ARTICLE HIGHLIGHTS
IgA nephropathy, being the most common cause of glomerular nephritis, is a significant concern to the human population. In this study, we have tried to comprehend the phytomolecules and also can provide sufficient insights into the molecular pathways which can be utilized in the treatment of IgA nephropathy. Conventional treatment of IgA nephropathy includes the use of corticosteroids which have lots of adverse effects such as abnormal metabolism, gastrointestinal disturbances, and compromise of the immune system. About 55% of the patient undergoes these adverse effects and the therapy needs to be discontinued. These phytomolecules based on extensive surveys can definitely be an alternative to the treatment with corticosteroids.
1. INTRODUCTION
Introduction immunoglobulin A nephropathy (IgAN), commonly known as Bergers disease, is the most commonly occurring primary glomerulonephritis in the world [1]. 1500,000–2000,000 new cases of IgA nephropathy get diagnosed globally per year [2]. Life expectancy is reduced between 6 and 10 years after its diagnosis [3]. Studies have reflected the probable indulgence of parasitic infections locally, mainly the helminthes [4,5]. Symptoms of the patient vary from asymptomatic to aggravating, with up to 25% of the patient suffering from end-stage renal disease (ESRD) within 20 years from diagnosis, requiring renal replacement therapy [6,7]. Proteinuria is a common outcome of IgAN and in a retrospective study, it was found that in patients with average proteinuria >0.5–1 g/day was connected with a poor outcome [8]. In patients with IgA nephropathy, aberrant glycosylation of IgA occurs due to which galactose deficient IgA is produced from the B-cells which can be attributed to the lack of the enzyme β-1,3 galactosyltransferase and thus attaching only a monosaccharide N acetyl galactosamine [9]. This aberrant glycosylation exposes the N acetylgalactosamine to the IgG or the IgA1 autoantibodies which induce the formation of immune complexes. These immune complexes after getting circulated in the blood ultimately get deposited in the subepithelial region of the glomerular cells of the kidney. This deposition triggers inflammation and fibrosis and ultimately damages the mesangial cells of the kidney producing IgA nephropathy [10]. This inflammation and fibrosis impact the mesangial cells of the kidney to lose its function due to heavy damage to its architecture leading to proteinuria and elevation of blood creatinine with or without hematuria.
2. INFLAMMATION IN IGA NEPHROPATHY
2.1. Toll-like Receptor (TLR)
The expression of TLR-4 protein in mucosal cells and B-cells is upregulated in IgA nephropathy which correlates with increased proteinuria in IgAN patients [11]. It was also found that TLR-4 activates nuclear factor κβ (NF-κB), a proinflammatory protein which migrates to the nucleus and increases the production of cytokines and chemokines perpetuating inflammation [12]. These cytokines and chemokines attract macrophage cells to the kidney cells and ultimately damage them after chronic inflammation.
2.2. Complement System
The complement system is an integral part of the innate immunity directed toward the elimination of the non-innate antigens. About 90% of the patient with IgA nephropathy were found to have C3 deposition in the mesangium following severe activation of the alternative and lectin pathways of the complement system [13]. The terminal pathway of the complement system which comprises membrane attack complex is also activated in IgAN which is understood by the fact that C9 neo antigen was present as a part of the membrane attack complex [14]. C3a and C5a activate the mesangial cell to produce B-cell activating factor (BAFF) which is a modulator of alternative complement pathway. An increase in mesangial C3 is directly associated with increased proteinuria which is the cause of fibrosis in mesangial cells. Thus, the overactivated complement system actually damages the kidney cells, leading to severe inflammation.
2.3. T-cells
In patients of IgAN, it was found that Th1 and Th2 balance shifts toward the latter which contributes to the progression of the disease [15]. Regulatory T-cells were reduced whereas T17 cells along with its cytokines like interleukin (IL)-17A correlated positively with increased proteinuria [16]. Th2 cells actually produce varieties of cytokines and chemokines which precipitate inflammation.
2.4. B-cells
Myeloid cells present in the mucosa secrete IL-12 and IL-8 and increase the release of interferon (IFN)-γ from the natural killer cells. An increase in the expression of BAFF is directly responsible for the production of aberrant IgA1 in patients with IgA nephropathy. It was found that the upregulation of BAFF and TLR is associated with increased production of autoantibodies-producing plasma cells in patients with IgAN [17]. Thus, the B-cells initiate the production of autoantibodies and trigger the inflammation process in IgA nephropathy.
2.5. Cytokines and Chemokines
IL-6 and tumor necrosis factor (TNF)-α are the cytokines responsible for the pathogenesis of IgAN and elevated IL-6 levels in urine are related to the aggressiveness of the disease [18]. Chemokines such as chemokine (C-C motif) ligand (CCL)2, CCL3, CCL4, and CCL5 are responsible for chemoattraction and proliferation of monocytes [19]. Transformation of acute to chronic inflammation is associated with the switching of chemokine (C-X-C motif) ligand (CXCL)8 chemokines to CCL2 chemokines [20]. It was also found that the presence of CXCL8 levels in the urine was high in the early phase of inflammation, whereas CCL2/monocyte chemoattractant protein 1 (MCP-1) levels were higher in chronic inflammation [21]. It has been observed that locally produced chemokines in mesangial cells of the kidney had an abundant amount of MCP-1/CCL2 which was present in patient with crescentic glomerulonephritis in advanced stages [20]. MCP-1/CCL2 upregulation in chronic inflammation is associated with transforming growth factor (TGF)-β pathway activation [22]. Cytokines such as IL-2, IL-4, IL-6, IL-12, IL-13, and IL-21 play a crucial role in aggravating renal inflammation [23]. TNF proteins are potent activators of NF-κB pathways mediated by Traf intracellular protein [24]. A summary of molecules how effect in inflammation that is present in Figure 1. Phytomolecules affecting inflammation in IgA nephropathy.
