbor-tristis showing antioxidant and anticancer activities. [Click here to view] |
REFERENCES
1. Uniyal SK, Singh KN, Jamwal P, Lal B. Traditional use of medicinal plants among the tribal communities of Chhota Bhangal, Western Himalaya. J Ethnobiol Ethnomed 2006;2(14):1–8.
2. Rangika BS, Dayananda PD, Peiris DC. Hypoglycemic and hypolipidemic activities of aqueous extract of flowers from Nyctanthes arbor-tristis L. in male mice. BMC Complement Altern Med 2015;15(1):1–9. CrossRef
3. Kiew R, Bass P. Nyctanthes is a member of the Oleaceae. Proc Indian Acad Sci (Plant Sci.) 1984;93(3):349–58.
4. Rani C, Chawla S, Mangal M, Mangal AK, Kajla S, Dhawan AK. Nyctanthes arbor-tristis Linn. (Night Jasmine): a sacred ornamental plant with immense medicinal potentials. Indian J Tradit Knowl 2012;11(3):427–35.
5. Agrawal J, Pal A. Nyctanthes arbor-tristis Linn—a critical ethnopharmacological review. J Ethnopharmacol 2013;146(3):645–58. CrossRef
6. Jain PK, Pandey A. The wonder of Ayurvedic medicine—Nyctanthes arbor-tristis. Int J Herbal Med 2016;4(4):9–17.
7. Latha PG, Suja SR, Shyamal S, Rajasekharan S. Some hepatoprotective garden plants. Nat Prod Radiance 2005;4(4):278–9.
8. Kumari TDS, Madhuri TDS, Charya MAS, Rao KS. Antioxidant and anticancer activities of Nyctanthes arbor-tristis. Int J Pharm Pharm Sci 2012;4(4):452–4.
9. Khatune NA, Islam ME, Rahman MAA, Mossaddik MA, Haque ME. In vivo cytotoxic evaluation of a new Benzofuran derivative isolated from Nyctanthes arbor-tristis L on Ehrlich Ascite carcinoma cells (EAC) in mice. J Med Sci 2003;9(2):169–73. CrossRef
10. Kanno S, Tomizawa A, Hiura T, Osanai Y, Shouji A, Ujibe M, et al. Inhibitory effects of Naringenin on tumor growth in human cancer cell lines and sarcoma S-180-implanted mice. Biol Pharm Bull 2005;28(3):527–30. CrossRef
11. Pandeti S, Sharma K, Bathula SR, Tadigoppula N. Synthesis of novel anticancer iridoid derivatives and their cell cycle arrest and caspase-dependent apoptosis. Phytomedicine 2014;21(3):333–9. CrossRef
12. Chidi BB, Pandeya S, Gharti KP Bharti L. Phytochemical screening and cytotoxic activity of Nyctanthes. Indian Res J Pharm Sci 2015;2(2):205–17.
13. Gupta P, Bajpai SK, Chandra K, Singh KL, Tandon JS. Antiviral profile of Nyctanthes arbor-tristis L. against encephalitis causing viruses. Indian J Exp Biol 2005;43:1156–60.
14. Kannan M, Singh AJAR, Kumar TTA, Jegatheswari P, Subburayalu S. Studies on immuno-bioactivities of Nyctanthes arbor-tristis (Oleaceae). Afr J Microbiol Res 2007;1:88–91.
15. Kannan M, Singh AJAR. An Immuno-Pharmacological investigation of Indian Medicinal Plant Nyctanthes arbor-tristis Linn. World Appl Sci J 2010;11(5):495–503.
