1. INTRODUCTION
On the whole, heavy metals are best defined as metals with a specific density >5 g/cm3 and can affect the environment and living organisms’ adversely [1]. Some heavy metals such as - arsenic (As), cadmium (Cd), mercury (Hg), and lead (Pb) can be toxic and detrimental to health even at trace concentrations [2]. There is no argument suggesting that other heavy metals such as copper (Cu), chromium (Cr), molybdenum, nickel (Ni), selenium, and zinc (Zn) forms important constituents in diets/foods for plants and humans. Perhaps, this can be beneficial if the metal elements are present in moderate amount. Fruits form an important part of human diets due to the composition of carbohydrates, proteins, vitamins, minerals, and trace elements [3]. The contamination of fruits with heavy metals from the soil and atmosphere question their quality and safety [4]. The consumption/ingestion of fruits with high concentrations of heavy metals (Ar, Cu, Cd, and Pb) has been implicated in the causation of gastrointestinal cancer [5]. Other anthropogenic sources such as waste incineration, industrial processes, and vehicular traffic emit heavy metals into the atmosphere [6].
The consumption/ingestion of contaminated fruits is the direct route where humans and animals are exposed to heavy metal [7,8]. Abandoned waste dumpsites have been used extensively for the cultivation of fruits crops. Furthermore, research has shown that fruits are capable of accumulating high levels of heavy metals from polluted soils through translocation [9]. It will be of great benefit if one appreciates that heavy metals are persistent and non-biodegradable [10]. They might be transported from soil to ground waters or taken up by crops. Based on this premise, the knowledge about metals to plants interactions is important for environmental safety and human health risk [2]. There has been an increasing quest for determining heavy metal levels in public food supplied. Though, heavy metals concentration in bio-available form may not be proportional to the total concentration in the soil ecology, that is because some of the heavy metals are either transform or found in combined state that render them inactive [11,12].
There has been loud and persuasive advocacy about the benefits of consuming foods from plants sources, especially fruits from the public health sector. In carrying out this advocacy, nobody was able to give a corresponding explanation about the health effect of ingesting toxic constituents in the fruits. At present, there is no regulation on the quality of fruits sold in most of Nigeria markets. It is well understood that the irrigation of crops (fruits) with pharmaceutical effluents, industrial discharges, and domestic wastewater has enriched the soils with heavy metals. Heavy metals are toxic to humans and consumption of these contaminated fruits could result in a much human health risk.
An elevated dietary iron intake enhances the incidence of carcinogen-induced mammary tumors in rats and estrogen-induced kidney tumors in Syrian hamsters [13]. Estrogen administration increases iron accumulation in hamsters and facilitates iron uptake by cells in culture. In humans, increased body stores of iron have been shown to increase the risk of several estrogen-induced cancers [14]. Increased incidence of congenital malformations in infants has also been associated with Zn deficiency in lactating mothers [15].
Pb toxicity has been linked to low intelligent quotients in children and found to affects several organs and systems - blood, central nervous system, kidneys, reproductive, and immune systems [16]. Cd in the body is known to affect the renal tubules and causes damage that results in proteinuria. Cd affects the enzymes responsible for reabsorption of proteins in kidney tubules [17], Cd reduces the activity of delta-aminolevulinic acid synthetase, arylsulfatase, alcohol dehydrogenase, and lipoamide dehydrogenase, respectively. Cr is associated with allergic dermatitis in humans [18]. Furthermore, hazard identification of biochemical and physical agents ensures that particular food or group of foods is free from substances that may induce negative health consequence. Hazard identification as it is applied to metals toxicity, aimed at evaluating the weight of evidence on post consumption of selected edible fruits to ascertain the extent of health risk on humans [19]. This judgment provides a quantitative/qualitative estimate of risk. It is established health-based guidance values such as - Acceptable Daily Intake (ADI) and Tolerable Daily Intake (TDI). This form the index where human exposure is considered to be without appreciable health risk [20]. We carry out this study to evaluate perceived human health risk associated with the consumption of fruits.
