1. INTRODUCTION
Dragon fruit is a lucrative crop in Vietnam, occupying 40,000 hectares of land and generating approximately $895 million annually [1]. However, the expansion of planting areas and the increasing demand for dragon fruit make these crops more vulnerable to pests and diseases [2,3]. The reduction in dragon fruit yields due to various diseases, particularly those caused by fungi, is a significant concern for dragon fruit plantations globally, as reported in previous studies [3,4]. It has been indicated that anthracnose on dragon fruit in Malaysia is attributed to Colletotrichum gloeosporioides [4]. In addition, it has been reported that Gilbertella persicaria has been identified as the etiological agent responsible for the occurrence of bloom rot in red-fleshed dragon fruit in Thailand [5].
Neoscytalidium dimidiatum is well-known for causing dragon fruit brown spot disease, which affects the plant’s stems, fruits, and flowers, generating brown patches and lesions that can lead to fruit rot and ultimately plant death [6]. On the stems, fruits, and flowers, dragon fruit brown spot disease manifests as small, irregularly shaped brown patches or lesions [7]. The blemishes can expand and combine, causing the damaged tissue to turn dark and finally rot. Fruits with a severe infection may develop cracks and become mushy and watery [8,9]. In Vietnam, brown spot disease is often observed in the rainy season when the weather is warm and humid [3]. The disease can cause significant yield losses and reduce fruit quality, resulting in economic losses for farmers [3]. The disease can be spread by contaminated planting materials, pruning tools, and wind-blown spores, and can be favored by poor plant hygiene practices [3]. However, the identification and management of pathogenic fungal strains on dragon fruit in Vietnam are still rarely documented.
Currently, the management of stem brown spot disease is predominantly dependent on the application of synthetic chemical fungicides. The frequent application of fungicides, which exhibit efficacy against fungicide-resistant strains of the pathogen, may pose a considerable hazard to human health and the environment, owing to their elevated levels of toxicity [10–12]. The potential of Trichoderma spp. as biocontrol agent against various plant fungal pathogens is widely acknowledged [13]. Trichoderma spp. inhibit fungal pathogens by competing for space, nutrition, and other resources, hence lowering the growth and survival of pathogens [14]. In addition, Trichoderma spp. are able to generate secondary metabolites, such as antibiotics and enzymes, that suppress the growth of pathogen fungus [15–17]. In addition, Trichoderma spp. might parasitize other fungi by growing on and penetrating their hyphae, resulting in inhibiting their growing [18]. Numerous investigations have exhibited the effectiveness of Trichoderma spp. in combating an extensive broad range of plant-associated fungi, encompassing Fusarium sp. [19], Rhizoctonia sp. [20], Phytophthora sp. [21], and Sclerotia sp. [22]. The utilization of Trichoderma spp. as biological control agent presents a viable and ecologically sound substitute to chemical pesticides [23]. The utilization of Trichoderma spp. in integrated pest management strategies offers cultivators the opportunity to reduce their reliance on chemical fungicides while minimizing the risk of developing fungal resistance strains [24].
The aim of this investigation was to achieve the following: (1) determination of the etiological agent accountable for brown spot disease on dragon fruit in Long An, Vietnam, and (2) assessment of Trichoderma spp. for their antagonistic attributes against the aforementioned pathogen responsible for the brown spot disease on dragon fruit.
2. MATERIALS AND METHODS
2.1. Isolation and Identification of the Pathogen Causing Brown Spot Disease in Dragon Fruit
A total of ten stem canker samples displaying symptoms of brown spot disease were gathered from a dragon fruit plantation located in the province of Long An, Vietnam. The specimens were preserved in a plastic container with ice during transit to the laboratory. The identification of fungal pathogens was conducted through tissue transplanting, following the methodology outlined by Pornsuriya et al. [25]. Tissue fragments measuring 2–3 mm, which was found to be infected, underwent a process of disinfection using a 0.5% solution of sodium hypochlorite. Following this, the fragments were rinsed and subsequently placed on water agar, then incubated at a temperature of 28°C for a period of 72 h [26]. Subsequently, the hyphal tips were transplanted onto a potato dextrose agar (PDA) medium to facilitate additional isolation. The utilization of Koch›s postulate was implemented in the experimentation process, wherein the inoculation of healthy dragon fruit leaves with isolated fungi was conducted to examine the potential causal relationship between the fungi and the manifestation of brown spot disease symptoms [27].
2.2. Morphology Study
The fungal isolate obtained from the second isolation purification was subjected to further analysis. A 5 mm mycelial disk was inoculated on PDA and kept under observation for daily monitoring. The cultivation took place at a temperature of 28°C and a relative humidity of 50–60% for 7 days. During this time, the colony color, radial expansion, and presence of conidia were recorded. To conduct a microscopic examination of the fungal isolate, mycelia were aseptically retrieved utilizing a sterile needle and subsequently deposited into a droplet of lactophenol cotton blue. The hyphal texture, phialides, and conidial shape were examined under a Nikon Eclipse E100 microscope at a magnification of ×1000. The morphological characteristics were identified through the utilization of descriptions provided by Watanabe (2010) and Humber (2012) [28,29].
