1. INTRODUCTION
Spodoptera litura (Fabricius) is a serious insect pest next to the pod borer Helicoverpa armigera (Hubner) with high reproductive potential, and its adult moths have the ability to migrate a large distance [1-3]. Now this pest is considered a most destructive insect pest, as almost 30% of the total insecticides throughout the world are used to control its population. This pest has a close association with many host crops and shows a polyphagous nature. That’s why it has many different names, such as beet armyworm, tobacco cutworm, tobacco budworm, cotton leaf worm, cotton cutworm, rice cutworm, pigweed caterpillar, and taro caterpillar. S. litura is distributed throughout the world, invading 112 species of plants belonging to 44 families, of which 40 species have been reported from India [4,5].
These insect pests are responsible for causing losses to vegetables, oilseeds, and pulses [6-8]. Vegetative parts such as leaves and reproductive parts like seed and fruits of host plants are fed by larvae [9]. The larvae caterpillars are the feeding stage that mainly causes a significant reduction in yield [10,11]. Larval forms of this pest are easily noticed in the field, as the destruction caused by them is easily visible to the naked eye. Mature larval instars of these insect pests skeletonized the leaves and caused defoliation, which weakened the plant’s capacity for photosynthesis [12,13].
In the absence of favorable hosts, S. litura migrates to other plants to complete their life cycle; this nature of the pest eventually makes it tough to control. It is also responsible for causing up to 70% yield losses in black Gram, followed by rice in Andhra Pradesh, India [14,15]. It is also responsible for causing 67% yield losses in groundnut. Due to its economic importance, chemical insecticide applications are the primary method used against S. litura throughout the crop growing seasons [16]. Researchers throughout the world have used different insecticides against this particular pest on different cultivated crops. Several novel insecticides have been registered to provide subsequent control of different insect pests [17,18]. These synthetic insecticides are very effective, but their indiscriminate use leads to many problems, such as the development of resistance against different insecticides in the genus Spodoptera [19,20], the resurgence of other minor pests in the fields, adverse effects on the useful biocontrol agents [21], environmental pollution in water, soil, and air, unwanted toxic chemical residue, and ultimately the whole disturbance in the agro-ecosystem [22,23].
This species of Spodoptera has attained the status of the major alarming insect pest on many crops and causes severe economic losses per year. A number of management strategies, such as physical, cultural, mechanical, chemical, botanical, and bio-pesticide, have been adopted at a small and large level to reduce the percent incidence of pest attacks and to protect the different crops [24].
Presently, entomopathogens are used as natural biocontrol agents [25-27] for the management of economically important horticultural and agricultural crop insect pests [28,29]. Among them, Bacillus thuringiensis var. kurstaki (Btk) is used against Helicoverpa armigera (Hubner) and Beauveria bassiana (Balsamo) [27], Metarhizium anisopliae [30], and Purpureocillium lilacinus against Spodoptera litura [31]. The need of the hour is to promote the use of bio- agents and bio-pesticides. The present study on the bio-efficacy of the different bio-pesticides will help in selecting an effective dose as well as effective bio-pesticides for economically and eco-friendly management of the tobacco caterpillar population in the field, as these insect pests are responsible for causing extensive damage in the field conditions.
2. MATERIALS AND METHODS
2.1. Availability of Biological Test Materials
The test material for three biopesticides, namely Bacillus thuringiensis (Mahastra, 0.5% W.P., International Panncea Limited), Beauveria bassiana (Daman, 1.0% W.P., International Panncea Limited), and Metarhizium anisopliae (Kalichakra, 1.0% W.P., International Panncea Limited), were purchased from the neighboring market (Solan) to perform the bioassay experiment against the 3rd and 4th instar larvae of S. litura.
2.2. Rearing of Test Insects in the Laboratory
The adult and larval stages of the test insect were collected from the agricultural field and brought to the laboratory. The rearing was done in the laboratory using the methodology followed by the methods of Tomar and Thakur [32].
2.3. Laboratory Bioassay Experiment
During this bioassay, new and fresh castor leaves were separated from the castor plants with the help of scissors. The collected leaves were washed properly and air-dried before being used in the bioassays. There were three different concentrations of each bio-pesticide prepared in the distilled water along with the control, and each treatment was replicated five times. The leaf dip method was used to perform the bioassays. Approximately 6 cm of equal-sized ten leaves were dipped into the prepared concentrations (0.5, 1.0, and 1.5%) of each bio-pesticide for 30 s and later kept on the tissue paper for an hour to dry them. Each treated leaf was then placed into the glass petri dish of 6 cm diameter with the help of forceps. Ten larvae per glass petri dish containing nine different concentrations of treated castor leaves were released through a brush, and the petri dishes were covered properly with the lid to avoid larval escape. The larval mortality using Abbott’s formula [33] was recorded at specific times (after 24, 48, 72, 96, and 120 h) after treatment.