2. MATERIALS AND METHODS
2.1 Fruits
Ten edible fruits were used in this study - Solanum lycopersicum (Tomatoes), Cucumis sativus (Cucumber), Solanum melongena (Nigerian Egggarden), Citrullus lanatus (Watermelon), Dacryodes edulis (African bush pear), Juglans regia (Walnut), Syzgium malaccense (Mountain apple), Annona muricata (Soursop), Pachycarpa kschum (Monkey kola), and Dennettia tripetala (Pepper fruit).
2.1.1 Chemicals/Reagents
The chemicals and reagents used in the study were of analytical grade and were sourced from chemical stores in Onitsha market, Anambra State and Multiuser Laboratory at the Department of Chemistry, Ahmadu Bello University Zaria.
2.1.2 Collection of fruit samples
Ten fruits were bought from Ubani market in Umuahia, Abia State, Nigeria. Ubani market is a centralized modern market in Umuahia where people come to buy and sell goods.
2.1.3 Preparation of fruit samples
The fruits samples were washed thoroughly with tap water followed by distilled water to remove adsorbed elements (pollutants). Samples were cut into small pieces and air dried for 48 h in a hot air oven at 100°C ± 1°C. Dried samples were pulverized to a fine powder and sieved with a 1 mm mesh. Each fruit sample was labeled (D1–D10). They were stored in a dry plastic container.
2.1.4 Digestion of fruits samples
To each fruit sample, 0.5 g of dried sample was digested with HNO3, and HClO4, in a 5:1 ratio until a transparent solution was obtained as described by World Health Organization/Food and Agriculture Organization [21]. The fruit digests were filtered and diluted to 25 ml with distilled water. The heavy metals in the acid digests of fruit samples were analyzed with atomic adsorption spectrophotometer (AAS).
2.1.5 Calibration of AAS
Standard solutions of the heavy metals, namely Pb, Cd, and Cu, cobalt (Co), Ni, and Zn, were purchased from Merck (Darmstadt, Germany). The standards reagents were prepared from the individual 1000 mg/l standards (Merck) supplied in 0.1N HNO3. A series of working standards were prepared from these standard reagents.
2.2 Determination of Heavy Metal Concentration in Fruit Samples
The concentration of heavy metals was analyzed using atomic absorption spectrophotometer, (Thermo Scientific Pvt. Ltd. India Model No. AA 303). A double beam and deuterium background hollow cathode lamps of Fe, Pb, Cd, Zn, and Cu was used at specific wavelengths. All samples were run in triplicates.
2.3 Determination of Daily Intake of Heavy Metals (DIM)
The DIM were determined using the method of Ali et al. [22]. DIM was calculated using equation 1.
2.3.1 DIM
Where: Cmetal is the metal concentration in vegetable in mg/kg.
D/fruits intake is the daily intake of fruit/vegetable (300 g/kg/day) (WHO, 2018).
BWaverage is the average body weight of a Nigerian 62 kg (WHO, 2018).
2.4 Determination of Non-Carcinogenic Risk (CR) Index
The non- CR assessments are conducted to estimate the potential health risks of pollutants using the target hazard quotient (THQ). The THQ values through the consumption of fruits were analyzed for each heavy metal, while the calculations were computed using the standard assumption for an integrated [23] risk analysis in line with Equation 2.
Where: DIM is the DIMs (Mg/Kg/person)
Reference dose (RfD) is the oral reference dose (mg/kg/d).
RfDs are based on 0.04, 0.02, 0.03, 0.7, 0.003, 0.001, 0.014, 0.3, and 0.004 mg/kg/day for Cu, Ni, Co, Fe, Cr, Cd, Mn, Zn, and Pb, respectively [23].
If the THQ value is equal to or >1, there is a potential health risk, and related <1, the exposed population is unlikely to experience any adverse health hazard. Conversely, if the THQ is interventions and protective measurements should be taken [24].
2.5 Determination of Hazard Index (HI)
To estimate the risk to human health through more than one heavy metal the concept of hazard index (HI) was initiated. The HI is the sum of the HQ for all HMs, which was calculated by the Equation 3 [25].