16. Devasree LD, Binuramesh C, Michael RD. Immunostimulatory effect of water-soluble fraction of Nyctanthes arbor-tristis leaves on the immune response in Oreochromis mossambicus (Peters). Aquac Res 2014;45(10):1581–90. CrossRef
17. Kirubakaran CJW, Subramani PA, Michael RD. Methanol extract of Nyctanthes arbor-tristis seeds enhances non-specific immune responses and protects Oreochromis mossambicus (Peters) against Aeromonas hydrophila infection. Res Vet Sci 2016;105:243–8. CrossRef
18. Bhalerao AR, Desai SK, Serathia BR, Vartak KM, Doshi GM. Acute and subacute models of inflammation of Nyctanthes arbor-tristis and Maharasnadi ghan. Int J Pharmacol Res 2011;1(1):14–24. CrossRef
19. Nirmal SA, Pal SC, Mandal SC, Patil AN. Analgesic and anti-inflammatory activity of β-sitosterol isolated from Nyctanthes arbor-tristis leaves. Inflammopharmacology 2012;20(4):219–24. CrossRef
20. Kakoti BB, Pradhan P, Borah S, Mahato K, Kumar M. Analgesic and anti-inflammatory activities of the methanolic stem bark extract of Nyctanthes arbor-tristis Linn. BioMed Res Int 2013;2013:1–6. CrossRef
21. Saxena RS, Gupta B, Saxena KK, Srivastava VK, Prasad DN. Analgesic, antipyretic and ulcerogenic activity of Nyctanthes arbor-tristis leaf extract. J Ethnopharmacol 1987;19(2):193–200. CrossRef
22. Pattanayak C, Datta PP, Prasad A, Panda P. Evaluation of anti-inflammatory activity of Nyctanthes arbor-tristis leaves. Int J Med Pharm Sci 2013;3(9):18–25. CrossRef
23. Phanindhra B, Raju BA, Vikas G, Anusha R, Deepika D. Effect of Nyctanthes arbor-tristis leaf extract against scopolamine-induced cognitive impairment in rats. Herva Pol 2015;60(4):34–49. CrossRef
24. Khan ZK, Manglani A, Shukla PK, Puri A, Saxena RP, Tandon JS. Effect of plant extracts and iridoid glucosides from Nyctanthes arbor-tristis against systemic candidiasis in mice. Int J Pharmacogn 1995;33(4):297–304.
25. Bhatia A, Kaur J. Nyctanthes arbor-tristis leaves extract as antagonizer of immunotoxic effects of chemical pesticides (experimental study). Int J Environ Stud 2001;58(2):197–215. CrossRef
26. Kirubakaran CJW, Alexander CP, Michael RD. Enhancement of non-specific immune responses and disease resistance on oral administration of Nyctanthes arbor-tristis seed extract in Oreochromis mossambicus (Peters). Aquac Res 2010;41(11):1630–9. CrossRef
27. Agrawal J, Shanker K, Chanda D, Pal A. Nyctanthes arbor-tristis positively affects immunopathology of malaria-infected mice prolonging its survival. Parasitol Res 2013;112:(7):2601–9. CrossRef
28. Thomas S, Nirmala JG, Narendhirakannan RT. Free radical scavenging activities of Nyctanthes arbor-tristis. L on adjuvant induced arthritis in rats. Br J Pharm Res 2013;3:536–47.
29. Rathore B, Paul B, Chaudhury BP, Saxena AK, Sahu AP, Gupta YK. Comparative studies of different organs of Nyctanthes arbor-tristis in modulation of cytokines in murine model of arthritis. Biomed Environ Sci 2007;20(2):154–9.
30. Goyal S, Sheth NR, Srivastava DN. Comparative evaluation of Nyctanthes arbor-tristis and Alstonia scholaris leaves extracts in Freund ’ s complete adjuvant induced arthritis in rats. Int J Pharm Biol Arch 2013;4(5):903–8.
31. Uroos M, Abbas Z, Sattar S, Umer N, Shabbir A, Rehman S, Sharif A. Nyctanthes arbor-tristis Ameliorated FCA-induced experimental arthritis: a comparative study among different extracts. Evid-Based Complement Altern Med 2017;2017:1–13. CrossRef
32. Puri A, Saxena R, Saxena RP, Saxena KC, Srivastava V, Tandon JS. Immunostimulant activity of Nyctanthes arbor-tristis L. J Ethnopharmacol 1994;42(1):31–7. CrossRef
33. Deshmukh RD, Pokharkar RD, Takate SB, Gite VN. Amelioration of CCL4-induced hepatosuppression by Nyctanthes arbor-tristis Linn leaves in Wistar albino rats. Pharmacologyonline 2007;1:203–8.
