1. INTRODUCTION
The contemporary world is under the influence of infectious diseases, especially in the developing countries despite advances in the modern medicine [1]. In Africa, plants are the main medications for primary health care [2,3]. According to the statistics, nearly 80% of the African population compared with 85% at the national level use traditional medicine [4]. Several species are used in this popular medicine for the treatment of oxidative, inflammatory, microbial, cardiovascular, renal, and viral diseases [5]. The lack of knowledge about the biological activities of the plants used is the basis of irrational doses leading to intoxication [6]. Controlling the pharmacodynamic properties of phytoconstituants requires the determination of phytochemical groups capable of generating pharmacological effects [7].
In Benin, low health coverage is associated with a strong predominance of the use of traditional pharmacopoeia. This finding translates into a ratio of one doctor per 10,000 inhabitants against a traditional medicine practitioner per 800 inhabitants. As a result, traditional medicine no longer appears as a complementary alternative medicine, but represents the basis for solving health problems in Benin [8]. Thus, the prospection of extracts administered empirically is essential in order to avoid, sometimes, tragic therapeutic accidents [9]. For this reason, several phytochemical investigations have been initiated in order to provide a scientific justification for the traditional use of medicinal plants [10]. The research has revealed more than 500 natural substances [11] that result in a structural diversity of bioactive molecules [12]. The floristic arsenal of Benin is full of impressive medicinal species, including Ocimum gratissimum, a plant with high medicinal value, antibacterial widely used empirically to prevent or cure several types of diseases [13]. It is legitimate to carry out a thorough phytochemical exploration of this plant in order to highlight the scientific proof of these traditional uses with the biomolecules present.
2. MATERIALS AND METHODS
2.1. Chemical
The chemicals and reagents used in this work such as the 2,2-diphenyl-1-picrylhydrazyl (DPPH), Folin–Ciocalteu reagent, ascorbic acid, potassium hexacyanoferrate, trichloroacetic acid, gallic acid, ascorbic acid, quercetin, sodium carbonate (Na2CO3), dimethyl sulfoxide (DMSO), aluminum chloride (AlCl3), solvent methanol, acetonitrile, and acetic acid were purchased from Sigma-Aldrich.
2.2. Collection of Plant Materials
The leafy stems of O. gratissimum were harvested in the garden of the Cotonou Airport, Littoral Department, in southern Benin in September 2017. A voucher specimen has been deposited under the registration number AA 6722/National Herbarium of Benin (HNB) at the herbarium of the University of Abomey-Calavi. The samples collected were washed and dried in shade under laboratory conditions. The dried plant was reduced into the fine powders using an electric grinder (MARLEX Electroline Excella), and stored at the room temperature in a dry place.
2.3. Preparation of the Extract
The extract was prepared using previously described method [14–16] with slight modifications. The dried fine powder of O. gratissimum was extracted by the maceration method. Solvent 500 ml ethanol (96%; v/v) were added to 100 g of O. gratissimum powder and were left to stand under agitation for 24 hours. Then, the solution obtained was filtered using a filter paper. The ethanol extract was concentrated under the reduced pressure using a rotavapor, and the obtained extract was preserved at 4°C for further use.
2.4. Preliminary Phytochemical Screenings
The O. gratissimum ethanolic extract was screened for the presence of secondary metabolites such as flavones, flavonols, anthocyanins, leucoanthocyanins, free anthracenics, O-heterosides, O-heterosides genin, C-heterosides, catechin tannins, gallic tannins, coumarins, alkaloids, steroids, saponosids, triterpenoids, cardenolides, quinone derivatives, cyanogenic derivatives, and mucilages as described by the previous method using an approach based on the differential color and precipitation reactions [17,18].
2.5. Quantitative phytochemical analysis
2.5.1. Total polyphenol contents
The polyphenol contents of the ethanolic extract were estimated according to the Folin–Ciocalteu method [19]. The tested extract was prepared at 1 mg/ml with the methanol solvent. Diluted 200 μl of sample were added to 1 ml of 1/10 diluted Folin–Ciocalteu reagent. After 4 minutes, 800 μl of the saturated sodium carbonate (75 g/l) was added. After 2 hours of incubation at the room temperature, the absorbance was measured at 765 nm. The standard calibration curve was plotted using gallic acid (y = 0.043 x − 0.051; R2 = 0.994). The mean of three readings was used and the results expressed as mg of gallic acid equivalent (GAE)/100 mg of extract [14,20].
2.5.2. Total flavonoid contents
Spectrophotometric method with aluminum trichloride (AlCl3) was used for the total flavonoid quantitation in the ethanolic extract of O. gratissimum [21]. The sample was prepared by dissolving extract in methanol (1 mg ml−1). Then, 1 ml of sample was added to 1 ml of the AlCl3 solution (2% in methanol). After 10 minutes of reaction, the absorbance was read at 430 nm against a blank which consists of a mixture of 1 ml of the extract solution and 1 ml of methanol without AlCl3. Quercetin was used as reference compound to produce the standard curve (y = 0.325x − 0.363; R2 = 0.995). The experiment was performed at triplicate and the results were expressed as mg of quercetin equivalent (mg QE)/100 mg of the extract.
