1. INTRODUCTION
In tropical and subtropical regions, cassava (Manihot esculenta Crantz) is ranked as the fourth most important food crop after rice, wheat, and maize providing source of livelihood for over 700 million people [1,2]. Recently, cassava has gained popularity among smallholder farmers due to its unique characteristics, such as adaptability to diverse environmental conditions, high nutrient and water use efficiency, low cost of propagation materials, tolerance to sporadic pest attack, and ability to be harvested piecemeal upon maturity [3].
Despite the vast production and utilization of cassava, biotic and abiotic factors form part of the constraints that limit full realization of the crop’s immense potential. The main biotic factors associated with cassava are pests (insects and mites), viral, bacterial, and fungal diseases. Fungal diseases form part of constraints that cause significant yield losses [4]. In spite of these adverse effects caused by phytopathogenic fungi, current research activities in cassava are focused more on viral and bacterial diseases as delineated in heightened research work on these diseases [5,6].
One of the most important fungal diseases in cassava is brown leaf spot (BLS) [7]. The disease is characterized by large, brown, necrotic spots appearing on older leaves, and the infected leaves have a tendency to drop early [8]. The disease is of worldwide distribution and is found in the most cassava fields in the lower canopy of crops that are more than 5-month old and is spread to new leaves and plants by wind or rain splash. The importance of BLS disease may be underestimated due to its being confined to the lower canopy leaves. However, the disease causes defoliation which may have a significant effect on yield, especially in the areas where cassava is extensively grown for commercial production. The disease covers a great surface area of the leaves and this could significantly reduce photosynthetic activities, thus reducing yields [9]. Losses in root yield in Africa are up to 30%, 23% in South America, and 17% in India [10]. Studies have been carried out in various regions, including China, Thailand, Asia, North- and Latin America, and some parts of Africa, showing etiology and epidemiology of BLS disease [11]. The disease is reported to result from infection by the fungus Cercospora heningsii. However, little has been done in Kenya to understand the causative pathogen(s) of the disease. The aim of this study was, therefore, to determine the causative agent for BLS disease in Kenya.
2. MATERIALS AND METHODS
2.1. Description of the study site and experimental materials
The study was conducted at the Kenya Agricultural and Livestock Research Organization (KALRO), Biotechnology Research Institute (BRI), Kabete. Infected leaf samples of field-grown cassava cultivars TME 204, TME 14, NASE 14, TME 7, and Ebwanateraka were obtained from KALRO BRI, KALRO Kakamega (Western Kenya), and KALRO Kandara (Central Kenya).
2.2. Collection of infected cassava leaf samples
Fifteen leaf samples showing strong symptoms were randomly collected from each of the cassava genotypes established at the study sites. Additional five samples were collected from healthy non-symptomatic cassava genotypes (TME 204, TME 14, TME 7, NASE 14, and Ebwanateraka) established in KALRO-BRI greenhouse to serve as negative controls. Samples were separately conserved in khaki envelopes which were appropriately labeled prior to transportation from the field to the laboratory. Five replicates of leaf samples per variety were maintained.
2.3. Isolation and purification of fungi
In this study, a modified version of fungal isolation procedure by Thilagam et al. [12] was used, in which potato dextrose agar (PDA) was used as the culturing medium The medium was prepared by mixing 39 g of PDA powder in 1,000 ml distilled water and autoclaved at 121ºC for 20 minutes. To avoid bacterial contamination, 20 ml of 50 mg/ml streptomycin and 12 ml of 10 mg/ml neomycin were added to 1,000 ml cooled medium (42°C). Approximately, 20 ml of medium was dispensed under laminar flow hood into sterile plastic petri dishes to form a layer of about 2 mm deep before isolation of fungal pathogens was performed.
Fungi were isolated by first washing the leaf samples with tap water and surface sterilizing in 70% ethanol for 30 seconds. Margins of necrotic leaf lesions were obtained using sterile scalpels and disinfected with 1.3% (v/v) sodium hypochlorite for 1 minute followed by rinsing three times in sterile distilled water. The leaf sections were dried using sterile blotting paper. The exercise was performed under aseptic conditions in which three leaf disks per plate for each sample were cultured on the medium. The plates were incubated at room temperature (23 ± 2ºC) for 7 days after which fungal colonies which had emerged from the leaf sections were individually sub-cultured on new PDA medium and incubated at room temperature for 21 days to allow for sporulation. This procedure yielded pure cultures that helped delineate both colony and morphological characteristics for each isolate.