34. Wagh AE, Yeotkar US, Nimbhorker MG, Deshmukh TA, Patil VR. Hepatoprotective activity of Nyctanthes arbor-tristis (l.). Orient Pharm Exp Med 2010;10(2):111–5. CrossRef
35. Vishwanathan M, Juvekar AR. Hepatoregenerative effect of Nyctanthes arbor-tristis Linn. on acetaminophen-induced oxidative damage in rats. Int J PharmTech Res 2010;2(2):1291–7.
36. Khatune NA, Mosaddik MA, Haque ME. Antibacterial activity and cytotoxicity of Nyctanthes arbor-tristis flowers. Fitoterapia 2001;72(4):412–4.
37. Mahida Y, Mohan JSS, Dubard R, Linn N. Screening of plants for their potential antibacterial activity against Staphylococcus and Salmonella spp . Nat Prod Radiance 2007;6(4):301–5.
38. Priya K, Ganjewala D. Antibacterial activities and phytochemical analysis of different plant parts of Nyctanthes arbor-tristis (Linn.). Res J Phytochem 2007;(4):61–7. CrossRef
39. Sathiya M, Parimala P, Muthuchelian K. Preliminary Phytochemical Screening and Antibacterial Studies on the Ethanolic Leaf Extract of Nyctanthes arbor-tristis Linn. Ethnobot Leaf 2008;12:337–42.
40. Manisha V, Neha S, Satish S. Antimicrobial activity of stem bark extracts of Nyctanthes arbor-tristis Linn. (Oleaceae). Int J Pharmacogn Phytochem Res 2009;1(1):12–4.
41. Verma NS, Dwivedi S, Panigrahi D, Gupta SK. Anti-bacterial activity of root bark of Nyctanthes arbor-tristis Linn. Int J Drug Discov Herbal Res 2011;1(2):61–2.
42. Satyal P, Paudel P, Poudel A, Setzer WN. Chemical composition and biological activities of essential Oil from leaf and bark of Nyctanthes arbor-tristis L . from Nepal. Open Access J Med Aromat Plants 2011;3(1):1–4.
43. Srinivasan KK, Goomber A, Kumar SS, Thomas AT, Joseph A. Phytochemical, antioxidant and antimicrobial study of flowers of Nyctanthes arbor-tristis Linn. Pharmacologyonline 2011;2:16–21.
44. Vyas A, Sarin R. Analysis of the phytochemical content and antimicrobial activity of Nyctanthes arbor-tristis. Int J Pharma Bio Sci 2013;4(1):201–6.
45. Aggarwal SG, Goyal S. Nyctanthes arbor-tristis against pathogenic bacteria. J Pharmacogn Phytochem 2013;2(3):124–7.
46. Shinde PR, Sali VA, Patil PS, Bairagi VA. Assessment of pharmacognostic, phytochemical and antibacterial potential of fruit of Nyctanthes arbor-tristis Linn. J Pharmacogn Phytochem 2014;2(6):203–12.
47. Show S, Banerjee S, Chakraborty I, Sikdar M. In vitro Comparision between antibacterial activity of Catharanthus roseus and Nyctanthes arbor-tristis on antibiotic-resistant Staphylococcus aureus strain. Indo Am J Pharm Res 2014;3(2):1746–51.
48. Gogoi N, Babu PJ, Mahanta C, Bora U. Green synthesis and characterization of silver nanoparticles using alcoholic flower extract of Nyctanthes arbor-tristis and in vitro investigation of their antibacterial and cytotoxic activities. Mater Sci Eng C 2015;46:463–9. CrossRef
49. Suresh V, Jaikumar S, Arunachalam G. Antidiabetic activity of ethanol extract of stem bark of Nyctanthes arbor-tristis Linn. Res J Pharm Biol Chem Sci 2010;1(4):311–7.
