1. INTRODUCTION
The consumer concerns about the use of synthetic antimicrobials to improve the quality of life led to a search for biodegradable compounds of natural origin [1]. The importance of biofilms control and the potential use of biosurfactants, as an antimicrobial agent, have enhanced the interest in these compounds, which are molecules with surfactant characteristics, produced by microorganisms and, although the similarity with petroleum-based surfactants, they are considered superior and more advantageous, because of their ecological and sustainable nature [2]. Structurally, they are amphiphilic molecules, whereas the hydrophobic moiety is a long chain of fatty acid, hydroxy acid, or α-alkyl β-hydroxy-acid, and the hydrophilic moiety is generally a carbohydrate, amino acid, cyclic peptide, phosphate, carboxylic acid, or alcohol [3].
Sophorolipids (SLs) are secondary metabolites classified as extracellular glycolipids, primary produced by yeast Candida bombicola, from carbohydrates and lipids, being excreted as a mixture of related chemical structures [4]. They are composed of a disaccharide sophorose (O-β-D-glucopyranosyl-2-1-β-D-glucopyranose) linked by a glycosidic bound between the carbon 1’ and the terminal (ω) or sub-terminal (ω-1) carbon of a fatty acid chain of 16 or 18 carbons [5]. They have no cytotoxicity and are accepted and approved by the Food and Drug Administration. Currently, they are the most applied biosurfactants in the industry, and the products are available in commercial level.
These metabolites are produced in two principal structural forms, acidic and lactonic [6], which results in changes in the physical-chemical and biological properties, responsible for the different applicabilities of these compounds [7]. In relation to the producing microorganisms, there are several species of yeasts that synthesize different profiles of SLs, highlighting C. bombicola, because of the high yields, which mainly produces SLs in the lactonic diacetylated form (6’, 6”) with monounsaturated fatty acids (C16 and C18), and in a minor extent, acidic non-acetylated or monoacetylated forms (6’’) [8]. Therefore, because of these structural characteristics, SLs have been reported with several applications, which are directly related to the composition of their acidic and lactonic forms. These applications are highlighted in agriculture, food, cosmetic, bioremediation, and biomedicine with antimicrobial activity [9,10].
2. ANTIMICROBIAL ACTIVITY OF SLs
The antimicrobial activity of SLs is related to the synergistic effect of their sugar and lipid portions (surfactant effect) [9,11]. This mechanism is characterized by changes or rupture in the cellular membrane, inducing the outpouring of their cytoplasmic contents and the consequent release of intracellular enzymes, for instance, malate dehydrogenase indicating the interaction of SLs with the cellular membrane [12,13]. Although the mechanism of action of biosurfactants is not well known, an activity of altering charge-charge properties is hypothesized, which may decrease the chances for bacteria to acquire antibiotic resistance [14].
The interactions between carbohydrates and bacterial membranes have been studied for years [15]; however, only recently, studies have attempted to show the impact of mono and disaccharides on the structure of membranes [16] such as the sophorose disaccharide present in the SL molecule, which are effective as a bactericidal agent, regardless if its lipid content is acidic or lactonic, being capable of inducing death of planktonic cells and biofilms of both Gram-positive and Gram-negative bacteria although the negative group presents a more complex cellular envelope both can be damaged by sophorose [17].
The peptidoglycan layer of Gram-positive bacteria is covered by polysaccharides, neutral acids, and proteins. The surface of the Gram-negative bacteria is constituted by lipopolysaccharides with neutral pH, but when the carboxylic and phosphate groups are ionized, they confer anionic charges. These negative charges make the bacterial membrane more hydrophilic [18] compared to Gram-positive bacteria. SLs, due to their amphiphilic characteristics, decrease the hydrophobicity of both bacterial groups, but because of the majority composition in fatty acids, they exhibit a greater tendency to hydrophobicity, leading to a more significant performance in hydrophobic microorganisms (Gram-positive) [19,20].
The antimicrobial activity of SLs depends on the concentration, treatment time, composition of fatty acids, and the predominance of acidic and lactonic forms [21,22], as well as the sugar group of the molecule [23]. Lactonic forms have better surface tension properties and antimicrobial activity [7]. Furthermore, it is known that the acetylated forms have better biological and physical-chemical properties [24]. Some of the studied bacteria tested along with the use of the glycolipid are mentioned in Table 1; it is possible to observe the differences in the minimal inhibitory concentration (MIC) depending on the target bacteria and the SL composition, which makes this MIC very variable in the literature because of these many factors.
