1. INTRODUCTION
Flemingia is a genus of flowering plants in the legume family, Fabaceae. Flemingia tuberosa (Figure 1), (Syn- Moghania tuberosa) commonly known as Birmova, is a plant species endemic to Western Ghats [13]. According to The United Nations Educational, Scientific and Cultural Organization (UNESCO), Western Ghats has been listed as a ‘World Heritage’. The document specifies that 54% of 650 tree species are endemic in the Western Ghats. Flemingia tuberosa is a runner with thick under shrub-like growth with delicate lilac pedicellate flowers and trifoliate leaves having hairy appearance [13]. Plants and their products are subject to the wide diversity in their phytochemical profile because of their varying meteorological conditions, maturity, post-harvest processing, storage, stability etc. Therefore it is extremely important to standardize these drugs based on their marker compounds using the suitable chromatographic technique in order to identify morphological and geographical variations [18].
A thorough literature survey revealed that a few species of Flemingia have therapeutic properties, among these, Flemingia strobilifera, Flemingia macrophylla, Flemingia chappar, Flemingia vestita belonging to the fabaceae family have been traditionally consumed to cure epilepsy, insomnia, ulcer, pain, swelling, intestinal infections and have been reported for the presence of flemichapparin (A, B, and C), β-sitosterol, quercitin, genistein, flemingins A-C, lupeol, etc. [1,8,17]. Scientific data on pharmacognostic parameters of Flemingia tuberosa remains an unexplored issue. Hence, setting up a pharmacognostic profile of the plant will aid in its standardization in terms of quality, purity and sample identification. In the current work, the parameters have been followed from ICMR to formulate a monograph for Flemingia tuberosa.
The available literature on phytochemical reports of the genus Flemingia reveals that the Flemingia species comprise mainly flavonoids [1]. A preliminary evaluation revealed the presence of a class of phytoconstituents which helped us in taking flavonoid for further evaluation since it is a group of plant secondary metabolites. Flavonoids have variable phenolic structures widely distributed in the plant kingdom. It is commonly reported being constituent of fruits, vegetables, grains, barks, roots, stems, flowers, tea and some beverages. Playing a role in decreasing risk of chronic diseases, flavonoids are associated with a diet rich in plant-derived foods. Some epidemiological studies have proved that ingestion of foods containing flavonoids can reduce the risk of developing cancer and cardiovascular diseases [4,11]. Tubers of Flemingia tuberosa has been reported to cure dysentery and vaginal discharge [7]. Kaempferol is a bio-marker reported for similar activities [19].
 | Figure 1: Photographic documentation of Flemingia tuberosa. [Click here to view] |
Therefore, the aim of this study was to standardize Flemingia tuberosa by carrying out the physicochemical, phytochemical and chromatographic evaluation. The quality control parameters were established for Flemingia tuberosa and kaempferol content in Flemingia tuberosa (different parts) was estimated and the content of kaempferol from aerial parts of Flemingia tuberosa and Flemingia vestita was compared using the chromatographic technique. Further, in order to evaluate the safety of an ethanolic extract of Flemingia tuberosa, an acute oral toxicity study was conducted on albino Wistar rats following OECD test guideline no. 420.
2. MATERIALS AND METHODS
2.1. Chemicals and Reagents
Kaempferol (98% purity, Figure 2) was procured from Sigma Aldrich chemical company (Steinheim, Germany) while all the chemicals used were of analytical grade and were procured from Merck Specialties Pvt. Ltd.
 | Figure 2: Structure of kaempferol. [Click here to view] |
2.2. Plant Sample
Flemingia tuberosa was collected from Rajapur, Maharashtra (average elevation of 72 meters) and Dr. M. M. Lekhak (Shivaji University, Kolhapur, Maharashtra) confirmed the taxonomic identification. The sample was thoroughly washed, cleaned and shade dried for a week. The material was then packed in absorbent paper, oven dried at 37°C for three days. The dried material was then powdered using a mixer-grinder and sieved through BSS mesh number 85.
2.3. Physicochemical Evaluation
Foreign organic matter, ash content (total ash, acid insoluble ash, and water-soluble ash) and extractive values were determined following standard Pharmacopoeial methods [10,12].