2.4. Identification of fungal isolates
Microscopic slides of each pure fungal isolate were prepared and viewed under light microscope for morphological characterization and identification. Using a sterile isolation needle, a small portion of each colony was picked and placed on a drop of lactophenol cotton blue dye on a sterilized glass slide. A clean cover slip was then placed gently on top to completely cover the sample and dye avoiding air bubbles. Microscopic features included type of spores produced and the fruiting body, and the form of hyphae, whether septate or aseptate, branching or non-branching. To aid identification, colony color, basic shape of colony, elevation, and the surface appearance were also used. Fungal pathogens were identified according to Barnett [13], Dugan [14], and Humber [15]. Five replicates per isolate were maintained for other downstream processes.
2.5. Inoculum preparation and plant inoculation for pathogenicity tests
The pathogenicity experiment was performed in the KALRO BRI greenhouses where plants were established and maintained. One cassava variety, TME 204 susceptible to BLS, was selected for this study; five plants were inoculated with each isolate. To achieve a completely randomized design, random numbers were generated using the “RAND” function of Microsoft excel and assigned to each plant such that each treatment was placed at random position within the greenhouse.
Inoculum was prepared following a modified version of the guidelines by Kirkhouse Trust [16]. The identified isolates, namely, Colletotrichum, Alternaria, and Cladosporium were separately scraped off the medium while being careful to avoid medium inclusion. The isolates were then homogenized through blending in sterile distilled water followed by straining through a sterile 0.9 mm pore strainer to obtain a spore suspension excluding much of the mycelia. The number of spores in the inoculum was ascertained using a hemocytometer, then adjusted to 1 × 106 spores/ml before dispensing in appropriately labeled hand sprayers in readiness for inoculation. The inoculum was prepared as single isolates as well as in combinations. Five cassava plants were spray-inoculated with fungal isolates 4 weeks after planting. Each inoculated plant was covered with a clear humidity bag for 48 hours to allow a conducive microenvironment for fungal infection. Assessment for symptom onset and disease development was done once a week for 4 weeks.
3. RESULTS
3.1. Identification of fungal pathogens causing cassava brown leaf spot disease
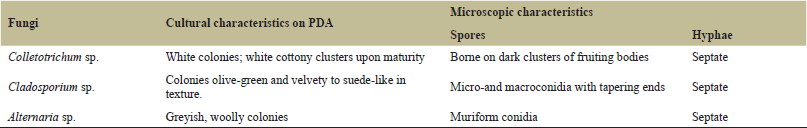
A total of 80 samples were collected from the fields: 25 from KALRO BRI, 25 from KALRO Kakamega (Western Kenya), and 30 KALRO Kandara (Central Kenya). From the entire population of the isolated and purified cultures, three fungal pathogens, Colletotrichum sp., Alternaria sp., and Cladosporium sp. were the most prevalent (Table 1), with no difference in regional prevalence of the three pathogens. These pathogens were selected as possible causative agents for brown leaf spot disease in cassava. Selection was based on three key factors: (i) culture characteristics delineated through microscopy and guided by identification manuals (ii) relative incidence of the isolates, and (iii) common isolate prevalence through the sampled areas. The cultural and morphological characteristics of the isolated fungi are listed in Table 2. Colletotrichum: Colonies were white in color and upon maturity; they appeared as white cottony clusters throughout the media. The spores were borne on dark clusters of fruiting bodies more evident on the back side of the culture (Fig. 1). Conidiophores, which emanated from a rather weak stroma, each bore septate conidia which appeared crescent shaped upon their release.
Cladosporium: The isolate in this study grew rather moderately and it matured to produce large amounts of non-septate macro and micro conidia. The colony was olive-green and velvety to suede-like in texture. Cladosporium produces erect, septate hyphae. Conidiophores were also septate and showed tree-like branching (Fig. 2).