2.5.3. Total tannin contents
The condensed tannins were determined here by the acidic vanillin method [22] reviewed and adapted to the laboratory conditions. This method is based on the ability of vanillin to react with tannic units in the presence of acid. The stock solution of O. gratissimum extract is prepared at a concentration of 1 mg ml−1 in methanol. Then, 400 ml of this stock solution were taken and then added to 3 ml of vanillin (4%) and 1,500 ml of hydrochloric acid (HCl). A methanol-acid mixture was used as a white. The sample mixture kept at 30°C for 20 minutes in a dark chamber. The absorbance is read at 500 nm. The contents of condensed tannins were determined using a calibration curve of gallic acid. The results were expressed in GAE.
2.6. HPLC—Analysis and Identification of Phenolic Compounds
The phenolic sets of elements analysis was carried out using a Ultimate 3000 High Performance Liquid Chromatography (HPLC) liquid chromatograph system (U-HPLC 3000, Dionex) equipped with a degasser, binary gradient pump, column thermostat, and a UV multi-wavelength detector (DAD-3000 RS and MWD-3000 RS). This method has been previously exploited by some researchers [23–25]. The flow rate was 1 ml min−1 and the injection volume was 20 μl. The stock solutions (100 mg/ml in methanol) of 13 standard sets of elements (ferulic acid, caffeic acid, chlorogenic acid, tannic acid, ellargic acid, gallic acid, syringic acid, luteolin, chrysin, rutin, hyperoside, quercetol, and isorhamnetin) were prepared and stored at 4°C. Appropriate dilutions of each of the standard sets of compounds were performed prior to analysis. The ethanol extract of the plant was also prepared at the concentration of 1 mg ml−1 in methanol. All samples solutions were filtered with filters of 0.2 μm pore sizes before injection. The chromatographic separation was carried out in a 150 × 4.6 mm, 5 μm Hypersil BDS C18 reversed phase column at ambient temperature. The gradient program was as follows: 20%–50% B to 0–20 minutes, 50%–70% B to 20–25 minutes, 70%–80% B to 25–30 minutes, 80%–20% B to 30–35 minutes, and 20% B 35–40 minutes. Chromleon v.6.80 Software (Dionex, Thermo Fisher Scientific) was used to collect and analyze the data. Phenolic compounds were identified according to their retention times by comparison with those of pure standards. Experiments were carried out in triplicate. Data are reported as mean ± standard deviation of three independent measurements.
2.7. Antioxidant Activities
2.7.1. Ferric reducing antioxidant power (FRAP)
The evaluation of the antiradical power by the FRAP method was carried out as previously described works [26,27]. The principle of this method was based on the ability of the extract to reduce the ferric ion (Fe3+) present in the potassium ferricyanide complex [K3Fe(CN)6] to ferrous ion (Fe2+) by an antioxidant. The reaction was translated by the change from the yellow color of ferric iron (Fe3+) to the blue-green color of ferrous iron (Fe2+). The intensity of this color was measured spectrophotometrically at 700 nm. Briefly, a volume of 2 ml of sample (100 μg/ml) was mixed with 2 ml of phosphate buffer (0.2 M, pH 6.6) and 2 ml of the aqueous solution (1%) of potassium hexacyanoferrate [K3Fe(CN)6]. The mixture was incubated at 50°C for 20 minutes followed by the addition of 2 ml of trichloroacetic acid (10 %). Then, the mixture was centrifuged at 3,000 rpm for 10 minutes to collect the top layer of the solution. A volume of 2 ml of the supernatant was then mixed with the same volume of distilled water and 0.4 ml of a freshly prepared aqueous solution of FeCl3 (0.1%) was added. After 10 minutes of reaction, the absorbance was read at 700 nm. The reducing power is deduced from the calibration curve established with ascorbic acid. The reducing power is expressed as ascorbic acid equivalent (AAE).
2.7.2. DPPH free radical scavenging activity
The antioxidant activity of the ethanolic extract of O. gratissimum was evaluated by the method previously described [27–29]. The principle of this method is based on the ability of the extracts to trap the radical DPPH (2,2-diphenyl-1-picrylhydrazyl). The stock solution of the extract was prepared at 1 mg/ml. Then, a serial dilution of 50% was made to obtain eight concentration ranges (1,000—7.8 μmg/ml). Then, 1.5 ml of a freshly prepared methanolic solution of DPPH (2%) was mixed with 0.75 ml of the extract solution. After 15 minutes of incubation in the dark, at room temperature, the absorbance of the mixture was read at 517 nm using a spectrophotometer (UV-1600 PC). White consisting of a mixture of 1.5 ml of methanol and 0.75 ml of extract solution. The positive control used was quercetin. All analyses were performed in triplet. The percentage of inhibition of DPPH radical was calculated according to the following formula:
Where IP = Inhibition percentage; Abs = absorbance; cont = blank; Ech = sample.
Nearly, 50% inhibitory concentration (IC 50%) was determined graphically by linear regression of the graphs, DPPH inhibition percentages as a function of different concentrations used [30].
2.8. Statistical Analysis
The statistical analysis of the result has been carried out using MS Excel software. The antiradical activity by the FRAP method has been estimated from the non-linear regression curve.