2.4. Phytochemical Evaluation
Phytochemical evaluation of some major groups of secondary metabolites (flavonoids, terpenoids, tannins, glycosides, and alkaloids) in Flemingia tuberosa (aerial part) was carried out by performing preliminary phytochemical tests as per the methods reported [6] and further the plant was subjected to phytochemical evaluation by successive soxhlet extraction with various organic solvents in order to analyze the percent extract of major class of compounds present in the plant extract as per the method reported [3]. In this study, UV-Visible spectroscopy was successfully used for quantitative determination of the phytochemical constituents viz., total flavonoids [9], total phenolics [14], total alkaloids [20], total saponins [23] and total terpenoids [2] which were found to be present in the plant samples during preliminary phytochemical study. Data on sensitivity and linearity for different markers used to determine the content of some major groups of phytochemical constituents (quercetin for total flavonoids, gallic acid for total phenolics, bismuth nitrate pentahydrate for total alkaloids, diosgenin for total saponins, and linalool for total terpenoids) in the plant sample is shown in Table 4A. Data on the content of total flavonoids, total phenolics, total alkaloids, total saponins and total terpenoids in the plant sample is represented in Table 4B.
 | Figure 3: HPTLC fingerprints of different parts of Flemingia tuberosa. [Click here to view] |
2.5. Chromatographic Characterization
The powdered plant material (1.0 g) was extracted with ethanol (10.0 mL), vortex mixed and sonicated for (1 min and 20 min respectively) followed by filtration through Whatman filter paper No. 1. The phytochemical fingerprint was developed for the filtrate using HPTLC. Also, separation and quantitation of kaempferol was established.
2.6. Preparation of Kaempferol Stock Solutions
Standard stock solution of kaempferol (1000 μg/mL) was prepared in methanol. The Serial dilution of the stock solution in methanol was carried out in order to prepare calibrant/ quality control samples.
2.7. Optimized Chromatographic Conditions
The HPTLC system used consisted of CAMAG TLC Scanner 4 supported by winCATS software version 1.4.7 equipped with CAMAG Linomat 5 sample spotter and CAMAG Reprostar 3 system for photo-documentation. A Denver analytical balance (Goettingen, Germany) was used to weigh the standard. Chromatographic separation of the phytochemical constituents was achieved on TLC plates (E. Merck) pre-coated with silica gel 60 F254 (0.2 mm thickness) on aluminium sheet support. To develop HPTLC fingerprints of ethanolic extract of different parts of Flemingia tuberosa, the samples (10.0 μL) were applied to the plate as a band of 7.0 mm wide and at a distance of 11.0 mm from the edges. Plate was developed up to a distance of 85.0 mm in CAMAG twin trough glass chamber pre-saturated with the mobile phase toluene: ethylacetate (9:1.2, v/v) for 15 min. After development, the plate was dried in a current of air at room temperature. For densitometric scanning, the source of radiation was a Deuterium lamp (254 nm), mercury lamp (366 nm). All measurements were performed at 22 ± 1°C. Photo-documentation of the plate was done at 254 nm and 366 nm. The plate was derivatised using 10% methanolic sulphuric acid and dried in oven preset at 110°C for 10 min. For densitometric scanning, the source of radiation was a mercury lamp (366 nm). All measurements were performed at 22 ± 1°C. Plate was photo-documented at 366 nm. The second time the similar plate was derivatised with 1% Anisaldehyde reagent and dried in oven preset at 110°C for 10 min. For densitometric scanning, the source of radiation was a white light (540 nm). All measurements were performed at 22 ± 1°C. Plate was photo-documented at 540 nm.
To separate kaempferol from Flemingia tuberosa (different parts) and Flemingia vestita (aerial part), samples (10.0 μL each) and kaempferol standard (100.0 μg/mL, 10.0 μL) were spotted on TLC plate as bands of 7.0 mm wide and at a distance of 11.0 mm from the edges under similar instrumental conditions. The plate was developed up to a distance of 85.0 mm in CAMAG twin trough glass chamber pre-saturated with mobile phase cyclohexane: ethyl acetate: formic acid (6:4.5:0.2, v/v/v) for 15 minutes. The plate was scanned and photo documented at 365 nm.