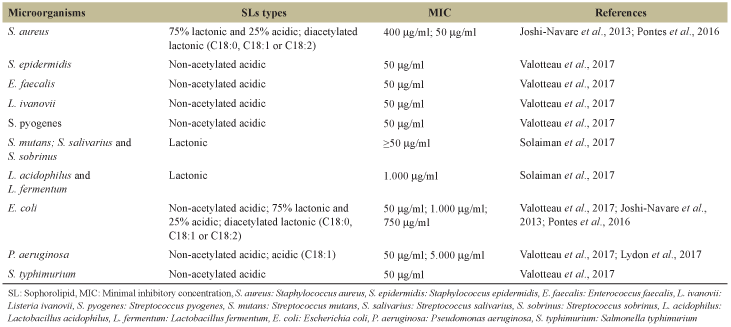 | Table 1: Minimum inhibitory concentrations of different types of SL against Gram-positive and Gram-negative bacteria (μg/ml) [Click here to view] |
SLs from C. bombicola produced in palmitic, stearic, and oleic acids were applied to Gram-positive bacteria (Enterococcus faecium, Aerococcus viridans, Staphylococcus xylosus, Staphylococcus cohnii, and Staphylococcus equorum) Gram-positive endospore forming (Bacillus licheniformis, Bacillus pumilus, and Bacillus mycoides) and Gram-negative bacteria (Pseudomonas luteola, Enterobacter cloacae, Enterobacter sakazakii, and Vibrio fluvialis), obtaining MIC from 4.88 μg/mL to 19.5 μg/mL, demonstrating effect in all bacteria studied [25].
Similar studies by different authors have shown that SLs from C. bombicola were also able to reduce Escherichia coli O157:H7 population. Applications with 0.5% and 1.0% of SL-oleic and SL-palmitic reduced planktonic cell cultures after 1–2 h of incubation. AWhile the use of only 0.1% of SL-stearic was sufficient to reduce the same bacteria after 2 h [26].
SLs produced by C. bombicola on coconut and corn oils were tested against Staphylococcus aureus and E. coli. The synthesized from corn oil was more efficient for E. coli, and coconut oil for S. aureus [21]; this demonstrates the varied action mechanism of different SLs as an antimicrobial agent in the various pathogenic strains. It was also tested by other authors in Bacillus subtilis and Pseudomonas aeruginosa, obtaining a MIC of 5.0 and 10.0 mg/mL, respectively [20].
Enterococcus faecalis and P. aeruginosa, bacteria responsible for nosocomial infections, were inhibited by purified acidic SLs from C. bombicola, predominantly non-acetylated (C18), at ≥5 mg/mL. At 20 mg/mL, an inhibitory effect on the growth of E. faecalis was observed, with no formation of colonies [22]. On the other hand, the mixture of SLs without purification, containing 75% of lactonic and 25% of acidic was effective against E. coli at 1 mg/mL and S. aureus at 15–150 μg/mL [27].
SLs produced from glucose, and lauryl alcohol was tested in Gram-negative bacteria (E. coli ATCC 8739 and P. aeruginosa ATCC 9027), Gram-positive (S. aureus ATCC 6358 and B. subtilis ATCC 6633), and yeast Candida albicans ATCC 2091 [28]. The results showed complete inhibition when compared to SL-oleic and SL-linolenic. The inhibition was 30 μg/mL for E. coli and 1 μg/mL for P. aeruginosa at 2 and 4 h, respectively, for S. aureus was 6 μg/mL, B. subtilis was 1 μg/mL, and C. albicans was 50 μg/mL, after 4 h of treatment.
The acidic and lactonic forms of SLs from Rhodotorula babjevae YS3 presented antifungal action against Colletotrichum gloeosporioides, Fusarium verticillioides, Fusarium oxysporum, Corynespora cassiicola, and Trichophyton rubrum verified by MIC of 62 μg/mL, 125 μg/mL, 125 μg/mL, ≥2000 μg/mL, and ≥1000 μg/mL, respectively [29].