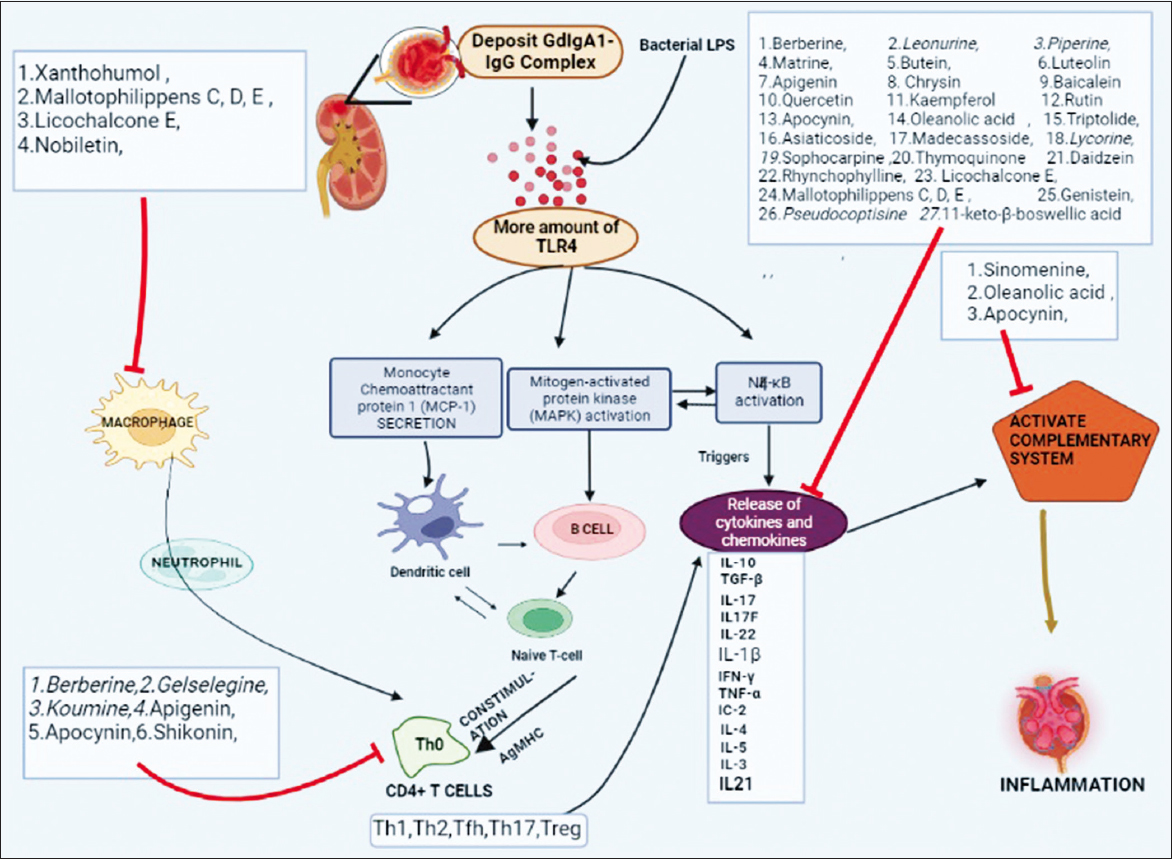 | Figure 1: Phytomolecules affecting inflammation in IgA nephropathy. Deposit immune complex increases the expression of TLR4 which further regulates MCP-1 secretion, MAPK activation, and NFκB activation. Naive T-cells convert into CD4+T-cells, and macrophage comes here through neutrophil. T-cells release various cytokines and chemokines (IL10, TGFβ, IL17, IL22, IL4, IL5, IL3, TNFα, and IL21) from Tfh, Th2, Th1, Th17, and Treg also activate complimentary system. Activated complimentary system and macrophage release lysozyme which also causes glomerular cell disruption and leads to glomerulus inflammation. Class-1 phytomolecules inhibited the release of cytokines and chemokines, Class-2 phytomolecules inhibited to activate the complimentary system, Class-3 phytomolecues inhibited T lymphocytes proliferation, and Class-4 phytomolecules helped to inactivate the macrophages. AgMHC: Antigen-presenting major histocompatibility complex, CCL: Chemokine (C-C motif) ligand, CXCL: Chemokine (C-X-C motif) ligand, LPS: Lipopolysaccharides, MAC: Membrane attack complex, MAPK: Mitogen-activated protein kinase, MBL: Mannose-binding lectin, MCP-1: Monocyte chemoattractant protein 1, Tfh: Follicular helper T-cells, Th: Helper T-cells, Treg: Regulatory T-cells. [Click here to view] |
3. FIBROSIS IN IGA NEPHROPATHY
As the inflammation continues to the chronic phase from the acute phase in renal mesangial cells, fibrosis becomes extremely essential as a tissue repair process. TGF-β is an inflammatory cytokine which regulates the process of fibrosis. It acts through TGF-β RI and RII receptors to mediate the process of fibrosis [25]. TGF-β interacts with transforming growth factor-β receptor II (TBRII) and transforming growth factor-β receptor I (TBRI) which activates the transducer protein Smad2 and Smad3. Smad 2 and Smad3 form a complex with Smad 4 [26] and are responsible for the production of fibrotic proteins such as collagen, integrins, connective tissue growth factors, and proteoglycans [27]. TGF-β1 can exert its effect through mitogen-activated protein kinases (MAPKs) including MEK and extracellular signal-regulated kinase (ERK) pathways [28,29]. Although TGF-β1 is not the only factor which influences the MAPK and ERK pathways, it is the most important one which causes massive fibrosis in patient with IgA nephropathy [30]. Along with it, TGF-β1 also exerts its action through P38, Janus kinase (JNK), and PI3K/AKT pathways to activate fibrotic proteins [31-33]. MAPK, P38, ERK/2, and PI3K/AKT are the pathways triggered by chronic inflammation and fibrosis-causing biomolecules such as TGF-β and bone morphogenetic protein. The process of fibrosis in the kidney cells actually damages the basement membrane of the glomerular cells, leading to the loss of kidney functions permanently. Such pathways when inhibited can reduce chronic inflammation followed by fibrosis. Phytomolecules inhibiting such pathways mentioned in this study in later sections can be very useful in reducing the later stages of inflammation associated with fibrosis such phytomolecules can be studied for the lookout whether it reduces fibrosis in IgA nephropathy. Gene expression studies or western blotting to estimate the proteins of these pathways can throw light on their antifibrotic activity in IgAN. A summary of molecules how effect in inflammation that is present in Figure 2. Phytomolecules affecting chronic inflammation and fibrosis.
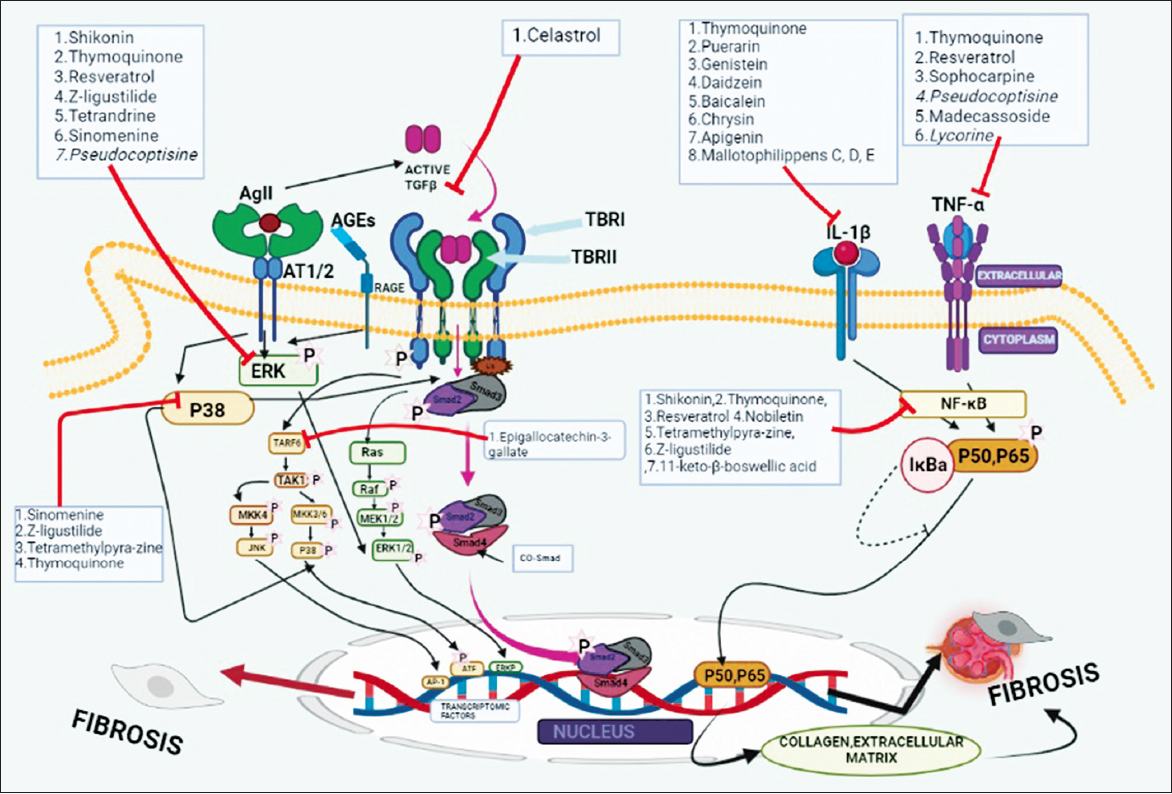 | Figure 2: Phytomolecules affecting chronic inflammation and fibrosis. First active TGF-β binds to TGF-β type TBRII receptor and forms TBRI-TBRII heteromeric complex and leads to Smad-2,3 phosphorylation, and then, it binds to Smad-4 then whole phosphorylation complex translocation into gene and transcription of ECM genes and produces various types of collagen which leads to fibrosis. Smad-7 is an inhibitory Smad and its acts in a negative mechanism with ubiquitin. TGF-β also activate through ERK pathway, TARF6-TAK1-MKK3/6-P38 pathway, TARF6-MKK4-JNK pathway, and TARF6-TAB1-NFκB pathway. NFκB -P50-P65 pathway is activated by IL-1 and TNF-α and gives a profibrotic response. Class-1 phytomolecules inhibited ERK pathway, Class-2 phytomolecules inhibited the expression of TGF-β, Class-3 phytomolecules reduced the expression of IL-1β pathway, Class-4 phytomolecules were inhibited the TNF-α, Class-5 phytomolecules were inhibited NF-κB pathway, Class-6 phytomolecules downregulate Tarf-6, and Class-7 phytomolecules P38 –MAPK Pathway. AGEs: Advanced glycation end products, AgII: Angiotensin II, AT1/2: Angiotensin II type 1/2 receptor, BMP: Bone morphogenetic protein, CD38: Cluster of differentiation, CoSmad: Common suppressor of mothers against decapentaplegic, ECM: Extracellular matrix, ERK: Extracellular signal-regulated kinase, GDF: Growth and differentiation factor, IkBa: Nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor alpha, I-Smad: Inhibitory suppressor of mothers against decapentaplegic, JNK: Janus kinase, LAP: Latency associated peptide, LLC: Large latent complex, LTBP: Latent TGFβ binding protein, MEK: Mitogen-activated protein kinase, NF-κβ: Nuclear factor κβ, RAGE: Receptor for advanced glycation end products, ROS: Reactive oxygen spices, SLC: Small latent complex, Smurf: Smad ubiquitination regulatory factor, TBRII: Transforming growth factor-β receptor II, TBRI: Transforming growth factor-β receptor I, TFR: Transferrin receptor, TGF: Transforming growth factor, Tnf: Tumor necrosis factor, and Ub: Ubiquitin. [Click here to view] |
4. EXISTING MEDICAL MANAGEMENT FOR IGA NEPHROPATHY
There is still no treatment known to modify the mesangial deposition of IgA. Available treatment options are mostly directed at downstream immune and inflammatory events in the glomerulus and the tubulointerstitial, which may lead to renal scarring. It is therefore likely that these are generic treatments with potential benefit in other chronic glomerular diseases [6].
