1. INTRODUCTION
Methicillin-resistant Staphylococcus aureus (MRSA) is well known as a public health problem worldwide [1]. Approximately 94,360 invasive MRSA infections were reported in the United States in 2005 which resulted in more than 18,000 deaths per year [1]. In Malaysia, MRSA infections in hospitals have increased from 17% in 1986 to 44.1% in 2007 [2]. This problem not only resulted in significant mortality and morbidity but also increase health-care costs [1]. MRSA Mu50 strain was first reported in Japanese hospitals [3]. It is highly resistant to beta-lactam antibiotic group and moderately resistant to vancomycin [4]. The changes in the structure of the bacterial cell wall resulted from the emergence of VRSA phenotype of Mu50 strain. Unlike MRSA strain, Mu50 strain has a thicker cell wall, resulting in more cell wall materials being released into the culture medium. An increase in the level of these materials eventually promotes the rates of autolysis [5]. The emergence of multi-resistant microorganisms sparked an interest in the search for natural sources as novel compounds with promising antimicrobial activity [6].
Canarium odontophyllum, which is known as “dabai” locally, is a famous indigenous fruit of Sarawak. C. odontophyllum belongs to the Burseraceae family and grows naturally in the wild environment [7]. The leaves from C. odontophyllum were found to have antimicrobial activity [8]. Recently, the acetone bark extract from C. odontophyllum was also reported to demonstrate bactericidal effect against S. aureus ATCC 25923 from minimum inhibitory concentration (MIC) and minimum bactericidal concentration (MBC) assays [9]. The present study, therefore, aims to investigate the antimicrobial activity of C. odontophyllum acetone stem bark extract against the resistant strain of S. aureus, namely MRSA Mu50 strain using time-kill assay (TKA) and post-antibiotic effect (PAE) assessments.
2. MATERIALS AND METHODS
2.1. Plant Extract
C. odontophyllum stem bark was collected from Kuching, Sarawak. All plant parts were authenticated and deposited in the Herbarium of the Universiti Kebangsaan Malaysia (UKM) with voucher specimen number UKMB 40052.
2.2. Preparation of Acetone Extract
The acetone extract of C. odontophyllum stem bark was prepared based on the previous study with some modification [10]. A total of 100 g of the dried C. odontophyllum stem bark was mixed with 500 ml absolute acetone. The mixture was shaken at 100 rpm for 24 h at room temperature. The mixture was then filtered through Whatman No. 1 filter paper. The residue was further extracted using 350 ml and 150 ml of absolute acetone. The filtrates were pooled together. The acetone solvent from the filtrates was removed using the rotary evaporator to form a pellet. The solvent was further removed by drying the pellet using a hot air-dryer. The pellet was then finely grounded using a mortar and pestle to produce a dry bark acetone crude extract.
2.3. Preparation of Antibacterial Agents
The acetone bark extract was dissolved in 10% dimethyl sulfoxide while vancomycin was dissolved in sterile distilled water. The stock solution for vancomycin was prepared at concentration 1 mg/ml and was stored at 4°C for further use.
2.4. Preparation of Bacterial Inoculum
Bacteria strain used in this experiment was MRSA Mu50 strain. The strain was obtained from the UKM Medical Molecular Biology Institute in Collaboration with Professor Keiichi Hiramatsu, Director of Infection Control Science Research Center of Juntendo University, Tokyo, Japan. The bacteria were subcultured on Mueller-Hinton agar (MHA) plate at 37°C for 24 h. About 2 or 3 single colonies with the same colony morphology were inoculated into 10 ml Mueller Hinton broth (MHB) and incubated at 37°C for 24 h. The size of inoculum was determined at optical density reading of 0.08 using spectrophotometer at 625 nm equivalent to 108 colony-forming unit (CFU)/ml. The inoculum was adjusted to 106 CFU/ml by diluting the inoculum with MHB at 1:100 ratio.