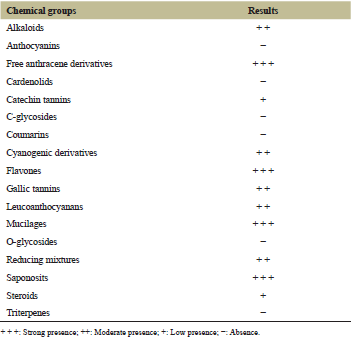 | Table 1: Phytochemical composition of ethanol extract of O. gratissimum. [Click here to view] |
3. RESULTS
3.1. Preliminary Phytochemical Screenings
The major classes of phytochemical contents, the ethanolic extract from O. gratissimum were assessed and the results are summarized (Table 1). The results of phytochemical screening show that in addition to the presence of the three major group of compounds (phenolic, nitrogen, steroid, and terpenoids), we have reported the cyanogenic derivatives, mucilages, and reducing mixtures too. The presence of phenolic mixtures results in the positive dosage of tannins (catechins and gallics), flavonoids (flavones), leuco-anthocyanins, and anthracenics (free) in variable proportions. The presence of nitrogen mixtures on one hand, steroids and terpenoids on the other hand is revealed by the positive exploration of alkaloids then steroids and saponosides, respectively. The presence of these different mixtures testifies to the medicinal potentialities of O. gratissimum.
.png) | Table 2: Total phenol, total flavonoid, and total tannin contents in ethanolic extract. [Click here to view] |
3.2. Total Yields of Polyphenol, Flavonoids, and Tannins
The assay of these molecules in the ethanolic extract revealed the presence of total tannins, total flavonoids, and total polyphenol in variable proportions (Table 2). These three desired compounds are molecules endowed with antibacterial and antioxidant activities. The analysis of the data shows that in 100 g of sample, the ethanolic extract contains 8.6 mgCE of total tannins, 13.71 mg QE of total flavonoids, and 56.59 mgGAE of total polyphenol, respectively. In a comparative approach, the amount of total flavonoids is almost twice the mass of total tannins. Indeed, the amount of total flavonoids harvested is more than four times inferior to the mass of total polyphenol obtained.
3.3. Chromatographic Profile of O. gratissimum
In this study, 13 phenolic mixtures have been searched in the ethanolic extract. The HPLC chromatogram obtained reflects the variation of the absorbance of the various constituents present in the sample as a function of time (Fig. 1). Most graphs are reproducible. The chromatogram is, therefore, acceptable. The chromatographic profile reveals the presence of eighteen distinct molecules of which nine identified and quantified. We have three flavonoids (chrysin, isorhamnetine, and quercetin) and six phenolic acids (tannic, ellargic, ferrulic, syringic, chlorogenic, and caffeic) of the quantified molecules. Among the flavonoids identified, chrysin (25.40 mAU) is the most abundant followed by quercetin (13.25 mAU). Phenolic acids have been qualitatively identified and showed low proportions. Given the number of standards used, several other peaks have not been identified. Among these mixtures two major peaks of respective masses 140.148 and 118.068 mAU have been found in the ethanolic extract.
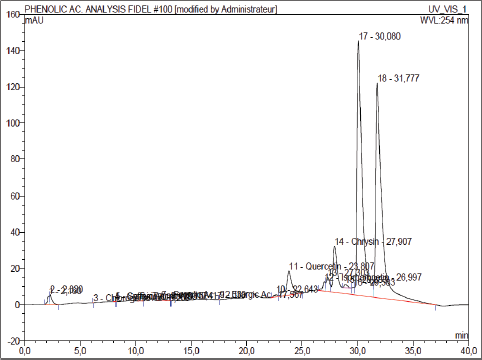 | Figure 1: HPLC chromatograms of O. gratissimum ethanolic extract. [Click here to view] |
3.4. Determination of Ferric Reducing Antioxidant Power
The FRAP results of the ethanolic extract reflects the reducing capacity of the extract according to the concentrations (Table 3). What emerges from the analysis of this result shows that the ferric reducing induced by the different concentrations of the ethanolic extract of O. gratissimum vary from 78.92 to 106.25 mMol AAE/g. As a rule, the increase of the reducing power of an extract is at the origin of the increase of the absorbance and testifies to the antioxidant potentialities of this extract.
3.5. Determination of DPPH Radical Scavenging Activity
The search for the antioxidant power of the ethanolic extract of O. gratissimum by the DPPH method revealed a dose–response relationship of the different concentrations tested according to the inhibition capacities of the registered DPPH radicals (Fig. 2).
 | Table 3: Ferric reducing antioxidant power of ethanolic extract of O. gratissimum. [Click here to view] |
In the case of the ethanolic extract of O. gratissimum, the curve has a curvilinear shape “S” composed of three phases. For the first phase between 7.8 and 125 μg ml−1, the curve has a weak evolution which results in a slow growth of the percentage of inhibition going from 05% to 20%. During the intermediate phase, which varies from 125 to 500 μg ml−1, the curve shows a strong evolution. This causes a rapid growth of the percentage of inhibition which passes from 20% to 75%. During the terminal phase which takes into account the range of concentrations 500 to 1,000 μg ml−1, the curve has a plateau and the percentage of inhibition remains almost invariable from 75% to 77%. What emerges from this summary analysis is that the antiradical potency of the ethanolic extract of O. gratissimum is very high in the range of concentrations 125 to 500 μg ml−1. This observation confirms the power of iron reduction by the extract of O. gratissimum which was obtained by the FRAP method and showed the antioxidant potential of this plant.