There is no cure for IgA nephropathy and no sure way of knowing what course the disease will take. Some people need only monitoring to determine whether the disease is getting worse [34]. Some of the treatment includes [35].
4.1. High Blood Pressure Medications
Taking angiotensin-converting enzyme inhibitors or angiotensin receptor blockers can lower blood pressure and reduce protein loss.
4.2. Corticosteroids
In some cases, corticosteroid medications, such as prednisone, and other potent drugs that suppress the immune response (immunosuppressant) might keep the immune system from attacking the glomeruli. These drugs can cause serious side effects, such as high blood pressure, high blood sugar, and increased risk of infection.
4.3. Diuretics
These drugs help to remove extra fluid from the blood. Removing the extra fluid can help improve blood pressure control.
Although the treatment is very essential, it is primarily aimed at delaying the progression of the disease, doubling creatinine levels, and increasing life expectancy [36]. However, the prognosis of the disease is still very poor, and new molecules are under study aiming for a better alternative to a steroid with low side effect [6].
5. NATURAL DRUG REMEDIES FOR IGA NEPHROPATHY
Corticosteroids and immunosuppressive medications are cornerstone therapies for glomerular diseases. However, these medications are associated with serious side effects [37]. Steroid therapy was associated with a 55% higher risk for adverse events [38]. Furthermore, resistance to therapy and relapse of disease after discontinuation of medication are common [6]. This triggers the thought of the search for a drug which is a better alternative to steroids aimed at total cure or at least with no significant adverse effect. Natural immunomodulatory drugs can be a significant alternative to corticosteroids which can be used in IgA nephropathy. A few phytomolecules with proven immunosuppressant activities have been researched and presented in the study in the quest for a better alternative to the patients with IgAN. Various phytomolecules discussed under this section were found to reduce cytokines such as IL-6, IL-2, IL-4, and TNFs, and also, chemokines such as CCL2/MCP-1 and CCL4 were also found to get reduced by various phytomolecules. Such cytokines are directly responsible for the activation of NF-kB pathway causing widespread inflammation by altering the production of various chemokines. Chemokines is responsible for the production of macrophage responsible for mesangial cell destruction in kidneys. Phytomolecules reducing the level of such cytokines and chemokines are thought to have an important role also in reducing inflammation-related destruction of kidney cells. Studies can definitely be designed with these phytomolecules in inducing inflammation in various IgA nephropathy model. Cytokine and chemokines are marker molecules of IgA nephropathy and if they are downregulated, they can be correlated with clinical markers such as albumin and creatinine levels in urine and can definitely suggest their IgA nephropathy-alleviating properties [39].
5.1. Phytomolecules with Possible Effect on Inflammation in IgA Nephropathy
Capsaicin is isolated from the plant Capsicum species. When capsaicin was studied in a model of lipopolysaccharide (LPS)-induced inflammation in non-differentiated intestinal porcine epithelial cell line-J2, it reduces the expression of TLR-4/NF-κB proteins [40,41].
Leonurine, an alkaloid isolated from the plant Herba leonuri, was found to have anti-inflammatory properties in human umbilical vein endothelial cells cell lines by inhibiting the NF-κB signaling pathway and thereby reduction of reactive oxygen species production [42,43].
Licochalcone E, a flavonoid isolated from Glycyrrhiza inflate, was found to inhibit the NF-κB pathway when it was studied for its anti-inflammatory effects in LPSs stimulated RAW 264.7 murine macrophage models [44].
Daidzein, an isoflavonoid isolated from Pueraria mirifica, possesses an anti-inflammatory effect and was found out when daidzein was studied on human macrophage cell lines stimulated by LPS causing inflammation. It was found to inhibit the activation of NF-κ bathway in those cells [45,46].
11-keto-β-boswellic acid is a phytomolecule isolated from the plant Boswellia carteri. The complement pathway and various cytokines such as TNF α and ILs were found to be inhibited when boswellic acid was studied in the endothelial cell line to demonstrate its anti-inflammatory activities. Boswellic acid is also studied clinically at pilot levels to demonstrate its activity against rheumatoid arthritis [47].
Oleanolic acid is isolated from the plant Luffa cylindrical which inhibits the complement pathway by downregulating C3 proteins and expression of proinflammatory genes such as IL-2, TNF-α, and IFN-gamma are also inhibited when they were studied in human clinical trials [48].
Gelselegine and koumine are two alkaloids isolated from the plant Gelsemium elegans which was found to possess anti-inflammatory activity by the inhibition of T lymphocyte proliferation in mice models [49,50]. As T lymphocyte proliferation is directly related to the production of T17 cells along with the production of IL17A and is responsible for the aggravation of proteinuria in IgAN, gelselegine and koumine are positive candidates to reduce inflammation and proteinuria in IgA nephropathy.
Triptolide is isolated from the herb Tripterygium wilfordii and was studied against various autoimmune and inflammatory conditions. It inhibits IL12, TNF-α, and IFN-γ in human clinical trials [48,51]. IL-12 and IFN-γ are directly responsible for the increase in macrophage production which causes destruction of podocyte and mesangial cells of the kidney. Thus, triptolide can definitely have a chance to neutralize the podocyte and mesangial cell destruction in IgA nephropathy kidney cells.
Piperine, isolated from the plant Piper longum, can be beneficial in alleviating IgA nephropathy as it was found to reduce the level of proinflammatory cytokines IL-1β, IL-6, and TNF-α when it was studied for cerebral ischemia in Wistar rat [52,53].
Being a natural isoquinoline alkaloid, berberine was studied for gene expression of cytokines in the spleen for Wistar rat models using the inducing agent LPS. It was found that berberine downregulated the genes for the production of IL-2. Such an outcome suggests the antiinflammatory activity of berberine by shifting the Th1/Th2 balance toward Th2 [54].
An alkaloid separated from the plant Uncaria rhynchophylla known as rhynchophylline was studied for its activity against neurodegeneration in microglial cell lines model using LPS as the inducing agent. MCP-1, IL-1β, and TNF-α all were found to get reduced resulting in a decrease of inflammation [55].
Matrine is an alkaloid isolated from the herb Sophora flavescens was investigated against LPSs induced lung injury in mice. It was found that matrine inhibited inflammatory mediators including TNF-α and IL-6. Thus, it can be a potential anti-inflammatory compound in any type of inflammation [56].
Butein can be isolated from various plants such as Semecarpus anacardium, Dalbergia odorifera, and Toxicodendron vernicifluum. Butein was found to inhibit the expression of genes responsible for the production of inflammatory chemokines such as TNF-α and IFN-gamma when studied in RAW 264.7 macrophage cells. All these studies were done to check its anti-inflammatory activity in adipose tissue in obesity-linked insulin resistance [57].
Mallotophillipens C, D, and E are three compounds isolated from Mallotus philippensis that were studied for IL-6 and IL-1 in murine macrophage cell lines using LPS as an inducing agent. It was found that it downregulated IL-6 and IL-1, and thus, it possessed anti-inflammatory effects [58].
Luteolin is a flavonoid isolated from the herb Lonicera japonica was found to possess anti-inflammatory properties by inhibition of the expression of TNF-α and IL-6 gene and studied in cell lines of mouse alveolar macrophage [59,60].
Apigenin is a compound which can be isolated from Cynodon dactylon and Salvia officinalis and possesses anti-inflammatory properties by inhibiting IL-1β, IL-8, and TNF-α when studied in LPS-stimulated human monocytic cell lines. Apigenin is also found to inhibit the transcription of NF-kB [61]. Apigenin when studied in SNF-1 mice against lupus was found to suppress the autoantibodies and also found to delay the development of severe glomerulonephritis [62].