Where: HI = Hazard Index
THQ = Target HQ
HQ for the locals (consumers) through the consumption of contaminated fruits were analyzed by finding the ratio of DIM to the oral RfD (for each metal). If the value of HQ is <1, then the exposed local population (consumers) is said to be safe if HQ is equal to or >1, it is considered unsafe for human health.
2.6 Determination of CR Index
CR indicates an incremental probability of an individual to developing cancer over a lifetime due to exposure to a potential carcinogen. Cancer risk over a lifetime exposure to Ni, Cr, Cd, and Pb was obtained by equation 4 using cancer slope factor (CSF).
Where: CSF is the oral carcinogenic slope factor of 0.0085, 0.38, 0.5, and 1.7 (mg/kg/day) for Pb, Cd, Cr, and Ni, respectively, and 1.5 (mg/kg/day) for as set by USEPA [23]. EDI is the estimated DIM. Acceptable risk levels for carcinogens range from 10−4 (risk of developing cancer over a human lifetime is 1 in 10,000) to 10−6 (risk of developing cancer over a human lifetime is 1 in 1,000,000).
2.7 Determination of Total CR
The cumulative cancer risk due to exposure to multiple sources of carcinogenic heavy metals through consumption of a particular type of fruit and was assumed, and the sum of the individual heavy metal increment risk was calculate following equation 5 [26].
Where CR is the cancer risk.
2.8 Statistical Analysis
The data were subjected to One-way analysis of variance. Significant differences were accepted at P < 0.05. Results are expressed in means ± standard errors of means. The analysis was computed using Statistical Product and Service Solutions software version 22.
3. DISCUSSION
Heavy metals are considered the most important constituents of pollution from the terrestrial environment due to toxicity, partitioning in the soil, and accumulation in plants [27]. Results in Table 1 show the concentration of heavy metals in fruits sold at Ubani market against together with their respective permissible limits. It was observed that the concentration of Cd in the fruits was above the permissible limit (0.001 ± 0.000 ppm). Their values range from 0.033 ± 0.002 in C. sativus to 0.042 ± 0.001 in D. tripetala. Cr and Cu concentration was also above the permissible limits 0.003 ± 0.000 and 0.010 ± 0.000, respectively, an exception of A. maricata where its Cr concentration was not detected. It was further observed that the concentration of iron and manganese was below their respective permissible limit - 5.000 ± 0.000 and 0.300 ± 0.000. Ni and Co concentration was also below their respective permissible limits. However, S. melongena, J. regia, Amuricata, P. kschum, and D. tripetala had no detectable concentration of Co, while Co concentration in S. melongena and A. muricata had no concentration. The concentration of Zn and Pb was not detected, an exception of J. regia (0.001 ± 0.000) - Zn concentration, which was below the permissible limit (2.000 ± 0.000 mg/kg). Toxic metals are commonly found in foods, drinking water, and air [28,29]. Metal poisoning occurs due to an accumulation of toxic metals in the body. Metals become toxic when the quantity is reasonably high such that its concentration may reason injury to the target cells. Heavy metals such as mercury, Pb, and Cd are mostly considered to elicit effective harmfulness to cells [29]. They cause damage at the cellular level by initiating oxidative stress, which is attributed to the causation/development of many diseases condition including mental retardation [30].
