1. INTRODUCTION
Hyaluronan or Hyaluronic Acid (HA) is an unbranched polysaccharide of repeating units of disaccharides with around 2000–25,000 units of D-glucuronic acid and N-acetyl-D-glucosamine [1]. Its secondary structure is a 2-fold helix consisting of intramolecular hydrogen bonds. It possesses a multitude of alcohol and carboxylic acid groups along with a single amide functional group facilitating numerous ways of chemical modifications. It was found that the protein content of this glycosaminoglycan was of great importance, as by removing the protein component, it was found that HA no longer retained its useful therapeutic properties [2]. HA behaves as a stiffened coil in dilute solutions, which allows it to be in a liquid state and shows viscoelastic properties in concentrated solutions. The formation of stiffened coils was attributed mainly to the intramolecular hydrogen bonding and also to the mutual electrostatic force of repulsion between the carboxyl groups. The size of the molecule was shown to decrease with an increase in the ionic concentration [3]. This molecule is synthesized on the plasma membrane by macrophages, tumor, and stromal cells [4], aided by three different classes of HA synthase (HAS) enzymes (HAS 1, 2, and 3), and simultaneously secreted into the pericellular space [5]. Being a crucial component of the extracellular matrix (ECM), HA is the major cause of varying morphologies in the cell [6]. It also plays a major role in signal transduction pathways and induces migratory and proliferative signals inside the cell [3,7]. During immunological inflammation reactions, there is an increase in the degradation of ECM components, including HA. As HA gets broken down into its oligomers, they interact with dendritic cells and induce the maturation and expression of macrophage genes to upregulate and maintain the intensity of the inflammatory response [3]. Since its production occurs on the plasma membrane, the cell can take up the biomolecule through phagocytosis, receptor-mediated endocytosis, or other methods [1]. HA molecules have an impressive water-holding capacity, as each can retain up to a thousand times its molecular weight due to its dynamic system of intramolecular hydrogen bonds and the flexibility of the molecule. This characteristic allows it to be used as a hydrant in moisturizers [3,8]. It has also been found useful in many other industries, including pharmaceutical industries by acting as a potent vehicle for drug delivery, and in the medical industry by serving as a pain-killer, anti-cancer, and anti-inflammatory agent. This review elaborates on the myriad applications of the HA molecule alone and in association with other molecules.
2. PRODUCTION
Commercially, HA is majorly used in pharmaceutical, cosmetic, food and biomedical industries due to its biocompatibility [9]. It is generally found in vertebrates and also in some bacteria like Streptococcus and Pastuerella multocida, which produces HA as a protective layer outside the cell as it helps protect the cell against any exterior damage and also helps in the attachment and colonization of the bacteria on the particular substrate [10].
2.1. Biosynthetic Pathway
The biosynthesis of HA is catalyzed by the action of an enzyme, namely, HAS. It is synthesized on the plasma membrane; whereas glycosaminoglycan is synthesized in the Golgi apparatus [11]. HAS has several isoforms depending on the organism in which it is present. In the case of humans, it is found in almost all organs but significantly present in the connective tissue. It is present in higher concentrations in the vitreous humor, umbilical cord, and synovial fluid of joints [12]. In the case of all natural producers, HA synthesis initially starts from the pathway of glycolysis. The two important molecules of glycolytic pathway glucose – 6 – phosphate and fructose – 6 – phosphate give rise to two other molecules UDP-GlcUA and UDP-GlcNAc, respectively, which majorly act as precursors for HA synthesis. Once these two molecules are available in the cell, the enzyme HAS uses these as substrates for the synthesis of HA intracellularly, which is then released into the ECM of the cell [12]. HAS structure elucidations have suggested that they consist of multiple membrane-spanning domains with intracellular loops toward the inner side of the cell membrane. It was in the bacteria group A Streptococci, that the first gene encoding the enzyme was identified. The operon included the gene hasA encoding the enzyme HAS, hasB gene coding for the enzyme UDP-Glucose dehydrogenase and hasC gene that codes for UDP-Glucose Pyrophosphorylase [11].
