1. INTRODUCTION
Multi-herbal formulations are predominantly used in Ayurveda for the maintenance of health and the management of diseases. However, these formulations are often very complex in their chemical composition, and their quality evaluation remains a major challenge in the Ayurveda drug industry. Significant differences can be seen in even a quick examination of multiple generic Ayurvedic formulas made and sold by various companies [1,2]. Ayurveda as a science and a drug industry, it is critical to adhere to strict and unwavering quality control guidelines to guarantee consistency and standards of formulations and products throughout the sector. Thus, standardization of herbal formulations is essential in order to assess the quality of drugs. Quality evaluation of herbal preparations is one of the fundamental requirements of industry and other organizations that deal with ayurvedic and herbal products. Regulatory bodies across the globe are forced to implement standards from input materials up to finished formulation. It is evident that the herbal industry needs to follow strict guidelines, and such regulations are necessary. According to WHO guidelines, an herbal product needs to be standardized with respect to safety before releasing it into the market. In this context, it is necessary to bring out a discussion on the need for better and more comprehensive standardization strategies for Ayurvedic formulations [3,4].
Prasarinyadi Kashayam is an Ayurvedic medicine that treats joint pain and vata disorders. It is primarily utilized in the Ayurvedic treatment of joint pain and musculoskeletal disorders. It is typically used to treat frozen shoulder and cervical spondylosis, headaches, pain, and stiffness in the lower back associated with lumbar spondylosis. The composition of the formulation is described in Table 1.
Table 1: Composition of Prasaranyadi Kashayam [6].
| S. No. | Sanskrit name | Botanical name | Quantity |
|---|
| 1. | Prasarini | Merremia tridentata | 1 Part |
| 2. | Masha | Vigna mungo | 1 Part |
| 3. | Bala | Sida cordifolia | 1 Part |
| 4. | Garlic (Rasona) | Allium sativum | 1 Part |
| 5. | Rasna | Alpinia galanga | 1 Part |
| 6. | Ginger (Sunthis) | Zingiber officinalis | 1 Part |
The present work is an attempt to develop the qualitative and quantitative standardization parameters for Prasarinyadi Kashayam in accordance with internationally acceptable standards. The results of this research can be used to monitor day-to-day quality control process during the manufacturing of these formulations to avoid product-to-product and batch-to-batch variations and to maintain the quality of finished formulations.
2. MATERIAL AND METHODS
2.1. Materials
HPLC and analytical grade chemicals were purchased from the commercial supplier and used in the study.
2.2. Procurement of Plant Material
The dried plant materials was purchased from verified commercial suppliers. The samples were visually inspected and validated for their morphological traits, then powdered using a mechanical blender at very low speed. The powdered samples were stored in airtight containers and kept in a dry place away from sunlight.
2.3. Preparation of Prasaranyadi Kashayam
The dried powder of plants was mixed in a previously described proportion [Table 1] to obtain the dried powder of Prasaranyadi Kashyam and the aqueous decoction (Kashyam/Kwath) was prepared as described by Nisteshwar and Vidyanath [5]. The prepared kwath solution was concentrated under vacuum to obtain a semisolid mass which was further dried at 40?C to obtain a powdered formulation and stored in airtight containers in a dry place away from sunlight until further use.
2.4. Physicochemical Evaluation
Physicochemical parameters such as total ash content, water-soluble ash, acid insoluble ash, loss on drying of the extract, pH was carried out on the dried extract according to the standard protocols [6,7].
2.5. Total Phenolic Contents
Total phenolic content was spectrophotometrically estimated using Folin-Ciocalteu reagent assays at 750 nm [8]. Gallic acid was used as a standard and the total phenolic contents were expressed as Gallic acid equivalents.
2.6. DPPH Radical Scavenging Assay
DPPH radical scavenging assay was performed as per the method described by Molole et al. [9]. In brief, 0.4 mL of test sample (PK) and gallic acid (50–400 μg/mL) were mixed with 2.6 mL of a methanolic solution of DPPH (0.1 mM). An equal amount of methanol (0.4 mL) and solution (2.6 mL) was used as a blank. The samples were all produced in triplicate, vortexed for one minute, and then incubated at 37°C in the dark for thirty minutes. The decrease in absorbance of each sample was measured against methanol as a blank on UV-Visible spectrophotometer at 517 nm.
All the experiments were performed in triplicate, and the results were expressed as mean ± SD.
2.7. HPTLC Method
The dried aqueous extract of Prasaranyadi kashayam was dissolved in water and subjected to HPTLC analysis. 10 uL of the extract was spotted on HPTLC silica gel 60F 254 (Merck) plate as bands of length 6 mm. The plates were developed using toluene, ethyl acetate, methanol, and formic acid (16, 14, 1, and 4) in the CAMAG twin-trough glass chamber previously saturated with the solvent for 30 min. After development, the plates were dried in an oven at 60°C and scanned at 254 nm and 366 nm.