In a comparative approach, on the concentrations range of 7.8 to 500 μg ml−1, the percentage inhibition of the radical DPPH of the ethanolic extract gradually increases from 05% to 78% but remains lower than ascorbic acid and quercetol which, respectively, increased from 11% to 80% and then from 43% to 80%. The percentage inhibition of the radical DPPH, ascorbic acid, and quercetol is approximately equal to 80% on the concentration range 500 to 1,000 μg ml−1. What emerges from this comparative exercise is that the percentage inhibition of the radical DPPH is dose-dependent and maximal for these three fractions on the concentration range 500 to 1,000 μg ml−1. In addition, it has great similarities across the curves with identical paces for the ethanolic extract of O. gratissimum and ascorbic acid, what brings more these two extracts closer. These results augur good prospects for the determination of the 50% inhibitory concentrations of extract of O. gratissimum and both standards.
 | Figure 2: DPPH scavenging capacity by ethanolic extract, ascorbic acid, and quercetin. [Click here to view] |
3.6. Determination of Inhibitory Concentrations at 50%
To better understand the antioxidant power of the extract and standards, the concentrations of each sample inhibiting 50% of the DPPH radicals of the reaction medium were calculated (Table 4). The exploitation of the results recorded shows a variation of inhibitory concentrations of 50% from one range to another. The 50% inhibitory concentration is high for extract of O. gratissimum (6.175 ± 0.0074), average for ascorbic acid (4.461 ± 0.003), but weak for quercetin (1.04 ± 0.001). It results from this analysis and the previous ones that the 50% inhibitory concentration of an extract is inversely proportional to its antiradical activity. What permits to rank first quercetin in terms of antiradical capacity followed by the ascorbic acid and finally the ethanolic extract of O. gratissimum.
4. DISCUSSION
The phytochemical profile of the O. gratissimum samples obtained by the standard screening technique revealed the presence of the three major phytochemical groups: phenolic mixtures, nitrogen mixtures, steroids and terpenoids. Phenolic molecules are present through catechin tannins, gallic tannins, flavones, free anthracene derivatives, and combined anthracenic derivatives specifically reducing mixtures. Nitrogen mixtures are represented by alkaloids. The group of steroids and terpenoids is signaled by the presence of steroids and saponosides. We also have the presence of cyanogenic derivatives and mucilages which are bioactive molecules whose classification is still in progress. These results are similar with those of [31]. Moreover, these results are partially consistent with the results of [32–34]. The results of the first author reveal, apart from the antimicrobials mentioned above, the presence of resins, cardiac glycosides, steroid terpenes, and carbohydrates at different concentrations in the extracts of fresh and dried leaves of the plant. The results of the second author are characterized by the absence of flavonoids and saponosides in the samples. Similarly, the presence of triterpenes, quinone derivatives, and anthocyanins coupled with the absence of cyanogenic derivatives revealed by the results of the third author is not consistent with the mapping of the standard screening obtained. In this same comparative exercise, the tri phytochemical tests (etheric, methanolic and aqueous) and phytochemical quadruples (hexanic, chloroformic, ethylacetatic, and n-butanolic) carried out by [10] followed by [35] respectively on the extracts of O. gratissimum revealed the presence of sterols, polyterpenes, polyphenols, coumarins, flavonoids, catechin tannins, reducing sugars, saponins, and alkaloids. These different results are also confirmed by [36–39] unlike the positive phlobatannin dosage and lactone. From these results, the samples of O. gratissimum dose are more suitable for the antibacterial and anti-inflammatory therapeutic properties attached to the active biomolecules contained in the plant. The divergences noted above can be explained by the origin of the samples, the vegetative stage, collection time and season, climatic conditions, edaphic properties, and dosage techniques in the laboratory. These justifications discussed here for differences in standard phytochemical results were confirmed by the work of [40]. The results of this author show that the rich bioactive molecules of the essential oils of O. gratissimum vary according to the vegetative stage, time of collection, light, nature of the soil, types of extraction, and methods of dosage.
 | Table 4: Determination of inhibitory concentrations at 50%. [Click here to view] |
The search for secondary molecules in the ethanolic extract of O. gratissimum was positive by the presence of total tannins, total flavonoids, and total phenolics in variable proportions. The results show that O. gratissimum contains mostly total phenolics (56.59 mgGAE/100 mg) than total flavonoids (13.71 mg QE/100 mg), and total tannins (8.6 mgCE/100 mg). In a comparative work, the content of total phenolic does not reflect the results related to [41] which record 32.56 ± 16.9 gGAE/100 g and 19.55 ± 3.3 gGAE/100 g total phenolic for the fresh and cooked leaves of O. gratissimum, respectively. This amount of total phenolic content revealed in the O. gratissimum sample is also well below the results published by [42] which found 168.2 ± 3.2 mgGAE/g. In the same logic, the content in total phenolic obtained does not corroborate with the results of [43,44] which report, respectively, 7.82 mgGAE/g and 11.3 mgGAE/g extract of O. gratissimum. Considering the total flavonoids content obtained, the same comparative approach reveals the percentage of 18.02 ± 4.78 recorded by [43] in the aqueous extract of O. gratissimum.