Chrysin is a natural flavonoids isolated from Picea crassifolia which was studied for alleviation of dextran sodium sulfate-induced colitis in mice. It was observed that chrysin decreases the production of TNFα leading to a decrease in inflammation, and thus, disruption of colonic architecture in mice was reversed [63,64]. Furthermore, inhibition of TNF-α-induced activation of NF-kβ was found when chrysin was studied in IEC-6 cell lines. Chrysin was also found to possess anti-inflammatory activity by decreasing the expression of proinflammatory cytokines such as TNFα, IL-4, and IL-6 in mast cells [65].
Baicalein is isolated from the plant Scutellaria altissima, which was found to reduce the protein expression of cytokines such as TNF-α and IL-1β when they were studied in benzopyrene-induced pulmonary carcinogenesis in Swiss albino mice [66]. When baicalein was studied for anti-inflammatory effect in RAW 264.7 cells, it was found to inhibit the TNF-alpha expression along with IL-1β. It also inhibited transcription factor NF-κB activation in RAW264.7 cells [67]. Baicalein when studied again with LPS-induced hyperpermeability and leukocyte migration, it was found to suppress TNF-α, IL-6, and NF-κB in cell lines [68-70].
Quercetin is a polyphenol isolated from grapes, onions, and apples which decrease MCP 1 and IL6 mRNA which can be a very important factor in alleviating inflammation in human cell lines [71]. When quercetin was studied in histological and microarray analysis in the aortas, it was found to reduce the levels of IL-1R and CCL8 which are the important factor of inflammation. Quercetin when studied in LPS-stimulated RAW264.7 cells, it was found to attenuate the NF-κB activation, and thus, it further proves its anti-inflammatory activities [72,73].
Curcumin is a diarylheptanoid isolated from Curcuma longa and has shown promising effect in several diseases such as type-II diabetes, arthritis, pancreatitis, neurodegenerative, and various other metabolic disorders [74]. It has anti-inflammatory effects such as a decrease in the expression of IL-1β, IL-6 was found against LPS-induced BV2 cells [75]. It was also found that the expression of TNF-α, IL-1, IL-6, and IL-12 levels was reduced by the treatment of curcumin in LPS-stimulated monocytes [76].
Genestein is a compound isolated from Genista tinctoria. When human astrocytes, cell lines were cultured and treated with amyloid protein to induce inflammation, genistein reduced the inflammation by inhibiting cytokines such as IL-1β and TNF-α [77].
Apocynin is an inhibitor of the NF-κB pathway by blocking the effect of TNF-α and IL-6 [78]. In lung inflammation induced in Wistar rats, apocynin reduced inflammation by inhibiting the cytokines IL-4, IL-13, and TNF-α [79].
Kaempferol is a polyphenol that is isolated from tomato, apple, etc., suppress the signaling of NF-κB and decrease the expression of MCP1 genes which were found out when kaempferol was studied in LPS-induced macrophage cells [45]. Kaemeferol reduces the levels of TNF-alpha, IL 1β, and also protein expression of MCP 1 in the aorta of rabbits when it was studied for high cholesterol diet-induced atherosclerosis [80].
Puerarin is an iso-flavonoid isolated from the plant Pueraria montana var. lobata and has been found to inhibit inflammation in cerebral ischaemia model rats. IL1-b and TNF-α in brain tissue were decreased when the rats pretreated with puerarin were subjected to inflammatory ischemia. It was also found to inhibit NF-κB activation and rat brain cells [81].
Madecassoside is isolated from the plant Centella asiatica. It has antiinflammatory effects as was found out against collagen-induced arthritis in mice. It was also found that TNF α, IL-6, and IL-10 levels were reduced in mice following the treatment of Madecassoside [82].
Asiaticoside is isolated from the plant Centella asiatica and reduces inflammation when studied in osteoarthritic models which emphasize that asiaticoside inhibits zymosan-induced cartilage degradation by the reduction in IL1β-induced PG synthesis [83].
Rutin is an active flavonoid compound isolated from Ruta graveolens and was found to inhibit TNF-α and IL-6 and thus possess anti-inflammatory properties when they were studied in human umbilical vein endothelial cell lines in which inflammation was induced by HMG b1 [84].
Xanthohumol is a flavonoid isolated from Humulus lupulus and was studied for its inhibitory effect on inflammation induced by LPSs and INF-γ in cell culture using mouse macrophagic RAW 264.7 cell lines. It was found that the expression of iNOS was decreased which suggests its potential as an anti-inflammatory compound [85].
From the above segments, it is evident that these phytomolecules have the ability to influence various proteins of the inflammatory pathways leading to various inflammatory diseases. However, these phytomolecules whether can influence the inflammatory proteins in the kidney leading to a decrease in IgA-related inflammation or not are yet to be tested. A summary of all these molecules is presented in Table 1. Phytomolecules affecting inflammation .
Table 1: Phytomolecules affecting inflammation.
| S. No. | Classification | Phytomolecules | Natural sources | Chemical structure | Diseases | Models studied | Findings | References |
|---|---|---|---|---|---|---|---|---|
| 1 | Alkaloids | Berberine | Coptis chinensis Franch | 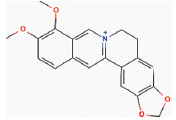 | Diabetes, Inflammatory diseases | Wister rat model | ↓levels of Th1 (TNF-α, IL-2), and Th2 (IL-4)] | [1] |
| Gelselegine | Gelsemium elegans | 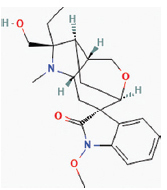 | Malignant tumor, Rheumatic arthritis | Mice model | ↓T lymphocyte proliferation | [2,3] | ||
| Leonurine | Leonurus japonicas Houtt |  | Rheumatoid arthritis, Inflammatory bowel disease | Human umbilical vein endothelial cells (HUVEC) (cell lines) | ↓TNF-α, IL-6, and upregulated IL-10 by inhibiting the expression of toll like receptors and the activation of NF-κB. | [4,5] | ||
| Piperine | Piper longum Linn |  | Diabetes, Arthritis, Multiple myeloma, Metabolic syndrome, Hypertension, Parkinson’s disease, Kidney diseases, Inflammatory diseases, and rhinopharyngitis. | Wistar rat | ↓Level of pro-inflammatory cytokines IL-β, IL-6, and TNF-α, expression of NF-κB. | [6,7] | ||
| Koumine | Gelsemium elegans |  | Rheumatoid arthritis, Neuropathic pain | Mice model | ↓T lymphocyte proliferation | [2] | ||
| Rhynchophylline | Uncaria rhynchophylla (Miq.) Jack |  | Anti-inflammatory, Neuroprotective, anti-Hypertensive, Anti-rhythmic, Angina | Microglia cell lines | ↓Production of pro-inflammatory cytokines, monocytes chemoattractant protein MCP-1, TNF-α, IL-β. ↓Phosphorylation of mitogen-activated protein kinases | [8] | ||
| Matrine | Sophora flavescens |  | Anti-allergic, Anti-inflammatory, Antiviral and Antifibrotic effects | Mice models | ↓Inflammatory mediators include TNF-α and IL-6. | [9] | ||
| 2 | Flavonoids | Butein | Semecarpus anacardium, Dalbergia odorifera, Toxicodendron vernicifluum |  | Anti-inflammatory, anticancer, antidiabetic, hypotensive, and neuroprotective effects. | RAW 264.7 macrophage model | ↓translocation of NF-κB, INF-γ, TNFα | [10] |
| Xanthohumol | Humulus lupulus |  | Anti-carcinogenic, free radical-scavenging, and anti-inflammatory activities, inflammatory bowel disease. | Mouse macrophagic RAW 264.7 cell lines | ↓Inflammation-induced by lipopolysaccharide and INF-γ | [11] | ||
| Mallotophilippens C, D, E | Mallotus philippinensis | 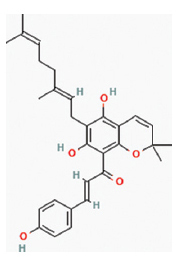 | Convulsion, pain, dysentery, diarrhea, inflammation, malaria, and infectious diseases. | Murine macrophage cell lines | ↓IL-6 and IL-β. Inactivate NF-κB. | [12] | ||
| Licochalcone E | Glycyrrhiza inflata | 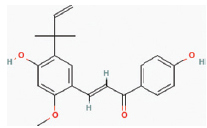 | Antioxidant, anti-inflammatory, anti-cancer, anti-bacterial, anti-diabetic, anti-obesity, anti-parasitic. | RAW 264.7 murine macrophage models. | ↓Secretion of pro-inflammatory cytokines IL-β, IL-6, and TNF-α, NF-κB pathway. | [13] | ||