2.5. Determination of MIC
The MIC of C. odontophyllum acetone stem bark extract against MRSA Mu50 strain was determined using two-fold serial microdilution method. Bacterial suspension and MHB were used as positive control, whereas the extract in MHB was used as negative control to monitor the medium sterility. The lowest concentration of the extracts with no turbidity showed after being incubated at 37°C for 24 h was taken as the MIC value. To further confirm the growth of the bacteria, triphenyl tetrazolium chloride (TTC) was added into each well and was then incubated for 20 min. Reduction of TTC to red formazan was directly proportional to the presence of bacteria. There was no growth of bacteria in the well that retains the yellow color of TTC, indicating the presence of antimicrobial activity which inhibited the bacterial growth [11]. The whole process was repeated using vancomycin. The experiment was run in triplicate.
2.6. Determination of MBC
Wells that showed no color changes were subcultured on MHA plate and incubated for 24 h at 37°C. MBC value was recorded at the lowest concentration that showed no obvious growth on the MHA plate. The whole process was repeated for vancomycin. The experiment was run in triplicate.
2.7. TKA Analysis
The time-kill curve of the extract was investigated using microbroth dilution assay at a concentration of ×1 MIC, ×2 MIC, ×4 MIC, and ×8 MIC. The growth control tube was only filled with MHB. All tubes were incubated at 37°C, and the viable count was performed at 0, 2, 4, 6, 8, and 24 h. At each hour, 10 μl of the mixture was removed from the tubes and followed by 10-fold serial microdilution by adding 90 μl of normal saline in each well. Then, 10 μl of the dilution was dropped onto the MHA plate with a total of five drops for each quadrant of the plate before being incubated at 37°C for 24 h. Time-mortality curves were plotted by obtaining colony count of bacteria ranged between 3 and 30 CFU/ml for each drop. The graph of log10 CFU/ml on the Y-axis and time (h) on the X-axis is plotted. Bactericidal activity was defined when the extracts showed a reduction of ≥3 log10 CFU/ml, whereas bacteriostatic activity was defined when the killing activity was <99.9% [12]. The whole process was repeated for vancomycin at ×1 MIC. The experiment was run in triplicate.
2.8. PAE Time Analysis
PAE time of the extract against MRSA Mu50 strain was determined using viable plate count method [13]. The treatment group was prepared at concentration ×10 MIC and bacterial suspension at a final inoculum of 106 CFU/ml. In this study, ×10 MIC was chosen to make sure the antibacterial agent reached the maximum effect of bactericidal activity within 1 h of exposure to bacteria [14]. The control group comprised of MHB in bacterial suspension. Both treatment and control groups were incubated in a 37°C water bath for 1 h. After 1:1000 dilution was achieved using MHB, the tubes were incubated at 37°C, and the viable count was performed at 0, 2, 4, 6, 8, and 24 h. At each hour, 10 μl of the sample was removed from the tubes, followed by 10-fold serial microdilution by adding 90 μl of normal saline in each well. A total of 10 μl of the dilution was dropped onto the MHA plate with 5 drops for each quadrant of the plate. The plates were incubated at 37°C for 24 h. PAE value was obtained from the plotted graph of log10 CFU/ml on the Y-axis against time (h) on the X-axis. The experiment was run in triplicate, and the whole process was repeated for vancomycin at ×10 MIC [15].
PAE was calculated using the following formula [14]:
PAE = T-C
T is the time required for the treated group to increase 1 log10 CFU/ml following dilution at 1:1000 and C is the time required for the control group to increase 1 log10 CFU/ml following dilution at 1:1000 [13].
2.9. Statistical Analysis
Each experiment was carried out in triplicate for MIC, MBC, TKA, and PAE. All the data were expressed as mean ± SD of three replicates.
