1. INTRODUCTION
From the past decades, the increase and spread of drug resistance to antibiotics is much more due to their misuse. As per the data of the Global Antimicrobial Resistance and Use Surveillance System of the WHO in 2020, more than 2,800,000 cases of anti-microbial resistance (AMR) infection and 35,000 demises were recorded each year in the United States. AMR strains such as Staphylococcus aureus, Pseudomonas aeruginosa, Acinetobacter baumannii, Listeria monocytogenes, Salmonella Typhimurium, and many more are a major threat to human health, globally. Among them, due to antibiotic resistance, the human foodborne pathogen S. aureus has become resistance towards antibiotics such as aminoglycosides, macrolides, tetracycline, chloramphenicol, lincosamides and even higher class of antibiotics such as methicillin and oxacillin [1]. Certain strains of S. aureus have also been earlier responsible for normal skin infections which can also lead to severe diseases such as meningitis, pleuropulmonary, pneumonia, bacteremia, nosocomial infections endocarditis, and post-surgical wound infections. [2] In addition, these methicillin-resistant S. aureus (MRSA) strains are strong biofilm formers and are very difficult to remove. Another Gram-positive pathogen that is responsible for the contamination of foods is L. monocytogenes which can colonize the surface of gastrointestinal tracts. It mainly infect spinal cord membranes followed by the bloodstream [3]. L. monocytogenes is a psychotropic foodborne pathogen with high risk due to its maximum mortality rates in individuals compared to other foodborne pathogens [4,5]. An illness caused by L. monocytogenes is called listeriosis, which covers mostly newborns, pregnant women, and individuals with compromised immune systems [6,7]. It has the maximum (90%) hospitalization rate and 20% of fatality rate in 2016 in European countries [8-10]. Furthermore, S. Typhimurium is also one of the major gram-negative foodborne pathogens which cause widespread contamination in food industries. It is responsible for causing a larger range of host tropism and that is one of the major reasons for salmonellosis all over the world in humans. Many research on the distribution pattern of antibiotic resistance in S. Typhimurium is helping in the selection of suitable antibiotics for Salmonella infections [7,11-17].
Another global concern related to AMR is biofilm formation by pathogenic microbes. Biofilm is a bunch of microorganisms that attach to surfaces and produce extracellular polysaccharides. It can be composed of different kinds of microorganisms such as bacteria, protozoa, fungi, and algae. They are generally formed on both living as well as nonliving surfaces and are observed in industrial, natural, and hospital settings. The main purpose of biofilm formation is to protect the microorganisms from a hostile environment or to act as a trap for nutrient acquisition [18]. In this study, we have focused on bacterial biofilm eradication. Bacteria are mostly found in a free-floating form in sessile states. The sessile stage of bacteria leads to biofilm formation. The biofilms on internal and external medical devices can be responsible for causing infection in patients and it is very much difficult to remove because of the increased resistance to biofilm-forming pathogens to antimicrobial agents [19-21]. In humans, 80% of microbial infections are caused by the formation of biofilms such as cystic fibrosis, endocarditis, periodontitis, osteomyelitis, rhinosinusitis, meningitis, prosthesis, kidney infections, and implantable device-related infections. A major hurdle faced in treating biofilms is their diagnosis, unavailability of biomarkers, and difficult to remove because of their high tolerance toward antibiotics [22].
The tremendous increase in AMR toward antibiotics suggests that, without taking a most crucial step, we are entering a “post-antibiotic era,” which means the therapeutic strategies which were used previously are now no longer applicable for infectious diseases. Therefore, current research is focused on investigating new and non-conventional therapy for the treatment of AMR infections such as vaccines, adjunctive, probiotics, and AMPs [23]. The major components of the innate immune system are AMPs and they play a crucial role in the host defense system against microorganisms; they are produced by bacteria, fungi, protozoa, algae, animals, and plants. Among them, a heterogeneous group of ribosomally synthesized AMPs from bacteria are known as bacteriocins and they can kill closely related microbes known as a narrow spectrum or a diverse range of microbes known as a broad spectrum. Bacteriocin has rapid action and a wide range of antimicrobial activity against both Gram-negative and Gram-positive bacteria which makes it important as a therapeutic agent. Furthermore, antibiotic resistance mechanisms create hindrance to the penetration of antibiotics which do not affect the AMPs [24,25]. Many of the AMPs are also active against bacterial biofilms [21,26-29]. Moreover, several researches have demonstrated a synergistic relationship between bacteriocin and antibiotics [30]. Therefore, we tried to determine whether the Enterocin MSW5 in combination with antibiotics has in vitro antimicrobial and antibiofilm activities alone or combined against S. aureus, L. monocytogenes, and S. Typhimurium.
2. MATERIALS AND METHODS
2.1. Materials
Nutrient Broth (NB), Luria Broth (LB), and Tryptic Soy Broth (TSB) were purchased from Himedia Laboratories (Mumbai, India). All other reagents and media were procured from Himedia Laboratories (Mumbai, India). The 96-well microtiter plate was purchased from Axiva Sichem Biotech (Delhi, India).
2.2. Bacterial Strains and Culture Conditions
The bacteriocin-producing strain Enterococcus faecalis MSW5 with accession number MW672393. S. aureus ATCC 6538, L. monocytogenes ATCC 13932, and Salmonella enterica subsp. enterica serovar Typhimurium ATCC 6539 indicator organisms were procured from the American Type Culture Collection and Gene Bank.
2.3. Antimicrobial Agents
Tetracycline, vancomycin, oxacillin, doxycycline, and linezolid were obtained from Hi-media, Mumbai, India. All five antibiotics were prepared in distilled water freshly as per Clinical and Laboratory Standards Institute (CLSI) guidelines. Enterocin MSW5 was produced from E. faecalis MSW5. Enterocin is purified using cold acetone extraction followed by ion-exchange chromatography using SP Sepharose fast flow cation exchanger (Sigma Aldrich Chemicals Pvt Ltd, Bangalore, India). The protein concentration of Enterocin MSW5 was determined in triplicate by Lowry methods using BSA (Hi-media, Mumbai, India) as a standard [31].