3. RESULTS AND DISCUSSION
3.1. Physicochemical Evaluation and Total Phenolic Contents
The physiochemical parameters give the primary quality validation data; for example, the ash value of herbal drugs represents the total mineral content and helps in the identification of possible adulteration with inorganic materials. The loss on drying refers to moisture content, which has to be foremost in avoiding microbial growth, thus conferring stability to the drug during storage. Phenolic content is a key parameter for estimating antioxidant capacity and demonstrating the therapeutic potential of the herbal drug. These are the parameters whose exact measurements determine the quality, purity, and efficacy of herbal medicines [7]. Physicochemical parameters, such as total ash content, water-soluble ash, acid-insoluble ash, loss on drying, pH, and total phenolic content, were evaluated for PK and the results have been summarized in Table 2.
Table 2: Physicochemical analysis of Prasaranyadi Kashayam.
| Sl. No. | Physicochemical Parameter | Results* |
|---|
| 1. | Total ash content | 3.28 ± 0.15 |
| 2. | Water soluble ash | 2.08 ± 0.09 |
| 3. | Acid insoluble ash | 0.024 ± 0.008 |
| 4. | LOD at 105?C | 8.60% (for dried extract) |
| 5. | pH in aqueous solution | 6.4 ± 0.3 |
| 6. | Total Phenolic content | 6.37 + 0.002 |
3.2. DPPH Radical Scavenging Assay
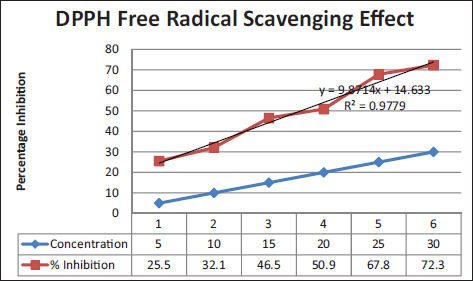
PK indicated good antioxidant activity in terms of percentage inhibition (72.30%) of DPPH radical [Figure 1].
3.3. HPTLC Figure Printing
HPTLC fingerprinting is a preferred analytical technique for the quality control and authentication of herbal drugs. It does not provide brief details about the chemical constituents present and helps in the identification and comparison of complex herbal formulations. Which, in turn, helps to ensure the consistency, and purity of herbal products. The HPTLC finger printing profile was developed in toluene, ethyl acetate, methanol, and formic acid (16, 14, 1, and 4) and the HPTLC chromatogram showed a total eight bands and corresponding peaks were recorded at respective Rf values as depicted in Figure 2.
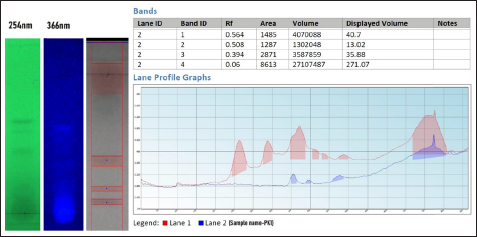 | Figure 2: HPTLC fingerprint profile of tannins in the leaf extract of Prasaranyadi Kashayam.
[Click here to view] |
4. CONCLUSION
This study aims to establish qualitative and quantitative standards for Prasaranyadi Kashayam. The findings from this research can be employed to oversee the quality control procedures in manufacturing these formulations, ensuring consistency and quality across different batches. The fingerprints created through HPTLC technique serve as valuable tools for identifying and quantifying key markers, as well as detecting any adulteration in PK preparations.
5. AUTHOR CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work. All the authors are eligible to be an author as per the international committee of medical journal editors (ICMJE) requirements/guidelines.
6. FINANCIAL SUPPORT AND SPONSORSHIP
There is no funding to report.
7. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
8. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
9. DATA AVAILABILITY
All the data is available with the authors and shall be provided upon request.
10. USE OF ARTIFICIAL INTELLIGENCE (AI)-ASSISTED TECHNOLOGY
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
11. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Balekundri, A., Mannur, V. Quality control of the traditional herbs and herbal products: a review. Futur J Pharm Sci. 2020;6:67. [CrossRef]
2. Wang H, Chen Y, Wang L, Liu Q, Yang S, Wang C. Advancing herbal medicine: enhancing product quality and safety through robust quality control practices. Front Pharmacol. 2023;14:1265178. [CrossRef]
3. Singh SK, Jha SK, Chaudhary A, Yadava RDS, Rai SB. Quality control of herbal medicines by using spectroscopic techniques and multivariate statistical analysis. Pharmaceut Biol. 2010;48(2):134-41. [CrossRef]
4. Hopkins AL. Network pharmacology: the next paradigm in drug discovery. Nat Chem Biol. 2008;4:682e90.
5. Tripathi A, Bankaitis VA. Molecular docking: from lock and key to combination lock. J Mol Med Clin Appl. 2017;2(10):10.
6. Nisteshwar K, Vidyanath R. Sahasrayogam: A popular book on Keraliya tradition of ayurvedic treatment. Banaras Ayurveda Series No. 20. 4th Ed, 2017.
7. Anonymous. Indian Herbal Pharmacopoeia. Publisher: Indian Drug Manufacturers? Association. 2002.
8. Anonymous. The Ayurvedic Pharmacopoeia of India. Publisher: Pharmacopoeia commission for indian medicine & homoeopathy, Ghaziabad. Vol IX, 2016.
9. Molole GJ, Gure A, Abdissa N. Determination of total phenolic content and antioxidant activity of Commiphora mollis (Oliv.) Engl. resin. BMC Chemistry. 2022;16:48. [CrossRef]