Alternaria: Colony was rapidly growing, woolly, and covered with grayish, short hyphae. The back side of the culture was typically black due to production of pigment (Fig. 3). The fungus possessed hyphae and conidiophores that were septate with muriform conidia (transverse and longitudinal septation). The conidia were produced singly or in acropetal chains at the apex of the conidiophores.
3.2. Development of symptoms on cassava plants
Inoculation of cassava plants with Colletotrichum sp. yielded typical chlorotic or necrotic spots for brown leaf spot disease (Figs. 4A and B) though the spot size was smaller compared to that observed in the field. Inoculation of cassava plants with Cladosporium sp. (Figs. 4C and D) also resulted in small leaf spots which were lumpy, thus giving the leaf a rough texture to touch. Alternaria sp. on the other hand showed yellowish spots with brown borders, almost similar to BLS symptoms but not as observed in the fields (Figs. 4E–G).
 | Table 1. Relative prevalence of brown leaf spot disease fungal pathogens isolated from infected leaves of field-grown cassava. [Click here to view] |
 | Table 2. Cultural and morphological characteristics of fungal pathogens isolated from infected leaves of field-grown cassava. [Click here to view] |
 | Figure 1. Cultural and morphological characteristics of Colletotrichum sp.: (A and B) front and back sides of culture on PDA medium; (C) fruiting bodies as seen under light microscope; (D) conidiophores bearing conidia; and (E) released crescent-shaped conidia. [Click here to view] |
 | Figure 2. Cultural and morphological characteristics of Cladosporium sp.: (A and B) front and back side of culture, respectively; (C and D) Conidiophores bearing conidia; and (E) Micro- and macroconidia upon release from conidiophores. [Click here to view] |
 | Figure 3. Cultural and morphological characteristics of Alternaria sp.: (A and B) front and back side of culture, respectively; (C) spores borne at the conidiophore terminus (arrow); and (D) mature spores. [Click here to view] |
Upon inoculation of healthy cassava plants with a combination of two of the fungal pathogens, Colletotrichum and Alternaria, Colletotrichum and Cladosporium (Figs. 5A–E), and Alternaria and Cladosporium (Figs. 6A–C), foliar disease symptoms appeared similar to infections with either of the combination, consisting of chlorotic or necrotic spots. The spots produced were not quite typical of BLS leaf spots since most appeared as plain brown necrotic lesions with a yellowish border as contrasted to BLS symptoms in which spots are brown with dark borders and often surrounded by indistinct yellow halo.
Inoculation of healthy cassava plants with a combination of all the three pathogenic fungi yielded symptoms typical of BLS disease as observed in the field (Fig. 7). Spots were brown with dark borders and often surrounded by an indistinct yellow halo, an indication that the three fungi are involved in BLS disease development.
4. DISCUSSION
Cassava brown leaf spot has been shown to be an important fungal disease of cassava due to the root yield losses reported globally [17]. This study has shown the causative agents of the disease in Kenya to be Colletotrichum sp., Alternaria sp., and Cladosporium sp. acting in synergism to produce brown leaf spots on the cassava leaves. The three pathogens produce phytotoxins which play an important role in plant colonization [18–20]. The indistinct yellow halo produced around the brown spot is due to the toxins produced by the advancing fungal mycelia [21].
The three pathogens have been shown to cause leaf spots in various plants including cassava (Manihot esculenta) [22,23,24]. The genus Colletotrichum comprises major and diverse plant pathogens and has been described as the main cause of anthracnose diseases in many plant crops globally. The genus has a diverse range of plant-pathogen interactions which is dependent on the Colletotrichum species, host species and physiological maturity and environmental conditions. There is a large number of species in the Colletotrichum species complexes, with many new species being described and many more likely to exist. Studies have shown that the same Colletotrichum species can be isolated from different hosts, as first reports in some hosts [24]. Colletotrichum has been reported to cause significant crop failure leading to food shortages especially for the poor subsistence farming population that depends on the crop for their livelihood [25].