3. RESULTS
3.1. Determination of MIC and MBC
The results of microbroth dilution and streak plate method of C. odontophyllum acetone bark extract and vancomycin against MRSA Mu50 strain are shown in Table 1. The MIC values of C. odontophyllum acetone stem bark extract and vancomycin against MRSA Mu50 strain are 1.563 mg/ml and 0.00195 mg/ml, respectively. On the other hand, the MBC value of C. odontophyllum acetone stem bark extract is twice its MIC value (3.125 mg/ml) indicating bacteriostatic effect. Vancomycin, which is bactericidal effect against MRSA Mu50 strain, has equal MIC and MBC value, which is 0.00195 mg/ml.
3.2. TKA Analysis
The rate killing analyses of C. odontophyllum acetone stem bark extract at concentrations of ×1 MIC, ×2 MIC, ×4 MIC, ×8 MIC, and at ×1 MIC of vancomycin are presented in Figure 1. At ×1 MIC and ×2 MIC of C. odontophyllum acetone stem bark extract, the bacteria did not exhibit 3log10 CFU/ml reduction, and after 6 h incubation period, bacterial colony regrowth was observed. The extract, however, showed a rate-killing pattern at ×4 MIC comparable with that of vancomycin at ×1 MIC throughout the 24 h incubation time without bacterial regrowth. At the highest concentration of the extract tested (×8 MIC), the bactericidal effect of the extract was apparent at 16 h against MRSA Mu50, indicating a concentration-dependent manner of the anti-MRSA agent.
3.3. PAE Time
As seen in Figure 2, the PAE time of C. odontophyllum acetone stem bark extract and vancomycin against MRSA Mu50 strain was 3.6 ± 0.51 h and 2.4 ± 0.68 h, respectively. The longer PAE time of C. odontophyllum acetone stem bark extract against MRSA Mu50 strain compared to vancomycin indicated that the growth inhibition of the bacterial strain exposed to the extract persisted over a longer duration of time than the standard antibiotic.
4. DISCUSSION
In this study, C. odontophyllum acetone stem bark extract was used against MRSA Mu50 strain with MIC value of 1.563 mg/ml. Meanwhile, the MBC value of this acetone stem bark extract is 3.125 mg/ml. The MBC value is twice the MIC value. MIC and MBC values of vancomycin against MRSA Mu50 are both 0.00195 mg/ml. This result showed that C. odontophyllum acetone stem bark extract possessed bacteriostatic effect while vancomycin possessed bactericidal effect against MRSA Mu50 strain.
Based on the previous study, C. odontophyllum acetone stem bark extracts showed a bactericidal effect against S. aureus ATCC 25923 [9]. Unfortunately, in this study, C. odontophyllum acetone stem bark extract showed bacteriostatic effect against MRSA Mu50. This may be because MRSA Mu50 strain is more virulent compared to S. aureus ATCC 25923. S. aureus ATCC 25923 is a clinical isolate that is sensitive to many types of antibiotics including methicillin [16]. S. aureus ATCC 25923 is known as methicillin-susceptible S. aureus (MSSA). Meanwhile, MRSA Mu50 strain used in this study is S. aureus isolate that is resistant to methicillin.
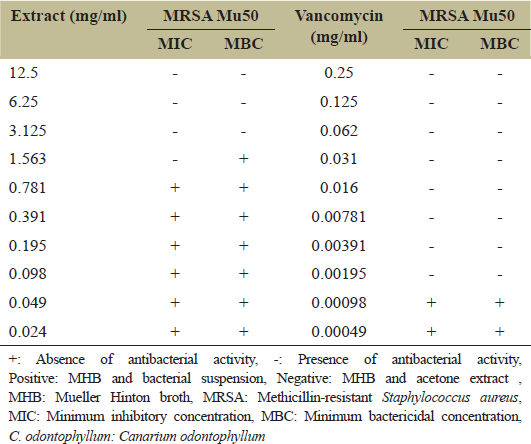 | Table 1: Determination of MIC and MBC value of C. odontophyllum acetone stem bark extract and vancomycin against MRSA Mu50 strain [Click here to view] |
In 1997, MRSA Mu50 strain has been identifying as vancomycin-intermediate S. aureus. It was first isolated from the surgical wound of Japanese infant. The patient infection does not respond to long-term vancomycin therapy [3]. In addition, a study done in 2003 showed that mortality rate of patients with MRSA bacteremia was higher compared to patient with MSSA bacteremia [17]. This supported the reason why C. odontophyllum acetone stem bark extract showed bacteriostatic effect against MRSA Mu50 strain instead of bactericidal effect against S. aureus ATCC 25923 [9]. However, the MIC value of C. odontophyllum acetone stem bark extract in the present study is higher compared to previously [9]. This might be due to the different bacteria strains that are being used. This finding is corresponding with the study of [18], where the MIC for MRSA strain is 32 mg/L, higher than MIC for MSSA strain which is 4 mg/L.