| 3 | Flavones | Luteolin | Lonicera japonica |  | Treat inflammation-related symptoms. | Cell lines of mouse alveolar macrophage | ↓Secretion of INF-γ, IL-6 | [14,15] |
| Apigenin | Cynodon dactylon, Salvia officinalis L., Portulaca oleracea, Mentha longifolia |  | Prevents neuronal apoptosis. Treatment for inflammatory disorders of the central nervous system, such as multiple sclerosis. | LPS-stimulated human monocytic cell lines SNF-1 mice model | ↓Expression of cytokines (IL-1α, TNF-α, IL-8). ↓Response of Th1 and Th17 cells | [16,17] | ||
| Chrysin | Picea crassifolia |  | Renal dysfunction and oxidative stress. Antidiabetic, anti-inflammatory, and antioxidant properties. | IEC-6 cells, Mice models | ↓Production OF TNFα, IL-β, and IL-6. | [18-20] | ||
| Baicalein | Scutellaria altissima L. |  | Rheumatoid arthritis, respiratory diseases, inflammatory bowel diseases, cardiovascular diseases, hepatitis, kidney diseases, and neurodegenerative diseases. | Benzopyrene-induced pulmonary carcinogenesis in Swiss albino mice, RAW 264.7 cells | ↓Production of inflammatory cytokine (IL-β, and TNF-α) by regulating NF-κB and ER-dependent pathway. | [21-23] | ||
| 4 | Flavonols | Quercetin | Dysosma veitchii Hemsl. et Wils |  | Anti-inflammatory, anti-oxidant, neuroprotective, and anti-allergic activities. Rheumatoid arthritis, inflammatory bowel disease, multiple sclerosis, and systemic inflammation. | LPS stimulated RAW264.7 cells | ↓Expression of pro-inflammatory cytokines, NF-κB, and CCL8. | [24-26] |
| Kaempferol | Found in various fruits and vegetables e.g., tea, tomato, cruciferous vegetables, apple, etc. |  | Inflammation-associated diseases, including allergies, arthritis, diabetes, and cardiovascular diseases like angiogenesis. | Macrophage cells lines Rabbit models | Suppression of the signaling of STAT-1, NF-κB, and AP-1. ↓Gene expression of monocyte chemotactic protein-1 (MCP-1). | [27,28] | ||
| Rutin | Ruta graveolens |  | Acute and chronic inflammation. Antifungal and anti-arthritic effects. | Human umbilical vein endothelial cells line | ↓Production of TNF-α, IL-and NF-κB, and extracellular regulated kinases | [29] | ||
| 5 | Isoflavones | Daidzein | Pueraria mirifica, Pueraria lobata, Glycine max |  | Osteoporosis, cardiovascular and cerebrovascular diseases. Anti-inflammatory effects. | Human macrophage cell lines | ↓TNF-α, IL-β, MCP-1, expression. | [27,30] |
| Genistein | Glycine max | 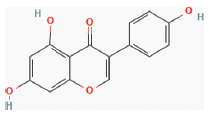 | Antioxidant, anti-inflammatory, antibacterial, and antiviral effects. | Human astrocytes cell lines | ↓IL-β and TNF-α production via activation of PPARs (peroxisome proliferator-activated receptors) | [31] | ||
| Puerarin | Pueraria lobata |  | Treatment of cardiovascular and cerebrovascular diseases, diabetes and diabetic complications, osteonecrosis, Parkinson’s disease, Alzheimer’s disease, endometriosis, and cancer. Immune diseases and inflammation. | Rat models | Inhibit NF-κB, IL-β, and activation of signal transducers and activators of transcription 3 (STAT3). | [32] | ||
| 6 | Phenols | Apocynin | Apocynum cannabinum L. |  | Atherosclerosis, rheumatoid arthritis, airway inflammation, stroke, and IBD | Wister rat models | ↓TNF-α and IL-6 , IL-4 , IL-13 , NF-κB pathway and CD4+and CD8+T cells production | [33,34] |
| 7 | Triterpenoid | Oleanolic acid | Luffa cylindrica, Phytolacca americana |  | Myocarditis, gut dysfunction, treat inflammatory disorders, and acute hepatitis. | Human clinical trials | ↓Level of IL-1α, IL-6, and TNF-α, complement the pathway through the inhibition of C3 convertase. | [35] |
| 8 | Diterpenoid | Triptolide | Tripterygium wilfordii |  | Treatment of immune-inflammatory disorders such as SLE, RA, psoriasis, and asthma. | Human clinical trials | ↓Lymphocyte activation and pro-inflammatory cytokines gene expression (IL-2, TNF-α, IFN-γ). ↓activation of transcription factors such as NF-κB and STAT3 | [36] |
| 9 | Triterpene glycoside | Asiaticoside | Centella asiatica |  | Treating skin diseases, syphilis, rheumatism, mental illness, epilepsy, hysteria, dehydration, and leprosy. A strong anti-inflammatory effect in osteoarthritis. | Mice models | ↓ILβ synthesis. | [37] |
| 10 | Triterpinoid saponin | Madecassoside | Centella asiatica |  | Anti-inflammatory and anti-aging agent. Inflammation, redness, dryness, or other skin conditions such as acne, rosacea. | Mice model | ↓Pro-inflammatory mediators such as TNF-α, and IL-6. | [38] |
| 11 | Triterpinoid | 11-keto-β- boswellic acid | Boswellia carteri | 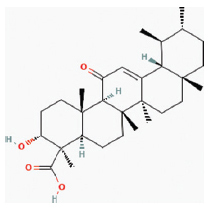 | A large number f inflammatory diseases including cancer, arthritis, chronic colitis, ulcerative colitis, Crohn’s disease, and bronchial asthma | Endothelial cell lines | ↓IL-1, IL-2, IL-4, IL-6, and IFN-γ through inhibition of NF-kB activation. | [39] |
| 12 | Diarylheptanoid | Curcumin | Curcuma longa (Turmeric) |  | Inflammatory bowel diseases, Rheumatoid Arthritis, Cystic Fibrosis, Non-Hodgkin’s, Lymphoma, Lung cancer | LPS-induced BV2 cell lines | ↓The expression of TNF-α, IL-1, IL-6, and IL-12 via LPS or PMA-stimulated monocytes, macrophages, dendritic cells, and spleen lymphocytes. | [40,41] |
| 13 | Alkaloid | Capsaicin | Capsicum species |  | Arthritis, neuropathic pain, gastrointestinal disorders, or cancer. | Cell–line (IPEC-J2) | ↓NF-κB pathway. | [42,43] |
5.2. Phytomolecules with Possible Effect on Fibrosis in IgA Nephropathy
Oxymatrine is an alkaloid isolated from Sophora flavescens; it was found to show antifibrotic effect on liver, pulmonary, and myocardial tissues through inhibition of TGF-β Smad signaling cascade [86]. Moreover, studies have shown that oxymatrine exhibits a protective effect by interacting with SIRT1/Nrf2/TGF-β signaling pathway in STZ-nicotinamide-induced diabetic cardiomyopathy rats [87]. Thus, oxymatrine is a promising molecule against IgA nephropathy-induced fibrosis which can be used to study its possible reduction of fibrosis in IgA nephropathy.
Shikonin, a naturally occurring naphthoquinone, is found in the root of the plant Lithospermum erythrorhizon. Studies have shown that shikonin has a significant effect in reducing ear edema induced by 12- o- tetradecanoylphorbol- 13- acetate in macrophage 264.7 cell lines. It was also found from the same study that shikonin decreased the activation of ERK and NF-κB pathways of inflammation and fibrosis [88].
Pseudocoptisine is a quaternary alkaloid isolated from the plant Corydalis turtschaninovii. It was studied that pseudocoptisine suppressed the cytokines such as TNFα and IL-6 when the murine macrophage RAW264.7 cells line was stimulated by LPS. It was also found that pseudocoptisine inhibited the activity of NF-κB by suppression of ERK and p38 pathways [89].
Z-ligustilide is an essential oil obtained from the plant Angelica sinensis. When LPS-stimulated RAW264.7 macrophages was used to study the anti-inflammatory activities, Z-ligustilide reduced the level of NF-κB and also inhibited the activation of p38 MAPK, ERK, and pathway which can be interpreted as Z-ligustilide can be an important phytomolecule against chronic inflammation and fibrosis [90].
Tetramethylpyrazine is isolated from the plant Ligusticum chuanxiong. When N9 cells were treated with tetramethylpyrazine and then inflammation was induced by LPS, it was found that p38 MAPK, ERK, and JNK pathways were inhibited. Thus, these molecules can be also promising one against chronic inflammation and fibrosis [91]. When tetramethylpyrazine was studied in rat models of spinal cord injury it was found that reduced expression of NF-κB, IL-18 was an important factor in the recovery of the rats following its treatment with tetramethylpyrazine [92].