2.3. Molecular Identification of Fungal Isolate
The fungal specimen underwent molecular identification through the utilization of universal internal transcribed spacer (ITS) primers and the “ITS” region. The ITS primers used were: ITS1 (5′-TCCGTAGGTGAACCTGCGG-3′) and ITS4 (5′-TCCTCCGCTTATTGATATGC-3′) [30], nuclear large subunit (LSU) primers were: LROR (5′-ACCCGCTGAACTTAAGC-3′) and LR5 (5′-TCCTGAGGGAAACTTCG-3′) [31], β-tubulin (tub) primers were: Bt1a (5′-TTCCC CGTCTCCACTTCTTCATG-3′) and Bt1b (5′-GACGAGATCGTTCATGTTGAACTC-3′) [32], translation elongation factor 1-α gene (TEF1α) primers were EF1F (5′-ATGGGTAAGGARGACAAGAC-3′) and EF1R (5′-GGARGTACCAGTSATCATGTT-3′) [33]. The T100 thermal cycler (Bio-Rad, Irvine, CA, USA) was utilized to conduct the polymerase chain reaction (PCR) reaction under the following cycling conditions: an initial denaturation at 95°C for 5 min, followed by 35 cycles of denaturation at 95°C for 1 min, annealing at 55°C for 1 min, extension at 72°C for 1 min, and a final extension at 72°C for 10 min. The PCR products were visualized by means of agarose gel electrophoresis, utilizing a 1% agarose gel that was stained with GelRed Loading Buffer (TBR, Viet Nam) and subsequently examined under UV light. The PCR amplicons were sequenced by Nam Khoa Biotek, Viet Nam, and the resulting sequences were analyzed for similarity using the Nucleotide Basic Local Alignment Search Tool (BLASTn) algorithm provided by the National Center for Biotechnology Information by comparing them to the GenBank database. The ITS-5.8S rDNA sequence was utilized for phylogenetic analysis through the implementation of the Clustal-W function and neighbor-joining method using MEGA 6.0 [34].
2.4. In Vitro Assessment of the Antagonistic Potential of Trichoderma spp. against N. dimidiatum
A dual culture assay was proposed by Sivan and Chet to assess the inhibitory effect of Trichoderma spp. isolates on the mycelial growth of N. dimidiatum [35]. A 5 mm disc derived from a 7-day-old culture of N. dimidiatum was positioned on the left side of a Petri dish with a diameter of 90 mm. A 5 mm disc derived from each isolate of Trichoderma sp. was placed at a distance of 3 cm on the opposite side of the dish, then incubated at 28°C for 7 days. The group consisting only of N. dimidiatum under cultivation was designated as the control group. The assessment of pathogen growth inhibition was conducted for 7 days and quantified as a percentage utilizing the prescribed formula:
GI (%) = (C − T)/C × 100
In this formula, GI represents the percentage of growth inhibition, C denotes the radial growth of the pathogen in the control, and T represents the radial growth of the pathogen in dual culture with the antagonist.
Following a 7-day of incubation, the region where isolates of Trichoderma spp. and N. dimidiatum interacted was carefully collected from the Petri dishes. The collected samples were then stained with lacto phenol cotton blue and observed under a light microscope at a magnification of ×1000 to investigate the interaction between the Trichoderma spp. and N. dimidiatum, with a particular focus on determining whether Trichoderma spp. came into contact with the hyphae of N. dimidiatum and formed coilings around them.
2.5. Effectiveness of Trichoderma spp. in Lowering Dragon Fruit Brown Spot Disease in the Pot Experiment
The study utilized a randomized complete block design that consisted of three treatments: pre-treatment, co-treatment, and post-treatment, each replicated three times, and each replication containing 10 dragon fruit plants [36,37]. For the pre-treatment, fungicides propineb 2.5% (according to the recommendation from the manufacture) or Trichoderma spp. was applied to the soil at a rate of 1 × 106 conidia/g for 3 days before N. dimidiatum inoculation at a rate of 1 × 104 conidia/g per pot. The co-treatment involved the simultaneous addition of propineb 2.5% or Trichoderma spp. to the soil at a rate of 1 × 106 conidia/g and N. dimidiatum inoculation at a rate of 1 × 104 conidia/g soil. In the post-treatment, the dragon fruit plants were first infected with N. dimidiatum at a rate of 1 × 104 conidia/g soil for 3 days before the addition of propineb 2.5% or Trichoderma spp. at a rate of 1 × 106 conidia/g per pot. The control group consisted of dragon fruit plants infected with N. dimidiatum at a rate of 1 × 104 conidia/g soil without any treatment with Trichoderma spp. The assessment of disease severity was conducted through the computation of the percentage of brown spot disease present on the dragon fruit plants, followed by the assignment of a severity rating on a scale ranging from 1 to 5, after a period of 7 and 14 days. The disease severity was assessed on a scale of 1–5, where a score of 1 represented the absence of symptoms and a score of 5 represented the presence of severe symptoms. The categorization of disease severity was established based on the percentage of disease symptoms observed. The levels were classified as no disease (<1%), level 1 (1–10%), level 2 (11–20%), level 3 (21–30%), level 4 (31–40%), and level 5 (>41%) [37].