2.4. Statistical Analysis
The analysis of variance (ANOVA, two-factor analysis) was used to evaluate the bio control efficacy of S. litura larvae. The median lethal concentrations (LC50) of bio-pesticides were calculated through probit analysis using OP statistics.
3. RESULTS
3.1. Rearing of Test Insect
The collected adults of S. litura were transferred to the chimney for oviposition. After oviposition, eggs were hatched, and the emerging larvae were fed upon the cabbage leaf. Up to the third instar, the larvae were placed together, but after that, they were transferred to separate vials in order to avoid cannibalism. A total of six larval instars were observed during this investigation. The populations of the 3rd and 4th instars were used for further bioassay experiments.
3.2. Laboratory Bioassay Experiment
The insecticidal attributes of different bioinsecticides at various concentrations were evaluated against the 3rd and 4th larval instars of S. litura in a bioassay study [Figure 1]. The data obtained from the bio-efficacy test revealed that both instars of S. litura were highly susceptible to bioinsecticide infection. Based upon the analysis of variance, the results revealed that in the 3rd instar larvae of S. litura, maximum 80 ± 0.28% larval mortality was observed after 120 h in the treatment concentration of 1.5% B. thuringiensis, followed by the highest concentration of 1.5% B. bassiana with 72 ± 0.21% mortality, and M. anisopliae caused 64 ± 0.20% larval mortality after 120 h exposure to these bioinsecticides (F statistics (F) = 1.16, degree of freedom (df) = 9, P < 0.05) [Figure 2]. The calculated median lethal concentration in probit analysis demonstrated the lowest LC50 = 1.22/10 larvae (95% fiducial limit: 4.71–11.45) with Pearson’s χ2 value of 0.68 amongst the 3rd instar larvae after 72 h [Table 1].
 | Figure 1: Laboratory rearing and bioassay study of Spodoptera litura: (a) Adult male; (b) Adult female; (c) Eggs in cluster; (d) Larvae feeding on leaves; (e) Different larval Instars; (f) Pupal stages under the stereozoom microscope; (g) Laboratory bioassay experiment; (h) Dead insect cadavers after the treatments. [Click here to view] |
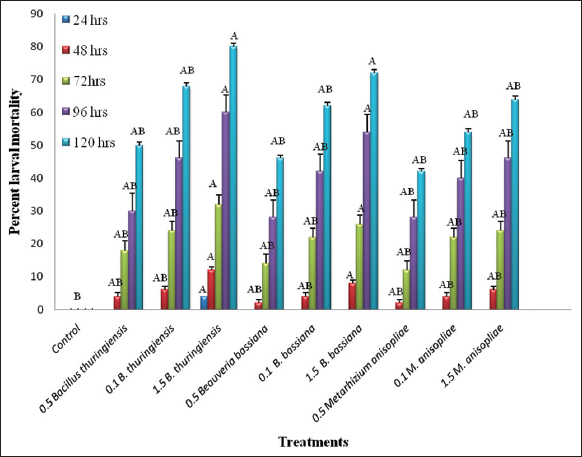 | Figure 2: Effect of applications of different bio insecticides on the mortality in 3rd instar larvae of Spodoptera litura. [Click here to view] |
Table 1: Calculated median lethal concentration (LC50) through probit analysis in Spodoptera litura larvae after treatment with bio insecticides.
| Larval Instar | Mortality after Hours | LC50 value | 95% Fiducial Limit | Pearson’s χ2 | |
|---|---|---|---|---|---|
| Upper limit | Lower limit | ||||
| 3rd Instar | 48 | 7.35 | 11.45 | 4.71 | 0.65 |
| 72 | 1.22 | 1.72 | 0.87 | 0.68 | |
| 4th Instar | 48 | 12.64 | 21.91 | 7.29 | 0.36 |
| 72 | 1.59 | 2.36 | 1.08 | 0.52 | |
*LC50: Mediam lethal concentration; Pearson’s c2 : Pearson’s Chi square value to determine the significant difference between the expected and observed values.