2.4. Determination of Minimum Inhibitory Concentration (MIC) of Enterocin MSW5
MIC of Enterocin MSW5 was determined using 96 well microtiter plate assay as suggested by Fugaban et al. [32] with slightly modifications against S. aureus ATCC 6538, L. monocytogenes ATCC 13932, and S. Typhimurium ATCC 6539 indicator organisms. For that, Sterile 100 μL of TSB was inoculated with 10% of actively grown cultures of indicator organisms. After that, Enterocin MSW5 was 2-fold serially diluted from 5.8 to 0.09 mg/mL. Further, 50 μL of these diluted Enterocin MSW5 samples were added in wells. The untreated well was considered as a positive control. Plates were incubated at 37°C for 24 h and cell density was measured at 595 nm in ELISA plate reader.
2.5. Determination of MIC of Antibiotics
MIC of antibiotics was determined using broth dilutions method as per CLSI guidelines against S. aureus ATCC 6538, L. monocytogenes ATCC 13932, and S. Typhimurium ATCC 6539 indicator organisms. For that, Sterile 100 μL of TSB was inoculated with 10% of actively grown cultures of indicator organisms. After that, five antibiotics (Doxycycline, Tetracycline, Vancomycin, Linezolid, and Oxacillin) were 2-fold serially diluted from 16 to 0.0156 μg/mL. Further, 50 μL of these diluted antibiotics samples were added in wells. The untreated was considered as a positive control [32]. Plates were incubated at 37°C for 24 h and cell density was measured at 595 nm in ELISA plate reader.
2.6. Determination of MIC of Enterocin MSW5, Antibiotics, and their Combinations by Checkerboard Assay
Synergy measurement using checkerboard analysis was used for the determination of the impact of the potency of the combination of two antimicrobial agents in comparison to their activities. The checkerboard assay was performed using a 96-well microtiter plate to find the fractional inhibitory concentration (FIC) index of antibiotics in combination with Enterocin MSW5 against each indicator organism. In microtiter plates, 100 μL of TSB was distributed into each well. In this method, 2-fold serially diluted antimicrobial agents were added in each well of the microtiter plate in the X- and Y-axis of an 8×8 matrix. Whereas Compound A was considered an antibiotic and Compound B was considered Enterocin MSW5. For each combination (A × B), each well of columns having 25 μL of antibiotics (A) which was serially double diluted in a basal medium along the X-axis, and rows of the same plates contained 25 μL of Enterocin MSW5 which was also serially double diluted in the same basal medium along the Y-axis. In each well, a 10% of inoculum size of S. aureus, L. monocytogenes, and S. Typhimurium with 1×108 CFU/ml cell density was added in separate plates. The plates were incubated at 37°C for 24 h and the next day cell density was measured at 595 nm in an ELISA reader (Multiskan Go, Thermo Scientific). The same procedure was repeated in triplicate sets for all five antibiotics against three indicator strains as mentioned above. MIC of antibiotics, Enterocin MSW5, and their combinations had been determined [2].
2.7. Determination of Fractional Inhibitory Concentration Index (FICI) of Enterocin MSW5 in Combination with Antibiotics
To check the synergistic effect of five different antibiotics and bacteriocin fractional inhibitory concentration (FIC) was determined. The formulas used to calculate the FICI are as follows:
 |
 |
The Fractional Inhibitory Concentration Index (FIC Index) for antibiotics and Enterocin MSW5 can be determined by the equation
 |
FIC results were interpreted, if FIC ≤0.5 was recognized as a synergistic effect, 0.5 < FIC ≤ 1 was assigned as an additive effect, 1 < FIC ≤ 4 indicate no interactive effect, and FIC >4 antagonistic effects in between two antimicrobial agents [33].
2.8. Determination of Inoculum Size for Potential Biofilm Formation of Pathogens
Biofilm formation of pathogens was determined using microtiter plate assay in triplicates [34]. Test organisms S. aureus, L. monocytogenes, and S. Typhimurium were activated in a sterile TSB medium at 37°C for 24 h. Next day, activated cultures were centrifuged and cell pellets were dissolved in sterile distilled water. The cell density of pathogens was adjusted to 1 × 108 CFU/mL. In a sterile microtiter plate, wells were filled with 200 μl of sterile TSB medium and indicator organisms were added in variable inoculums size ranges from 2% to 12% of the total media volume in respective wells, and plates were incubated for 24 h at 37°C. Further, crystal violate staining was carried out, microtiter plate was decanted gently followed by the addition of 200 μL methanol for biofilm fixation and allowed for 1 min to react. After that, 200 μL D/W was added for washing purposes. Then, 200 μL of crystal violet (0.1% w/v) stain was added and allowed to react for 2–3 min. Further, the plates were decanted gently, and cell density was measured at 595 nm using an ELISA reader by adding 200 μL of 33 % glacial acetic acid.
2.9. Determination of Medium for Biofilm Potential of Pathogens
Test organisms S. aureus, L. monocytogenes, and S. Typhimurium were activated in a sterile TSB medium and incubated overnight at 37°C. Further, the cultures were centrifuged and cell pellets were dissolved in sterile distilled water. The cell density of pathogens was adjusted to 1 × 108 CFU/mL. In a sterile microtiter plate, 200 μL sterile NB, Luria Bertani broth (LB), and Tryptic Soybean casein Broth (TSB) medium were added in triplicates [34]. Media-containing wells were inoculated with 10% activated indicator cultures and plates were incubated for 24 h at 37°C. The next day, as described earlier staining procedure was carried out and cell density was measured at 595 nm using an ELISA reader.
2.10. Use of Enterocin MSW5 in Combination with Antibiotics for Eradication of Biofilm
In a sterile Microtiter plate, sterile TSB medium (100 μL) was added and each well was inoculated with 10% of activated indicator organisms with cell density of 1 × 108 CFU/mL in each separate plate. In each well 25 μL of filter-sterilized Enterocin MSW5, antibiotics, and their combinations were added as per their MIC and sub-MIC values. The untreated well was considered a positive control. This same procedure was carried out for Oxacillin, Vancomycin, Tetracycline, Doxycycline, and Linezolid with Enterocin MSW5. Plates were then overnight incubated at 37°C. The next day staining procedure was carried out as described earlier and cell density was measured at 595 nm using an ELISA reader. Percent Biofilm eradication was calculated as per the given formula [21].
 |
Where, Initial OD: OD of positive control; Test OD: OD of treated wells with antimicrobials.
2.11. Statistical Analysis
All the experiments were performed in triplicate. Data were analyzed using one-way analysis of variance (ANOVA) followed by Tukey’s test using GraphPad Prism 8.0. Differences between mean values were considered significant at P ≤ 0.05.