Tetrandrine is a bisbenzylisoquinoline alkaloid isolated from Stephania tetrandra. Tetrandrine when studied in primary rat mesangial cells in the glomerulo nephritis model, it was found to inhibit the activation of ERK/NF-κB pathways suggesting that tetrandrine can be useful against chronic inflammation and fibrosis [93].
Sinomenine is the plant herb separated from the plant Sinomenium acutum. Sinomenine when studied in human cell lines against rheumatoid arthritis, it was found to reduce inflammation by inhibiting the release of inflammatory chemokines such s CCL2 and CXCL8 [94]. When it was studied in the human mast cell line, it was found to inhibit the activation of effector molecules of ERK, P38, and MAPK pathways. These results can be encouraging considering the fact that cinomenine can also be active in alleviating fibrosis [95].
Epigallocatechin-3-gallate is isolated from the plant Camellia sinensis. When epigallocatechin-3- gallate where studied for antiinflammatory effect in human pulmonary alveolar epithelial cell lines, it was found that epigallocatechin-3- gallate reduced the MAPK activation along with reduced formation of TRAF-2 complex. Thus, it can be an important phytomolecule against inflammation and fibrosis [96].
Resveratrol is isolated from the herb japonica, grape, and nuts. When resveratrol was studied in mouse colonic mucosa where dextran sulfate sodium was used to induce inflammation, it reduced inflammation by deactivating NF-κB and ERK pathways [97]. Several studies of resveratrol have been made to alleviate inflammation including clinical trials, but the existing data recommends more research on resveratrol before it can be used at higher doses beyond the dietary sources [98].
Thymoquinone is a monoterpene obtained from the plant Nigella sativa has shown several promising effects in a number of diseases including asthma, hypertension, and influenza through its anti-inflammatory and antioxidant properties. Studies have shown that thymoquinone interacts with numerous molecular pathways such as p38 MAPK, NF-κB, PI3K/AKT, and Nrf2 to exert its anti-inflammatory and antioxidant activities. In a study involving human RA fibroblast-like synoviocytes where LPS was used to induce inflammation, it was found that thymoquinone significantly reduced the serum levels of IL-1β, TNF-α through downregulation of MAPK p38, ERK1/2, and NF-kB, respectively. When thymoquinone was studied in rats in experimental models of rheumatoid arthritis, such an outcome was also observed [99].
Celastrol is a triterpenoid isolated from the root extract of Tripterygium wilfordii. Studies have revealed that it has significantly decreased interstitial fibrosis, collagen deposition, and mesangial matrix expansion through a decrease in the expression of TGF-β, PI3K/AKT, and mTOR pathway in HFD/STZ-induced diabetic nephropathy in rats [100,101]. Nobiletin is a flavonoid obtained in abundant amounts from the citrus fruits such as orange and grapefruits. It was found to inhibit cytokines such as NF-κB and MAPK against LPS-induced inflammation and fibrosis against RAW264.7 macrophages [102].
Sophocarpine is isolated from the plant Sophora alopecuroides L. When sophocarpine was studied for its affectivity against cachexia on RAW264.7 cells and murine primary macrophage, it was found that both TNFα and IL-6 production was reduced in the cells [103]. It was also found that sophocarpine attenuated the activation of p35 MAP kinase when it was studied for its anti-inflammatory effect in LPS induced RAW264.7 cells. It suggests that it may have activity against fibrosis [104].
Lycorine is isolated from the herb Lycoris radiata when studied in tumor 264.7 cell lines, it was found that lycorine inhibited TNFα and IL-1 released from the cells. It was also observed that LPS-induced activation of p38 pathways was also inhibited by lycorine which suggests that it can also be studied for anti-fibrotic activities [105].
The above-mentioned phytomolecules have a promising effect in reducing IgA nephropathy-related fibrosis. We already know that fibrosis in the mesangial cells of the kidney makes it lose its normal architecture leading to a decrease in kidney function permanently. Prevention of fibrosis in IgA nephropathy is a very important aspect relating to slowing the progression of the disease to the end stage. A summary of all these molecules is presented in Table 2. Phytomolecules affecting in fibrosis.
Table 2: Phytomolecules affecting chronic inflammation and fibrosis.
| S. No. | Classification | Phytomolecules | Biological sourses | Chemical structure | Diseases | Models studied | Findings | References |
|---|---|---|---|---|---|---|---|---|
| 1 | Alkaloids | Pseudocoptisine | Corydalis turtschaninovii Besser |  | Inflammatory bowel disease, diarrhea, acute enteritis. Anti-inflammatory effect. | Macrophage RAW264.7 cell lines | ↓The phosphorylation of ERK and p38 inhibits NF-κB activation. ↓Pro-inflammatory mediators level (TNF-α, and IL-6). | [86] |
| Lycorine | Lycoris radiate |  | Pulmonary fibrosis inhibits inflammation, fibrosis, oxidative stress, and apoptosis. | Tumor 264.7 cell lines | ↓TNFα and IL. Inactivate p38 pathways. | [102] | ||
| Sophocarpine | Sophora alopecuroides L. |  | Isoproterenol-induced kidney injury by Suppressing Inflammation, Apoptosis, Oxidative Stress, and Fibrosis | Murine macrophage RAW264.7 cell lines | ↓Production TNF-α and IL-6. Attenuated p35 MAP kinase. | [100,101] | ||
| Sinomenine | Sinomenium acutum (Thunb.) Rehd.etWils |  | Asthma, rheumatoid arthritis, and various inflammatory diseases. | Human mast cell line (HMC1) | ↓The activation of effector molecules of ERK, P38, and MAPK pathways | [91,92] | ||
| Tetrandrine | Stephania tetrandra |  | Anti-inflammatory, anti-angiogenic, and anti-fibrotic effects. Pulmonary fibrosis. | Rat models | ↓ERK/NF-κB signaling and inhibits activation of mesangial cells. | [90] | ||
| 2 | Essential oils | Z-ligustilide | Angelica sinensis (Oliv.) Diels | 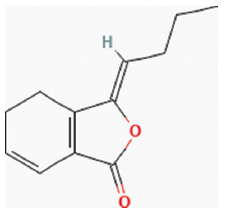 | Pulmonary inflammation, anticancer, anti-inflammatory, anti-oxidant. | Macrophages RAW264.7 cell lines | ↓NF-kB, p38 MAPK, ERK signal pathways. | [87] |
| Tetramethylpyrazine | Ligusticum chuanxiong Hort |  | Hepatic fibrosis, treat renal fibrosis and CKD. | Rat models | ↓NF-κB, IL-18 production. ↓ERK and JNK pathways Block the phosphorylation of p38 mitogen-activated protein kinase. | [88,89] | ||
| 3 | Flavones | Nobiletin | Citrus nobilis Lour, Citrus aurantium L. |  | Chronic colitis, chronic kidney disease, anti-inflammatory, and antifibrotic effects. | Macrophages RAW264.7 cell lines | Blocking NF-κB and MAPK signaling pathways. | [99] |
| 4 | Flavanols | Epigallocatechin-3- gallate | Camellia sinensis L. |  | Anti-inflammatory, anticarcinogenic, antimicrobial, and immunomodulating effects. Diabetic nephropathy, hypoglycemic, and antifibrotic activity. | Human pulmonary alveolar epithelial cell lines | Inhibit reactive oxygen species generation, MAPKs phosphorylation, adhesion molecules expression signal transducers, and activators of transcription 3 (STAT-3). Inhibit TRAF-2 complex. | [93] |
| 5 | Stilbenes | Resveratrol | Resveratrol fallopia |  | Rheumatoid arthritis, an anti-inflammatory, antioxidant, and anticancer agent. Prevented the progression of fibrosis. | Mouse model | Deactivating NF-κB and ERK pathways. ↓Levels of IL-8, TNFα, IFN-γ, and IL-1α. | [94,95] |
| 6 | Quinones | Thymoquinone | Nigella sativa L |  | Myocardial and perivascular fibrosis induced by chronic lipopolysaccharide. Restores liver fibrosis and improves oxidative stress status in lipopolysaccharide-induced inflammation. Asthma, hypertension, and influenza. | Rat models | Inhibit IL-β, TNF-α, MMP-13. Blocking phosphorylation of MAPK p38, ERK1/2, and NF-kBp65. | [96] |
| Shikonin | Lithospermum erythrorhyzon Sieb. etZucc. |  | Improved early-stage diabetic retinopathy. Suppressing fibrosis, inflammation, and apoptosis. | Macrophagic 264.7 cell lines | ↓The activation of ERK, and NF-κB pathways. Inhibit Th1 Th2 cytokines. | [85] | ||
| 7 | Terpenoid | Celastrol | Tripterygium wilfordii |  | Chronic inflammatory diseases, Autoimmune diseases, and cancer. | Rat models | ↓The expression of TGF-β, PI3K/AKT, and mTOR pathway. ↓Proteasome activity and topoisomerase II. | [97,98] |
6. CONCLUSION
In this study, we tried to highlight the molecules which inhibit inflammation and fibrosis in various types of cells when studied in vivo or in vitro. Suppression of proteins such as IL1 and TNF-alpha by many of the phytomolecules can be of great interest with the possibility of reducing the inflammation in mesangial and podocyte cells of the kidney of IgA nephropathy subjects. However, few phytomolecules with their ability to affect the expression of p38, MAPK, ERK 1/2, and TGF-β pathways along with MCP-1/CCL2 cytokines can be very effective in reducing the chronic inflammation and fibrosis in the inflamed kidney, leading to the reversal of inflammation-related proteinuria and creatinine levels in subjects of IgA nephropathy. Thus, the search for novel molecules which can reverse the inflammation-related elevation of proteinuria can be addressed from this study with the possibility of alleviation of symptoms of IgA nephropathy. Oleanolic acid, berberine, apegenine, rutin, diadzein, epigallocatechin-3- gallate, oxymatrine, sinomenine, and celastrol are more effective in alleviating symptoms of IgA nephropathy at least in in vitro and animal studies. Quercetin, curcumin, genistein, and resveratrol have already been tested in human clinical trials with a satisfactory result in alleviating symptoms of IgA nephropathy.