HAS is grouped into two classes; Class 1, which is a single-domain integral membrane protein, and Class 2, which is a two-domain/soluble anchored membrane protein. Class 1 enzymes are generally ubiquitously present in the bacterium Streptococcus, vertebrates, and viruses, and are comparatively more complex than Class 2. On the other hand, Class 2 HAS enzyme has been found in P. multocida [13]. This classification of the HAS enzymes is based on the structural topology and amino acid sequence homology. Structural homology was observed between class 1 HAS of eukaryotes and class 1 HAS of microorganisms – it was made by assembling five transmembrane domains containing a motif protein on the glycosyltransferase domain in the cytoplasmic region which has a key role in the binding of UTP – sugars to the enzyme which is essential for the polymerization of HA [11]. As glucose-6-P acts as the major precursor for the synthesis of HA, the first step involves the conversion of glucose to glucose-6-P, which is catalyzed by the enzyme hexokinase. Further, the reactions are split into two pathways to form the monomers important for the production of HA, namely glucuronic acid and N-acetyl glucosamine. The first pathway resulting in the formation of glucuronic acid starts with the conversion of glucose-6-P into glucose-1-P, catalyzed by the enzyme phosphoglucomutase. Pyrophosphorylase, an enzymatic product of gene hasC, leads to the conversion of glucose-1-P to UDP-glucose, that on further oxidation gets converted to glucuronic acid, the first precursor of HA. This reaction is catalyzed by the enzyme UDP-glucose dehydrogenase encoded in the gene hasB. In the second pathway which involves the formation of the N-acetyl glucosamine, the glucose-6-P formed is then transformed to fructose-6-P with the help of the enzyme phosphoglucoisomerase encoded by the gene hasE. A set of enzymes called aminotransferases marks the amino group on fructose-6-P and the transfer of the glutamine residue results in the formation of glucosamine-6-P, the modification of which leads to the formation of glucosamine-1-P. Glucosamine-1-P is further acetylated and phosphorylated, leading to the formation of the second precursor. The enzyme HAS polymerizes both these precursors, finally forming the polymer HA [14,15] as depicted in Figure 1.
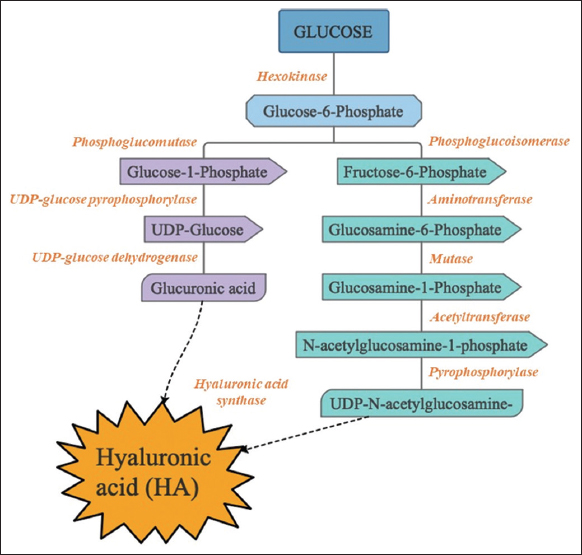 | Figure 1: Biosynthetic pathway of Hyaluronic acid. [Click here to view] |
2.2. Hyaluronan in Morphogenesis
HA-rich matrix maintains a hydrated environment that enables cell migration by interacting with cells via hyaluronic acid binding proteins (HABP). This can occur in granulation tissue and the initial inflammation stage in wound repair. Fibroblasts reach the wound site to produce pro-inflammatory cytokines that lead to the production of HA by endothelial cells. This promotes adhesion and thus the functioning of lymphocytes that are cytokine-activated, leading to an immune response at that site. Separation of cells from the matrix and mitosis is also facilitated by HA, thus promoting cell proliferation [16]. Cell migration can be prevented by the high binding of pericellular HA in cartilage with membrane proteoglycans. HA/HABP interactions also mediate the proliferation, differentiation, and maintenance of dormancy of stem cells like hematopoietic stem cells (HSCs), mesenchymal stem cells (MSCs), and even human embryonic stem cells (hESCs). Interactions between receptor for HA-mediated motility (RHAMM) and HA/CD44 are essential for the HSCs to migrate into and for homing into the bone marrow where they remain undifferentiated [17]. When MSCs are stimulated by platelet-derived growth factor, they increase the expression of CD44, which facilitates their migration by interactions with extracellular HA. This mechanism is vital for localization of MSCs for wound healing and regeneration of tissues. In terms of medicine, stem cell delivery using HA matrices improves cell survival and slowly releases them as the HA matrix is subject to enzymatic degradation. Neural stem cell (NSC) maturation and proliferation in the stroke cavity is promoted by the delivery of HA hydrogel-containing NSCs. By interacting with its signaling receptors, HA can maintain the cell structural integrity, lead to inflammatory response, and stimulate tissue repair.
2.3. Strategies to Improve Industrial Production
The large-scale manufacturing of HA is predominantly based on extraction from tissues of animals, bacterial fermentation using bacterial strains, and enzyme technology. The extraction of HA from animal tissue is one of the traditional methods used and is still followed for commercial production, but its use is limited due to certain disadvantages – the endogenous hyaluronidase activity and the harsh conditions used for extraction causing the degradation of the HA produced within the animal tissue. Contamination risks with viruses also pose a major problem [18]. Although the extraction protocols have been modified over the years, low yield is another major limitation of this method. Over the last two decades, the production of HA using bacterial fermentation technology has emerged as a mature technology. HA from microorganisms was found to be feasible in terms of its biocompatibility and non-immunogenic nature that is due to its structural similarity across various species [19].