3. RESULTS
3.1. Determination of the MIC of Enterocin MSW5
The MIC of Enterocin MSW5 was determined by microdilution method using 96 well microtiter plate. The MIC of Enterocin MSW5 was 0.362 ± 0.00 mg/mL for both S. aureus and S. Typhimurium while 0.725 ± 0.00 mg/mL for L. monocytogenes. This implies that S. aureus and S. Typhimurium were comparatively more susceptible to Enterocin MSW5 than L. monocytogenes.
3.2. Determination of the MIC of Antibiotics
In the present study, we have selected broad spectrum antibiotics based on their mode of action; oxacillin and vancomycin can inhibit cell wall synthesis of Gram-positive bacteria whereas tetracycline, doxycycline, and linezolid can inhibit the protein synthesis. As per CLSI guidelines, the MICs of five different antibiotics such as tetracycline, vancomycin, oxacillin, doxycycline, and linezolid were determined against S. aureus, L. monocytogenes, and S. Typhimurium and are tabulated in Table 1. Tetracycline and doxycycline were the most potent antibiotics among five antibiotics with 1 ± 0.00 μg/mL MIC whereas oxacillin was comparatively less potent antibiotic with 4 ± 0.00 μg/mL MIC against S. aureus. Similarly, antibiotics inhibiting protein synthesis work well against another Gram-positive pathogen L. monocytogenes, where the MIC of tetracycline and linezolid was low (1 ± 0.00 μg/mL). Furthermore, tetracycline antibiotic also works well against gram-negative pathogen S. Typhimurium, where the MIC of tetracycline was low (1 ± 0.00 μg/mL). However, the MIC of oxacillin and linezolid was more (8 ± 0.00 μg/mL) against S. Typhimurium.
Table 1: MIC of antibiotics against indicator organisms.
| Antibiotics | MIC of antibiotics (μg/mL) | ||
|---|---|---|---|
| Staphylococcus aureus ATCC 6538 | Salmonella typhimurium ATCC 6539 | Listeria monocytogenes ATCC 13932 | |
| Tetracycline | 1±0.00a | 1±0.00A | 1±0.00I |
| Oxacillin | 4±0.00c | 8±0.00D | 8±0.00III |
| Doxycycline | 1±0.00ab | 2±0.00B | 2±0.00II |
| Linezolid | 2±0.00ab | 8±0.00D | 1±0.00I |
| Vancomycin | 4±0.00c | 4±0.00C | 8±0.00III |
Mean±SD. Different letters above the columns denote statistically significant differences at P<0.05 by Tukey’s test. SD: Standard deviation, MIC: Minimum inhibitory concentration, ATCC: American Type Culture Collection
3.3. Evaluation of the Synergistic Effect of Antibiotics and Enterocin MSW5
The Fractional Inhibitory Concentration Index (FICI) was determined for all the combinations of Enterocin MSW5 and antibiotics. In this study, the FICI value was <0.5 for all the antibiotics in combination with Enterocin MSW5 against S. aureus, L. monocytogenes, and S. Typhimurium which indicated synergism was observed between all the five antibiotics and Enterocin MSW5. In the case of S. aureus, the best synergism was observed with linezolid in combination with Enterocin MSW5 with FICI, 0.25 followed by doxycycline (FICI 0.263), vancomycin (FICI 0.279), tetracycline (FICI 0.310), and oxacillin (FICI 0.498) as mentioned in Table 2. Likewise, tetracycline-Enterocin MSW5 combination has also shown potential synergism against S. Typhimurium. The FICI value for this combination was 0.249, which was less compared to the other four antibiotic combinations. While, other four antibiotics also showed synergism with enterocin MSW5 in sequence doxycycline (FICI 0.251), vancomycin (FICI 0.255), linezolid (FICI 0.263), and oxacillin (FICI 0.507) [Table 3]. Furthermore, best synergism was observed with linezolid in combination with Enterocin MSW5 with FICI 0.125 for L. monocytogenes. Furthermore, tetracycline, doxycycline, and vancomycin have shown synergism with enterocin MSW5 with FICI 0.139 followed by oxacillin (FICI 0.155) for L. monocytogenes [Table 4]. Hence, these results depict that the MIC of both Enterocin MSW5 as well as antibiotics were decreased when utilized in combinations compared to individual. Moreover, the efficacy of Enterocin MSW5 increased toward indicator pathogens when utilized in combinations with antibiotics.
Table 2: Fraction inhibitory concentration of enterocin MSW5 in combination with antibiotics against Staphylococcus aureus American Type Culture Collection 6538.
| Enterocin MSW5 in combination with antibiotics | MIC of A in presence of B (μg/mL) | MIC of B in presence of A (mg/mL) | FIC Aa | FIC Bb | FICIc | Activityd |
|---|---|---|---|---|---|---|
| Enterocin MSW5+Tetracycline | 0.062±0.00a | 0.090±0.00A | 0.062 | 0.248 | 0.310 | Synergic effect |
| Enterocin MSW5+Oxacillin | 1±0.00d | 0.090±0.00A | 0.250 | 0.248 | 0.498 | Synergic effect |
| Enterocin MSW5+Doxycycline | 0.062±0.00a | 0.090±0.00A | 0.015 | 0.248 | 0.263 | Synergic effect |
| Enterocin MSW5+Linezolid | 0.012±0.00b | 0.090±0.00A | 0.007 | 0.248 | 0.255 | Synergic effect |
| Enterocin MSW5+Vancomycin | 0.125±0.00c | 0.090±0.00A | 0.031 | 0.248 | 0.279 | Synergic effect |
a FIC A=MIC of A in presence of B (μg/mL)/MIC of A alone (μg/mL),
b FIC B=MIC of B in presence of A (mg/mL)/MIC of B alone (mg/mL),
c FICI=FIC A+FIC B),
d Activity=S: FICI≤0.5, AD: 0.5≤FIC≤1, No interaction (I): FIC≤4, Antagonistic effect (A): FIC>4. Mean±SE. Different letters above the columns denote statistically significant differences at P<0.05 by Tukey’s test. A: Antibiotics, B: Enterocin MSW5, SE: Standard error, MIC: Minimum inhibitory concentration, FIC: Fractional inhibitory concentration, FICI: FIC index, AD: Additive effect
Table 3: Fraction inhibitory concentration of enterocin MSW5 in combination with antibiotics against Salmonella typhi American Type Culture Collection 6539.