The disease index and its corresponding control efficacy were documented using the formula [38]:
 |
3. RESULTS
3.1. Isolation and Identification of the Fungus Responsible for Dragon Fruit Brown Spot Disease
Fungal strains were obtained from branches of dragon fruit affected by brown spot disease in Duc Hoa district, Long An province, Viet Nam. The stems of the dragon fruit exhibited sunken brown spots, which indicated the presence of the brown spot disease [Figure 1a]. Tissue samples with spots were collected and isolated on PDA medium for further investigation. After 10 days of cultivation, the colonies changed in color from white and cottony to yellowish-brown or dark brown, and the colony surface became rough or woolly [Figure 1b]. The conidia observed were unicellular, cylindrical, slightly curved, egg-shaped, or elliptical [Figure 1c]. The results of the BLAST analysis indicated that the fungal strain in question exhibited complete homology with N. dimidiatum. The phylogenetic tree constructed using ITS rDNA indicated that the fungal strain under investigation and N. dimidiatum are closely related, as they were found to be on the same branch. In contrast, the other Neoscytalidium species displayed a greater genetic distance from them, as depicted in Figure 1d. Moreover, the DNA sequence of the fungal strain was utilized to construct a maximum likelihood (ML) tree, employing the tub [Figure 1e] and LSU [Figure 1f] regions. The results demonstrated that the fungal strain clustered within the same clade as N. dimidiatum, aligning with the phylogenetic tree based on the ITS region. The pathogenicity of the fungal strain was confirmed using Koch’s postulates, where incubating healthy dragon fruit leaves with N. dimidiatum conidia resulted in brown spot disease. The re-isolated fungus has a comparable morphology to the previously isolated N. dimidiatum.
 | Figure 1: Characterization of Neoscytalidium dimidiatum isolated from brown spot disease-infected dragon fruit. (a) Dragon fruit branches with brown spot disease. (b) Macroscopic morphology of N. dimidiatum isolates in potato dextrose agar culture medium. (c) N. dimidiatum conidia under microscope at ×1000 magnification. (d) Phylogenetic tree constructed from the internal transcribed spacer gene sequences, (e) tub gene sequences, and (f) large subunit gene sequences of N. dimidiatum in this study and other known N. dimidiatum spp. strains from the GenBank database. In this investigation, N. dimidiatum was highlighted in bold. [Click here to view] |
3.2. Screening of Antagonist Trichoderma spp. Strain against N. dimidiatum in Vitro
Fifteen isolates of Trichoderma spp. were procured from diverse plantations, comprising 5 isolates from soil samples of dragon fruit (TDF), 5 from pepper plantations (TP), and 5 from cocoa soils (TC). In vitro, the dual culture assay was employed to evaluate the antagonistic activity of various strains of Trichoderma spp. against N. dimidiatum, as presented in Table 1. Our findings indicated that the Trichoderma spp. exhibited discernible antagonistic properties against N. dimidiatum, as manifested by the expansion and colonization of the pathogen’s mycelium.
Table 1: Effectiveness of antagonistic Trichoderma spp. against Neoscytalidium dimidiatum in dual culture.
| Sample code | Sampling material | Sampling location | % inhibition | ||
|---|---|---|---|---|---|
| 2 days | 5 days | 7 days | |||
| TDF1 | Dragon fruit rhizosphere | Long An province, Viet Nam | 5.41de | 35.79c | 54.74d |
| TDF2 | 19.94a | 57.28ab | 88.35a | ||
| TDF3 | 15.84ab | 37.97c | 56.53d | ||
| TDF4 | 7.24c-e | 36.51c | 58.06cd | ||
| TDF5 | 15.05ab | 65.64a | 86.65ab | ||
| TP1 | Pepper rhizosphere | Dak Lak Province, Viet Nam | 10.15b-d | 36.15c | 55.05d |
| TP2 | 4.13e | 35.79c | 52.27d | ||
| TP3 | 19.19ab | 41.31c | 65.61cd | ||
| TP4 | 5.41de | 34.53c | 57.93d | ||
| TP5 | 17.58ab | 37.23 c | 58.91d | ||
| TC1 | Cocoa rhizosphere | Dak Lak Province, Viet Nam | 11.99a-c | 61.13ab | 85.05a |
| TC2 | 1.65e | 35.24c | 57.93cd | ||
| TC3 | 14.90ab | 50.45a-c | 69.45b-d | ||
| TC4 | 13.91a-d | 38.70bc | 67.05cd | ||
| TC5 | 14.76ab | 39.62c | 75.46a-c | ||
Statistically significant differences between groups (P<0.05) are denoted by different lowercase letters (a-e).