The high content of the samples in secondary molecules, especially total phenolic, testifies to the pharmacological potential of the plant. As a result, the FRAP method shows that the ethanolic extract of O. gratissimum has an interesting anti-radical potential. This results in the reduction of ferric iron to ferrous ion, through the significant variation of optical densities (0.365–0.4995) from one concentration to another inducing an antioxidant capacity that varies from 78.92 to 106.25 mg AAE/g extract. The highest optical density value read in this study (0.4995) is significantly lower than that recorded by [42] whose average is 0.74 ± 0.04. As confirmation by the DPPH method, the antiradical activity of the ethanolic extract ranges from 5% to 77% over the concentration range of 7.8 to 1,000 μg ml−1. The best antioxidant activity (20%–75%) is observed in the concentration range 12.5 to 500 μg ml−1, but less than the antiradical activities of the quercetol and ascorbic acid standards over the whole range of concentrations (7.8–1,000 μg ml−1). Apart from the differences in concentrations, these results are similar to those of [33] which have a free radical scavenging activity of 8% to 79% for fresh, baked leaves of O. gratissimum for the extract concentrations ranging from 7.8 to 1,000 μg ml−1. These values are significantly higher than those of [41]. From the evaluation of antioxidant capacities, these authors found inhibition percentages of 42.5% (±16.61) and 26.83% (±5.2), respectively, for fresh and cooked leaves of O. gratissimum. The antiradical properties of this plant are also confirmed by [43] who showed, at the end of their work, that O. gratissimum contains a strong capacity to neutralize the radical DPPH. This permits to the phytocompounds of the plant under study a very significant antiradical power. The same study reveals that the antiradical capacity of this plant was also reported by [45] following the FRAP binary method coupled of DPPH. As regard to the results of [38], the antioxidant capacity of O. gratissimum induced by the reducing potential of phenolic compounds is a dependent dose. This reducing power would be due to the liberation of hydrogen atoms capable of reacting with free radicals, which causes the blocking of chain reactions [46]. For [47] and after them [48], plants extracts containing phenolic mixtures such as beneficial basil are very good antioxidants. The results of the anti-free radical capacity of O. gratissimum also corroborate with those of [45] by the binary method of coupled DPPH of FRAP. According to the results of [38], the antioxidant capacity of O. gratissimum based on the reducing potential of phenolic mixtures has been dose-dependent.
Based on these results, the deduction of 50% inhibitory concentrations gave 6.175, 4.461 and 2.031 mg ml−1 for O. gratissimum, ascorbic acid, and quercetin, respectively. This IC 50% value recorded for O. gratissimum in the range 4.03 to 6.04 mg ml−1 has been reported by [33]. However, the value of IC 50% of the extract obtained in this study is much greater than the result of [44] which is 83.10−3 mg ml−1.
In a comparison, the clinical tests of the acetate extract of O. gratissimum on Plasmodium falciparum gave 50 % inhibitory concentrations varying between 1,800 and 1,930 μg ml−1 [49] quoted by [2]. The results show that O. gratissimum, a plant with high antioxidant and rich in phenol, may be advisable in the fight against many of the chronic diseases remarkably spread today among the human beings [41].
Exploration of the HPLC profile initiated to deepen the standard screening and specific dosages permitted the identification and quantification of three flavonoids (quercetin, chrysin, and isorhamnetin) and six phenolic acids (ferric, ferrulic, tannic, syringic, caffeic, and chlorogenic). It should be emphasized that three of the nine compounds identified and quantified by the HPLC technique namely caffeic acid, syrigic acid, and ferrulic acid were also highlighted in the results of [42] by the same assay approach. In a comparative approach [38] observed phytochemical profiles of O. gratissimum varies according to the type of extraction. The results of these authors confirm the presence of molecules responsible for biological effects, especially phenolic acids. On the other hand, these results are divergent from the chromatographic profiles obtained by [50–52]. This variation between the results mainly from that of the analysis programs was used. Biomolecules revealed by chromatography are recognized for their antioxidant, anti-inflammatory, antimutagenic, antibacterial, antiviral, and cytotoxic properties [53,54]. Indeed, flavonoids strengthen the inner lining of blood vessels and promote the disappearance of varicose veins [18]. This justifies the traditional use of O. gratissimum against epistaxis. In the literature, catechin tannins, polyphenols, and alkaloids taken in this order are recognized for their hemostatic, antihypertensive, and antimalarial activities [10]. These molecules being present in O. gratissimum explains the use of this plant to fight these types of diseases mentioned. In addition, phenolic and catechin tannins on the one hand and alkaloids on the other hand have bactericidal and anti-ulcerous properties. This explains the use of this plant as antibiotic and anticholeric. Flavonoids and polyterpenes, respectively, of diuescent and deworming remedies [10]. These molecules present in O. gratissimum then justify its use for these types of diseases. Flavonoids and glycosides give plants anti-asthmatic and oxytocic properties [55]. This also explains the use of O. gratissimum to fight asthma and facilitate childbirth. The antiradical power of O. gratissimum is due to the presence of phenolic mixtures such as flavonoids, tannins, and coumarins. The therapeutic potential of these molecules has been demonstrated [56,57]. These mixtures also have the antimicrobial and anti-inflammatory properties [58–60]. The presence of these molecules justifies the traditional use of O. gratissimum in the fight against several types of diseases.