Cladosporium sp. has also been implicated as an important pathogen causing leaf spots in plants, including spinach, pecan nuts, cucumber, and peach [26]. It can also exist as a saprophyte, living on decaying plant tissue. Conidiophores are usually tall and upright with branching at the apex. Conidia are produced singly or in chains. Studies conducted on Cladosporium were limited on the role of Cladosporium in cassava brown leaf spot disease but its implication in other plant leaf spots and high prevalence in this study made it deserve a deeper investigation. Inoculation of cassava leaves with Cladosporium yielded tiny, sunken spots on the upper side of leaf which were rough to touch on the leaf underside. The exhibited symptoms were similar to spots caused by the fungus on cucumber plant [27].
 | Figure 4. Foliar symptoms upon inoculation of cassava cultivar TME 204 with (A and B) Colletotrichum; (C and D) Cladosporium; (E, F, and G) Alternaria; (H) water; and (I) non-inoculated plants. [Click here to view] |
 | Figure 5. Foliar symptoms upon inoculation of cassava cultivar TME 204 with combinations of (A, B, and C) Colletotrichum and Alternaria and (D and E) Colletotrichum and Cladosporium symptoms. [Click here to view] |
There were no detailed studies on the role of Alternaria sp. in cassava brown leaf spot disease reported in literature nonetheless, it was prevalent in cassava samples studied and produced typical BLS symptoms upon single or mixed inoculation on healthy cassava leaves. Alternaria species have been shown to be important pathogens of a wide variety of crops such as potato and tomato where they cause severe blight [28]. It can, therefore, be concluded that synergism of the three isolates is required for development of BLS disease symptoms.
 | Figure 6. Foliar symptoms upon inoculation of cassava cultivar TME 204 with combinations of Cladosporium and Alternaria. [Click here to view] |
 | Figure 7. Foliar symptoms (A–E) in cassava cultivar TME 204 upon inoculation with a combination of the three isolates of: Colletotrichum, Alternaria and Cladosporium. (A and B) Front side of leaf and (C, D, and E) back side of leaf. [Click here to view] |
5. CONCLUSION
The results from the study indicate that cassava BLS disease results from the combination of Colletotrichum, Cladosporium, and Alternaria pathogens and that the symptoms are a result of synergistic effect of three pathogens. This information is important in designing BLS disease management programs in Kenya.
ACKNOWLEDGMENT
The authors would like to thank Kenya Agricultural and Livestock Research Organization for providing laboratory and greenhouse facilities used in this study, and the Virus Resistant Cassava for Africa (VIRCA) project for partially funding the study.
REFERENCES
1. Stone GD. Both sides now: fallacies in the genetic-modification wars, implications for developing countries, and anthropological perspectives. Curr Anthropol 2002;43(4):611–30.
2. Sanginga N. Root and tuber crops (Cassava, Yam, Potato and Sweet Potato). In Proceedings of the `an action plan for African Agricultural Transformation Conference', Dakar, Senegal, 2015, p. 21–3.
3. Tan SL. Cassava—silently, the tuber fills. UTAR Agric Sci J 2015;1(2):12–24.
4. Knogge W. Fungal infection of plants. Plant Cell 1996;8:1711–22.
5. Food and Agriculture Organization. Handbook of mycological methods, pp 1–33, 2013. Available via http://www.fao.org/fileadmin/user_upload/agns/pdf/coffee/Annex-F.2.pdf (accessed 22 July 2019).
6. Homenauth O, P-DeSouza S. Pests and diseases of cassava in Guyana. Rural Enterprise and Agricultural Development Project, International Fund for Agricultural Development, 2011. Available via http://www.newgmc.com/gmc_docs/brochures/pests%20%20diseases%20of%20cassava%20in%20guyana.pdf (accessed 22 July 2019).
7. Ayesu-Offei EN, Antwi-Boasiako C. Production of Microconidia by Cercospora henningsii Allesch, Cause of Brown Leaf Spot of Cassava (Manihot esculenta Crantz ) and Tree Cassava (Manihot glaziovii Muell. -Arg.). Ann Bot 1996;78:653–57.
8. Pei YL, Shi T, Li CP, Liu XB, Cai JM, Huang GX. Distribution and pathogen identification of cassava brown leaf spot in China. Genet Mol Res 2014;13(2):3461–73.