The discontinuation of N. dimidiatum hyphae growth was observed after 5 days of co-culture, whereas the Trichoderma spp. exhibited continued growth until complete coverage of the pathogen colony and the entire plate was achieved [Figure 2a]. The efficacy of Trichoderma spp isolates fight against N. dimidiatum was recorded to as 52.27–88.35% after 7 days of co-incubation, with TDF2, TDF5, and TC1 isolates showing the highest efficacy at 88.35%, 86.65%, and 85.05%, respectively [Table 1]. The antagonistic phenomenon of Trichoderma spp. was further studied by microscopy observations, which revealed an interaction between Trichoderma spp. and N. dimidiatum. Trichoderma spp. isolates produced hyphal branches that were directed toward N. dimidiatum colonies. The hyphae of Trichoderma TDF2, TDF5, and TC1 came into contact with and formed coilings with the hyphae of N. dimidiatum, ultimately leading to the inhibition of the growth of N. dimidiatum hyphae [Figure 2b-d]. Three Trichoderma strains (TDF2, TDF5, and TC1) were chosen for further experimentation based on their high efficacy in antagonism, as indicated by the results of the in vitro experiments.
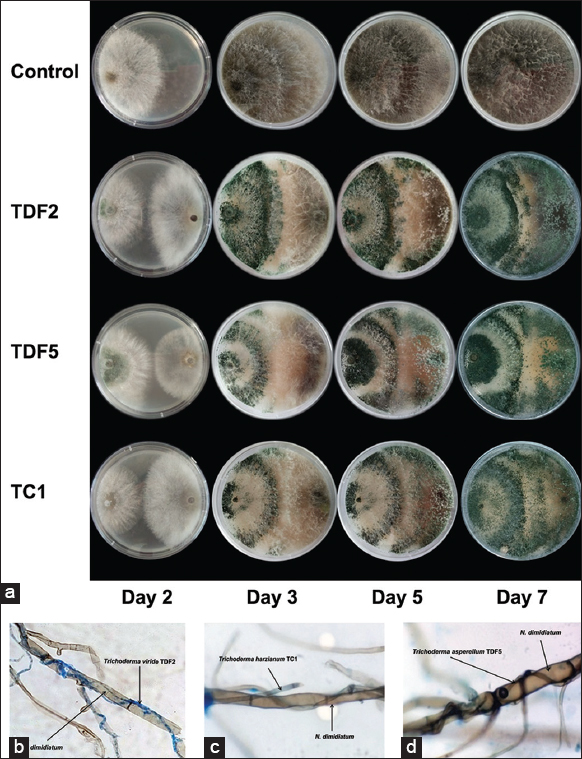 | Figure 2: (a) Representative photos of three antagonistic Trichoderma spp. against Neoscytalidium dimidiatum in dual culture. Representative photos of the interaction between TDF2. (b) TDF5 (c), and TC1 (d), hyphae with Neoscytalidium dimidiatum. [Click here to view] |
3.3. Molecular Identification of Isolated Fungi
The genomic DNA of three isolates of Trichoderma spp., which demonstrated antagonistic activity against the brown spot pathogen N. dimidiatum, was amplified using the PCR method with an estimated size of 600 base pairs. Following this, the amplified DNA fragments underwent sequencing and were subsequently subjected to homology comparison using the BLAST. The ITS gene regions of both the isolates and reference sequences sourced from GenBank were utilized to develop phylogenetic trees. The findings obtained from the analysis of sequencing and homology comparison indicate that Trichoderma isolates TDF2, TDF5, and TC1 pertain to Trichoderma viride, Trichoderma asperellum, and Trichoderma harzianum, correspondingly. The construction of a phylogenetic tree utilizing both the isolate strains and reference strains provided confirmation of the proximity between the two groups [Figure 3a]. To enhance the reliability of the molecular identification process, additional genetic regions, namely tub [Figure 3b] and TEF1α [Figure 3c] regions were examined. The analysis of these regions yielded consistent results that aligned with the initial findings based on the ITS rDNA analysis.