In recent years, strains of Streptococci sp. are generally used from which promising yields of HA have been obtained on the industrial scale. Media optimization and strain improvement strategies are employed to increase the yield of HA. Streptococcus zooepidemicus is the commonly used strain for HA production. Hence, understanding the biosynthetic pathway is important as it provides information regarding the precursors, intermediates, and enzymes involved in the process of formation of HA. Since stages of cell growth and synthesis of HA share the same intermediates, there exists competition for the same, and therefore specific growth at high rate is not favorable for HA production. However by optimizing the fermentation conditions and media, the HA titer was found to be improved. The combined use of both batch and fed-batch culture was shown effective for HA synthesis; the two-stage culture strategy seemed to increase production of HA by 32% [20]. Improved HA synthesis has been obtained by strain improvement using random mutagenesis and by selecting the strain containing depolymerizing enzyme which decreases the molecular weight of the enzyme and also reduces streptolysin toxin. Fermentation using such strains which were non-hemolytic and hyaluronidase free increased the molecular weight of HA 6–7g/L [11]. Studies on the large-scale production of HA have to resolve problems like selective separation of UDP to prevent product inhibition, development of sugar nucleotide substrate using simple, low-cost technologies, large scale production of robust enzymes for the synthesis of HA, etc. [21,22]. In vivo, the HAS catalyzes the conversion of nucleotide sugars UDP-GlcNAc and UDP-GlcUA to produce HA [11]. Class 1 HASs are less suitable for the production of HA as these enzymes are integral membrane proteins and research on these would require performance on membrane fraction, which is less suitable for large-scale production [23]. Class 2 HASs produced by P. multocida are better enzymes for the production of HA; this enzyme has shown intracellular location when expressed in Escherichia coli [24].
3. APPLICATIONS
The uses of HA span across various biomedical fields. Cell growth, adhesion, migration, and proliferation are some of the biological processes aided by HA, other than inflammatory responses, joint lubrication, structural stability of organs, embryonic development [25], angiogenesis, tumor progression, and metastasis [4]. It also regulates many other biological processes concerned with inflammation, immune response, and tissue damage. The biocompatibility, biodegradability, and susceptibility of HA to chemical modification have proven to be helpful for researchers to develop HA-based biomaterials that have higher prospects in clinical and therapeutic applications. The construction of hydrogel matrices utilizes HA. It also acts as a tool in tissue engineering, a vehicle for drug delivery, and a tissue filler or a surgical device. As opposed to synthetic polymers such as polyethylene glycol (PEG), HA-based materials can enhance the biological activity of cells by facilitating changes in cellular behavior, like interactions with HA-based biomaterials [25]. The HA characteristic of binding to cell surface receptors can be exploited for drug-targeting purposes. The interaction and binding of cell surface receptors with HA lead to activation of signaling pathways that are involved in cell functioning, inflammatory response, wound healing and tissue development, tumor growth, and progression [26]. An increase in the levels of HA is also a considerable biomarker to predict the progression of COVID-19 [27]. High molecular weight HA has anti-inflammatory effects that cause downregulation of apoptotic pathways and provide protection to the respiratory epithelial cells [28]. It causes reduction in the production of pro-inflammatory cytokines during T-cell mediated liver injury [29]. It promotes maintenance and inhibits proliferation in regulatory T-cells [30].