7. ACKNOWLEDGMENT
The authors are thankful to the Department of Pharmacology, Sanaka Educational Trust’s Group of Institutions, Durgapur, West Bengal, India, for providing essential facilities for carrying out this work.
8. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to the conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
9. FUNDING
There is no funding to report.
10. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
11. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
12. DATA AVAILABILITY
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
10. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. McGrogan A, Franssen CF, De Vries CS. The incidence of primary glomerulonephritis worldwide:A systematic review of the literature. Nephrol Dial Transplant 2011;26:414-30. [CrossRef]
2. Schena FP, Nistor I. Epidemiology of IgA nephropathy:A global perspective. Semin Nephrol 2018;38:435-42. [CrossRef]
3. Jarrick S, Lundberg S, Welander A, Carrero JJ, Höijer J, Bottai M, et al. Mortality in IgA nephropathy:A nationwide population-based cohort study. J Am Soc Nephrol 2019;30:866-76. [CrossRef]
4. Kiryluk K, Li Y, Scolari F, Sanna-Cherchi S, Choi M, Verbitsky M, et al. Discovery of new risk loci for IgA nephropathy implicates genes involved in immunity against intestinal pathogens. Nat Genet 2014;46:1187-96. [CrossRef]
5. Sallustio F, Curci C, Di Leo V, Gallone A, Pesce F, Gesualdo L. A new vision of IgA nephropathy:The missing link. Int J Mol Sci 2019;21:189. [CrossRef]
6. Barratt J, Feehally J. Treatment of IgA nephropathy. Kidney Int 2006;69:1934-8. [CrossRef]
7. D'Amico G. Natural history of idiopathic IgA nephropathy:Role of clinical and histological prognostic factors. Am J Kidney Dis 2000;36:227-37. [CrossRef]
8. Donadio JV, Bergstralh EJ, Grande JP, Rademcher DM. Proteinuria patterns and their association with subsequent end-stage renal disease in IgA nephropathy. Nephrol Dial Transplant 2002;17:1197-203. [CrossRef]
9. Kerr MA. The structure and function of human IgA. Biochem J 1990;271:285-96. [CrossRef]
10. Tomana M, Novak J, Julian BA, Matousovic K, Konecny K, Mestecky J. Circulating immune complexes in IgA nephropathy consist of IgA1 with galactose-deficient hinge region and antiglycan antibodies. J Clin Invest 1999;104:73-81. [CrossRef]
11. Chang S, Li XK. The role of immune modulation in pathogenesis of IgA nephropathy. Front Med (Lausanne) 2020;7:92. [CrossRef]
12. Yang S, Tamai R, Akashi S, Takeuchi O, Akira S, Sugawara S, et al. Synergistic effect of muramyldipeptide with lipopolysaccharide or lipoteichoic acid to induce inflammatory cytokines in human monocytic cells in culture. Infect Immun 2001;69:2045-53. [CrossRef]
13. Evans DJ, Williams DG, Peters DK, Sissons JG, Boulton-Jones JM, Ogg CS, et al. Glomerular deposition of properdin in Henoch-Schönlein syndrome and idiopathic focal nephritis. Br Med J 1973;3:326-8. [CrossRef]
14. Lee HJ, Choi SY, Jeong KH, Sung JY, Moon SK, Moon JY, et al. Association of C1q deposition with renal outcomes in IgA nephropathy. Clin Nephrol 2013;80:98-104. [CrossRef]
15. He L, Peng Y, Liu H, Yin W, Chen X, Peng X, et al. Th1/Th2 polarization in tonsillar lymphocyte form patients with IgA nephropathy. Ren Fail 2014;36:407-12. [CrossRef]
16. Lin FJ, Jiang GR, Shan JP, Zhu C, Zou J, Wu XR. Imbalance of regulatory T cells to Th17 cells in IgA nephropathy. Scand J Clin Lab Invest 2012;72:221-9. [CrossRef]
17. Muto M, Manfroi B, Suzuki H, Joh K, Nagai M, Wakai S, et al. Toll-like receptor 9 stimulation induces aberrant expression of a proliferation-inducing ligand by tonsillar germinal center B cells in IgA nephropathy. J Am Soc Nephrol 2017;28:1227-38. [CrossRef]
18. He JW, Zhou XJ, Lv JC, Zhang H. Perspectives on how mucosal immune responses, infections and gut microbiome shape IgA nephropathy and future therapies. Theranostics 2020;10:11462-78. [CrossRef]
19. Gan PY, Steinmetz OM, Tan DS, O'Sullivan KM, Ooi JD, Iwakura Y, et al. Th17 cells promote autoimmune anti-myeloperoxidase glomerulonephritis. J Am Soc Nephrol 2010;21:925-31. [CrossRef]
20. Wada T, Yokoyama H, Matsushima K, Kobayashi K. Chemokines in renal diseases. Int Immunopharmacol 2001;1:637-45. [CrossRef]
21. Segerer S, Henger A, Schmid H, Kretzler M, Draganovici D, Brandt U, et al. Expression of the chemokine receptor CXCR1 in human glomerular diseases. Kidney Int 2006;69:1765-73. [CrossRef]
22. Schneider A, Panzer U, Zahner G, Wenzel U, Wolf G, Thaiss F, et al. Monocyte chemoattractant protein-1 mediates collagen deposition in experimental glomerulonephritis by transforming growth factor-beta. Kidney Int 1999;56:135-44. [CrossRef]
23. Xu Z, Zan H, Pone EJ, Mai T, Casali P. Immunoglobulin class-switch DNA recombination:Induction, targeting and beyond. Nat Rev Immunol 2012;12:517-31. [CrossRef]
24. Wajant H, Henkler F, Scheurich P. The TNF-receptor-associated factor family:Scaffold molecules for cytokine receptors, kinases and their regulators. Cell Signal 2001;13:389-400. [CrossRef]
25. Ito Y, Goldschmeding R, Kasuga H, Claessen N, Nakayama M, Yuzawa Y, et al. Expression patterns of connective tissue growth factor and of TGF-beta isoforms during glomerular injury recapitulate glomerulogenesis. Am J Physiol Renal Physiol 2010;299:F545-58. [CrossRef]
26. MassaguéJ. TGFβ signalling in context. Nat Rev Mol Cell Biol 2012;13:616-30. [CrossRef]
27. Verrecchia F, Chu ML, Mauviel A. Identification of novel TGF-beta/Smad gene targets in dermal fibroblasts using a combined cDNA microarray/promoter transactivation approach. J Biol Chem 2001;276:17058-62. [CrossRef]
28. Dong X, Zhao B, Iacob RE, Zhu J, Koksal AC, Lu C, et al. Force interacts with macromolecular structure in activation of TGF-β. Nature 2017;542:55-9. [CrossRef]
29. Sureshbabu A, Tonner E, Allan GJ, Flint DJ. Relative roles of TGF-β and IGFBP-5 in idiopathic pulmonary fibrosis. Pulm Med 2011;2011:517687. [CrossRef]
30. Haramaki R, Tamaki K, Fujisawa M, Ikedo H, Haramaki N, Okuda S. Steroid therapy and urinary transforming growth factor-β1 in IgA nephropathy. Am J Kidney Dis 2001;38:1191-8. [CrossRef]
31. Edlund S, Landström M, Heldin CH, Aspenström P. Transforming growth factor-beta-induced mobilization of actin cytoskeleton requires signaling by small GTPases Cdc42 and RhoA. Mol Biol Cell 2002;13:902-14. [CrossRef]