Heavy metals are included in many fertilizers and pesticides as micronutritional or biocidal components; they may be represented as naturally occurring contaminants. Most of the fruits samples collected from Ubani market contained detectable levels of the studied heavy metals. The accumulation of these heavy metals in fruits and vegetables may suggest health risk to the consumers, especially for a population with high consumption rates [31]. Thus, estimating daily intake or “tolerable intake” is widely used to describe safety levels of intake of heavy metals. Cr has been described as an essential trace element in humans and some animals [32]. The daily intake for Cr in this study was below the established TDI of 1.5E-01 [33]. The concentration of Pb and Zn in most fruits in Table 1 was not detected; hence, their daily intake was calculated. Some researchers reasoned that the concentration of Cd, Cr, and Cu in some edible fruits samples were usually above the permissible limits as listed by Gowd at al. [34]’ in view of this, the route of elemental absorption and trans-location was reported by Adewole and Uchegbu [35] that fruits accumulate Cd from soil much more efficiently than any other heavy metals. Cr is a mineral found in fruits which plays a role in the metabolism of nutrients through its impact on insulin. Cr picolinate is the form found in dietary supplements [36]. Cu is also an essential micronutrient which functions as a biocatalyst and in humans, it is required for body pigmentation, and helps to maintain a healthy central nervous system and prevent anemia [3]. However, slight increases in the levels of Cu and Cr (beyond the permissible limits) may interfere with physiological processes. For instance, chronic exposure to Cu and Cr can damage the liver and kidney [35]. The data in Table 2 show the DIM in fruit sold in Ubani market. It was observed that the daily intake of Cd in all the fruits was below the TDI (4.0E-02 mg/kg/day) in D. edulis and C. sativus having the highest and least values of daily intake of Ni from S. melongena, J. regia, A. muricata, P. kschum, and D. tripetala, respectively. However, the daily intake of Ni from S. lycopersicum, C. sativus, C. lunatus, D. edulis, and S. malaceense, respectively, was below the TDI of Ni (2.0E-02 mg/kg/day). Their ranges vary from 4.5E-05 mg/kg/day in C. lanatus to 3.3E-04 in D. edulis. It was also recorded that with the exception of C. lanatus and A. muricata which indicates no daily intake of Co, other fruits indicated that the daily intake of Co from their consumption was below the TDI which varies from 5.0E-05 (mg/kg/day) in P. kschum to 4.1E-02 (mg/kg/day) in S. lycopersium.
THQ through the consumption of fruits and vegetables is a measure of chemical contaminants. It is not a measure of risk but indicates a level of concern [24]. The observed THQ of manganese was approximately 1 indicating that consumers of Aciagrion africanum, Talinum triangulare, and Gongronema latifolium from Ubani are exposed to the health risk of manganese. The fruits studied indicate no adverse health effect because the THQ is <1; hence, it is safe for consumption (Table 3). Li Z [37], reported that the highest THQ value poses a relatively higher potential health risk to human [37].
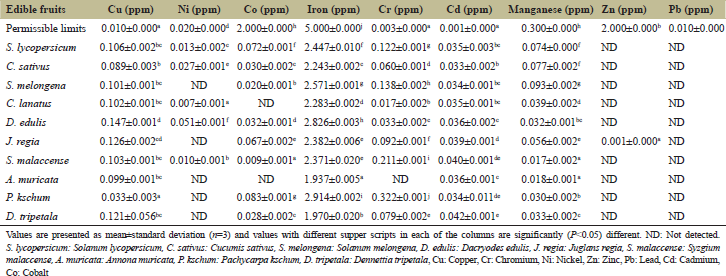 | Table 1: Heavy metals in fruits sold at Ubani Market in Umuahia, against their respective permissible limits in fruits [Click here to view] |
 | Table 2: DIM from fruits sold in Ubani market Umuahia (mg/kg/day) [Click here to view] |
Results in Table 3 show the THQ of heavy metals of fruits sold at Ubani market. There was no THQ for Pb and Zn, except J. regia for Zn. A. muricata had no THQ for Cr, Co, and Ni. Furthermore, S. melongena, J. regia, P. kschum, and D. tripetala had no THQ for the Ni. It was observed that Cu, iron, Cd, and manganese had THQ in all fruits which were <1. The highest range of THQ was noted in Cd which ranged from C. sativus (0.213) to D. tripetala (0.271). Table 4 represents the HI of heavy metals of fruits sold at Ubani market. It was observed that none of the fruits had HI ≥ 1. All fruits had HI <1, which ranges from A. muricata (0.2740) to S. lycoporsicum (0.3525). The HI of all fruits in Table 4 indicates no adverse health effect because the HI is <1.