| Enterocin MSW5 in combination with antibiotics | MIC of A in presence of B (μg/mL) | MIC of B in presence of A (mg/mL) | FIC Aa | FIC Bb | FICIc | Activityd |
|---|---|---|---|---|---|---|
| Enterocin MSW5+Tetracycline | 0.015±0.00a | 0.090±0.00A | 0.001 | 0.248 | 0.249 | Synergic effect |
| Enterocin MSW5+Oxacillin | 0.062±0.00b | 0.181±0.00B | 0.007 | 0.500 | 0.507 | Synergic effect |
| Enterocin MSW5+Doxycycline | 0.015±0.00a | 0.090±0.00A | 0.003 | 0.248 | 0.251 | Synergic effect |
| Enterocin MSW5+Linezolid | 0.015±0.00a | 0.090±0.00A | 0.015 | 0.248 | 0.263 | Synergic effect |
| Enterocin MSW5+Vancomycin | 0.015±0.00a | 0.090±0.00A | 0.007 | 0.248 | 0.255 | Synergic effect |
a FIC A=MIC of A in presence of B (μg/mL)/MIC of A alone (μg/mL),
b FIC B=MIC of B in presence of A (mg/mL)/MIC of B alone (mg/mL),
c FICI=FIC A+FIC B,
d Activity=S: FICI≤0.5, AD: 0.5≤FIC≤1, No interaction (I): FIC≤4, Antagonistic effect (A): FIC>4. Mean±SE. Different letters above the columns denote statistically significant differences at P<0.05 by Tukey’s test. A: Antibiotics, B: Enterocin MSW5, MIC: Minimum inhibitory concentration, FIC: Fractional inhibitory concentration, FICI: FIC index, AD: Additive effect, SE: Standard error
Table 4: Fraction inhibitory concentration of enterocin MSW5 in combination with antibiotics against Listeria monocytogenes American Type Culture Collection 13932.
| Enterocin MSW5 in combination with antibiotics | MIC of A in presence of B (μg/mL) | MIC of B in presence of A (mg/mL) | FIC Aa | FIC Bb | FICIc | Activityd |
|---|---|---|---|---|---|---|
| Enterocin MSW5+Tetracycline | 0.015±0.00a | 0.090±0.00A | 0.015 | 0.124 | 0.139 | Synergic effect |
| Enterocin MSW5+Oxacillin | 0.25±0.00c | 0.090±0.00A | 0.031 | 0.124 | 0.155 | Synergic effect |
| Enterocin MSW5+Doxycycline | 0.030±0.00b | 0.090±0.00A | 0.015 | 0.124 | 0.139 | Synergic effect |
| Enterocin MSW5+Linezolid | 0.015±0.00a | 0.090±0.00A | 0.001 | 0.124 | 0.125 | Synergic effect |
| Enterocin MSW5+Vancomycin | 0.015±0.00a | 0.090±0.00A | 0.015 | 0.124 | 0.139 | Synergic effect |
a FIC A: MIC of A in presence of B (μg/mL)/MIC of A alone (μg/mL),
b FIC B: MIC of B in presence of A (mg/mL)/MIC of B alone (mg/mL),
c FICI: FIC A+FIC B,
d Activity=S: FICI≤0.5, AD: 0.5≤FIC≤1, No interaction (I): FIC≤4, Antagonistic effect (A): FIC>4. Mean±SE. Different letters above the columns denote statistically significant differences at P<0.05 by Tukey’s test. A: Antibiotics; B: Enterocin MSW5, AD: Additive effect, SE: Standard error, MIC: Minimum inhibitory concentration, FIC: Fractional inhibitory concentration, FICI: FIC index
3.4. Determination of Biofilm Potential
3.4.1. Determination of inoculum size for potential biofilm formation
To determine inoculum size for potential biofilm formation, 96 well microtiter plate assay was performed in triplicates. In the case of S. aureus and L. monocytogenes, maximum biofilm formation was observed at 10% inoculum size while in case of S. Typhimurium, maximum biofilm formation was observed at 12% inoculum size [Figure 1].
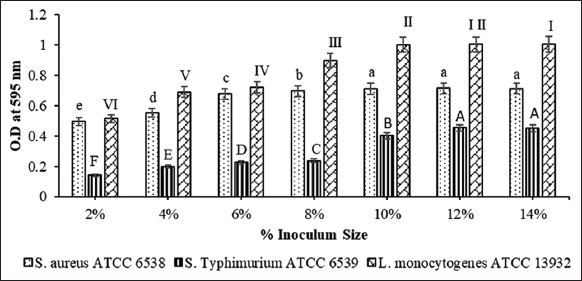 | Figure 1: Determination of inoculum size of pathogens for potential biofilm formation. Different letters above the bars denote statistically significant differences at P < 0.05 by Tukey’s test. [Click here to view] |
3.4.2. Determination of biofilm potential using LB, NB, and TSB media
Different growth media such as LB, NB, and TSB have been assessed for potential biofilm formation using microtiter plate assay. Among them, at 10% inoculum size with TSB media, maximum biofilm formation was observed for indicator organisms S. aureus, and L. monocytogenes while in case of S. Typhimurium, maximum biofilm formation observed in same TSB media but at 12 % of inoculum size [Figure 2].
 | Figure 2: Determination of media for potential biofilm formation of pathogens (where NB: Nutrient Broth, LB: Luria Broth, TSB: Tryptic Soy Broth). Different letters above the bars denote statistically significant differences at P < 0.05 by Tukey’s test. [Click here to view] |
3.5. Eradication of Biofilm using Antibiotics, Enterocin MSW5, and their Combination
The biofilm eradication study was carried out using different antimicrobials (antibiotics and Enterocin MSW5) by microtiter plate assay. They were applied individually and in combination. When antimicrobials were utilized individually at their MIC and sub-MIC concentration, less biofilm eradication was observed. When antibiotics were used alone, biofilm eradication was 27.86 ± 0.78% for tetracycline against S. aureus, 24.31 ± 0.81% for tetracycline against S. Typhimurium and 38.58 ± 0.96% for linezolid against L. monocytogenes. Similarly, when biofilm eradication was studied alone with Enterocin MSW5, eradication was 53.21 ± 0.75% at MIC; 30.72 ± 0.99% at sub-MIC concentration for S. aureus, it was 30.02 ± 0.81% at MIC; 21.50 ± 0.70% at sub-MIC for S. Typhimurium and 42.18 ± 0.75% at MIC; 28.05 ± 0.87% at sub-MIC for L. monocytogenes [Figure 3]. Then, biofilm eradication was studied using five different antibiotics combined with Enterocin MSW5, the maximum biofilm eradication was observed in the case of tetracycline (68.08 ± 0.75%) against S. aureus and (53.76 ± 0.70%) against S. Typhimurium, and 96.97 ± 0.90% against L. monocytogenes in case of linezolid. Further, potential biofilm eradication was also observed at the sub-MIC concentration of these antimicrobials when they were applied in combination. The biofilm eradication was 40.19 ± 0.67% for tetracycline against S. aureus, 33.99 ± 0.72% for tetracycline against S. Typhimurium, and 88.36 ± 0.90% for linezolid against L. monocytogenes [Figure 4]. Thus, the overall results depict that the combinatorial effect of antibiotics and Enterocin MSW5 is significant for biofilm eradication in comparison to individual antimicrobial agents.