3.1. Hyaluronan in Osteoarthritis (OA) Treatment
Matrix metalloproteinase 13 (MMP13) is the main protein that has been shown to be degraded in the ECM in the chondrocytes of osteoarthritic patients. HA has helped in the treatment of OA by downregulating MMPs through CD44, the adhesion molecule in a normal OA chondrocyte [31]. Different concentrations of HA have shown varying efficacies. Pre-treatment with Interleukin-1 beta (IL-1β) and monensin has depicted enhanced activity of HA while pre-treatment with IM7 has resulted in the reversal of the HA inhibitory effect. HA has also been shown to block phosphorylation of p38 induced by IL-1β at lower concentrations [32]. Intra articular HA blended with platelet-rich plasma (PRP) deemed a major advancement in the treatment of OA than treatment with PRP alone. HYAJOINT Plus is a combination of single cross-linked HA with a single PRP. There is significant reduction in the VAS pain, Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain and WOMAC stiffness exceeding clinical significance [33]. HA conjugated with tyramine and gelatin combined with epigallocatechin-3-gallate (EPCG) was used to prepare injectable composite hydrogel. Such HA-EPCG conjugated hydrogel greatly enhanced the degree of adhesion. Hydrogels like HA-EPCG can quaff a large amount of water and on injection, generate an anti-inflammatory microenvironment that influences regeneration [34]. Astragalus polysaccharides (APS) and HA were encapsulated in the nanostructure; APS was used as a therapeutic agent and hydrogel-encapsulated HA was used to deliver the agent. The upgrading of the functions of MMPs, particularly MMP9 and MMP13 induced by IL-1β were diminished by this nanostructure with much less cytotoxicity. The cytokines and proteinase in the chondrocytes were protected and their survival was prominently enhanced by the APS-HA loaded nanoparticles [35]. HA-based polymers were also synthesized by combining sulfo-dibenzocyclooctyne-PEG4-amine linkers and poly N-isopropylacrylamide (PNIPAM). Such derivatives prepared by PNIPAM grafting were unique, thermoreversible, capable of self-assembly, and enabled facilitated injection at room temperature. HA-L-PNIPAM could be successfully used as a viscosupplement treatment for OA thanks to its high viscoelastic value combined with enhanced resistance to enzymatic and reactive oxygen species-mediated degradation. In light of their versatility, they can also be used for a variety of different other applications as well [36]. There also exist different types of HA with different biocompatibility. Of the three kinds of injectable HA, namely, Avian-CL-HA (obtained from rooster comb and is a cross-linked HA), Avian-HA (derived from rooster comb), and Bio-HA (biological fermentation), Avian-HA and Bio-HA demonstrated similar levels of biocompatibility while immunostimulatory activity was seen more in Avian-CL-HA injections. One of the reasons for this is its cross-linking structure or an appreciable amount of (1 → 3)-β-D-glucan impurity in the formulation [37]. Some of the mechanisms by which HA alleviates OA are represented in Figure 2.
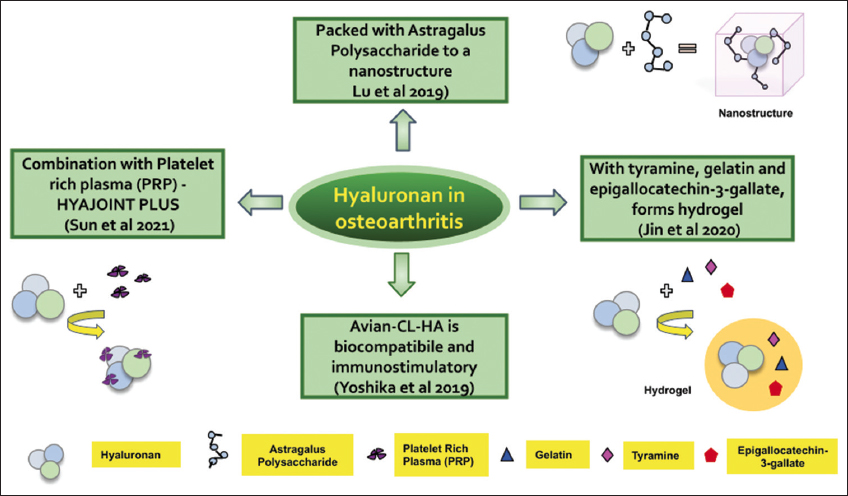 | Figure 2: Hyaluronan modified with other molecules for effective treatment of osteoarthritis. [Click here to view] |
3.2. Hyaluronan in Ophthalmology
HA is a naturally occurring constituent of the eye and is found in conjunctiva, corneal epithelium, lacrimal glands, and tears [38]. Hence, ophthalmic products with HA in the formulations are biocompatible and are commonly used viscosurgical devices to lubricate and protect the ocular cavities. It is also used during surgical procedures to replenish vitreous fluid during ophthalmic operations. It is the main ingredient in products such as eye drops and artificial tears [38]. Derivatives of HA like urea-crosslinked HA (HA-CL) and HA-cysteine ethyl ester are being studied for the formulation of eye drops with enhanced ocular residence time. Drug intravitreal injections have wide applications used in ophthalmology, but the short-term effect of the drugs leads to a need for repeated injections, which can cause side effects. The drug can be encapsulated into a hydrogel mixed with HA. This results in a gel that is highly biocompatible, facilitates a more stable release of drugs, and can sustain drug effects for more than 6 months. Nanoparticles can also be administered through intravitreal injection [39].