The cancer risks of heavy metals in fruits sold at Ubani market is shown in Table 5. All the fruits show no high cancer risk except P. kschum (1.1E-03) for Cr. Pb indicated no cancer risk in all the studied fruits. The cancer risk of Cd ranges from A. muricata < C. sativus < S. melingena < C. lanatus < S. lycopersicum = D. edulis < J. regia < S. malaccense = P. kschum < D. tripetala. All studied fruits samples had cancer risk of Cr which was within the acceptable cancer risk except A. muricata that was not indicated. In the case of Ni, S. melongena, J. regia, A. muricata, P. kschum, and D. tripetala had no cancer risk. CR is estimated and expressed as a probability of contracting cancer over a lifetime of 70 years. The average value of cancer risk for all studied vegetables did not show carcinogenicity thereby rendering it safe for consumption. However, in comparison with established guideline values, data from this study are suggestive that P. kschum collected from Ubani may not be safe for consumers as it shows high cancer risk and as such consumers have the probability of contracting cancer over an extended lifetime of 70 years. An acute oral dose of Zn may cause symptoms such as tachycardia, vascular shock, dyspeptic nausea, vomiting, diarrhrea, pancreatitis, and damage of hepatic parenchyma cells [38]. When high levels of Zn are ingested, inhibition of Cu absorption through interaction with metallothionein at the brush border of the intestinal lumen may occur. Both Cu and Zn may bind to the same metallothionein protein, but Cu has a higher affinity for metallothionein than Zn and readily displaces Zn from metallothionein protein [39]. Cu complexes with metallothionein are retained in the mucosal cell, relatively unavailable to be transferred to the plasma, and can be excreted in feces when the mucosal cells are sloughed off. Thus, an excess of Zn may result in a decreased bioavailability of dietary Cu and the development of Cu deficiency [40]. On the other hand, Zn deficiency has been associated with dermatitis, anorexia, growth retardation, poor wound healing, hypogonadism with impaired reproductive capacity, impaired immune function, and depressed mental function [41,42].
 | Table 3: THQ s of heavy metals of fruits sold in Ubani market Umuahia [Click here to view] |
 | Table 4: HI of heavy metals of fruits sold in Ubani market ‑ Umuahia [Click here to view] |
 | Table 5: Cancer risks of heavy metals in fruits sold at Ubani market in Umuahia [Click here to view] |
 | Table 6: Total cancer risk of heavy metals in fruits sold at Ubani market in Umuahia [Click here to view] |
The analysis in Table 6 represents the total cancer risk of heavy metals in fruits sold at Ubani market. It was observed that all the fruits had its cancer risk below 10−6, - Incremental life cancer risk [ILCR]<10−6. The fruit samples S. lycopersicum, C. sativus, D. edulis and S. malaccense were within the same range. The fruit samples - A. muricata and C. lanatus had the highest total cancer risk of Pb. Following up with this analysis, one can infer that some of the fruits are safe, while some are unsafe for consumption. The big question is how can one identify a particular fruit that is laden with heavy metals? That is why this investigation was conducted to create consumer awareness to avert any possible health hazard that may arise from the consumption of/fruit with heavy metals.
4. CONCLUSION
The fruit samples used in this study are safe for consumption except (P. kschum) which showed a high concentration of Cr and high cancer risk rendering it unsafe for consumption. The findings on the ADI, HQ, and HI revealed that consumption of G. latifolium could pose CRs to human health due to the high concentration of Cu, Ni, Cr, Cd, and manganese.
COMPETING INTEREST
The authors hereby declare no conflicts of interest regarding the publication of this investigation.
AUTHOR’S CONTRIBUTIONS
All authors contributed equally.
REFERENCES
1. Järup L. Hazards of heavy metal contamination. Br Med Bull 2003;68:167-82. CrossRef
2. Divrikli U, Horzum N, Soylak M, Elci L. Trace heavy metal contents of some spices and herbal plants from western Anatolia, Turkey. Int J Food Sci Technol 2013;41:712-6. CrossRef
3. Sobukola OP, Adeniran OM, Odedairo AA, Kajihausa OE. Heavy metal levels of some fruits and leafy vegetables from selected markets in Lagos, Nigeria. Afr J Food Sci 2007;4:389-93.