Similarly, the analysis of variance was determined for the 4th instar larvae of S. litura. The results revealed that in the 4th instar larvae of S. litura, maximum 76 ± 0.23% larval mortality was observed after 120 h in the treatment concentration of 1.5% B. thuringiensis, followed by the highest concentration of 1.5% B. bassiana having 70 ± 0.21% mortality, and M. anisopliae causing 64 ± 0.20% larval mortality after 120 h exposure to these bioinsecticides (F = 1.10, df = 9, P < 0.05) [Figure 3]. The calculated median lethal concentration in probit analysis demonstrated the lowest LC50 = 1.59/10 larvae (95% FL: 1.08-2.36) with Pearson’s χ2 value of 0.52 amongst the 4th instar larvae after 72 h. The P-value in this study was <0.05, indicating that all the concentrations of bioinsecticide inoculum were statistically significant and all the other treatments were superior to the control.
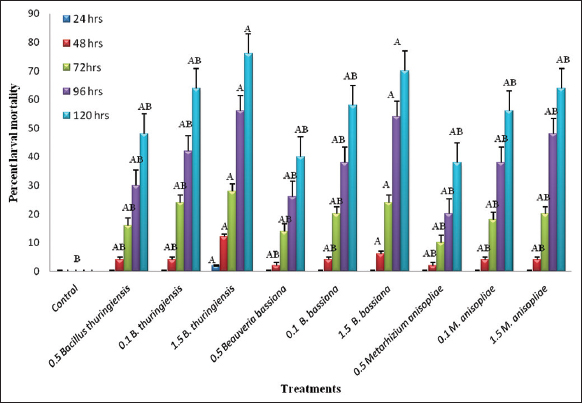 | Figure 3: Effect of applications of different bio insecticides on the mortality in 4th instar larvae of Spodoptera litura. [Click here to view] |
4. DISCUSSION
Insect pests are known for causing substantial damage to agricultural produce since ancient times. S. litura is a polyphagous insect pest known to damage many crops, including vegetables, oilseeds, and pulses, throughout the world. Though the use of synthetic insecticides is very effective in controlling the population of S. litura, it is also hazardous to other non-targeted organisms, including human beings. The excessive use of synthetic insecticides also resulted in pest resistance and resurgence problems. This has raised the need for some alternative that must be eco-safe. The use of biopesticides is such an alternative that can solve all these issues. The present study demonstrated the management of the armyworm using three biopesticides.
The test materials, namely B. bassiana, M. anisopliae, and B. thuringiensis formulations were evaluated in the laboratory on 3rd and 4th instar larvae of S. litura larvae at different doses. The results revealed that in 3rd instar larvae of S. litura, maximum 80 ± 0.28% larval mortality was observed in the inoculum of B. thuringiensis, followed by B. bassiana with 72 ± 0.21% mortality, and M. anisoplae with 64 ± 0.20% larval mortality after 120 h exposure at the highest dose of 1.5%. The 4th instar larvae of S. litura showed maximum 76 ± 0.23% larval mortality caused by B. thuringiensis, followed by B. bassiana 70 ± 0.21% and M. anisoplae 64 ± 0.20% at inoculum 1.5% after 120 h exposures. Almost similar results were observed by Malarvannan et al. (2010), who reported that larvae of S. litura showed a small amount of pupation when treated with four different concentrations, viz., 2.4 × 104, 2.4 × 105, 2.4 × 107, and 2.4 × 106 conidia/mL of B. bassiana. They further reported that the emergence of healthy moth was minimum at 2.4 × 104 spore concentrations, while egg laying was totally stopped at the highest concentration [34]. The results of the present investigation are also in accordance with Kaur et al. (2011), who used three concentrations of B. bassiana, i.e., 2.03 × 108, 4.03 × 106, and 1.47 × 105 spores/mL, against the 2nd, 3rd, and 4th larval stages of S. litura to check the virulence of B. bassiana, and significantly higher mortality in treatments than control [35]. Besides mortality, sub-lethal effects were also observed in larvae that endured fungal infection. Similar observations were obtained by Freed et al. (2012), who reported the efficacy of M. anisopliae against the third instar larvae of Spodoptera exigua by isolating M. anisopliae from the soil of a cotton field and observed that it caused 87.5% mortality in the larvae of S. exigua in laboratory and semi-field conditions [36].