 | Figure 3: Biofilm eradication of pathogens by antimicrobials (Enterocin MSW5 and antibiotics alone) at their minimum inhibitory concentration (MIC) and sub-MIC values. (a) Staphylococcus aureus biofilm eradication by antimicrobials (b) Salmonella Typhimurium biofilm eradication by antimicrobials (c) Listeria monocytogenes biofilm eradication by antimicrobials. Different letters above the bars denote statistically significant differences at P < 0.05 by Tukey’s test. [Click here to view] |
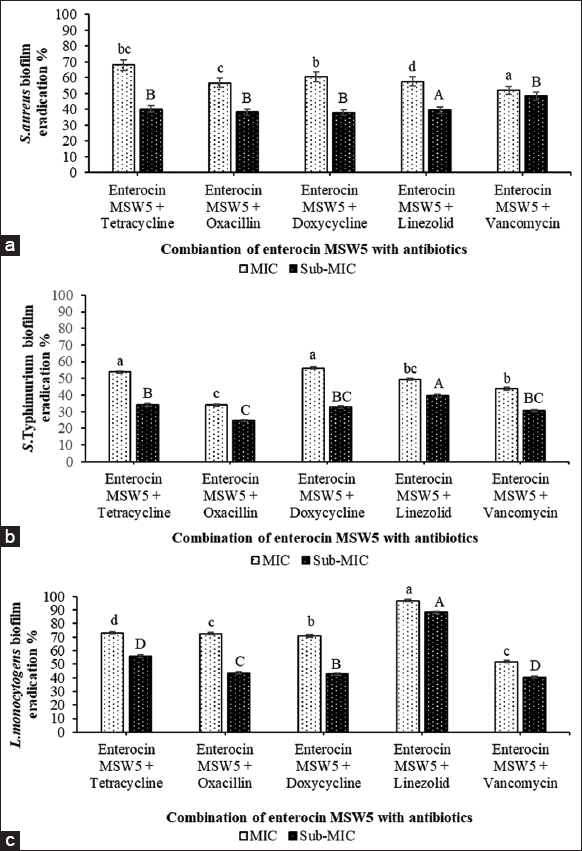 | Figure 4: Biofilm eradication of pathogens by a combination of Enterocin MSW5 and antibiotics at their minimum inhibitory concentration (MIC) and sub-MIC values. (a) Staphylococcus aureus biofilm eradication by a combination of antimicrobials (b) Salmonella Typhimurium biofilm eradication by a combination of antimicrobials (c) Listeria monocytogenes biofilm eradication by a combination of antimicrobials. Different letters above the bars denote statistically significant differences at P < 0.05 by Tukey’s test. [Click here to view] |
4. DISCUSSION
In the present work, we studied the in vitro activities of conventional antibiotics and Enterocin MSW5 alone and in combinations against various pathogenic strains. We used purified Enterocin MSW5 which was obtained from isolate E. faecalis MSW5. Enterocin is a small molecular weight AMP which have ability to inhibit closely related Gram-positive bacteria and also in some cases Gram-negative bacteria. It is basically cationic heat stable peptide synthesized by ribosomes and contains 20–60 amino acids. Enterocin is stable over a wide range of pH and heat. They are classified in to four classes such as lantibiotic (Class I), non-lantibiotic (Class II), cyclic Enterocin (Class III), and Enterocin with high molecular weight (Class IV). The antimicrobial mode of action of Enterocin is different than antibiotics. They inhibit the growth of bacteria by different mechanisms like perforating on target cells, inhibiting protein synthesis by interacting with ribosomes or tRNA, inhibiting peptidoglycan synthesis, and directly degrading target cell DNA [35]. The MIC of this Enterocin MSW5 was same for S. aureus and S. Typhimurium, respectively, while slightly higher for L. monocytogenes. Similar kind of result was noted by another group of researchers and Enterocin TJUQ1 isolated from pickled Chinese celery, MIC was 5.26 μg/ml against L. monocytogenes CMCC 1595 and 46.50 μg/mL against S. aureus [36]. Similarly, other researchers have also found that Enterocin RM6 from E. faecalis has potential activity against foodborne pathogens like B. cereus ATCC 14579, L. monocytogenes, and MRSA S. aureus, but it has no activity against Gram-negative bacteria such as Yersinia enterocolitica, S. Typhimurium, and Escherichia coli [37]. These results indicate bacteriocins produced by Gram-positive bacteria can significantly inhibit the growth of closely related strains [38].