4. Yusuf AA, Arowolo TA, Bamgbose O. Cadmium, copper and nickel levels in vegetables from industrial and residential areas of Lagos city, Nigeria. Food Chem Toxicol 2003;41:375-8. CrossRef
5. TürkdoÄŸan MK, Kilicel F, Kara K, Tuncer I, Uygan I. Heavy metals in soil, vegetables and fruits in the endemic upper gastrointestinal cancer region of Turkey. Environ Toxicol Pharmacol 2003;13:175-9. CrossRef
6. Czarnek K, Terpiłowska S, Siwicki AK. Selected aspects of the action of cobalt ions in the human body. Cent Eur J Immunol 2015;40:236‑42. CrossRef
7. Sajjad K, Robina F, Shagufta S, Mohammed AK, Maria S. Health risk assessment of heavy metals for population via consumption of vegetables. World Appl Sci J 2009;6:1602-6.
8. Tsafe AI, Hassan LG, Sahabi DM, Alhassan Y, Bala BM. Evaluation of heavy metals uptake and risk assessment of vegetables grown in Yargadama of Northern Nigeria. J Basic Appl Sci Res 2012;2:6708‑14.
9. Benson NU, Ebong GA. Heavy metals in vegetables commonly grown in a tropical garden ultisol. J Sustain Trop Agric Res 2005;16:77-80.
10. Khadeeja R, Sobin A, Umer R, Muhammad I, Saadia H, Tehreema I, et al. Comparison of proximate and heavy metal contents of vegetables grown with fresh and wastewater. Pak J Bot 2013;45:391‑40.
11. Nwachukwu MA, Feng H, Allinor J. Assessment of heavy metal pollution in soil and their implications within and around mechanic villages. Int J Environ Sci Technol 2010;7:347-58. CrossRef
12. Opaluwa OD, Aremu MO, Ogbo LO, Magaji JI, Odiba IE, Ekpo EK. Assessment of heavy metals in water, fish and sediments from UKE stream, Nasarawa state, Nigeria. Curr World Environ 2012;7:213-20. CrossRef
13. Chishti KA, Khan FA, Hassan SS. Estimation of heavy metals in the seeds of blue and white capitulum’s of Silybum marianum grown in various districts of Pakistan. J Basic Appl Sci 2011;7:45-9.
14. Liehr JG, Jones JS. Role of iron in estrogen-induced cancer. Curr Med Chem 2001;8:839-49. CrossRef
15. Silbergeld EK. Facilitative mechanisms of lead as a carcinogen. Mutat Res 2003;533:121-33. CrossRef
16. Neal AP, Guilarte TR. Mechanisms of heavy metal neurotoxicity: Lead and manganese. J Drug Metab Toxicol 2012;S5:1-13.
17. Manahan SE. Toxicological Chemistry and Biochemistry. 3rd ed. Boca Raton: CRC Press, Limited Liability Company (LLC); 2003.
18. Scragg A. Environmental Biotechnology. 2nd ed. Oxford, UK: Oxford University Press; 2006.
19. Osma E, Serin M, Leblebici Z, Aksoy A. Heavy metals accumulation in some vegetables and soils in Istanbul. Ekoloji 2012;21:1-8. CrossRef
20. Idodo-Umeh G, Ogbeibu E. Bioaccumulation of heavy metals in cassava tubers and plantain fruits grown in soils impacted with petroleum and non-petroleum activities. Resour J Environ Sci 2010;4:33. CrossRef
21. World Health Organization/Food and Agriculture Organization. Codex Alimentarius Commission. Food Additives and Contaminants. Joint FAO/WHO Food Standards Programme. No. ALINORM 01/12A; 2001. p. 1-289.
22. Ali Z, Hadi R, Amir HM. Health risk assessment of heavy metals in vegetables in an endemic esophageal cancer region in Iran. Health Scope 2018;7:e12340.
23. USEPA. Treatment Technologies for Site Cleanup: An Annual Status Report 2007. 12th ed. Washington, DC, USA: Solid Waste and Emergency Response (5203P); 2010.