 | Figure 3: Phylogenetic tree constructed from the internal transcribed spacer gene sequences (a), translation elongation factor 1α gene sequences (b), and tub gene sequences (c) of Trichoderma spp. in this study and other known Trichoderma spp. strains from the GenBank database. In this investigation, Trichoderma isolates was highlighted in bold. [Click here to view] |
3.4. Antagonistic Activity of Trichoderma spp against Brown Spot Disease Causes by N. dimidiatum in Pot Experiment
The activity of isolated strains of Trichoderma spp. against the brown spot pathogen N. dimidiatum was investigated under different incubation conditions. The establishment pre-treatment involved applying Trichoderma spp. strains to the base of dragon fruit trees 3 days before N. dimidiatum infection, while in the co-treatment groups, dragon fruit plants were co-fertilized with Trichoderma spp. and N. dimidiatum. Finally, during the post-treatment groups, dragon fruit plants were infected with N. dimidiatum for 3 days and then treated with Trichoderma spp. The infection group showed a disease index of 4.01–6.41 after 7–14 days, indicating the presence of brown spot disease on dragon fruit branches [Figure 4]. The highest reduction in the disease index was observed in the Trichoderma spp. pre-treatment group [Figure 4], with a relative control effect of 69.53–72.47% [Table 2]. This pre-treatment outperformed the propineb pre-treatment group, which had a relative control effect of 66.28% [Table 2]. Under the co-treatment conditions, the disease index reduction was approximately 51.46–82.23% in the Trichoderma spp.-treated groups and 83.75% in the propineb-treated group [Table 2]. However, Trichoderma spp. co-treatment groups were less effective than the pre-treatment groups [Figure 4 and Table 2]. Post-treatment with Trichoderma spp. did not show obvious antifungal activity against N. dimidiatum, with a relative control effect of 18.69–34.11%, while propineb showed a relative control effect of 63.13% [Table 2]. These findings suggest that the pre-incubation treatment of Trichoderma spp. is a highly effective strategy for controlling brown spot disease in dragon fruit. However, further studies are needed to determine the underlying mechanisms of Trichoderma spp. antifungal activity and optimize the treatment protocols for better disease management.
 | Figure 4: Effectiveness of Trichoderma isolates in the management of brown spot disease in dragon fruit caused by Neoscytalidium dimidiatum infection in pot experiment. Data are presented as the means of triplicate analysis ± standard deviation. *Indicate significant differences compared with the infection group. [Click here to view] |
Table 2: Relative control effect of antagonistic Trichoderma spp. against brown spot disease in dragon fruit caused Neoscytalidium dimidiatum infection.
| Treatment | Relative control effect | |
|---|---|---|
| 7 days | 14 days | |
| Pre-treatment | ||
| Propineb | 34.21b-d | 66.28a-c |
| TDF2 | 69.29ab | 75.71a |
| TDF5 | 56.29abc | 71.98ab |
| TC1 | 64.39abc | 70.25a-c |
| Co-treatment | ||
| Propineb | 77.96a | 77.89a |
| TDF2 | 50.99a-d | 45.23de |
| TDF5 | 43.67b-e | 49.13d |
| TC1 | 38.09c-e | 60.99cd |
| Post-treatment | ||
| Propineb | 45.98b-d | 63.13b-d |
| TDF2 | 19.11e | 19.15ef |
| TDF5 | 0.67e | 4.75f |
| TC1 | 15.42de | 7.29f |
Statistically significant differences between groups (P<0.05) are denoted by different lowercase letters (a-e).
4. DISCUSSION
The cultivation of dragon fruit is significantly challenged by the brown spot disease caused by N. dimidiatum, which leads to compromised yield and quality [39]. Our investigation has effectively isolated and identified N. dimidiatum as the etiological factor responsible for the brown pot disease of dragon fruit in Long An province, Vietnam. Cultural identification of the fungus was based on its characteristic features, such as dark brown to black colonies with a velvet-like texture on PDA medium [8]. These characteristics are consistent with previous reports of N. dimidiatum in dragon fruit [8]. Molecular identification was based on the ITS region of the fungal genome, which is a widely used DNA barcode for fungal identification. The ITS region is highly variable among different fungal species, making it a useful tool for species-level identification [40]. Our results showed that the ITS sequences obtained from the isolated fungal cultures had a high sequence identity with previously reported sequences of N. dimidiatum.
Several measures can be implemented to manage the N. dimidiatum-related disease in dragon fruit cultivation, such as removing infected leaves and controlling humidity, which has proven to be effective [3]. Whilst fungicides have demonstrated potential in managing brown spot disease, their application can yield detrimental consequences for both the environment and human well-being and may facilitate the emergence of fungus-resistant strains [41]. The filamentous fungi belonging to the Trichoderma spp. have been the subject of considerable scientific research due to their potential as a biocontrol agent against various plant pathogens [42]. Trichoderma spp. exhibit various mechanisms to inhibit plant pathogens, such as nutrient and space competition, antibiosis synthesis, and induction of systemic resistance in plants [16,43,44]. The perceived relationship between the biocontrol agent and the pathogen is often characterized as antagonistic due to their direct interaction. Consequently, the present investigation employed a dual culture assay to evaluate the isolates of Trichoderma spp. with regard to their antagonistic impact on N. dimidiatum pathogens. Among 15 isolates of Trichoderma spp. tested, T. viride (TDF2), T. asperellum (TDF5), and T. harzianum (TC1) showed significant antagonistic activity against N. dimidiatum. Furthermore, our study indicated the Trichoderma spp.’s mycoparasitic mechanism [45], which involves physically interacting with N. dimidiatum. Specifically, the hyphae of Trichoderma TDF2, TDF5, and TC1 coiled around the hyphae of N. dimidiatum, potentially restricting its growth and limiting its ability to infect plants. The results indicate that Trichoderma exhibits significant promise as a biocontrol agent for combating plant pathogens and can serve as a crucial component of sustainable agriculture.