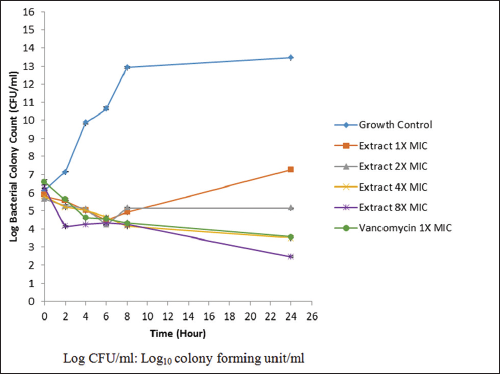 | Figure 1: Time-kill assay of Canarium odontophyllum acetone stem bark extract and vancomycin against methicillin-resistant Staphylococcus aureus (MRSA) Mu50 strain. Growth control curve that represented MRSA Mu50 strain showed increased in bacterial colony growth proportionally with the incubation time [Click here to view] |
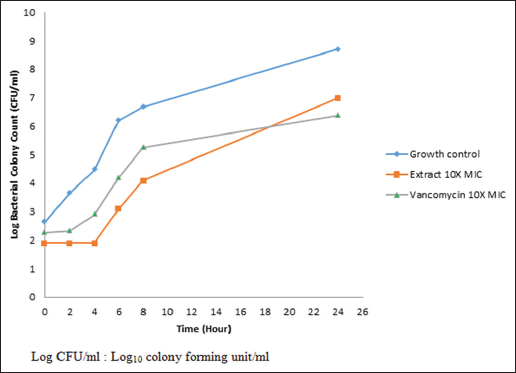 | Figure 2: Post-antibiotic effect of Canarium odontophyllum acetone stem bark extract and vancomycin against methicillin-resistant Staphylococcus aureus Mu50 strain [Click here to view] |
From the results, C. odontophyllum acetone stem bark extract showed lower antibacterial effect than vancomycin, where the MIC value of acetone extract was greater than MIC value of vancomycin. This study is corresponded with a study of [19], in which C. odontophyllum leaf acetone and methanol extract showed lower antibacterial activity compared to vancomycin. This may be because the stem bark extract has a variety of compounds while the standard antibiotics are pure compounds [20]. Plant secondary metabolites are compounds that play no role in the maintenance of the plant life process but very important for the plant to withstand stress, protect from harmful microorganism, and interact with the environment for their adaptation [21]. Antimicrobial activity of C. odontophyllum acetone leaf extract resulted from the presence of secondary metabolites such as terpenoid, saponin, tannin, flavonoid and phenolic compounds [10]. Previous study also reported the existence of phenolic compound and tannins in the stem barks of C. album [22].