Synergistic actions of diacetylated lactonic SLs (SL-oleic) with cefaclor and tetracycline have been described, demonstrating that the activity of conjugated antibiotics was enhanced for E. coli ATCC 8739 and S. aureus ATCC 29737 [27]. SL conjugated with caprylic acid (0.8%) increased the inhibition of P. aeruginosa PAO1, B. subtilis NCTC 10400, S. aureus ATCC 9144, and E. coli NCTC 10418 [30]. In another study, the same authors verified the combination of SLs and rhamnolipids (0.04%/0.01%) against biofilms of P. aeruginosa ATCC 15442, S. aureus ATCC 9144, and a mixed culture of both, obtaining positive results about the synergism of this molecule with different compounds [31].
The activity of SLs from C. bombicola was compared with thiamine dilauryl sulfate (TDS) in the presence of alcohol against Salmonella spp. and Listeria spp. The lactonic SLs presented superior antimicrobial activity in Listeria spp. than in Salmonella spp. The populations of Listeria spp. were reduced from 7.2 log CFU/mL to an undetectable level after treatment of 1 min with 0.1% (w/v) of single-layer perceptron and TDS in the presence of ethanol (20%). TDS was more effective than SLs against Salmonella spp. and Listeria spp., but both are capable of causing cell lysis; demonstrating that SLs and TDS in the presence of ethanol can be used to inactivate pathogens, especially Gram-positive bacteria [12].
3. CONCLUSION
This review presented the potentials of this glycolipid and their applications as an antibacterial and antifungals agent. SLs can be used to repair infectious diseases, as therapeutic agents, sanitizers, and germicides in several sectors, highlighting the main bacteria of foodborne illness and contamination, both Gram-positive and Gram-negative, can be inhibited by antimicrobial activity of SLs produced by C. bombicola. Considering the significance of the development of new sustainable strategies, combined with the importance of controlling the formation of biofilms and being a non-toxic product, SLs present a promising perspective for an excellent antimicrobial agent.
4. ACKNOWLEDGMENTS
The authors would like to thank The Coordination of Training of Higher Education Graduate Foundation (CAPES) and The National Council for Scientific and Technological Development (CNPq) for the financial support.
5. REFERENCES
1. Zhang H, Wu J, Guo X. Effects of antimicrobial and antioxidant activities of spice extracts on raw chicken meat quality. Food Sci Hum Wellness Beijing Acad Food Sci 2016;5:39-48. Crossref
2. Chen M, Dong C, Penfold F, Thomas RK, Smyth TJ, Perfumo A, et al. Adsorption of sophorolipid biosurfactants on their own and mixed with sodium dodecyl benzene sulfonate, at the air/water interface. Langmuir 2011;27:8854-66. Crossref
3. Desai JD, Banat IM. Microbial production of surfactants and their commercial potential. Microbiol Mol Biol Rev MMBR 1997;61:47-64.
4. Cooper DG, Paddock DA. Production of a biosurfactant from torulopsis-bombicola. Appl Environ Microbiol 1984;47:173-6.
5. Asmer HJ, Lang S, Wagner F, Wray V. Microbial production, structure elucidation and bioconversion of sophorose lipids. J Am Oil Chem Soc 1988;65:1460-6. Crossref
6. Morya VK, Park JH, Kim TJ, Jeon S, Kim EK. Production and characterization of low molecular weight sophorolipid under fed-batch culture. Bioresourc Technol 2013;143:282-8. Crossref
7. Paulino BN, Pessôa MG, Mano MC, Molina G, Neri-Numa IA, Pastore GM. Current status in biotechnological production and applications of glycolipid biosurfactants. Appl Microbiol Biotechnol 2016;100:10265-93. Crossref
8. Minucelli T, Ribeiro-Viana RM, Borsato D, Andrade G, Cely MV, Oliveira MR, et al. Sophorolipids Production by Candida bombicola ATCC 22214 and its potential application in soil bioremediation. Waste Biomass Valorization 2017;8:743-53. Crossref
9. Glover RE, Smith RR, Jones MV, Jackson SK, Rowlands CC. An EPR investigation of surfactant action on bacterial membranes. FEMS Microbiol Lett 1999;177:57-62.
10. Oliveira MR, Magri A, Baldo C, Camilios-Neto D, Minucelli T, Celligoi MA. Review: Sophorolipids a promising biosurfactant and its applications. Int J Adv Biotechnol Res 2015;6:161-74.
11. Lang S, Katsiwela E, Wagner F. Antimicrobial effects of biosurfactants. Fett Wiss Technol Fat Sci Technol1989;91:363-6.