9. Moses E, Oppong A, Lamptey JNL. Reaction of local accessions of cassava to diseases in Southern Ghana. Afr Crop Sci J 2015;23(1):27–34.
10. Powbunthorn K, Abudullakasim W, Unartngam J. Assessment of the severity of brown leaf spot disease in cassava using image analysis, presented at the international conference of the Thai society of agricultural engineering, Chiangmai, Thailand, 2012 August 4–5, pp. 2–8.
11. Lozano JC, Booth RH. Diseases of Cassava. J PANS 1976;20.
12. Thilagam R, Kalaivani G, Hemalatha N. Isolation and identification of phytopathogenic fungi from infected plant parts. Int J Curr Pharm Res 2018;10(26):26–8.
13. Barnett HL. Illustrated genera of imperfect fungi, brief description of 302 genera with drawings illustrating a typical species. 2nd edition, Burgess Publishing Co., Minneapolis, MN, 1960.
14. Dugan FM. The identification of fungi: an illustrated introduction with keys, glossary, and guide to literature. American Phytopathological Society (APS Press), St. Paul, MN, 2006.
15. Humber RA. Fungi: identification. In: Lacey LA (ed.). Manual of techniques in insect pathology, Academic Press, Cambridge, MA, pp 153–85, 1997.
16. Kirkhouse Trust. Isolation of a fungal pathogen and producing inoculum, 2012. Available via https://www.youtube.com/watch?v=F92GEhJaubc&t=118s (Accessed 20 November 2017).
17. Hillocks RJ, Wydra K. Bacterial, fungal, and nematode disease. Cassava Biol Prod Util 2002;261–80.
18. Yoshida S, Hiradate S, Fujii Y, Shirata A. Colletotrichum dematium produces phytotoxins anthracnose lesions of mulberry leaves. Biochemistry and Cell Biology. 1999;90(3):285–291.
19. Meena M, Gupta SK, Swapnil P, Zehra A, Dubey MK, Upadhyay RS. Alternaria toxins: potential virulence factors and genes related to pathogenesis. Front Microbiol 2017;8:1–14.
20. Alwatban MA, Hadi S, Moslem MA. Mycotoxin production in Cladosporium species influenced by temperature regimes. J Pure Appl Microbiol 2014;8(5):4061–9.
21. Cab International [Internet]. Brown leaf spot of cassava: Mycosphaerella henningsii [updated 2019]. Available via https://www.plantwise.org/knowledgebank/datasheet/35299 (Accessed May 2019).
22. Batta Y. Alternaria leaf spot disease on cucumber: susceptibility and control using leaf disk assay. An-Najah University Journal for Research (Natural Sciences). 2003;17(2):271–279.
23. Scruggs AC, Butler SC, Quesada-Ocampo LM. First Report of Cladosporium Leaf Spot of Spinach Caused by Cladosporium variabile in North Carolina. The American Phytopathological Society (APS) [Internet]. 2014. Available from https://apsjournals.apsnet.org/doi/10.1094/PDIS-05-14-0474-PDN
24. De Silva DD, Crous PW, Ades PK, Hyde KD, Taylor PWJ. Life styles of Colletotrichum species and implications for plant biosecurity. Fungal Biol Rev 2017;31:155–68.
25. Fokunang CN, Dixon AGO, Ikotun T, Tembe EA, Akem CN, Asiedu R. Anthracnose: an economic disease of dassava in Africa. Pak J Biol Sci 2001;4:920–5.
26. Bensch K, Braun U, Groenewald Z, Crous PW. The genus Cladosporium. Stud Mycol 2012;72(1):1–401.
27. Thomma BP, Esse HP, Crous PW, Wit PJ. Pathogen profile Cladosporium fulvum (syn. Passalora fulva), a highly specialized plant pathogen as a model for functional studies on plant pathogenic Mycosphaerellaceae. Mol Plant Pathol 2005;6(4):379–93.
28. Stammler G, Böhme F, Philippi J, Miessner S, Tegge V. Pathogenicity of Alternaria-species on potatoes and tomatoes. Fourteenth euroblight workshop limassol—Cyprus; 2013. Report no 16