Based on TKA graph, acetone extract of C. odontophyllum stem bark at ×1 MIC showed bacteriostatic activity by the reduction of bacterial colony <2 log10 CFU/ml during the first 6 h of incubation. However, bacterial colony increases from 6 to 24 h of incubation. Acetone extract of C. odontophyllum stem bark at ×2 MIC also showed a bacteriostatic effect during the first 6 h of incubation. However, the number of bacterial colonies increased until 8 h and reached a plateau until 24 h incubation time. Bacteriostatic effect also showed at ×4 MIC concentration when the number of bacterial colony decreases <3 log10 CFU/ml. Based on Figure 1, there was no increase in the number of bacterial colony as happened at ×1 MIC concentration and ×2 MIC concentration. Reduction of bacterial colony growth by 3 log10 CFU/ml was showed at 16 h for concentration ×8 MIC. This showed the bactericidal effect of the C. odontophyllum acetone stem bark extract against MRSA Mu50 strain. This result indicates that the plant extract showed bactericidal effect only at highest concentration. This could be due to MRSA which is a powerful isolate that does not respond to many commonly used antibiotics [23]. The rate of killing bacteria depends on extract concentration and duration of exposure [24]. This result showed that C. odontophyllum acetone stem bark extract gives concentration-dependent antibacterial activity toward MRSA Mu50 strain. This corresponds with the previous study which showed that honeybee venom activity against MRSA was concentration dependent, in which an increase of the agent concentration leads to progressive reduction of the bacterial colonies [25].
A TKA analysis for vancomycin at ×1 MIC showed that this antibiotic has bactericidal effect against MRSA Mu50 strain, where the bacterial colony was reduced by 3 log10 CFU/ml during 24 h of incubation. Vancomycin exhibited bactericidal effect with time-dependent killing activity [26]. In this study, vancomycin showed time-dependent killing activity due to a number of bacterial colony that reduces proportionally to the time.
The antimicrobial activity of C. odontophyllum acetone stem bark extract was studied further by looking at PAE time. In this study, PAE time of C. odontophyllum acetone stem bark extract was 3.6 ± 0.51 h, while vancomycin was 2.4 ± 0.68 h. Based on PAE time, C. odontophyllum acetone stem bark extract showed prolonged PAE compared to vancomycin, which suggested that plant extract residing in MRSA Mu50 strain was longer than vancomycin. This result is supported by Basri and Khairon [27], who showed that PAE for gall acetone extract Quercus infectoria was longer than vancomycin. Bioactive compound or secondary metabolites in the plant have antimicrobial activity [21]. Tannin in gall of Q. infectoria inhibits the growth of bacteria by interacting with proteins, enzymes as well as bacterial membrane [13]. Tannin is one of the bioactive compounds in C. odontophyllum acetone leaf extract that gives antimicrobial effect [19]. Thus, we can conclude that antimicrobial effect on MRSA Mu50 strain maybe due to tannin content in C. odontophyllum acetone stem bark extract.
Our result showed that PAE time of vancomycin was shorter than PAE time of C. odontophyllum acetone stem bark extract. Vancomycin showed that bactericidal effect of β-lactam antibiotics depends on time. This result is supported by Basri and Khairon [27] that showed shorter PAE time of vancomycin. This is why the frequency of taking this type of antibiotic or dose interval is important. In contrast, the PAE time is usually longer for an antibiotic that showed dose-dependent manner than for time-dependent antibiotic [28].
The struggle between humankind and microbes was begun since his existence on this earth. Fossil records in Mesopotamia (Iraq) reported that the human lived 60,000 years ago has used a medicinal plant named Hollyhock (Alcea rosea L.) in their life. This suggested that plant may be the first weapon used by ancient human to counter the human diseases. Plants are known to have various biochemical and pharmaceutical properties. Many of these so-called “medicinal plants” are expected to be an alternative source of new antimicrobials in the future [29].
5. CONCLUSION
C. odontophyllum acetone stem bark extract has the potential to be an antimicrobial agent against the MRSA Mu50 strain. TKA results showed that C. odontophyllum acetone stem bark extract has a dose-dependent effect: At ×8 MIC, the extract showed bactericidal effect on MRSA Mu50 strain. PAE results showed that C. odontophyllum acetone stem bark extract has longer PAE time compared to vancomycin. However, from MIC and MBC tests, C. odontophyllum acetone stem bark extract showed bacteriostatic effect instead of bactericidal effect. The incubation time should be extended from 24 to 36 h or 48 h to determine if the plant extract showed bactericidal effect on MRSA Mu50 strain. From this study, C. odontophyllum acetone stem bark extract has the potential to be an antimicrobial agent against MRSA Mu50 strain in addition with a few more assays or tests to further investigate the antimicrobial activity of this plant extract.