The results of the present investigation are also similar to the previous work done by Agrawal and Simon (2017), who used B. bassiana against different larval stages of S. litura using different concentrations of 1%, 2%, 3%, 4%, and 5% in 2.3 × 106 conidia/mL. The results revealed that 91.66, 90.00, 88.33, 78.77, 66.11, and 49.99 percent mortality were shown by the highest dose at 5% 2.3 × 106 conidia of B. bassiana in 1st, 2nd, 3rd, 4th, 5th, and 6th instar larvae [37]. Similar observations were obtained by Narvekar et al. (2018), who studied the bioefficacy of B. thuringiensis against the 3rd instar larval stage of S. litura by using different host plants. They observed that B. thuringiensis on okra showed 96.67% mortality, followed by cowpea (90.00%), whereas this bio-insecticide on sweet potato was significantly inferior and least effective (6.67%), respectively [38]. Huange et al. (2018) reported the efficiency of B. thuringiensis CAB109 on S. exigua larvae using sublethal concentrations of 0, 102, 103, 104, 105, and 106 colony-forming units (CFU) per mL, and after 7 days of treatment, mortality rates were 5.0, 8.3, 15.0, 23.3, 36.0, and 55.0%, respectively [39]. Earlier, Thakur et al. (2022) applied individual and combined treatments of the biopesticides H. bacteriophora, B. thuringiensis, and B. bassiana against S. litura larvae. They also reported 96% and 92% mortality in S. litura larvae upon treatment with B. thuringiensis and B. bassiana, respectively [8].
5. CONCLUSION
The different concentrations of bio-insecticides were applied against the 3rd and 4th instar larvae of S. litura. It can be concluded from the results that both larval instars were highly susceptible to these bio-insecticides. B. thuringiensis was found to be the most effective biocontrol agent, followed by B. bassiana and M. anisopliae. Larvae treated with entomopathogenic fungus and bioinsecticides exhibit various morphological abnormalities. Infected larval cadavers were highly contaminated, with inclusion bodies appearing blackish in color. Also, with the enhancement of treatment concentrations and exposure durability, larval mortality also increased. These bioinsecticides can be included in integrated pest management programs as they do not pose any toxic hazards to the environment. Further studies are required to explore its potential in insect pest management through field experiments.
6. ACKNOWLEDGMENTS
The study was carried out in the Zoology Laboratory at Eternal University, Baru Sahib, Himachal Pradesh. The authors are thankful to the Vice Chancellor of Eternal University, Baru Sahib, for providing the necessary laboratory facilities.
7. AUTHORS CONTRIBUTION
AS and SJ conducted the experiment and wrote the manuscript. PT helped in conducting surveys to collect the insects from fields and with statistical analysis. SS, AKS, SC, MK, SS, SR and ANY reviewed the manuscript. NT gave the concept and drafted the manuscript. All authors have read and reviewed the manuscript.
8. FUNDING
There is no funding to report.
9. CONFLICTS OF INTEREST
The authors declare that there is no conflict of interest.
10. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
11. DATA AVAILABILITY
All the data is available with the authors and shall be provided upon request.
12. PUBLISHER’S NOTE
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors and the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Anjali Y, Manoj K, Chauhan AK. Biology of tobacco caterpillar (Spodoptera litura Fab.) on different host plants. Asian J Anim Sci2014;9:111-2.
2. Murtaza G, Ramzan M, Sabir MW, Shafiq M, Shahid M, Maroof A, et al. Effect of host plant on the biology of Spodoptera litura. Indian J Entomol 2020;82:123-6. [CrossRef]
3. Ramzan M, Ilahi H, Umar AB, Nasir M, Zahid MK, Rukh S, et al. Biological parameters of armyworm, Spodoptera litura and toxicity of three insecticides against 3rd instars larvae under laboratory conditions. Indian J Pure Appl Biosci 2021;9:12-7. [CrossRef]
4. Chari M, Patel S. Cotton leaf worm Spodoptera litura Fabricius, its biology and integrated control measures. Cotton Dev 1983;13:7-8.