12. Zhang X, Ashby R, Solaiman DK, Uknalis J, Fan X. Inactivation of Salmonella spp. and Listeria spp. by palmitic, stearic, and oleic acid sophorolipids and thiamine dilauryl sulfate. Front Microbiol 2016;7:1-11.
13. Kulakovskaya E, Baskunov B, Zvonarev A. The antibiotic and membrane-damaging activities of cellobiose lipids and sophorose lipids. J Oleo Sci 2014;63:701-7.
14. Díaz De Rienzo MA, Banat IM, Dolman B, Winterburn J, Martin PJ. Sophorolipid biosurfactants: Possible uses as antibacterial and antibiofilm agent. New Biotechnol 2015;32:720-6.
15. Quiocho FA. Carbohydrate-binding proteins: Tertiary structures and protein-sugar interactions. Ann Rev Biochem 1986;55:287-315.
16. Moiset G, López CA, Bartelds R, Syga L, Rijpkema E, Cukkemane A, et al. Disaccharides impact the lateral organization of lipid membranes. J Am Chem Soc 2014;136:16167-75.
17. Valotteau C, Banat IM, Mitchell CA, Lydon H, Marchant R, Babonneau R, et al. Antibacterial properties of sophorolipid-modified gold surfaces against Gram positive and Gram negative pathogens. Coll Surf B Biointerf 2017;157:325-34.
18. Abu-Lail NI, Camesano TA. Role of lipopolysaccharides in the adhesion, retention, and transport of Escherichia coli JM109. Environ Sci Technol 2003;37:2173-83.
19. Pontes C, Santos MA, Ribeiro MH, Gonçalves L, Bettencourt AF, Ribeiro IA. Can Sophorolipids prevent biofilm formation on silicone catheter tubes? Int J Pharm 2016;513:697-708.
20. Hoa NL, Loan LQ, Eun-Ki K, Ha TT, Duy ND, Khanh HQ, et al. Production and characterization of sophorolipids produced by Candida bombicola using sugarcane molasses and coconut oil. Asia Pac J Sci Technol 2017;22:66-75.
21. Morya VK, Park JH, Kim TJ, Jeon S, Kim EK. Production and characterization of low molecular weight sophorolipid under fed-batch culture. Bioresour Technol 2013;143:282-8.
22. Lydon HL, Baccile N, Callaghana B, Marchanta R, Mitchella CA, Banat IM. Adjuvant antibiotic activity of acidic sophorolipids with potential for facilitating wound healing. Antimicrob Agents Chemother 2017;61:e02547-16.
23. Shah V, Badia D, Ratsep P. Sophorolipids having enhanced antibacterial activity. Antimicrob Agents Chemother 2007;51:397-400.
24. Lang S, Brakemeier A, Heckmann R, Spockner S, Rau U. Production of native and modified sophorose lipids. Chim Oggi 2000;18:76-9.
25. Solaiman DK, Ashby RD, Bier M, Caglayan P. Antibacterial activity of sophorolipids produced by Candida bombicola on gram-positive and gram-negative bacteria isolated from salted hides. J Am Leather Chem Assoc 2011;111:358-64.
26. Zhang X, Ashby RD, Solaiman DK, Liu Y, Fan X. Antimicrobial activity and inactivation mechanism of lactonic and free acid sophorolipids against Escherichia coli O157:H7. Biocatal Agric Biotechnol 2017;11:176-82.
27. Joshi-Navare K, Khanvilkar P, Prabhune A. Jatropha oil derived sophorolipids: Production and characterization as laundry detergent additive. Biochem Res Int 2013;2013:1-11.
28. Dengle-Pulate V, Chandorkar P, Bhagwat S, Prabhune AA. Antimicrobial and SEM studies of sophorolipids synthesized using lauryl alcohol. J Surf Detergents 2014;17:543-52.
29. Sen S, Borah SN, Bora A, Deka S. Production, characterization, and antifungal activity of a biosurfactant produced by Rhodotorula babjevae YS3, microbial cell factories. Biomed Cent 2017;16:95.
30. Díaz De Rienzo MA, Stevenson PS, Marchant R, Banat IM. Effect of biosurfactants on Pseudomonas aeruginosa and Staphylococcus aureus biofilms in a BioFlux channel. Appl Microbiol Biotechnol 2016;100:5773-9.
31. Díaz De Rienzo MA, Stevenson PS, Marchant R, Banat IM. Pseudomonas aeruginosa biofilm disruption using microbial surfactants. J Appl Microbiol 2016;120:868-76.