3. SAFETY EVALUATION
Safety study of an ethanolic extract of Flemingia tuberosa was conducted on albino Wistarrats Wistar rats (female, 200-225 g) approval no.: RM-160110-01). The rats were fasted for 4 hours prior dosing and approximately 2 hours post dosing, water was provided ad libitum. The animals were orally administered with extract of Flemingia tuberosa (Ethanol was evaporated to dryness at 78°C in a boiling water bath [Innovative Instruments, Mumbai, India] since the boiling point of ethanol is 78.37°C and the complete removal of ethanol was confirmed by ‘no smell’ of the alcohol. The dose was calculated with reference to the body weight of each animal, weighed individually and suspended in distilled water). The animals were examined individually during the first 30 min for all reflexes, periodically during the first 48 h with special attention given during the first 4 h (short-term toxicity) and daily thereafter for a total of 14 days (long-term toxicity) for alteration from general behavior and clinical symptoms like alteration of skin and fur texture, ptosis, excessive salivation, breathing problems, diarrhea etc. Daily body weight, food, and water intake records were also maintained. The results were compared with the control group (orally administered with distilled water).
4. STATISTICAL ANALYSIS
Microsoft Excel-2007 was used to determine mean, standard deviation (SD), relative standard deviation (RSD) and mean difference during the analysis.
5. RESULTS AND DISCUSSION
During the various metabolic processes phytochemicals, which are chemical compounds, are synthesized [16]. In order to ensure both identity and purity proximate analysis of the plant materials can be done [5]. The foreign matter involves matter other than source plant while the ash values represent the organic content and crude fibre gives an idea about excessive woody material for judging the purity [21]. Extractive values indicate the approximate measure of phytochemical constituents of the crude drug [5]. Proximate parameters such as foreign organic matter, ash values (total, acid insoluble and water soluble) and extractive values (ethanol soluble, water-soluble and ether soluble) of Flemingia tuberosa were determined and results are shown in (Table 1A). The water-soluble extractive value was found to be more as compared to ethanol soluble extractive value suggesting the presence of more polar components in the raw material (approx. 20%). In the preliminary phytochemical evaluation (Table 1B), flavonoids, phenolics, and terpenoids were found to be present in the methanolic extract of Flemingia tuberosa, whereas alkaloids and saponins were found to be absent. Soxhlet extraction revealed that the plant was rich in Quaternary alkaloids and N-oxides followed by, fats and waxes, terpenoids and phenolics and other alkaloids.
 | Table 1A: Proximate analysis of Flemingia tuberosa. [Click here to view] |
 | Table 1B: Phytochemicals in Flemingia tuberosa detected as per preliminary phytochemical tests. [Click here to view] |
.png) | Table 2: Number of peaks obtained from different parts of Flemingia tuberosa using different derivatising reagents. [Click here to view] |
Phytochemical evaluation of the plant is also used as a quality control tool in the medicinal plants research [3]. Flemingia tuberosa has not been reported for the presence of any phytoconstituents. Chromatographic fingerprints are considered to be one of the most important and acceptable approaches for quality evaluation of herbal preparations and to analyze a particular group of compounds or to focus on the analysis of plant sample [15,22,24]. HPTLC fingerprint was developed from ethanolic extracts of different parts of Flemingia tuberosa using toluene: ethyl acetate (9:1.2, v/v) as a mobile phase (Figure 3) and it peaks as shown in Table 2.
 | Figure 4A: Chromatographic plate of standard kaempferol with different parts of Flemingia tuberosa. [Click here to view] |
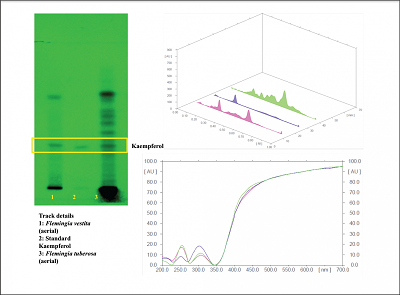 | Figure 4B: Chromatographic plate of standard kaempferol with aerial part of Flemingia tuberosa and Flemingia vestita. [Click here to view] |
 | Table 3A: Validation of kaempferol using HPTLC. [Click here to view] |
 | Table 3B: Content of kaempferol in different parts of Flemingia tuberosa. [Click here to view] |
 | Table 3C: Comparative account of kaempferol content in Flemingia tuberosa and Flemingia vestita. [Click here to view] |
Based on the fact that kaempferol is a phytoestrogen reported to have therapeutic activities, in this research work, an HPTLC method for characterization of Flemingia tuberosa in terms of kaempferol content has been developed. For separation of kaempferol from different parts of Flemingia tuberosa (Figure 4A) and the comparison of the content with Flemingia vestita (Figure 4B), cyclohexane: ethyl acetate: formic acid (6:4.5:0.2, v/v/v) was used as mobile phase which gave well-resolved band of kaempferol at Rf = 0.27. Presence of kaempferol in the sample was confirmed by overlay as well as spectral scan.