50. Husain A, Tiwari U, Sharma V, Kumar A, Rais N. Effect of Nyctanthes arbor-tristis Linn. leaves against streptozotocin-induced oxidative stress in rats. Int J Pharma Prof Res 2010;1(1):10–3.
51. Panda SK, Patra N, Sahoo G, Bastia AK, Dutta SK. Anti-diarrheal activities of medicinal plants of Similipal Biosphere Reserve, Odisha, India. Int J Med Aromat Plants 2012;2(1):123–34.
52. Saini P, Gayen P, Kumar D, Nayak A, Mukherjee N, Mukherjee S, Pal BC, Babu SPS. Antifilarial effect of ursolic acid from Nyctanthes arbor-tristis: molecular and biochemical evidences. Parasitol Int 2014;63(5):717–28. CrossRef
53. Simonsen HT, Nordskjold JB, Smitt UW, Nyman U, Palpu P, Joshi P, Varughese G. In vitro screening of Indian medicinal plants for antiplasmodial activity. J Ethnopharmacol 2001;74(2):195–204. CrossRef
54. Tuntiwachwuttikul P, Rayanil K, Taylor WC. Chemical constituents from the flowers of Nyctanthes arbor-tristis. Sci Asia 2003;29:21–30. CrossRef
55. Misra P, Pal NL, Guru PY, Katiyar JC, Tandon JS. Antimalarial activity of traditional plants against erythrocytic stages of Plasmodium berghei. Int J Pharmacogn 1991;29(1):19–23. CrossRef
56. Ghiware NB, Nesari TM, Gond NY. Clinical validation of Piper nigrum and Nyctanthes arbor-tristis formulation for antimalarial activity. J Res Edu Indian Med 2007;13(1):33–8.
57. Karnik S, Tathed P, Antarkar D, Godse C, Vaidya R, Vaidya A. Antimalarial activity and clinical safety of traditionally used Nyctanthes arbor-tristis Linn. Indian J Tradit Knowl 2008;7(2):330–4.
58. Kumari P, Sahal D, Jain SK, Chauhan VS. Bioactivity guided fractionation of leaves extract of Nyctanthes arbor-tristis (Harshringar) against P falciparum. PLoS One 2012;7(12):1–7. CrossRef
59. Shukla AK, Patra S, Dubey VK. Iridoid glucosides from Nyctanthes arbor-tristis result in increased reactive oxygen species and cellular redox homeostasis imbalance in Leishmania parasite. Eur J Med Chem 2012;54:49–58. CrossRef
60. Godse CS, Tathed PS, Talwalkar SS, Vaidya RA, Amonkar AJ, Vaidya AB, Vaidya ADB. Antiparasitic and disease-modifying activity of Nyctanthes arbor-tristis Linn. in malaria: an exploratory clinical study. J Ayurveda Integr Med 2016;7(4):238–48. CrossRef
61. Tandon JS, Vandita S, Guru PY. Iridoids: a new class of leishmanicidal agents from Nyctanthes arbor-tristis. J Nat Prod 1991;54(4):1102–4. CrossRef
62. Talakal TS, Dwivedi SK, Sharma SR. In vitro and in vivo antitrypanosomal potential of Nyctanthes arbor-tristis leaves. Pharma Biol 2000;38(5):326–9. CrossRef
63. Mathew N, Anitha MG, Bala TSL, Sivakumar SM, Narmadha R, Kalyanasundaram M. Larvicidal activity of Saraca indica, Nyctanthes arbor-tristis, and Clitoria ternatea extracts against three mosquito vector species. Parasitol Res 2009;104(5):1017–25. CrossRef
64. Patil SV, Patil CD, Salunkhe RB, Salunke BK. Larvicidal activities of six plants extracts against two mosquito species, Aedes aegypti and Anopheles stephensi. Trop Biomed 2010;27(3):360–5.