Further, the synergism between Enterocin MSW5 and five different antibiotics was studied using a checkerboard assay, and their FICI value was <0.5 for all the combinations. Among them all the antibiotic combinations with Enterocin MSW5, linezolid has shown maximum two-way synergism against Gram-positive pathogens S. aureus (FICI = 0.25) and L. monocytogenes (FICI = 0.125). Hanchi et al. [2] established the two-way synergy for durancin and vancomycin against S. aureus ATCC 700699 with FICI 0.3. Likewise, combination studies reported for the cationic peptide Enterocin CRL35, FICI was <0.5 for antibiotics, erythromycin, chloramphenicol, and tetracycline but it was not for cefalexin, ciprofloxacin, nalidixic acid, ampicillin, or vancomycin against Listeria innocua 7 [39]. A similar set of experiments using L. monocytogenes FBUNT was accomplished by researchers, where the MIC of Enterocin CRL35 alone was observed at 1.6 ng/ml that was reduced to 0.8 ng/mL when the Enterocin CRL35 was utilized in combination with bacitracin, gramicidin, and monensin antimicrobials [40]. In our study, we also observed a similar kind of observation, the MIC of five different antibiotics was high but when they were used in combination with Enterocin MSW5, MIC was reduced and FICI value was 0.255 in case of combination of tetracycline and Enterocin MSW5 against S. Typhimurium. Likewise, Sharma et al. [41], have determined the synergy effect of ampicillin, penicillin, and ceftriaxone with Enterocin E20c and it has given <0.5 FICI against Salmonella enterica. Similarly, the use of bacteriocin produced by Pediococcus pentosaceus ST44AM in combination with ciprofloxacin have shown synergism with each other against Listeria ivanovii subsp. ivanovii ATCC 19119 [42]. These results revealed that bacteriocin may create a pore in the cell membrane which allow the penetration of antibiotic within the cell and shows its inhibitory effect [42]. Bacteriocin can bind with cell receptors such as lipid II and other cell wall precursors, the mannose phosphotransferase system, undecaprenyl pyrophosphate phosphatase, the maltose ABC transporter, and Zn-dependent metallopeptidase which can creates pore in cell membrane of bacteria [35]. Thus, the benefit of use of the combinatorial therapy of two antimicrobials with a different mechanism of action may result in a more lethal activity against pathogenic microorganisms compare to individual use. Moreover, the use of antibiotics with Enterocin might enhance each other’s inhibitory effects, thereby possibly decreasing the probability of the development of resistance either to the Enterocin or antibiotics [43-45].
Another major global concern of antimicrobial resistance is biofilm formation by AMR bacteria. Both types of bacteria, Gram-negative and positive such as S. aureus, L. monocytogenes, S. Typhimurium, E. faecalis, E. coli, Staphylococcus epidermidis, Staphylococcus viridans, Proteus mirabilis, Klebsiella pneumoniae, and P. aeruginosa can form biofilms. Therefore, we have studied the biofilm formation assay of three foodborne pathogens, S. aureus, L. monocytogenes, and S. Typhimurium using a microtiter plate assay. At 10% inoculum size of S. aureus and L. monocytogenes maximum biofilm formation whereas, S. Typhimurium had shown maximum biofilm formation at 12% inoculum size. The ability of formation of biofilm was considered as OD570 < 0.120, there is no biofilm-formation, 0.120 < OD570 < 0.240, weak biofilm formation, and OD570 > 0.240, strong biofilm formation [46,47]. According to these studies, S. aureus, S. Typhimurium, and L. monocytogenes pathogens were found to be strong biofilm formers. Medium composition is perhaps the most imperative factor which can influence biofilm formation by bacteria under laboratory in vitro conditions. For this work, we selected three different media such as LB, NB, and TSB for the investigation of potential biofilm formation by S. aureus, S. Typhimurium, and L. monocytogenes. Among them, TSB media showed maximum biofilm formation than LB and NB media. A similar kind of study was carried out by Cruz et al. [48], S. aureus biofilm formation was observed in Muller Hinton (MH) broth, Tryptic Soy Glucose (TSG), TSB, brain heart infusion glucose, and brain heart infusion (BHI). Among them, significant biofilm formation was observed with TSB. Likewise, a study was carried out for S. Typhimurium and L. monocytogenes by Ranin et al. [49], they observed that diluted TSB (1/20-TSB) has shown maximum biofilm formation of Salmonella sps.(0.51 ± 0.177) and BHI (0.326 ± 0.06) for L. monocytogenes. All these results revealed that the presence of carbohydrates plays a significant role in biofilm formation [48] and the potential biofilm formation observed in the TSB medium might be due to the amino acids present in the media as a main nutrient component, as well as, it contains enough amount of glucose (2.5 g/l) as a carbohydrate source [50].
Antibiofilm activities of clinically available antibiotics are becoming a significant part of treating infections that occur due to biofilms, such as microbial infections, including cystic fibrosis, endocarditis, periodontitis, osteomyelitis, rhinosinusitis, non-healing chronic wounds, kidney infections, meningitis, prosthesis and infections related to an implantable device, catheter-associated infections, or wound infections. However, due to AMR development in bacteria, it becomes hard to eradicate the biofilm using antibiotics on the bases of their MIC. As a solution, we studied the in vitro activities of antibiotics (Tetracycline, vancomycin, oxacillin, doxycycline, and linezolid) and Enterocin MSW5 alone and in combinations against S. aureus, S. Typhimurium, and L. monocytogenes. Enterocin MSW5 has shown 53.51 ± 0.75%, 30.02 ± 0.81%, and 42.18 ± 0.75% eradication of S. aureus, S. Typhimurium, and L. monocytogenes biofilm, respectively at their MIC value. However, with the use of Enterocin MSW5 in combination with antibiotics, eradication was increased for all the combinations. Moreover, Enterocin MSW5 and antibiotics were also used in combination at their sub-MIC concentration which has shown potential biofilm eradication of all the three pathogens. Similarly, Field et al. [51] have found synergistic relationship between nisin + chloramphenicol and nisin + penicillin against S. aureus biofilm but in contrast, no synergistic effect was found for nisin and vancomycin combinations. Whereas, Dosler and Gerceker [52] have confirmed synergism between nisin and vancomycin against MRSA and MSSA biofilms. Similar results were also observed when vancomycin paired with bacteriocins from E. faecalis ST651ea, ST7119ea, or ST7319a, all the combinations have shown synergism against Enterococcus faecium VRE19 biofilm; while in the case of L. monocytogenes, only ST651ea and ciprofloxacin combination has shown synergism [32]. Along similar lines, the combination of micrococcin P1, garvicin KS, and penicillin G has also significantly eradicated the biofilm of S. aureus by causing severe cell damage [53].
5. CONCLUSION
A combination of Enterocin MSW5 with five different antibiotics has shown a synergistic effect against three food borne pathogens, S. aureus, S. Typhimurium, and L. monocytogenes. Their combinations have also shown potential antibiofilm activity and can significantly eradicate the biofilm of three food borne pathogens at their MIC and sub–MIC concentrations. Therefore, in the future, this combinatorial therapy can be used in the therapeutic sector and might help to solve the problem of multidrug resistance across a wide range of bacterial populations.