3.3. Hyaluronan as a Biomaterial
Due to the ubiquitous and biocompatible nature of HA, it is selectively used in three-dimensional applications such as tissue engineering, bioprinting, and regeneration of tissues. The 2D culture of tissues created many issues with visualizing and analysis of the phenotype of the tumor, as their morphology seemed different. This was remedied by culturing them in a microenvironment created by a microcarrier HA bead that replicate the conditions in vivo [40]. Initially, HA or its hydrogels could not be used for organ regeneration or bioprinting, since it could not retain its structure in 3D. Hence, HA was modified using Au NPs to enable cross-linking, so that it hardens slowly in 96 h, giving enough time for the arrangement and processing of the tissue to be grown [40]. Regeneration of bone tissue has also been attempted using a 3D polyetheretherketone scaffold coated with methylacrylated hyaluronic acid hydroxyapatite [41].
HA can be used effectively as a wound-healing agent or a dermal filler by harnessing the regenerative capacity of amniotic fluid-derived stem cells. The principle behind this was thought to be paracrine signaling, and any process that increases this signaling could improve wound healing. Photocrosslinkable HA hydrogels are being manufactured for this process, to enhance the effectiveness and reduce the time taken to heal in murine systems [40].
3.4. Other Biomedical Applications
HA has been used for drug delivery as it is non-toxic, has low molecular weight and is easily biodegradable [42], and can be used in different formulations such as micro and nanoemulsions, nanolipid carriers, ethosomes, niosomes and polymeric micelles. NP-associated HA carries small molecular drugs or nucleotides within an organic matrix such as lipid, PLGA, PEI, chitosan, or inorganic materials like iron, silica, and gold [43]. Hydrogel formed from HA stays intact, thus allowing the easy release of insulin into the surrounding tissue [44]. Medication-related osteonecrosis of the jaw can be prevented by vascular endothelial growth factor locally delivered through a heparin-HA hydrogel carrier [45]. HA-curcumin conjugate can be used for the treatment of diabetic wounds [46]. Fragmented and native HA has been successfully used to treat autoimmune diseases due to its anti-inflammatory and immune-tolerant nature [1,47], by targeting the CD44 receptor for chronic autoimmune diseases such as Type 1 diabetes, and for other autoimmune diseases such as rheumatoid arthritis and multiple sclerosis [48]. Nanoforms of HA have also been used in cancer therapy by CD44 receptor-mediated cell internalization of the conjugate; intracellular enzymes hydrolyze the HA derivatives, releasing the drug inside the targeted cancer cells [49-51].
4. CHALLENGES
HA has many complex biological functions but also has certain risks when used exogenously in the body. Its connection with inflammation, cancer, and other human pathologies complicates the process of formulations and clinical trials. Various precautions must be followed to establish long-term safety of HA-based products and biomaterials. Intra-articular HA injections are commonly used to treat symptomatic knee OA, where viscosupplementation is associated with an increased risk of serious side events [52,53]. In case of some tumors like the pancreatic ductal adenocarcinomas, HA has been found to act as a fuel for cancer cells by triggering the hexosamine production biosynthetic pathway [54]. In the tumor microenvironment, HA has been found to show dual characteristics. It is thought that increased rates of catabolism of HA could lead to more accumulation of low molecular weight HA which in turn paves way for inflammatory and precancerous microenvironment [55]. There are also concerns about cell delivery by employing HA-based biomaterials in tissue engineering and cancer therapy, since they may trigger cell migration and hence the metastasis of remaining tumorous cells after chemotherapy. The use of HA may not promote, but rather prolong proliferation of remnant tumor cells, which is a crucial metric in verifying the use of HA to improve chemotherapy techniques. The HA-based biomaterials designed for in vivo employment degrade rapidly due to enzymatic, ROS, and hydrolysis processes. The HA polymer has a swelling capacity that limits its mechanical stability and impairs its effectiveness for cell growth. Fragmented or low molecular weight HA levels are associated with various diseases such as pulmonary fibrosis and cancer [56].
5. FUTURE PERSPECTIVES
HA has applications in pharmaceutical, medicinal, and cosmetic products as it has extraordinary and diverse properties. From HA polymers to materials of clinical significance, there have been huge developments in HA-related fields. The use of microorganisms for the biosynthetic production of HA paves the way for the optimization of the biotechnological processes and the use of safe hosts. This provides an opportunity to regulate the molecular weight of HA by metabolic engineering that can be used to refine and manage the molecular weight of the HA. These advancements in the production of HA will further help research in enhancing the prevailing medical products and novel therapies in the coming years [12]. The HA metabolism, fragmentation, dose regimen for diverse therapeutic responses, mechanisms, and relevant downstream pathways remains poorly understood, limiting the broad use of the HA. The key factors that control the molecular weight during HA biotechnological synthesis are yet to be studied and clarified to create strategies for producing more consistent and size-defined HA. Further study on metabolic engineering is needed to enhance the production of HA and identify biosynthetic pathways with cost-efficiency and higher sustainability.