65. Mishra RK, Mishra V, Pandey A, Tiwari AK, Pandey H, Sharma S, Pandey AC, Dikshit A. Exploration of anti-Malassezia potential of Nyctanthes arbor-tristis L. and their application to combat the infection caused by Mala s1 a novel allergen. BMC Complement Altern Med 2016;16(1):1–14. CrossRef
66. Jamdagni P, Khatri P, Rana JS. Green synthesis of zinc oxide nanoparticles using flower extract of Nyctanthes arbor-tristis and their antifungal activity. J King Saud Univ Sci 2018;30(2):168–75. CrossRef
67. Gupta RS, Kachhawa JBS, Sharma R. Antispermatogenic effects of Nyctanthes arbor-tristis in male albino rats. Physiology 2006;261–73.
68. Tripathi S, Tripathi PK. Antistress activity of Nyctanthes arbor-tristis fruits in rats. Mol Clin Pharmacol 2013;4(1):53–8.
69. Mishra V, Shukla A, Pandeti S, Barthwal MK, Pandey HP, Palit G, Narender T. Arbortristoside-A and 7-O-trans-cinnamoyl-6β-hydroxyloganin isolated from Nyctanthes arbor-tristis possess anti-ulcerogenic and ulcer-healing properties. Phytomedicine 2013;20(12):1055–63. CrossRef
70. Tripathi S, Tripathi PK, Vijayakumar M, Rao CV, Singh PN. Anxiolytic activity of leaf extract of Nyctanthes arbor-tristis in experimental rats. 2010;457–63.
71. Sopi RB, Khan MFH. Bronchodilatory effect of ethanolic extract of the leaves of Nyctanthes arbor-tristis. Pharmacogn Res 2013;5(3):169–72. CrossRef
72. Bharti M, Saxena RC, Baghel OS, Saxena R, Apte KG. Wound healing activity of leaf of Nyctanthes arbor-tristis (Linn.). J Health Sci 2007;2(10):111–4.
73. Paul BN, Saxena AK. Depletion of tumor necrosis factor-c in mice by Nyctanthes arbor-tristis. J Ethnopharmacol 1997;56:153–8. CrossRef
74. Gadgoli C, Shelke S. Crocetin from the tubular calyx of Nyctanthes arbor-tristis. Nat Prod Res 2010;24(17):1610–5. CrossRef
75. Deshmukh A, Dongre S. Natural yellow colour from Corolla of Nyctanthes arbor-tristis Linn . for dyeing and painting on cotton and silk for value addition. Biolife 2015;3(1):353–7. CrossRef
76. Paul BN, Prakash A, Kumar S, Yadav AK, Mani U, Saxena AK, Sahu AP, Lal K, Dutta KK. Silica induced early fibrogenic reaction in lung of mice ameliorated by Nyctanthes arbor-tristis extract. Biomed Environ Sci 2002;15:215–22.
77. Saxena RS, Gupta B, Lata S. Tranquilizing, antihistaminic and purgative activity of Nyctanthes arbor-tristis leaf extract. J Ethnopharmacol 2002;81:321–5. CrossRef
78. Chitravanshi VC, Singh AP, Ghoshal S, Krishna Prasad BN, Srivastava V, Tandon JS. Therapeutic action of Nyctanthes arbor-tristis against caecal amoebiasis of rat. Pharm Biol 1992;30(1):71–5. CrossRef
79. Verma N, Kaur J, Bhatia A. Stimulation of acetylcholinesterase activity with Nyctanthes arbor-tristis leaves extract in the malathion-treated immunosuppressed mice. Int J Environ Stud 2001;58(5):645–54. CrossRef
80. Nirmal SA, Pal SC, Mandal SC. Mast cell stabilizing and bronchodilatory activity of Nyctanthes arbor-tristis bark. Phytother Res 2012;2(1):234–42. CrossRef
81. Das RK, Gogoi N, Bora U. Green synthesis of gold nanoparticles using Nyctanthes arbor-tristis flower extract. Bioprocess Biosyst Eng 2011;34(5):615–9. CrossRef
82. Basu S, Maji P, Ganguly J. Rapid green synthesis of silver nanoparticles by aqueous extract of seeds of Nyctanthes arbor-tristis. Appl Nanosci 2016;6(1):1–5. CrossRef
83. Saratha R, Vasudha VG. Inhibition of mild steel corrosion in 1N H2SO4 medium by acid extract of Nyctanthes arbor-tristis leaves. E-J Chem 2015;5(1):14–23.