5. CONCLUSION
Ocimum gratissimum is used in various forms in the biological control and especially preservation and treatment of many types of diseases. These therapeutic and biological properties are induced by various active molecules. The standard phytochemical screening shows the presence of nitrogen, phenolic, steroid, terpenoid mixtures cyanogenic derivatives, mucilages, and reducing agents. The dose of the secondary molecules reveals the presence of total tannins, total flavonoids, and total phenolics. The chromatographic profile reveals the presence of eighteen distinct molecules of which nine identified and quantified divided into three flavonoids and six phenolic acids. The antiradical power of the ethanolic extract is very interesting in the concentration range of 125–500 μg ml−1. The molecules present are endowed with antibacterial, anti-inflammatory, and antioxidant activities. The richness of the plant in bioactive molecules explains the scientific aspect of its excessive use by the population for various applications in the traditional medicine.
COMPETING INTERESTS
The authors declare that they have no competing interests.
ACKNOWLEDGMENTS
The authors acknowledged all those who contributed to the realization of this work.
REFERENCES
1. Traoré Y, Ouattara K, Yéo D, Doumbia I, Coulibaly A. Recherche des activités antifongique et antibactérienne des feuilles d’Annona senegalensis Pers. (Annonaceae). J Appl Biosci 2012;(58):4234–42.
2. Bla KB, Trébissou JND, Bidié AP, Assi YJ, Zirihi-Guédé N, Djaman AJ. Etude ethnopharmacologique des plantes antipaludiques utilisées chez les Baoulé- N’Gban de Toumodi dans le Centre de la Côte d’Ivoire. J Appl Biosci 2015;(85):7775–83.
3. Etame LG, Yinyang J, Okalla Ebongue C, Makondo BV, Ngaba GP, Mpondo Mpondo E, et al. Etude de la toxicité aigüe et subaigüe de l’extrait au vin des graines de Carica papaya Linn. J Appl Biosci 2017;120:12077–85.
4. WHO/MSP. Renforcement du rôle de la médecine traditionnelle dans le système de santé au Benin, p 47, 2013.
5. Bayala B. Activité anticancéreuse des huiles essentielles de plantes médicinales du Burkina Faso sur le cancer de la prostate et le glioblastome. Thèse de Doctorat de l’Université de Ouagadougou, p 166, 2014.
6. Mangambu MJD, Mushagalusa KF, Kadima NJ. Contribution à l’étude phytochimique de quelques plantes médicinales antidiabétiques de la ville de Bukavu et ses environs. J Appl Biosci 2014;(75):6211–20.
7. Bentabet N, Boucherit-Otmani Z, Boucherit K, Ghaffour K. Preliminary phytochemical study of leaves and roots of Fredolia aretioides, endemic plant of Algeria. Scholars Research Library. Der Pharm Chem 2014;6(1):443–7.
8. Ministère de la Santé/Programme National de la Pharmacopée et de la Médecine Traditionnelles au Bénin (MS/PNPMTB). La pharmacopée et la médicine traditionnelle au Bénin: état des lieux et perspectives, p 37, 2013.
9. Mangambu M. Taxonomie, biogéographie et écologie des ptéridophytes de l’écosystème forestier des montagnes du Parc National de Kahuzi-Biega à l’Est de la R.D.C. Thèse de doctorat. Université d’Anvers, Belgique, p 463, 2013.
10. N’guessan K, Kadja B, Zirihi GN, Traoré D, Aké-Assi L. Screening phytochimique de quelques plantes médicinales ivoiriennes utilisées en pays Krobou (Agboville, Côte-d’Ivoire). Sci Nat 2009;6(1):1–15.
11. Abdelrazag H. Etude phytochimique et activité biologique de la plante Limoniastrum guyonianum. Diplôme de Magister. Université Kasdi Merbah, Ouargla, Algeria, p 111, 2013.
12. Djahra AB. Etude phytochimique et activité antimicrobienne, antioxydante, antihépatotoxique du Marrube blanc ou Marrubium vulgare L. Thèse de Doctorat en science, option Biologie Végétale, Université Badji Mokhtar, Annaba, Algérie, p 114, 2014.
13. Kpodékon MT, Boko KC, Mainil GJ, Farougou S, Sessou P, Yèhouénou B, et al. Composition chimique et test d’efficacité in vitro des huiles essentielles extraites de feuilles fraîches du basilic commun (Ocimum basilicum) et du basilic tropical (Ocimum gratissimum) sur Salmonella enterica sérotype Oakland et Salmonella enterica sérotype Legon. J Soc Ouest-Afr Chim 2013;(035):41–8.
14. Sharmin S, Kabir MT, Nasiful IM, Jamiruddin MR, Rahman I, Rahman A, et al. Evaluation of antioxidant, thrombolytic and cytotoxic potentials of methanolic extract of Aporosa wallichii Hook.f. leaves: an unexplored phytomedicine. J Appl Pharm Sci 2018;8(07):051–6.
15. Lagnika L, Amoussa AMO, Adjileye ARA, Laleye A, Sanni A. Antimicrobial, antioxidant, toxicity and phytochemical assessment of extracts from Acmella uliginosa, a leafy-vegetable consumed in Benin, West Africa. BMC Complement Altern Med 2016;16(34):11. CrossRef
16. Abdullahi HY, Musa A, Safiya BB, Abdullahi BN. Antihyperglycaemic and antihyperlipidaemic activities of ethanol leaf extract of eleusine coracana (Linn) gaertn. in lloxan-induced hyperglycaemic rats. J Appl Pharm Sci 2018;8(07):028–32.
17. Houghton PJ, Raman A. Laboratory handbook for the fractionation of natural extracts, Ed Chapman and Hall, New York, NY, p 208, 1998. CrossRef
18. Dougnon TV. Evaluation des propriétés hémostatiques de la sève de Jatropha multifida L. (Euphorbiaceae). Mémoire de Master Professionnel, UAC/EPAC, p 75, 2011.