6. CONCLUSION
Hyaluronic acid, a biomolecule once known only to be the main part of the acrosome in sperms, is now being vastly used in a variety of fields ranging from cosmetics to medical industries. The molecular weight of HA, which serves as both an advantage and disadvantage, can be controlled and manipulated according to its usage in the future, thereby increasing its value manifold. So far, this compound used either in the crude form or in conjugation with other molecules, seems to have pros that far outweigh its cons without much debate. The field of nanotechnology has helped a long way in the therapeutic applications of many biomolecules including HA. Due to the ongoing research on this biomolecule, every aspect of it, ranging from its production to its applications can be made much more efficient with probable new discoveries along the way.
7. ACKNOWLEDGMENTS
The authors are grateful to the Department of Life Sciences, CHRIST (Deemed to be University), Bangalore, for the support extended for the successful completion of this work.
8. AUTHOR CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
9. FUNDING
There is no funding to report.
10. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
11. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
12. DATA AVAILABILITY
All the data is available with the authors and shall be provided upon request.
13. PUBLISHER’S NOTE
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors and the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
14. USE OF ARTIFICIAL INTELLIGENCE (AI)-ASSISTED TECHNOLOGY
The authors declares that they have not used artificial intelligence (AI)-tools for writing and editing of the manuscript, and no images were manipulated using AI.
REFERENCES
1. Kotla NG, Bonam SR, Rasala S, Wankar J, Bohara RA, Bayry J, et al. Recent advances and prospects of hyaluronan as a multifunctional therapeutic system. J Control Release 2021;336:598-620. [CrossRef]
2. Hargittai I, Hargittai M. Molecular structure of hyaluronan:An introduction. Struct Chem 2008;19:697-717. [CrossRef]
3. Garg HG, Hales CA. Chemistry and Biology of Hyaluronan. 1st ed. Amsterdam Boston:Elsevier;2004.
4. Karousou E, Parnigoni A, Moretto P, Passi A, Viola M, Vigetti D. Hyaluronan in the cancer cells microenvironment. Cancers (Basel) 2023;15:798. [CrossRef]
5. Skandalis SS, Karalis T, Heldin P. Intracellular hyaluronan:Importance for cellular functions. Semin Cancer Biol 2020;62:20-30. [CrossRef]
6. Garantziotis S, Savani RC. Hyaluronan biology:A complex balancing act of structure, function, location and context. Matrix Biol 2019;78-79:1-10. [CrossRef]
7. Carvalho AM, Reis RL, Pashkuleva I. Hyaluronan receptors as mediators and modulators of the tumor microenvironment. Adv Healthc Mater 2023;12:e2202118. [CrossRef]
8. Kobayashi T, Chanmee T, Itano N. Hyaluronan:Metabolism and function. Biomolecules 2020;10:1525. [CrossRef]
9. Wang Y, Hu L, Huang H, Wang H, Zhang T, Chen J, et al. Eliminating the capsule-like layer to promote glucose uptake for hyaluronan production by engineered Corynebacterium glutamicum. Nat Commun 2020;11:3120. [CrossRef]
10. Zhang L, Huang H, Wang H, Chen J, Du G, Kang Z. Rapid evolution of hyaluronan synthase to improve hyaluronan production and molecular mass in Bacillus subtilis. Biotechnol Lett 2016;38:2103-8. [CrossRef]
11. Boeriu CG, Springer J, Kooy FK, van den Broek LA, Eggink G. Production methods for hyaluronan. Int J Carbohydr Chem 2013;2013:624967. [CrossRef]
12. de Oliveira JD, Carvalho LS, Gomes AM, Queiroz LR, Magalhães BS, Parachin NS. Genetic basis for hyper production of hyaluronic acid in natural and engineered microorganisms. Microb Cell Fact 2016;15:119. [CrossRef]
13. Agarwal G, Krishnan KV, Prasad SB, Bhaduri A, Jayaraman G. Biosynthesis of hyaluronic acid polymer:Dissecting the role of sub structural elements of hyaluronan synthase. Sci Rep 2019;9:12510. [CrossRef]
14. Ucm R, Aem M, Lhb Z, Kumar V, Taherzadeh MJ, Garlapati VK, et al. Comprehensive review on biotechnological production of hyaluronic acid:Status, innovation, market and applications. Bioengineered 2022;13:9645-61. [CrossRef]
15. Shah MV, Badle SS, Ramachandran KB. Hyaluronic acid production and molecular weight improvement by redirection of carbon flux towards its biosynthesis pathway. Biochem Eng J 2013;80:53-60. [CrossRef]
16. Blunck D, Schöffski O. Hyaluronic acid treatment versus standard of care in chronic wounds in a German setting:Cost-effectiveness analysis. Health Sci Rep 2023;6:e969. [CrossRef]
17. Munjal A, Hannezo E, Tsai TY, Mitchison TJ, Megason SG. Extracellular hyaluronate pressure shaped by cellular tethers drives tissue morphogenesis. Cell 2021;184:6313-25.e18. [CrossRef]
18. Murado MA, Montemayor MI, Cabo ML, Vázquez JA, González MP. Optimization of extraction and purification process of hyaluronic acid from fish eyeball. Food Bioprod Process 2012;90:491-8. [CrossRef]
19. Suri S, Schmidt CE. Photopatterned collagen-hyaluronic acid interpenetrating polymer network hydrogels. Acta Biomater 2009;5:2385-97. [CrossRef]
20. Liu L, Liu Y, Li J, Du G, Chen J. Microbial production of hyaluronic acid:Current state, challenges, and perspectives. Microb Cell Fact 2011;10:99. [CrossRef]
21. Kooy FK, Ma M, Beeftink HH, Eggink G, Tramper J, Boeriu CG. Quantification and characterization of enzymatically produced hyaluronan with fluorophore-assisted carbohydrate electrophoresis. Anal Biochem 2009;384:329-36. [CrossRef]
22. Kooy FK. Enzymatic Production of Hyaluronan Oligo- and Polysaccharides. Dissertation PhD Thesis. Wageningen University;2010.