6. AUTHORS’ CONTRIBUTIONS
All authors have equally contributed to this manuscript. Material preparation, data collection, and analysis were performed by Mansi Shukla and Dhruv Valand. Shilpa Gupte analyzed these data and necessary inputs were given for the design of the manuscript. The first draft of the manuscript was written by Mansi Shukla and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
7. FUNDING
The authors declare that no grants, funds, or other financial support were received during the preparation of this manuscript.
8. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
9. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
10. DATA AVAILABILITY
Data and material described in this study are available from the authors upon request and availability.
11. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Nikaido H. Multidrug resistance in bacteria. Annu Rev Biochem 2009;66:119-46. [CrossRef]
2. Hanchi H, Hammami R, Gingras H, Kourda R, Bergeron MG, Hamida JB, et al. Inhibition of MRSA and of Clostridium difficile by durancin 61A:Synergy with bacteriocins and antibiotics. Future Microbiol 2017;12:205-12. [CrossRef]
3. Ramaswamy V, Cresence VM, Rejitha JS, Lekshmi MU, Dharsana KS, Prasad SP, et al. Listeria- review of epidemiology and pathogenesis. J Microbiol Immunol Infect 2007;40:4-13.
4. Bennion JR, Sorvillo F, Wise ME, Krishna S, Mascola L. Decreasing listeriosis mortality in the United States, 1990-2005. Clin Infect Dis 2008;47:867-74. [CrossRef]
5. Yi L, Li X, Luo L, Lu Y, Yan H, Qiao Z, et al. A novel bacteriocin BMP11 and its antibacterial mechanism on cell envelope of Listeria monocytogenes and Cronobacter sakazakii. Food Control 2018;91:160-9. [CrossRef]
6. Baka M, Noriega E, Mertens L, Van Derlinden E, Van Impe JF. Protective role of indigenous Leuconostoc carnosum against Listeria monocytogenes on vacuum packed Frankfurter sausages at suboptimal temperatures. Food Res Int 2014;66:197-206. [CrossRef]
7. Martín I, Rodríguez A, Delgado J, Córdoba JJ. Strategies for biocontrol of Listeria monocytogenes using lactic acid bacteria and their metabolites in ready-to-eat meat-and dairy-ripened products. Foods 2022;11:542. [CrossRef]
8. CFSAN. Assessment of the Relative Risk to Public Health from Food borne Listeria monocytogenes Among Selected Categories of Ready-to-Eat Foods. Foods. United States:CFSAN;2001. 1-27. Available from:http://www.foodsafety.gov/~dms/lmrisksu.html
9. Eurosurveillance editorial team. The European Union summary report on trends and sources of zoonoses, zoonotic agents and food-borne outbreaks in 2010. EFSA J. 2012;10(3):2597. doi:10.2903/j.efsa.2012.2597 https://doi.org/10.2903/j.efsa.2012.2597
10. Iseppi R, Camellini S, Sabia C, Messi P. Combined antimicrobial use of essential oils and bacteriocin bacLP17 as seafood biopreservative to control Listeria monocytogenes both in planktonic and in sessile forms. Res Microbiol 2020;171:351-6. [CrossRef]
11. Hohmann EL. Nontyphoidal salmonellosis. Food Saf 2001;32:263-9. [CrossRef]
12. Herikstad H, Motarjemi Y, Tauxe RV. Salmonella surveillance:A global survey of public health serotyping. Epidemiol Infect 2002;129:1-8. [CrossRef]
13. Peterson AT, Soberón J. Species distribution modeling and ecological niche modeling:Getting the concepts right. Nat Conserv 2012;10:102-7. [CrossRef]
14. Lan R, Reeves PR, Octavia S. Population structure, origins and evolution of major Salmonella enterica clones. Infect Genet Evol 2009;9:996-1005. [CrossRef]
15. Majowicz SE, Musto J, Scallan E, Angulo FJ, Kirk M, O'Brien SJ, et al. The global burden of nontyphoidal Salmonella gastroenteritis. Clin Infect Dis 2010;50:882-9. [CrossRef]
16. Hendriksen RS, Vieira AR, Karlsmose S, Lo Fo Wong DM, Jensen AB, Wegener HC, et al. Global monitoring of Salmonella serovar distribution from the world health organization global foodborne infections network country data bank:Results of quality assured laboratories from 2001 to 2007. Foodborne Pathog Dis 2011;8:887-900. [CrossRef]
17. Wang X, Biswas S, Paudyal N, Pan H, Li X. Antibiotic resistance in Salmonella typhimurium isolates recovered from the food chain through national antimicrobial resistance monitoring system between 1996 and 2016. Front Microbiol 2019;10:985. [CrossRef]
18. Sandholm TM, Wirtanen G. Biofilm formation in the industry?:A review. Ind Water Treat 2009;?8:37-41.
19. Donlan RM. Biofilm formation:A clinically relevant microbiological process. Clin Infect Dis 2001;33:1387-92. [CrossRef]