After conducting an in vitro screening of Trichoderma spp. that showed potential as antagonists against N. dimidiatum, a potted evaluation was performed to evaluate the effectiveness of Trichoderma spp. in protecting dragon fruit from brown spot disease caused by N. dimidiatum. These findings suggest that Trichoderma spp. displays significant potential as a biocontrol agent for the mitigation of brown spot disease in dragon fruit plants. The results of the study indicate that pre-treatment with Trichoderma spp. proved to be the most efficient method, as it facilitated the establishment of competitive advantage of Trichoderma spp. over the pathogen in the soil [46], resulting in the high relative control effect of TDF2, TDF5, and TC1 on the damping-off of brown pot disease in dragon fruit leaves reached 75.71%, 71.98%, and 70.25%, respectively, much higher control efficiency compared to propineb (66.28%). According to prior research, T. asperellum, T. spirale, T. koningiopsis, and T. reesei have been identified as potential agents for combating Rigidoporus microporus, the causative agent of white root rot disease (WRD) in Hevea brasiliensis, which lower the disease index by 70% [46]. The findings of this study suggest that the isolates of Trichoderma spp. that were selected for their antagonistic properties exhibit robust disease control capabilities and hold significant promise for employment in biocontrol measures, particularly in the context of countering N. dimidiatum pathogens.
Co-treatment of Trichoderma spp. and N. dimidiatum also showed potential in reducing brown spot disease index. However, the protective effect of TDF2 and TDF5 against N. dimidiatum was decreased compared to pre-treatment, suggesting that the timing of Trichoderma spp. application might be critical for the management of N. dimidiatum infection. Additionally, post-treatment with Trichoderma was found to be less effective in reducing N. dimidiatum infection in dragon fruit. Previous research has demonstrated that the timing of Trichoderma spp. application is crucial for protecting grapevine wounds. Specifically, early application within 5–6 h after pruning has been shown to be more effective in preventing pathogen infection, while the effectiveness significantly decreases after 48 h post-pruning [47]. These findings underscore the importance of early intervention for preventing brown spot disease development cause by N. dimidiatum infection.
5. CONCLUSION
To summarize, the cultivation of dragon fruit is considerably impeded by the brown spot disease instigated by N. dimidiatum, which results in reduced yield and quality, thereby culminating in economic setbacks for cultivators. The present investigation effectively determined N. dimidiatum as the etiological factor responsible for the brown spot ailment on dragon fruit in Long An, Viet Nam. Furthermore, we demonstrated the potential of Trichoderma spp. as a biocontrol agent in mitigating the growth of N. dimidiatum. Our results indicated that Trichoderma spp. displays considerable potential in managing brown spot disease in dragon fruit, thus making a valuable contribution to the progress of sustainable agriculture. The optimal timing of Trichoderma spp. application is of paramount importance, and prompt intervention is imperative to avert the onset of disease. The aforementioned discoveries possess significant ramifications for the administration of brown spot ailment in dragon fruit farming and have the potential to aid in the establishment of sustainable and environmentally conscious agricultural methodologies.
6. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to the conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
7. FUNDING
There is no funding to report.
8. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
9. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
10. DATA AVAILABILITY
All data generated or analyzed during this study are included in this manuscript.
11. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Paull RE, Chen NJ. Overall Dragon Fruit Production and Global Marketing. Taiwan:FFTC;2019.
2. Van Hoa N, Hieu NT, Hanh TT, Uyen DT, Dien LQ. Emerging infectious diseases and insect pests of dragon fruit, passionfruit, citrus, longan. Management 2016;3:87-100.