23. Pang B, Wang H, Huang H, Liao L, Wang Y, Wang M, et al. Enzymatic production of low-molecular-weight hyaluronan and its oligosaccharides:A review and prospects. J Agric Food Chem 2022;70:14129-39. [CrossRef]
24. Jing W, DeAngelis PL. Dissection of the two transferase activities of the Pasteurella multocida hyaluronan synthase:Two active sites exist in one polypeptide. Glycobiology 2000;10:883-9. [CrossRef]
25. Dicker KT, Gurski LA, Pradhan-Bhatt S, Witt RL, Farach-Carson MC, Jia X. Hyaluronan:A simple polysaccharide with diverse biological functions. Acta Biomater 2014;10:1558-70. [CrossRef]
26. Petrey AC, de la Motte CA. Hyaluronan, a crucial regulator of inflammation. Front Immunol 2014;5:101. [CrossRef]
27. Yang S, Ling Y, Zhao F, Li W, Song Z, Wang L, et al. Hymecromone:A clinical prescription hyaluronan inhibitor for efficiently blocking COVID-19 progression. Signal Transduct Target Ther 2022;7:91. [CrossRef]
28. Jiang D, Liang J, Fan J, Yu S, Chen S, Luo Y, et al. Regulation of lung injury and repair by Toll-like receptors and hyaluronan. Nat Med 2005;11:1173-9. [CrossRef]
29. Nakamura K, Yokohama S, Yoneda M, Okamoto S, Tamaki Y, Ito T, et al. High, but not low, molecular weight hyaluronan prevents T-cell-mediated liver injury by reducing proinflammatory cytokines in mice. J Gastroenterol 2004;39:346-54. [CrossRef]
30. Bollyky PL, Falk BA, Wu RP, Buckner JH, Wight TN, Nepom GT. Intact extracellular matrix and the maintenance of immune tolerance:High molecular weight hyaluronan promotes persistence of induced CD4+CD25+regulatory T cells. J Leukoc Biol 2009;86:567-72. [CrossRef]
31. Julovi SM, Ito H, Nishitani K, Jackson CJ, Nakamura T. Hyaluronan inhibits matrix metalloproteinase-13 in human arthritic chondrocytes via CD44 and P38. J Orthop Res 2011;29:258-64. [CrossRef]
32. Julovi SM, Ito H, Hiramitsu T, Yasuda T, Nakamura T. Hyaluronan inhibits IL-1beta-stimulated collagenase production via down-regulation of phosphorylated p38 in SW-1353 human chondrosarcoma cells. Mod Rheumatol 2008;18:263-70. [CrossRef]
33. Sun SF, Lin GC, Hsu CW, Lin HS, Liou IS, Wu SY. Comparing efficacy of intraarticular single crosslinked Hyaluronan (HYAJOINT Plus) and platelet-rich plasma (PRP) versus PRP alone for treating knee osteoarthritis. Sci Rep 2021;11:140. [CrossRef]
34. Jin Y, Koh RH, Kim SH, Kim KM, Park GK, Hwang NS. Injectable anti-inflammatory hyaluronic acid hydrogel for osteoarthritic cartilage repair. Mater Sci Eng C Mater Biol Appl 2020;115:111096. [CrossRef]
35. Lu G, Du L, Lu J, Jin L. Nanoparticles containing hyaluronan acid and Astragalus polysaccharides for treating osteoarthritis. Int J Polym Sci 2019;2019:8143528. [CrossRef]
36. Porcello A, Gonzalez-Fernandez P, Jordan O, Allémann E. Nanoforming hyaluronan-based thermoresponsive hydrogels:Optimized and tunable functionality in osteoarthritis management. Pharmaceutics 2022;14:659. [CrossRef]
37. Yoshioka K, Katayama M, Nishiyama T, Harada K, Takeshita S, Kawamata Y. Biocompatibility study of different hyaluronan products for intra-articular treatment of knee osteoarthritis. BMC Musculoskelet Disord 2019;20:424. [CrossRef]