5. Kergoat GJ, Goldstein PZ, Le Ru B, Meagher RL Jr., Zilli A, Mitchell A, et al. A novel reference dated phylogeny for the genus Spodoptera Guenée (Lepidoptera:Noctuidae:Noctuinae):New insights into the evolution of a pest-rich genus. Mol Phylogenet Evol 2021;161:107161. [CrossRef]
6. Pogue MG, Passoa S. Spodoptera ochrea (Lepidoptera:Noctuidae):A new host record (Asparagus) from Peru and description of the female genitalia. Ann Entomol Soc Am 2000;93:1019-21. [CrossRef]
7. Saleem M, Hussain D, Ghouse G, Abbas M, Fisher SW. Monitoring of insecticide resistance in Spodoptera litura (Lepidoptera:Noctuidae) from four districts of Punjab, Pakistan to conventional and new chemistry insecticides. Crop Prot 2016;79:177-84. [CrossRef]
8. Thakur N, Tomar P, Sharma S, Kaur S, Sharma S, Yadav AN, et al. Synergistic effect of entomopathogens against Spodoptera litura (Fabricius) under laboratory and greenhouse conditions. Egypt J Biol Pest Control 2022;32:39. [CrossRef]
9. Yooboon T, Pengsook A, Ratwatthananon A, Pluempanupat W, Bullangpoti V. A plant-based extract mixture for controlling Spodoptera litura (Lepidoptera:Noctuidae). Chem Biol Technol Agric 2019;6:5. [CrossRef]
10. Rukundo P, Karangwa P, Uzayisenga B, Ingabire JP, Waweru BW, Kajuga J, et al. Outbreak of fall armyworm (Spodoptera frugiperda) and its impact in Rwanda agriculture production. In:Sustainable Management of Invasive Pests in Africa. Berlin:Springer;2020. 139-57. [CrossRef]
11. Thakur N, Tomar P, Kaur J, Kaur S, Sharma A, Jhamta S, et al. Eco-friendly management of Spodoptera litura (Lepidoptera:Noctuidae) in tomato under polyhouse and field conditions using Heterorhabditis bacteriophora Poinar, their associated bacteria (Photorhabdus luminescens), and Bacillus thuringiensis var. kurstaki. Egypt J Biol Pest Control 2023;33:7. [CrossRef]
12. Waterhouse D, Norris K. Spodoptera litura (Fabricius). In:Biological Control:Pacific Prospects. Canberra:Australian Centre for International Agricultural Research;1987. 250-9.
13. Yadav SK, Patel S. Bioactivity of some plant extracts against larvae of Spodoptera litura (Fab.) and Athalia proxima lugens(Klug.) under laboratory conditions. J Entomol Zool Stud 2017;5:1430-3.
14. Chari MS, Ramaprasad G. Integrated Control of Pests on Tobacco. In:Integrated Pest Control- Progress and Perspectives. Proceedings National Symposium Association for Advancement of Entomology, Trivandrum, India;1987. 42-52.
15. Kulkarni K. Bio Ecology and Management of Spodoptera litura (F.) (Lepidoptera:Noctuidae) on Groundnut (Arachis hypogaea L.). Ph. D. Thesis;1989.
16. Rao MS, Manimanjari D, Rao AC, Swathi P, Maheswari M. Effect of climate change on Spodoptera litura Fab. on peanut:A life table approach. Crop Prot 2014;66:98-106. [CrossRef]
17. Cordero RJ, Bloomquist JR, Kuhar TP. Susceptibility of two diamondback moth parasitoids, Diadegma insulare (Cresson) (Hymenoptera;Ichneumonidae) and Oomyzus sokolowskii (Kurdjumov) (Hymenoptera;Eulophidae), to selected commercial insecticides. Bio Control 2007;42:48-54. [CrossRef]
18. Brück E, Elbert A, Fischer R, Krueger S, Kühnhold J, Klueken AM, et al. Movento an innovative ambimobile insecticide for sucking insect pest control in agriculture:Biological profile and field performance. Crop Prot 2009;28:838-44. [CrossRef]
19. Denholm I, Rowland MW. Tactics for managing pesticide resistance in arthropods:Theory and practice. Annu Rev Entomol 1992;37:91-112. [CrossRef]
20. Ahmad M, Arif MI, Ahmad M. Occurrence of insecticide resistance in field populations of Spodoptera litura (Lepidoptera:Noctuidae) in Pakistan. Crop Prot 2007;26:809-17. [CrossRef]
21. Saxena H, Ahmad R. Field evaluation of Beauveria bassiana (Balsamo) Vuillemin against Helicoverpa armigera (Hubner) infecting chickpea. J Bio Control 1997;11:93-6.