20. Jiang X, Pace JL. Microbial Biofilms. United States:John Wiley and Sons;2005.
21. Mataraci E, Dosler S. In vitro activities of antibiotics and antimicrobial cationic peptides alone and in combination against methicillin-resistant Staphylococcus aureus biofilms. Antimicrob Agents Chemother 2012;56:6366-71. [CrossRef]
22. Khatoon Z, McTiernan CD, Suuronen EJ, Mah TF, Alarcon EI. Bacterial biofilm formation on implantable devices and approaches to its treatment and prevention. Heliyon 2018;4:e01067. [CrossRef]
23. Mahlapuu M, Håkansson J, Ringstad L, Björn C. Antimicrobial peptides:An emerging category of therapeutic agents. Front Cell Infect Microbiol 2016;6:194. [CrossRef]
24. Hancock RE, Sahl HG. Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat Biotechnol 2006;24:1551-7. [CrossRef]
25. Yeung AT, Gellatly SL, Hancock RE. Multifunctional cationic host defence peptides and their clinical applications. Cell Mol Life Sci 2011;68:2161-76. [CrossRef]
26. Dean SN, Bishop BM, van Hoek ML. Natural and synthetic cathelicidin peptides with anti-microbial and anti-biofilm activity against Staphylococcus aureus. BMC Microbiol 2011;11:114. [CrossRef]
27. De La Fuente-Núñez C, Korolik V, Bains M, Nguyen U, Breidenstein EB, Horsman S, et al. Inhibition of bacterial biofilm formation and swarming motility by a small synthetic cationic peptide. Antimicrob Agents Chemother 2012;56:2696-704. [CrossRef]
28. Kharidia R, Liang JF. The activity of a small lytic peptide PTP-7 on Staphylococcus aureus biofilms. J Microbiol 2011;49:663-8. [CrossRef]
29. Pompilio A, Scocchi M, Pomponio S, Guida F, Di Primio A, Fiscarelli E, et al. Antibacterial and anti-biofilm effects of cathelicidin peptides against pathogens isolated from cystic fibrosis patients. Peptides 2011;32:1807-14. [CrossRef]
30. Brumfitt W, Salton MR, Hamilton-Miller JM. Nisin, alone and combined with peptidoglycan-modulating antibiotics:Activity against methicillin-resistant Staphylococcus aureus and vancomycin-resistant enterococci. J Antimicrob Chemother 2002;50:731-4. [CrossRef]
31. Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem 1951;193:265. [CrossRef]
32. Fugaban JI, Bucheli JE, Holzapfel WH. Assessment of bacteriocin-antibiotic synergy for the inhibition and disruption of biofilms of Listeria monocytogenes and vancomycin-resistant Enterococcus. Microbiol Res 2022;13:480-99. [CrossRef]
33. Gutierrez J, Barry-Ryan C, Bourke P.The antimicrobial efficacy of plant essential oil combinations and interactions with food ingredients. Int J Food Microbiol 2008;124:91-7. [CrossRef]
34. Hassan M, Kjos M, Nes IF, Diep DB, Lotfipour F. Natural antimicrobial peptides from bacteria:Characteristics and potential applications to fight against antibiotic resistance. J Appl Microbiol 2012;113:723-36. [CrossRef]
35. Wu Y, Pang X, Wu Y, Liu X, Zhang X. Enterocins:Classification, synthesis, antibacterial mechanisms and food applications. Molecules 2022;27:2258. [CrossRef]
36. Qiao X, Du R, Wang Y, Han Y, Zhou Z. Purification, characterization and mode of action of enterocin, a novel bacteriocin produced by Enterococcus faecium TJUQ1. Int J Biol Macromol 2020;144:151-9. [CrossRef]
37. Huang E, Zhang L, Chung Y, Zheng Z, Yousef AE. Characterization and application of enterocin RM6, a Bacteriocin from Enterococcus faecalis. Biomed Res Int 2013;2013:206917. [CrossRef]
38. Lu Y, Aizhan R, Yan H, Li X, Wang X, Yi Y, et al. Characterization, modes of action, and application of a novel broad-spectrum bacteriocin BM1300 produced by Lactobacillus crustorum MN047. Braz J Microbiol 2020;51:2033-48. [CrossRef]
39. Minahk CJ, Dupuy F, Morero RD. Enhancement of antibiotic activity by sub-lethal concentrations of enterocin CRL35. J Antimicrob Chemother 2004;53:240-6. [CrossRef]
40. Salvucci E, Hebert EM, Sesma F, Saavedra L. Combined effect of synthetic enterocin CRL35 with cell wall, membrane-acting antibiotics and muranolytic enzymes against Listeria cells. Lett Appl Microbiol 2010;51:191-5. [CrossRef]
41. Sharma P, Rashid M, Kaur S. Novel enterocin E20c purified from Enterococcus hirae 20c synergised with ß-lactams and ciprofloxacin against Salmonella enterica. Microb Cell Fact 2020;19:98. [CrossRef]
42. Bucheli JE, Fugaban JI, Holzapfel WH, Todorov SD. Combined action of antibiotics and bacteriocins against vancomycin-resistant enterococci. Microorganisms 2022;10:1423. [CrossRef]
43. Scheffers DJ, Tol MB. LipidII:Just another brick in the wall?PLoS Pathog 2015;11:e1005213. [CrossRef]
44. Kouwen TR, Trip EN, Denham EL, Sibbald MJ, Dubois JY, Van Dijl JM. The large mechanosensitive channel MscL determines bacterial susceptibility to the bacteriocin sublancin 168. Antimicrob Agents Chemother 2009;53:4702-11. [CrossRef]
45. Cotter PD. An 'Upp'-turn in bacteriocin receptor identification. Mol Microbiol 2014;92:1159-63. [CrossRef]
46. Christensen GD, Simpson WA, Younger JJ, Baddour LM, Barrett FF, Melton DM, et al. Adherence of coagulase-negative staphylococci to plastic tissue culture plates:A quantitative model for the adherence of staphylococci to medical devices. J Clin Microbiol 1985;22:996-1006. [CrossRef]
47. Di Rosa R, Creti R, Venditti M, D'Amelio R, Arciola CR, Montanaro L, et al. Relationship between biofilm formation, the enterococcal surface protein (Esp) and gelatinase in clinical isolates of Enterococcus faecalis and Enterococcus faecium. FEMS Microbiol Lett 2006;256:145-50. [CrossRef]
48. Cruz CD, Shah S, Tammela P. Defining conditions for biofilm inhibition and eradication assays for Gram-positive clinical reference strains. BMC Microbiol 2018;18:173. [CrossRef]
49. Ranin L, Svabic M, Stepanovic S. Biofilm formation by Salmonella spp. and Listeria monocytogenes on plastic surface. Lett Appl Microbiol 2004;38:428-32. [CrossRef]
50. Chen X, Thomsen CR, Winkler H, Xu Y. Influence of biofilm growth age, media, antibiotic concentration and exposure time on Staphylococcus aureus and Pseudomonas aeruginosa biofilm removal in vitro. BMC Microbiol 2020;20:264. [CrossRef]
51. Field D, Connor RO, Cotter PD, Ross RP, Hill C. In vitro activities of nisin and nisin derivatives alone and in combination with antibiotics against Staphylococcus biofilms. Front Microbiol 2016;7:508. [CrossRef]
52. Dosler S, Gerceker AA. In vitro activities of nisin alone or in combination with vancomycin and ciprofloxacin against methicillin-resistant and methicillin-susceptible Staphylococcus aureus strains. Chemotherapy 2012;57:511-6. [CrossRef]
53. Kranjec C, Ovchinnikov KV, Grønseth T, Ebineshan K, Srikantam A, Diep DB. A bacteriocin-based antimicrobial formulation to effectively disrupt the cell viability of methicillin-resistant Staphylococcus aureus (MRSA) biofilms. npj Biofilms Microbiomes 2020;6:58. [CrossRef]