38. Fallacara A, Vertuani S, Panozzo G, Pecorelli A, Valacchi G, Manfredini S. Novel artificial tears containing cross-linked hyaluronic acid:An in vitro re-epithelialization study. Molecules 2017;22:2104. [CrossRef]
39. Yu Y, Lau LC, Lo AC, Chau Y. Injectable chemically crosslinked hydrogel for the controlled release of bevacizumab in vitreous:A 6-month in vivo study. Transl Vis Sci Technol 2015;4:5. [CrossRef]
40. Serban MA, Skardal A. Hyaluronan chemistries for three-dimensional matrix applications. Matrix Biol 2019;78-79:337-45. [CrossRef]
41. Ferroni L, D'Amora U, Leo S, Tremoli E, Raucci MG, Ronca A, et al. PEEK and hyaluronan-based 3D printed structures:Promising combination to improve bone regeneration. Molecules 2022;27:8749. [CrossRef]
42. Juhašcik M, Kovácik A, Huerta-Ángeles G. Recent advances of hyaluronan for skin delivery:From structure to fabrication strategies and applications. Polymers (Basel) 2022;14:4833. [CrossRef]
43. Sakurai Y, Harashima H. Hyaluronan-modified nanoparticles for tumor-targeting. Expert Opin Drug Deliv 2019;16:915-36. [CrossRef]
44. Jiang D, Liang J, Noble PW. Hyaluronan in tissue injury and repair. Annu Rev Cell Dev Biol 2007;23:435-61. [CrossRef]
45. Sharma D, Hamlet S, Vaquette C, Petcu EB, Ramamurthy P, Ivanovski S. Local delivery of hydrogel encapsulated vascular endothelial growth factor for the prevention of medication-related osteonecrosis of the jaw. Sci Rep 2021;11:23371. [CrossRef]
46. Manju S, Sreenivasan K. Conjugation of curcumin onto hyaluronic acid enhances its aqueous solubility and stability. J Colloid Interface Sci 2011;359:318-25. [CrossRef]
47. Khan N, Niazi ZR, Rehman FU, Akhtar A, Khan MM, Khan S, et al. Hyaluronidases:A therapeutic enzyme. Protein Pept Lett 2018;25:663-76. [CrossRef]
48. Nagy N, Kuipers HF, Marshall PL, Wang E, Kaber G, Bollyky PL. Hyaluronan in immune dysregulation and autoimmune diseases. Matrix Biol 2019;78-79:292-313. [CrossRef]
49. Mattheolabakis G, Milane L, Singh A, Amiji MM. Hyaluronic acid targeting of CD44 for cancer therapy:From receptor biology to nanomedicine. J Drug Target 2015;23:605-18. [CrossRef]
50. Lee SY, Kang MS, Jeong WY, Han DW, Kim KS. Hyaluronic acid-based theranostic nanomedicines for targeted cancer therapy. Cancers (Basel) 2020;12:940. [CrossRef]
51. Liu M, Tolg C, Turley E. Dissecting the dual nature of hyaluronan in the tumor microenvironment. Front Immunol 2019;10:947. [CrossRef]
52. Rutjes AW, Jüni P, da Costa BR, Trelle S, Nüesch E, Reichenbach S. Viscosupplementation for osteoarthritis of the knee:A systematic review and meta-analysis. Ann Intern Med 2012;157:180-91. [CrossRef]
53. Hunter DJ. Viscosupplementation for osteoarthritis of the knee. N Engl J Med 2015;372:1040-7. [CrossRef]
54. Kim PK, Halbrook CJ, Kerk SA, Radyk M, Wisner S, Kremer DM, et al. Hyaluronic acid fuels pancreatic cancer cell growth. Elife 2021;10:10.7554/eLife.62645. [CrossRef]
55. Liu Y, Li L, Wang L, Lu L, Li Y, Huang G, et al. “Two-faces”of hyaluronan, a dynamic barometer of disease progression in tumor microenvironment. Discov Oncol 2023;14:11. [CrossRef]
56. Walker K, Basehore BM, Goyal A, Zito PM. Hyaluronic acid. In:StatPearls. Treasure Island, FL:StatPearls Publishing;2023.