A validated method was used and was found to be rapid, specific, precise, sensitive and rugged using ICH guidelines (Table 3A). The content of kaempferol in different parts of Flemingia tuberosa is tabulated (Table 3B). All the parts of Flemingia tuberosa contain kaempferol as seen in Table 3B. Although the content of kaempferol was maximum in roots, aerial parts of the plant were used for further research work. As the current work aims at the use of renewable parts like aerial parts which would not hamper the proliferation of these endemic species. Therefore, the study was concentrated on the use of aerial parts despite kaempferol being present more in roots. Thus, underground parts have been avoided for the future pharmacological studies so as to prevent the exploitation of the plant being an endemic species. In further studies, the aerial part has been checked for its efficacy studies. Thus, the aim of the study to show that the biomarker content is in enough quantity in aerial part has been justified. Safety of Flemingia tuberosa was established using acute toxicity study in albino Wistar rats. Oral administration of the plant treatment showed no significant change in the body weight, food and water intake in comparison to the animals of the control group.
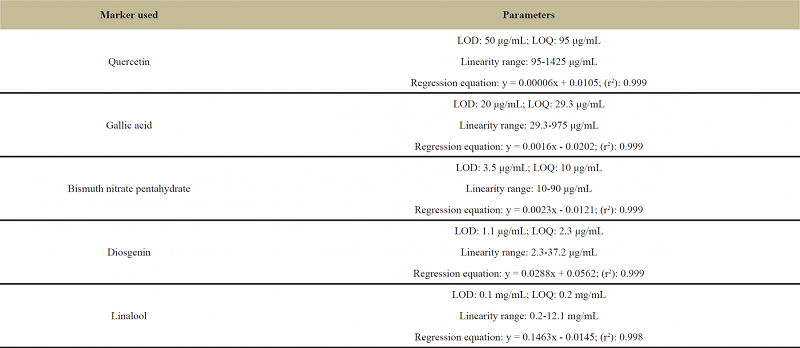 | Table 4A: Data on sensitivity and linearity of various reference markers used to determine the content of major groups of phytochemicals in Flemingia tuberosa. [Click here to view] |
.png) | Table 4B: Quantitative data on total flavonoids, total phenolics, total alkaloids, total saponins and total terpenoids in Flemingia tuberosa. [Click here to view] |
6. CONCLUSION
Chemoprofiling of Flemingia tuberosa established in this project work can be used for the correct identification of the species from other species of Flemingia. Kaempferol content has been quantitated from various parts of the plant with roots showing the maximum content of kaempferol. Kaempferol content for the two species of Flemingia, viz., Flemingia tuberosa and Flemingia vestita have been compared wherein Flemingia tuberosa shows a higher amount of kaempferol content. These methods can also be applied to various plant matrices and polyherbal formulations containing Kaempferol. Using such validated methods, Flemingia tuberosa with precise quality can be encouraged in herbal industries and cultivation of Flemingia tuberosa can be encouraged for its commercial use. The data generated through this research work can also be used as a monograph, which will be highly beneficial while using Flemingia tuberosa in the formulations.
7. ACKNOWLEDGMENT
We would like to express our gratitude towards Prof. S. R. Yadav (Shivaji University, Kolhapur) for helping us in the identification of Flemingia tuberosa in wild. A part of this work has been presented at 4th International Congress of the Society for Ethnopharmacology in Bardoli, Surat, India during February 23-25, 2017.
8. CONFLICT OF INTEREST
Nil.
9. REFERENCES
1. Ghalot K, Lal VK, Jha S. Phytochemical and Pharmacological potential of Flemingia Roxb. ex W.T.Aiton (Fabaceae). International Journal of Phytomedicine 2011; 3:294-307.
2. Ghorai N, Chakraborty S, Gucchait S, Saha SK, Biswas S. Estimation of total Terpenoids concentration in plant tissues using a monoterpene, Linalool as standard reagent 2012; 5.