3. Hieu NT, Hoa N. Management strategies of major pitaya diseases in Vietnam. Management 2015;1:87-100.
4. Mohd MH, Salleh B, Zakaria L. Identification and molecular characterizations of Neoscytalidium dimidiatum causing stem canker of red-fleshed dragon fruit (Hylocereus polyrhizus) in Malaysia. J Phytopathol 2013;161:841-9. [CrossRef]
5. Ruangwong OU, Kunasakdakul K, Wonglom P, Dy KS, Sunpapao A. Morphological and molecular studies of a rare mucoralean species causing flower rot in Hylocereus polyrhizus. J Phytopathol 2022;170:214-20. [CrossRef]
6. Al Raish SM, Saeed EE, Sham A, Alblooshi K, El-Tarabily KA, AbuQamar SF. Molecular characterization and disease control of stem canker on royal poinciana (Delonix regia) caused by Neoscytalidium dimidiatum in the United Arab Emirates. Int J Mol Sci 2020;21:1033. [CrossRef]
7. Lan GB, He ZF, Xi PG, Jiang ZD. First report of brown spot disease caused by Neoscytalidium dimidiatum on Hylocereus undatus in Guangdong, Chinese Mainland. Plant Dis 2012;96:1702. [CrossRef]
8. Dy KS, Wonglom P, Pornsuriya C, Sunpapao A. Morphological, molecular identification and pathogenicity of Neoscytalidium dimidiatum causing stem canker of Hylocereus polyrhizus in southern Thailand. Plants (Basel) 2022;11:504. [CrossRef]
9. Zakaria L. Fungal and oomycete diseases of minor tropical fruit crops. Horticulturae 2022;8:323. [CrossRef]
10. Yang LN, He MH, Ouyang HB, Zhu W, Pan ZC, Sui QJ, et al. Cross-resistance of the pathogenic fungus Alternaria alternata to fungicides with different modes of action. BMC Microbiol 2019;19:205. [CrossRef]
11. Zubrod JP, Bundschuh M, Arts G, Bru?hl CA, Imfeld G, Kna?bel A, et al. Fungicides:An overlooked pesticide class?Environ Sci Technol 2019;53:3347-65. [CrossRef]
12. Steinberg G, Gurr SJ. Fungi, fungicide discovery and global food security. Fungal Genet Biol 2020;144:103476. [CrossRef]
13. Ty?kiewicz R, Nowak A, Ozimek E, Jaroszuk-?cise?J. Trichoderma:The current status of its application in agriculture for the biocontrol of fungal phytopathogens and stimulation of plant growth. Int J Mol Sci 2022;23:2329. [CrossRef]
14. Pandey V, Shukla A, Kumar J. Physiological and molecular signalling involved in disease management through Trichoderma:An effective biocontrol paradigm. In:Current Trends in Plant Disease Diagnostics and Management Practices. Berlin:Springer;2016. 317-46. [CrossRef]
15. Rajani P, Rajasekaran C, Vasanthakumari M, Olsson SB, Ravikanth G, Shaanker RU. Inhibition of plant pathogenic fungi by endophytic Trichoderma spp. through mycoparasitism and volatile organic compounds. Microbiol Res 2021;242:126595. [CrossRef]
16. Meng X, Miao Y, Liu Q, Ma L, Guo K, Liu D, et al. TgSWO from Trichoderma guizhouense NJAU4742 promotes growth in cucumber plants by modifying the root morphology and the cell wall architecture. Microb Cell Fact 2019;18:148. [CrossRef]
17. Silva JC, Salgado JC, Vici AC, Ward RJ, Polizeli ML, Guimarães LH, et al. A novel Trichoderma reesei mutant RP698 with enhanced cellulase production. Braz J Microb 2020;51:537-45. [CrossRef]
18. Poveda J. Trichoderma as biocontrol agent against pests:New uses for a mycoparasite. Biol Control 2021;159:104634. [CrossRef]
19. Chen J, Zhou L, Din IU, Arafat Y, Li Q, Wang J, et al. Antagonistic activity of Trichoderma spp. against Fusarium oxysporum in rhizosphere of Radix pseudostellariae triggers the expression of host defense genes and improves its growth under long-term monoculture system. Front Microbiol 2021;12:579920. [CrossRef]
20. Abbas A, Mubeen M, Zheng H, Sohail MA, Shakeel Q, Solanki MK, et al. Trichoderma spp. Genes involved in the biocontrol activity against Rhizoctonia solani. Front Microbiol 2022;13:884469. [CrossRef]
21. La Spada F, Stracquadanio C, Riolo M, Pane A, Cacciola SO. Trichoderma counteracts the challenge of Phytophthora nicotianae infections on tomato by modulating plant defense mechanisms and the expression of crinkler, necrosis-inducing Phytophthora protein 1, and cellulose-binding elicitor lectin pathogenic effectors. Front Plant Sci 2020;11:583539. [CrossRef]
22. Da Silva LR, Muniz PH, Peixoto GH, Luccas BE, da Silva JB, de Mello SC. Mycelial inhibition of Sclerotinia sclerotiorum by Trichoderma spp. volatile organic compounds in distinct stages of development. Pak J Biol Sci 2021;24:527-36. [CrossRef]
23. Zin NA, Badaluddin NA. Biological functions of Trichoderma spp. for agriculture applications. Ann Agric Sci 2020;65:168-78. [CrossRef]
24. Lorito M, Woo SL. Trichoderma:A multi-purpose tool for integrated pest management. In:Principles of Plant-Microbe Interactions:Microbes for Sustainable Agriculture. Berlin:Springer;2015. p. 345-53. [CrossRef]
25. Pornsuriya C, Chairin T, Thaochan N, Sunpapao A. Identification and characterization of Neopestalotiopsis fungi associated with a novel leaf fall disease of rubber trees (Hevea brasiliensis) in Thailand. J Phytopathol 2020;168:416-27. [CrossRef]
26. Daengsuwan W, Wonglom P, Sunpapao A. First report of Lasiodiplodia theobromae causing spadix rot in Anthurium andraeanum. J Phytopathol 2020;168:129-33. [CrossRef]
27. Grimes DJ. Koch's Postulates-Then and Now. Vol. 1. Washington, D.C.:Microbe-American Society for Microbiology;2006. 223-8. [CrossRef]
28. Watanabe T. Pictorial Atlas of Soil and Seed Fungi:Morphologies of Cultured Fungi and Key to Species. United States:CRC Press;2010.