6. ACKNOWLEDGMENT
This project was supported by GUP-2016-036 under Universiti Kebangsaan Malaysia Research University Grant.
7. REFERENCES
1. Green BN, Johnson CD, Egan JT, Rosenthal M, Griffith EA, Evans MW, et al. Methicillin-resistant Staphylococcus aureus: An overview for manual therapists(). J Chiropr Med 2012;11:64-76. CrossRef
2. Lim KT, Hanifah YA, Yusof MY, Ito T, Thong KL. Comparison of methicillin-resistant Staphylococcus aureus strains isolated in 2003 and 2008 with an emergence of multidrug resistant ST22: SCCmec IV clone in a tertiary hospital, Malaysia. J Microbiol Immunol Infect 2013;46:224-33. CrossRef
3. Hiramatsu K, Katayama Y, Matsuo M, Sasaki T, Morimoto Y, Sekiguchi A, et al. Multi-drug-resistant Staphylococcus aureus and future chemotherapy. J Infect Chemother 2014;20:593-601. CrossRef
4. Ohta T, Hirakawa H, Morikawa K, Maruyama A, Inose Y, Yamashita A, et al. Nucleotide substitutions in Staphylococcus aureus strains, mu50, mu3, and N315. DNA Res 2004;11:51-6. CrossRef
5. Avison MB, Bennett PM, Howe RA, Walsh TR. Preliminary analysis of the genetic basis for vancomycin resistance in Staphylococcus aureus strain mu50. J Antimicrob Chemother 2002;49:255-60. CrossRef
6. Hayashi MA, Bizerra FC, Da Silva PI Jr. Antimicrobial compounds from natural sources. Front Microbiol 2013;4:195. CrossRef
7. Chua HP, Nicholas D, Suzalyna M. Phenolic and flavonoid contents and antioxidant activities of selected dabai (Canarium odontophyllum) genotypes. J Trop Agric Food Sci 2014;42:105-14.
8. Basri DF, Sandra V. Synergistic interaction of methanol extract from Canarium odontophyllum Miq. leaf in combination with oxacillin against methicillin-resistant Staphylococcus aureus (MRSA) ATCC 33591. Int J Food Microbiol 2016;2016:1-6.
9. Basri DF, Mustari NF, Alamin ZA, Zin NM. Stem bark of Canarium odontophyllum Miq. (Dabai) as potential source of antimicrobial agent. Malays J Med Sci 2017;15:1-6.
10. Basri DF, Saidi N, Mahari H, Saari S, Santhanam J. Preliminary screening for antimicrobial activity of the pulp of Canarium odontophyllum Miq. (Dabai) fruit. Glob J Pharmacol 2014;8:213-20.
11. Moussa SH, Tayel AA, Al-Hassan AA, Farouk A. Tetrazolium/formazan test as an efficient method to determine fungal chitosan antimicrobial activity. J Mycol 2013;2013:1-7. CrossRef
12. Drago L, De Vecchi E, Lombardi A, Nicola L, Valli M, Gismondo MR, et al. Bactericidal activity of levofloxacin, gatifloxacin, penicillin, meropenem and rokitamycin against Bacillus anthracis clinical isolates. J Antimicrob Chemother 2002;50:1059-63. CrossRef
13. Amman V, Basri DF, Huyop F. Determination of the post-antibiotic effect (PAE) of combinations of extracts from galls of Quercus infectoria with vancomycin against methicillin-resistant Staphylococcus aureus (MRSA). Afr J Biotechnol 2011;10:18274-8.