22. Cuthbertson A, Murchie A. European red spider mite-an environmental consequence of persistent chemical pesticide application. Int J Environ Sci Technol 2005;2:287-90. [CrossRef]
23. Ahmad M, Mehmood R. Monitoring of resistance to new chemistry insecticides in Spodoptera litura(Lepidoptera:Noctuidae) in Pakistan. J Econ Entomol2015;108:1279-88. [CrossRef]
24. Murtaza G, Ramzan M, Ghani MU, Munawar N, Majeed M, Perveen A, et al. Effectiveness of different traps for monitoring sucking and chewing insect pests of crops. Egypt Acad J Biol Sci2019;12:15-21. [CrossRef]
25. Revathi K, Chandrasekaran R, Thanigaivel A, Arunachalam Kirubakaran S, Senthil-Nathan S. Biocontrol efficacy of protoplast fusants between Bacillus thuringiensis and Bacillus subtilis against Spodoptera litura Fabr. Arch Phytopathol Pflanzenschutz 2014;47:1365-75. [CrossRef]
26. Chaudhari C, Chandele A, Pokharkar D, Dethe M, Firake D. Pathogenicity of different isolates of entomopathogenic fungus, Nomuraea rileyi (Farlow) Samson against tobacco caterpillar, Spodoptera litura (Fabricius). Proc Natl Acad SciIndia BBio Sci 2016;86:1001-7. [CrossRef]
27. Yang F, González JC, Williams J, Cook DC, Gilreath RT, Kerns DL. Occurrence and ear damage of Helicoverpa zea on transgenic Bacillus thuringiensis maize in the field in Texas, US and its susceptibility to Vip3A protein. Toxins 2019;11:2-15. [CrossRef]
28. Thakur N, Tomar P, Kaur S, Jhamta S, Thakur R, Yadav AN. Entomopathogenic soil microbes for sustainable crop protection. In:Soil Microbiomes for Sustainable Agriculture:Functional Annotation. Berlin:Springer;2021. 529-71. [CrossRef]
29. Tomar P, Thakur N, Yadav AN. Endosymbiotic microbes from entomopathogenic nematode (EPNs) and their applications as biocontrol agents for agro-environmental sustainability. Egypt J Biol Pest Control 2022;32:80. [CrossRef]
30. Sahayaraj K, Subash N, Allingham RW, Kumar V, Avery PB, Mehra LK, et al. Lethal and sublethal effects of three microbial biocontrol agents on Spodoptera litura and its natural predator Rhynocoris kumarii. Insects 2018;9:2-14. [CrossRef]
31. Thakur N, Kaur S, Tomar P, Thakur S, Yadav AN. Microbial biopesticides:Current status and advancement for sustainable agriculture and environment. In:New and Future Developments in Microbial Biotechnology and Bioengineering. Netherlands:Elsevier;2020. 243-82. [CrossRef]
32. Tomar P, Thakur N. Biocidal potential of indigenous isolates of entomopathogenic nematodes (EPNs) against tobacco cutworm, Spodoptera lituraFabricius (Lepidoptera:Noctuidae). Egypt J Biol Pest Control 2022;32:107. [CrossRef]
33. Abbott WS. A method of computing the effectiveness of an insecticide. J Econ Entomol 1925;18:265-7. [CrossRef]
34. Malarvannan S, Murali P, Shanthakumar S, Prabavathy V, Nair S. Laboratory evaluation of the entomopathogenic fungi, Beauveria bassiana against the Tobacco caterpillar, Spodoptera litura Fabricius (Noctuidae:Lepidoptera). J Biopestic2010;3:126-31.
35. Kaur S, Kaur HP, Kaur K, Kaur A. Effect of different concentrations of Beauveria bassiana on development and reproductive potential of Spodoptera litura (Fabricius). J Biopestic2011;4:161-8.
36. Freed S, Saleem MA, Khan M, Naeem M. Prevalence and effectiveness of Metarhizium anisopliaeagainst Spodoptera exigua (Lepidoptera:Noctuidae) in southern Punjab, Pakistan. Pak J Zool 2012;44:753-8.
37. Agrawal S, Simon S. Efficacy of Beuveria bassiana on different larval instars of tobacco caterpillar (Spodoptera lituraFab). Int J Curr Microbiol Appl Sci 2017;6:1992-6. [CrossRef]
38. Narvekar P, Mehendale S, Golvankar G, Karmarkar M, Desai S. Comparative biology of Spodoptera litura (Fab.) on different host plants under laboratory condition. Int J Chem Stud 2018;6:65-9.
39. Huang S, Li X, Li G, Jin D. Effect of Bacillus thuringiensis CAB109 on the growth, development, and generation mortality of Spodoptera exigua (Hübner) (Lepidoptera:Noctuidea). Egypt J Biol Pest Control 2018;28:2-15. [CrossRef]