24. Wang X, Sato T, Baoshan X. Health risk of heavy metals to the general public of Tianjin, China via consumption of vegetables and fish. Sci Total Environ 2005;350:28-37. CrossRef
25. Gezahegn WW, Srinivasulu A, Aruna, B, Banerjee S, Sudarshan M, Narayana PV, et al. Study of heavy metals accumulation in leafy vegetables of Ethiopia. IOSR J Environ Sci Toxicol Food Technol 2017;11:57-68. CrossRef
26. Mwegoha WJ, Kihampa C. Heavy metal contamination in agricultural soils and water in Dar es Salaam city, Tanzania. Afr J Environ Sci Technol 2010;4:763-9.
27. Alam MG, Snow ET, Tanaka A. Arsenic and heavy metal contamination of vegetables grown in Samta village, Bangladesh. Sci Total Environ 2003;308:83-96. CrossRef
28. Singh A, Sharma RK, Agrawal M, Marshall FM. Risk assessment of heavy metal toxicity through contaminated vegetables from wastewater irrigated area of Varanasi, India. Int Soc Trop Ecol 2007;51:375-87.
29. Jaishankar M, Mathew BB, Shah MS, Gowda KR. Biosorption of few heavy metal ions using agricultural wastes. J Environ Pollut Hum Health 2014;2:1-6.
30. Kim HL, Seo YR. Molecular and genomic approach for understanding the gene-environment interaction between Nrf2 deficiency and carcinogenic nickel-induced DNA damage. 2012;28:1959-67.
31. Díez S, Delgado S, Aguilera I, Astray J, Pérez-Gómez B, Torrent M, et al. Prenatal and early childhood exposure to mercury and methylmercury in Spain, a high-fish-consumer country. Arch Environ Contam Toxicol 2009;56:615-22. CrossRef
32. Nguta JM. Essential Trace Elements: Trace Elements in Human and Animal Health. Saarbrücken, Germany: LAP, Lambert Academic Publishing; 2010.
33. USDOE. The Risk Assessment Information System (RAIS). Oak Ridge, TN, USA: U.S. Department of Energy Oak Ridge Operations Office (ORO); 2011.
34. Gowd SS, Reddy MR, Govil PK. Assessment of heavy metal contamination in soil at Jajmau (Kanpur) and unnao industrial areas of the Ganga plain, Uttar Pradesh, India. J Hazardous Mater 2010;174:113-21. CrossRef
35. Adewole MB, Uchegbu LU. Properties of soils and plants uptake within the vicinity of selected automobile workshops in Ile-Ife, Southwestern Nigeria. Ethiop J Environ Stud Manage 2010;3:23-8.
36. Vupputuri S, He J, Muntner P, Bazzano LA, Whelton PK, Batuman V. Blood lead level is associated with elevated blood pressure in blacks. Hypertension 2003;41:463-8. CrossRef
37. Li Z, Zhang D, Wei Y, Luo L, Dai T. Risk assessment of trace elements is cultured from freshwater fishes from Jiangxi Provence, China. Environ Monit Assess 2014;186:2185-94. CrossRef
38. Salgueiro MJ, Zubillaga M, Lysionek A, Sarabia M, Caro R. Zinc as an essential micronutrient. Nutr Res 2000;20:737-55. CrossRef
39. Krezel A, Maret W. Different redox states of metallothionein/thionein in biological tissue. Biochem J 2007;402:551-8. CrossRef
40. Gyorffy EJ, Chan H. Copper deficiency and microcytic anemia resulting from prolonged ingestion of over-the-counter zinc. Am J Gastroenterol 1992;87:1054-5.
41. Prasad AS. Discovery of human zinc deficiency: 50 years later. J Trace Elements Med Biol 2012;26:66-9. CrossRef
42. Adah CA, Abah J, Ubwa ST, Ekele S. Soil availability and uptake of some heavy metals by three staple vegetables commonly cultivated along the South bank of river benue, Makurdi, Nigeria. Int J Environ Bioenerg 2013;8:56-67.