3. Harborne JB. 2007. Phytochemical methods-A guide to modern techniques of plant analysis. Delhi: Springer Private Ltd.
4. Hertog MGL, Feskens EJM, Kromhout D, Hollman PCH, Katan MB. Dietary antioxidant flavonoids and risk of coronary heart disease: the Zutphen Elderly Study. The Lancet 1993; 342:1007-1011. Crossref
5. Kataria S, Bhardwaj S, Middha A. Standardization of medicinal plant materials. International Journal of Research in Ayurveda & Pharmacy 2011; 2:4.
6. Khandelwal KR. 2008. Practical Pharmacognosy: Techniques and Experiments, India: Nirali Prakashan.
7. Kirthikar KR, Basu BD. 1993. Indian medicinal plants. Dehradun, India: Bishen Singh Mahendra Pal Singh: 220.
8. Madan S, Gullaiya S, Singh GN, Kumar Y. Flemingiastrobilifera: review on phytochemistry and pharmacological aspects. International Journal of Phytopharmacology 2013; 4(4):255-262.
9. Marinova D, Ribarova F, Atanassova M. Total phenolics and total flavonoids in Bulgarian fruits and vegetables. Journal of the University of Chemical Technology and Metallurgy 2005; 40(3):255-260.
10. Mukherjee PK. 2007. Quality control of herbal drugs-an approach to evaluation of botanicals. New Delhi: Business Horizons Publishers.
11. Neuhouser ML. Dietary flavonoids and cancer risk: evidence from human population studies. Nutrition and cancer 2004; 50(1):1-7. Crossref
12. Indian Pharmacopoeia. 2010. Government of India “Ministry of Health & Family Welfare” The Controller & Publication. Delhi, India: 2186-2188.
13. Subrahmanya Prasad K, Biju P, Raveendran K. Flemingia tuberose Dalz.(Fabaceae)-a new addition to the flora of Kerala, India. Journal of Threatened Taxa 2011; 3(2):1550-1552.
14. Rajopadhye AA, Upadhye AS. Determination of phenolic content and in vitro antioxidant potential of ethanol extract of seven sources of Ayurvedic drug ‘Pittapapda.’ 2013.
15. Romanik G, Gilgenast E, Przyjazny A, Kamiński M. Techniques of preparing plant material for chromatographic separation and analysis. Journal of biochemical and biophysical methods 2007; 70(2):253-261.
16. Seasotiya L, Siwach P, Malik A, Bai S, Bharti P, Dalal S. Phytochemical evaluation and HPTLC fingerprint profile of Cassia fistula. International journal of advances in Pharmacy, biology and chemistry 2014; 604-611.
17. Shailajan S, Kumaria S, Pednekar S, Menon S, Choudhury H, Matani A. Estrogenic Potential of Flemingia vestita Benth Tubers in Ovariectomized Rat Model. Pharmacognosy Journal 2016; 8:1. Crossref
18. Shailajan S, Kumaria S, Pednekar S, Menon S, Joshi H, Matani A. Chromatographic evaluation of a phytoestrogen Genistein from Flemingia vestita Benth: An endemic plant of Northeast India. Pharmacognosy Communications 2014; 4(4):2.
19. Singh G, Kumar P. Phytochemical study and screening for antimicrobial activity of flavonoids of Euphorbia hirta. International Journal of Applied and Basic Medical Research 2013; 3(2):111. Crossref
20. Sreevidya N, Mehrotra S. Spectrophotometric method for estimation of alkaloids precipitable with Dragendorff’s reagent in plant materials. Journal of AOAC International 2003; 86(6):1124-1127.
21. Syed HM, Kunte SP, Jadhav BA, Salve RV. Extraction and characterization of papaya seed oil. International Journal of Applied Physical and Bio-Chemical Research 2012; 2:3343.
22. Tistaert C, Dejaegher B, Heyden YV. Chromatographic separation techniques and data handling methods for herbal fingerprints: a review. Analytica chimica acta. 2011; 690(2):148-161. Crossref
23. Uematsu Y, Hirata K, Saito K, Kudo I. Spectrophotometric determination of saponin in Yucca extract used as food additive. Journal of AOAC International 2000; 83(6):1451-1454.
24. Zhao Y, Kao C-P, Wu K-C, Liao C-R, Ho Y-L, Chang Y-S. Chemical compositions, chromatographic fingerprints and antioxidant activities of Andrographis Herba. Molecules 2014; 19(11):18332-18350. Crossref