84. Kumar KCS, Müller K. Medicinal plants from Nepal; II. Evaluation as inhibitors of lipid peroxidation in biological membranes. J Ethnopharmacol 1999;64(2):135–9. CrossRef
85. Dasgupta N, De B. Antioxidant activity of some leafy vegetables of India: a comparative study. Food Chem 2007;101(2):471–4. CrossRef
86. Rathee JS, Hassarajani SA, Chattopadhyay S. Antioxidant activity of Nyctanthes arbor-tristis leaf extract. Food Chem 2007;103(4):1350–7. CrossRef
87. Akki KS, Krishnamurthy G, Naik HSB. Phytochemical investigations and in vitro evaluation of Nyctanthes arbor-tristis leaf extracts for antioxidant property. J Pharm Res 2009;2(4):752–5.
88. Sasikumar JM, Mathew GM, Darsini DTP. Comparative studies on antioxidant activity of methanol extract and flavonoid fraction of Nyctanthes arbor-tristis leaves. EJEAFChe 2010;9(1):227–33.
89. Meghashri S, Gopal S. Biochemical characterization of radical scavenging polyphenols from Nyctanthes arbor-tristis. J Pharm Bioallied Sci 2012;4(4):341. CrossRef
90. Savarimuthu MJ, Kalirajan A, Padmalatha C, Singh AJAR. In vitro antioxidant evaluation and total phenolics of methanolic leaf extracts of Nyctanthes arbor-tristis L. Chin J Nat Med 2013;11(5):484–7. CrossRef
91. Kumari TDS, Charya MAS. Phytochemistry, anti-cancer and anti-inflammatory activities of solvent leaf extracts of Nyctanthes arbor-tristis. Int J Pharm Sci Res 2017;8(4):1654–63.
92. Vankar PS. Antioxidant activity of the flower of Nyctanthes arbor-tristis L. Int J Food Eng 2008;4(8). CrossRef
93. Nagavani V, Rao RKV, Ch RK, Rao RT. In-vitro screening of Nyctanthes arbor-tristis flowers for antioxidant activity and identification of polyphenols by RP-HPLC. Pharmacologyonline 2010;2:57–78.
94. Mishra AK, Upadhyay R, Chaurasia JK, Tiwari KN. Comparative antioxidant study in different flower extracts of Nyctanthes arbor-tristis (L.) (Oleaceae): an important medicinal plant. Brazil J Bot 2016;39(3):813–20. CrossRef
95. Hussain A, Ramteke A. Flower extract of Nyctanthes arbor-tristis modulates glutathione level in hydrogen peroxide treated lymphocytes. Pharmacogn Res 2012;4(4):230.
96. Pandey RS. Anti-oxidant activity of Nyctanthes arbor-tristis Linn . Asian J Pharm Life Sci 2012;2(2):360–4.
97. Khanapur M, Avadhanula RK, Setty OH. In vitro antioxidant, antiproliferative, and phytochemical study in different extracts of Nyctanthes arbor-tristis flowers. BioMed Res Int 2014;2014:1–10.
98. Thakur MK, Jaiswal P. The investigation on total phenolic content and In vitro antioxidant potential of different plant parts of Nyctanthes arbor-tristis (night jasmine). Int J Pharm Sci Res 2017;8(8):3547–51.