19. Li HB, Cheng KW, Wong CC, Fan KW, Chen F, Jiang Y. Evaluation of antioxidant capacity and total phenolic content of different fractions of selected microalgae. Food Chem 2007;102(3):771–6. CrossRef
20. Wolfe K, Wu X, Liu RH. Antioxidant activity of apple peels. J Agric Food Chem 2003;51(3):609–14. CrossRef
21. Nadhiya K, Vijayalakshmi K. Evaluation of total phenol, flavonoid contents and in vitro antioxidant activity of benincasa Hispida fruit extracts. Int J Pharm Chem Biol Sci 2014;4(2):332–8.
22. Ibrahima MSA, Attakpa SE, Béhanzin GJ, Amoussa AMO, Lagnika L, Guinnin F, et al. Antioxidant and free radical scavenging activity of various extracts of Boerhavia diffusa Linn. (Nictaginaceae). Pharm Chem J 2018;5(2):62–71.
23. Koné D. Enquête ethnobotanique de six plantes médicinales maliennes, extraction, identification d’alcaloïdes, caractérisation, quantification de polyphénols: étude de leur activité antioxydante. Thèse de Doctorat, Université Paul Verlaine et Bamako, p 188, 2009.
24. Bangou, MJ. Etude phytochimique et activités biologiques des tiges feuillées de Lantana camara L. et de Lippia chevalieri Moldenke : deux verbenaceae du Burkina Faso. Thèse de Docteur de l’Université de Ouagadougou, p 199, 2012.
25. Tlili N. Procédés de séparation multi colonnes continus : Extension à la chromatographie à gradient de solvant. Thèse de Doctorat, Université de Lorraine, Lorraine, France, p 229, 2013.
26. Saeed N, Khan MR, Shabbir M. Antioxidant activity, total phenolic and total flavonoid contents of whole plant extracts Torilis leptophylla L. BMC Complement Altern Med 2012;12(221):11. CrossRef
27. Amoussa AMO, Sanni A, Lagnika L. Antioxidant activity and total phenolic, flavonoid and flavonol contents of the bark extracts of Acacia ataxacantha. J Pharmacogn Phytochem 2015;4(2):172–8.
28. Velazquez E, Tournier HA, de Bushiazzo MP, Saavedra G, Schinella GR. Antioxidant activity of Paraguayan plants extracts. Fitoterapia 2003;74:91–7. CrossRef
29. Kansole MMR, Hilou A, Millogo J, Nacoulma O. Ethnobotany and ethnopharmacognosy of Lamiaceae species from Central Burkina Faso: Leucas martinicensis (Jacquin) R. Brown, Hoslundia opposita Vahl and Orthosiphon pallidus Royle Ex Benth. Am J Ethnomed 2015;2(4):219–32.
30. Asmaa N, Boubakeur S, Boulanouar B, Mohamed M. Comparative study of antioxidant activity index by the tests of cerium and DPPH: application on three local medicinal plants. Int J Innov Appl Stud 2015; 13(3): 681-690.
31. Kpètèhoto HW, Hessou S, Dougnon VT, Johnson RC, Boni G, Houéto EEM. Etude ethnobotanique, phytochimique et écotoxicologique de Ocimum gratissimum Linn (Lamiaceae) à Cotonou. J Appl Biosci 2017;(109):10609–17.
32. Junaid SA, Olabodé AO, Onwuliri FC, Okwori AEJ, Agina SE. The antimicrobial properties of Ocimum gratissimum extracts on some selected bacterial gastrointestinal isolates. Afr J Biotechnol, 2006;5(22):2315–21.
33. Agbéssy K. Phytochimie, activité antioxydante et teneur en ETM de trois légumes feuilles consommés au sud-Benin. Mémoire de Master en biochimie, UAC/FAST, p 48, 2018.
34. Kouchadé SA, Adjatin AR, Adomou AC, Dassou HG, Akoègninou A. Phytochimiques des plantes médicinales utilisées dans la prise en charge des maladies infantiles au Sud-Bénin. Eur Sci J 2017;13(3):471–88. CrossRef
35. N’guessan HA, Dago DCE, Mamyrbékova-Békro JA, Békro YA. CCM d’extraits sélectifs de 10 plantes utilisées dans le traitement traditionnel de l’hypertension artérielle en Côte d’Ivoire. Eur J Sci Res 2011;66(4):575–85.
36. Ojo OA, Oloyede OI, Olarewaju OI, Ojo AB, Ajiboye BO, Onikanni SA. Toxicity studies of the crude aqueous leaves extracts of Ocimum gratissimum in albino rats. J Environ Sci Toxicol Food Ttechnol 2013;6(4):34–9.
37. Okon UA, Obembe AO, Olubobokun TH, Owu DU. Anti-gastric ulcerative activity of Ocimum gratissimum in streptozotocin-induced diabetic rats. J Sci Res Rep 2013;2(1):408–22.
38. Ajayi AM, Naluwuge A, Buyinza P, Luswata I. Comparative physicochemical, phytochemical and acute toxicity studies of two Ocimum species in Western Uganda. J Med Plants Res 2017;11(1):1–10. CrossRef
39. Talabi JY, Makanjuola SA. Proximate, phytochemical, in vitro antimicrobial properties of dried leaves from Ocimum gratissimum. Prev Nutr Food Sci 2017;22(3):191–4.