14. Craig WA, Gudmundsson S. In: Lorian V. The Postantibiotic Effect. Vol. 4. Baltimore, Md: The Williams and Wilkins Co.; 1996. p. 296-329.
15. Athamna A, Athamna M, Medlej B, Bast DJ, Rubinstein E. In vitro post-antibiotic effect of fluoroquinolones, macrolides, β -lactams, tetracyclines, vancomycin, clindamycin, linezolid, chloramphenicol, quinupristin/dalfopristin and rifampicin on Bacillus anthracis. J Antimicrob Chemother 2004;53:609-15.
16. Treangen TJ, Maybank RA, Enke S, Friss MB, Diviak LF, Karaolis DK, et al. Complete genome sequence of the quality control strain Staphylococcus aureus subsp. Aureus ATCC 25923. Genome Announc 2014;2:e01110-14.
17. Cosgrove SE, Sakoulas G, Perencevich EN, Schwaber MJ, Karchmer AW, Carmeli Y, et al. Comparison of mortality associated with methicillin-resistant and methicillin-susceptible Staphylococcus aureus bacteremia: A meta-analysis. Clin Infect Dis 2003;36:53-9. CrossRef
18. Von Ah U, Wirz D, Daniels AU. Rapid differentiation of methicillin-susceptible Staphylococcus aureus from methicillin-resistant Staphylococcus aureus and MIC determinations by isothermal microcalorimetry. J Clin Microbiol 2008;46:2083-7. CrossRef
19. Basri DF, Hafidzah N, Nor M. Phytoconstituent screening and antibacterial activity of the leaf extracts from Canarium odontophyllum Miq. Am J Plant Sci 2014;5:2878-88. CrossRef
20. Gatsing D, Christine FN, Bridget FN, Kuiate JR, Tchouanguep FM. Antibacterial activity, bioavailability and acute toxicity evaluation of the leaf extract of Alchornea cordifolia (Euphobiaceae). Int J pharmacol 2010;63:173-82.
21. Ramakrishna A, Ravishankar GA. Influence of abiotic stress signals on secondary metabolites in plants. Plant Signal Behav 2011;6:1720-31. CrossRef
22. Zhang LL, Lin YM. Tannins from canarium album with potent antioxidant activity. J Zhejiang Univ Sci B 2008;9:407-15. CrossRef
23. Kaur DC, Chate SS. Study of antibiotic resistance pattern in methicillin resistant Staphylococcus aureus with special reference to newer antibiotic. J Glob Infect Dis 2015;7:78-84. CrossRef
24. Chatterjee SK, Bhattacharjee I, Chandra G. In vitro synergistic effect of doxycycline ofloxacin in combination with ethanolic leaf extract of Vangueria spinosa against four pathogenic bacteria. Indian J Med Res 2009;130:475-8.
25. Han SM, Kim JM, Hong IP, Woo SO, Kim SG, Jang HR, et al. Antibacterial activity and antibiotic-enhancing effects of honeybee venom against methicillin-resistant Staphylococcus aureus. Molecules 2016;21:79. CrossRef
26. Jacqueline C, Caillon J, Le Mabecque V, Miegeville AF, Donnio PY, Bugnon D, et al. In vitro activity of linezolid alone and in combination with gentamicin, vancomycin or rifampicin against methicillin-resistant Staphylococcus aureus by time-kill curve methods. J Antimicrob Chemother 2003;51:857-64. CrossRef
27. Basri DF, Khairon R. Pharmacodynamic interaction of Quercus infectoria galls extract in combination with vancomycin against MRSA using microdilution checkerboard and time-kill assay. Evid Based Complement Alternat Med 2012;2012:493156.
28. Levison ME. Pharmacodynamics of antimicrobial drugs. Infect Dis Clin North Am 2004;18:451-65, vii.
29. Abdallah EM. Plants: An alternative source for antimicrobials. J App Pharm Sci 2011;1:16-20.