29. Humber RA. Identification of entomopathogenic fungi. In:Manual of Techniques in Invertebrate Pathology. United States:Academic Press;2012. 151-87. [CrossRef]
30. Tangthirasunun N, Poeaim S, Soytong K, Sommartya P, PopoonsakS. Variation in morphology and ribosomal DNA among isolates of Metarhizium anisopliae from Thailand. J Agric Technol 2010;6:317-29.
31. Vilgalys R, Hester M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 1990;172:4238-46. [CrossRef]
32. Van der Nest A, Wingfield MJ, Ortiz PC, Barnes I. Biodiversity of Lecanosticta pine-needle blight pathogens suggests a Mesoamerican Centre of origin. IMA Fungus 2019;10:2. [CrossRef]
33. Karlsson I, Edel-Hermann V, Gautheron N, Durling MB, Kolseth AK, Steinberg C, et al. Genus-specific primers for study of Fusarium communities in field samples. Appl Environ Microbiol 2016;82:491-501. [CrossRef]
34. Thi HN, Le KY, Thien ND, Nguyen TD, Do AD. Insecticidal activity of isolated Purpureocillium lilacinum PL1 against whitefly, Bemisia tabaci Gennadius (Hemiptera:Aleyrodidae) on cassava plantations in southern Viet Nam. Egypt J Biol Pest Control 2023;33:44. [CrossRef]
35. Sivan A, Chet I. Degradation of fungal cell walls by lytic enzymes of Trichoderma harzianum. Microbiology 1989;135:675-82. [CrossRef]
36. Ghanbarzadeh B, Safaie N, Goltapeh EM, Danesh YR, Khelghatibana F. Biological control of Fusarium basal rot of onion using Trichoderma harzianum and Glomus mosseae. J Crop Prot 2016;5:359-68. [CrossRef]
37. Tuan LN, Du BD, Ha LD, Dzung LT, Van Phu D, Hien NQ. Induction of chitinase and brown spot disease resistance by oligochitosan and nanosilica-oligochitosan in dragon fruit plants. Agric Res 2019;8:184-90. [CrossRef]
38. Liu Y, He P, He P, Munir S, Ahmed A, Wu Y, et al. Potential biocontrol efficiency of Trichoderma species against oomycete pathogens. Front Microbiol 2022;13:974024. [CrossRef]
39. Xie HH, Long L, Huang S, Mao L, Huang Q, Wang L, et al. First report of black spot caused by Neoscytalidium dimidiatum on sisal in Guangxi, China. Plant Dis 2021;105:701. [CrossRef]
40. Badotti F, de Oliveira FS, Garcia CF, Vaz AB, Fonseca PL, Nahum LA, et al. Effectiveness of ITS and sub-regions as DNA barcode markers for the identification of Basidiomycota (Fungi). BMC Microbiol 2017;17:42. [CrossRef]
41. Deising HB, Reimann S, Pascholati SF. Mechanisms and significance of fungicide resistance. Braz J Microbiol 2008;39:286-95. [CrossRef]
42. Sood M, Kapoor D, Kumar V, Sheteiwy MS, Ramakrishnan M, Landi M, et al. Trichoderma:The “secrets“of a multitalented biocontrol agent. Plants 2020;9:762. [CrossRef]
43. Ahluwalia V, Kumar J, Rana VS, Sati OP, Walia S. Comparative evaluation of two Trichoderma harzianum strains for major secondary metabolite production and antifungal activity. Nat Prod Res 2015;29:914-20. [CrossRef]
44. Masi M, Nocera P, Reveglia P, Cimmino A, Evidente A. Fungal metabolites antagonists towards plant pests and human pathogens:Structure-activity relationship studies. Molecules 2018;23:834. [CrossRef]
45. Harman GE, Howell CR, Viterbo A, Chet I, Lorito M. Trichoderma species-opportunistic, avirulent plant symbionts. Nat Rev Microbiol 2004;2:43-56. [CrossRef]
46. Go WZ, Chin KL, H'ng PS, Wong MY, Lee CL, Khoo PS. Exploring the biocontrol efficacy of Trichoderma spp. against Rigidoporus microporus, the causal agent of white root rot disease in rubber trees (Hevea brasiliensis). Plants (Basel) 2023;12:1066. [CrossRef]
47. Mutawila C, Halleen F, Mostert L. Optimisation of time of application of Trichoderma biocontrol agents for protection of grapevine pruning wounds. Aust J Grape Wine Res 2016;22:279-87. [CrossRef]