40. Yayi E, Moudachirou M, Chalchat JC. Chemotyping of three Ocimums pecies from Benin: Ocimum basilicum, canum, gratissimum. J Essent Oil Res 2001;13(1):13–7. CrossRef
41. Assogba KF, N’Danikou S, Honfoga J, Sossa CV, Mensah A, Simon S. Ocimum gratissimum L. (Tchiayo): Du jardin à la table, Fiche technique INRAB, p 3, 2015.
42. Hakkim LF, Arivazhagan G, Boopathy R. Antioxidant property of selected Ocimum species and their secondary metabolite content. J Med Plants Res 2008;2(9):250–7.
43. N’Guessan AHO, Déliko CED, Mamyrbékova-Békro JA, Békro Y-A. Teneurs en composés phénoliques de 10 plantes médicinales employées dans la tradithérapie de l’hypertension artérielle, une pathologie émergente en Côte d’Ivoire. Rev de génie ind 2011;(6):55–61.
44. Gontijo DC, Fietto LC, Leite JPV. Phytochemical evaluation and antioxidant and antimutagenic and toxicological activity of aqueous extract of leaves of Ocimum gratissimum L. Rev Bras Plantas Med 2014;16(4):874–80. CrossRef
45. Ferreira IC, Baptista P, Vilas-Boas M, Lillian B. Free-radical scavenging capacity and reducing power of wild edible mushrooms from northeast Portugal: individual cap and stipe activity. Food Chem 2007;(100):1511–6. CrossRef
46. Dabire C, Nébie RHC, Belanger A, Nacro M, Sib SF. Effet du séchage de la matière végétale sur la composition chimique de l’huile essentielle et l’activité antioxydante d’extraits de Ocimum basilicum L. Int J Biol Chem Sci 2011;5(3):1082–95. CrossRef
47. Agbodan KA, Dotse K, Koumaglo KH. Activités anti oxydantes des H.E de trois plantes aromatiques acclimatées au Togo. Int J Biol Chem Sci 2014;8(3):1103–10. CrossRef
48. Agbodan KA, Dotse K, Koumaglo KH, Saloufou KI. Effets d’un acide fort sur la réaction de la DPPH avec deux huiles essentielles de deux plantes aromatiques acclimatées au Togo. Int J Biol Chem Sci 2015;9(6):2932–9. CrossRef
49. Abiodun O, Gbotosho G, Ajaiyeoba E, Happi T, Falade M, Wittlin S, et al. In vitro antiplasmodial activity and toxicity assessment of some plants from Nigerian ethnomedicine. Pharm Biol 2010;49(1):9–14. CrossRef
50. Salawu SO, Ogundare AO, Ola-Salawu BB, Akindahunsi AA. Antimicrobial activities of phenolic containing extracts of some tropical vegetables. Afr J Pharm Pharmacol 2011;5(4):486–92.
51. Casanova LM, da Silva D, Sola-Penna M, Camargo LM, Celestrini DM, Tinoco LW, et al. Identification of chicoric acid as a hypoglycemic agent from Ocimum gratissimum leaf extract in a biomonitoring in vivo study. Fitoterapia 2014;(93):132–41. CrossRef
52. Bonta KR. Application of HPLC and ESI-MS techniques in the analysis of phenolic acids and flavonoids from green leafy vegetables. J Pharm Anal 2017;7(6):349–64. CrossRef
53. Kelebek H, Selli S, Gubbuk H, Gunes E. Comparative evaluation of volatiles, phenolics, sugars, organic acids and antioxidant properties of Sel-42 and Tainung papaya varieties. Food Chem 2015;(173):912–9. CrossRef
54. Penchev PI. Etude des procédés d’extraction et de purification de produits bioactifs à partir de plantes par couplage de techniques séparatives à basses et hautes pressions. Thèse de Doctorat de l’Université de Toulouse, p 239, 2010.
55. Adjanohoun E. et Aké-Assi L. Contribution au recensement des plantes médicinales de Côte d’Ivoire. Ministère de la recherche scientifique, p 359, 1979.
56. Mamadou RS, Moussa I, Sessou P, Yehouenou B, Agbangnan PDC, Illagouma AT, et al. Etude phytochimique, activités antiradicalaire, antibactérienne et antifongique d’extraits de Sebastiania chamaelea (L.) Müll. Arg. J Soc Ouest-Afr Chim 2014;(037):10–7.
57. Sharma P, Vidyasagar G, Singh S, Ghule S, Kumar B. Antidiarrheal activity of leaf extract of Celosia argentea in experimentally induced diarrhea in rats. J Adv Pharmacol Technol Res 2010;1(1):41–8.
58. Ogundaré AO, Adetuyi FC, Akinyosoye FA. Antimicribial activity of Vernonia tenoreana. Afr J Biotechnol 2006;5(18):6.
59. Gulcin I, Huyut ZO, Elmastas M, Aboul-Enein HY. Radical scavenging and antioxydant activity of tannic acid. Arabian J Chem 2010;3(1):43–53. CrossRef
60. Bidié AP, N’guessan BB, Yapo AF, N’guessan JD, Djaman AJ. Activités antioxydantes de dix plantes médicinales ivoirienne. Sci Nat 2011;8(1):1–11.