32. Kim SI, Kwak JH, Na HJ, Kim JK, Ding Y, Choi ME. Transforming growth factor-beta (TGF-beta1) activates TAK1 via TAB1-mediated autophosphorylation, independent of TGF-beta receptor kinase activity in mesangial cells. J Biol Chem 2009;284:22285-96. [CrossRef]
33. Li Y, Tan X, Dai C, Stolz DB, Wang D, Liu Y. Inhibition of integrin-linked kinase attenuates renal interstitial fibrosis. J Am Soc Nephrol 2009;20:1907-18. [CrossRef]
34. Chapter 10:Immunoglobulin A nephropathy. Kidney Int Suppl (2011) 2012;2:209-17. [CrossRef]
35. Gutiérrez E, Carvaca-Fontán F, Luzardo L, Morales E, Alonso M, Praga M. A personalized update on IgA nephropathy:A new vision and new future challenges. Nephron 2020;144:555-71. [CrossRef]
36. Floege J, Rauen T, Tang SC. Current treatment of IgA nephropathy. Semin Immunopathol 2021;43:717-28. [CrossRef]
37. Glassock RJ. Glomerular disease:Targeted steroid therapy for IgA nephropathy. Nat Rev Nephrol 2017;13:390-2. [CrossRef]
38. Lv J, Xu D, Perkovic V, Ma X, Johnson DW, Woodward M, et al. Corticosteroid therapy in IgA nephropathy. J Am Soc Nephrol 2012;23:1108-16. [CrossRef]
39. Kalliakmani P, Nakopoulou L, Tsakas S, Gerolymos M, Papasotiriou M, Goumenos DS. Urinary interleukin-6 (IL-6) and transforming growth factor (TGF-β) levels in corticosteroidtreated patients with IgA nephropathy. Clin Nephrol 2011;76:144-50. [CrossRef]
40. Zhao X, Dong B, Friesen M, Liu S, Zhu C, Yang C. Capsaicin attenuates lipopolysaccharide-induced inflammation and barrier dysfunction in intestinal porcine epithelial cell line-J2. Front Physiol 2021;12:715469. [CrossRef]
41. Kim CS, Kawada T, Kim BS, Han IS, Choe SY, Kurata T, et al. Capsaicin exhibits anti-inflammatory property by inhibiting IkB-a degradation in LPS-stimulated peritoneal macrophages. Cell Signal 2003;15:299-306. [CrossRef]
42. Liu XH, Pan LL, Yang HB, Gong QH, Zhu YZ. Leonurine attenuates lipopolysaccharide-induced inflammatory responses in human endothelial cells:Involvement of reactive oxygen species and NF-kB pathways. Eur J Pharmacol 2012;680:108-14. [CrossRef]
43. Song X, Wang T, Zhang Z, Jiang H, Wang W, Cao Y, et al. Leonurine exerts anti-inflammatory effect by regulating inflammatory signaling pathways and cytokines in LPS-induced mouse mastitis. Inflammation 2015;38:79-88. [CrossRef]
44. Lee HN, Cho HJ, Lim DY, Kang YH, Lee KW, Park JH. Mechanisms by which licochalcone E exhibits potent anti-inflammatory properties:Studies with phorbol ester-treated mouse skin and lipopolysaccharide-stimulated murine macrophages. Int J Mol Sci 2013;14:10926-43. [CrossRef]
45. Hämäläinen M, Nieminen R, Vuorela P, Heinonen M, Moilanen E. Anti-inflammatory effects of flavonoids:Genistein, kaempferol, quercetin, and daidzein inhibit STAT-1 and NF-kappaB activations, whereas flavone, isorhamnetin, naringenin, and pelargonidin inhibit only NF-kappaB activation along with their inhibitory EF. Mediators Inflamm 2007;2007:45673.
46. Alderton WK, Cooper CE, Knowles RG. Nitric oxide synthases:Structure, function and inhibition. Biochem J 2001;357:593-615. [CrossRef]
47. Ammon HP. Boswellic acids in chronic inflammatory diseases. Planta Med 2006;72:1100-16. [CrossRef]
48. Brinker AM, Ma J, Lipsky PE, Raskin I. Medicinal chemistry and pharmacology of genus Tripterygium (Celastraceae). Phytochemistry 2007;68:732-66. [CrossRef]
49. Xu YK, Liao SG, Na Z, Hu HB, Li Y, Luo HR. Gelsemium alkaloids, immunosuppressive agents from Gelsemium elegans. Fitoterapia 2012;83:1120-4. [CrossRef]
50. Zuo M, Liu Y, Sun Z, Lin L, Tang Q, Cheng P, et al. An integrated strategy toward comprehensive characterization and quantification of multiple components from herbal medicine:An application study in Gelsemium elegans. Chin Herb Med 2021;13:17-32. [CrossRef]
51. Duan H, Takaishi Y, Momota H, Ohmoto Y, Taki T, Tori M, et al. Immunosuppressive terpenoids from extracts of Tripterygium wilfordii. Tetrahedron 2001;57:8413-24. [CrossRef]
52. Vaibhav K, Shrivastava P, Javed H, Khan A, Ahmed ME, Tabassum R, et al. Piperine suppresses cerebral ischemia-reperfusion-induced inflammation through the repression of COX-2, NOS-2, and NF-kB in middle cerebral artery occlusion rat model. Mol Cell Biochem 2012;367:73-84. [CrossRef]
53. Son DJ, Akiba S, Hong JT, Yun YP, Hwang SY, Park YH, et al. Piperine inhibits the activities of platelet cytosolic phospholipase A2 and thromboxane A2 synthase without affecting cyclooxygenase-1 activity:Different mechanisms of action are involved in the inhibition of platelet aggregation and macrophage inflammatory response. Nutrients 2014;6:3336-52. [CrossRef]
54. Lin WC, Lin JY. Berberine down-regulates the Th1/Th2 cytokine gene expression ratio in mouse primary splenocytes in the absence or presence of lipopolysaccharide in a preventive manner. Int Immunopharmacol 2011;11:1984-90. [CrossRef]
55. Song Y, Qu R, Zhu S, Zhang R, Ma S. Rhynchophylline attenuates LPS-induced pro-inflammatory responses through down-regulation of MAPK/NF-κB signaling pathways in primary microglia. Phytother Res 2012;26:1528-33. [CrossRef]
56. Zhang B, Liu ZY, Li YY, Luo Y, Liu ML, Dong HY, et al. Antiinflammatory effects of matrine in LPS-induced acute lung injury in mice. Eur J Pharm Sci 2011;44:573-9. [CrossRef]
57. Wang Z, Lee Y, Eun JS, Bae EJ. Inhibition of adipocyte inflammation and macrophage chemotaxis by butein. Eur J Pharmacol 2014;738:40-8. [CrossRef]
58. Daikonya A, Katsuki S, Kitanaka S. Antiallergic agents from natural sources 9. Inhibition of nitric oxide production by novel chalcone derivatives from Mallotus philippinensis (Euphorbiaceae). Chem Pharm Bull (Tokyo) 2004;52:1326-9. [CrossRef]
59. Chen CY, Peng WH, Tsai KD, Hsu SL. Luteolin suppresses inflammation-associated gene expression by blocking NF-kappaB and AP-1 activation pathway in mouse alveolar macrophages. Life Sci 2007;81:1602-14. [CrossRef]
60. Ziyan L, Yongmei Z, Nan Z, Ning T, Baolin L. Evaluation of the anti-inflammatory activity of luteolin in experimental animal models. Planta Med 2007;73:221-6. [CrossRef]
61. Nicholas C, Batra S, Vargo MA, Voss OH, Gavrilin MA, Wewers MD, et al. Apigenin blocks lipopolysaccharide-induced lethality in vivo and proinflammatory cytokines expression by inactivating NF-kappaB through the suppression of p65 phosphorylation. J Immunol 2007;179:7121-7. [CrossRef]
62. Kang HK, Ecklund D, Liu M, Datta SK. Apigenin, a non-mutagenic dietary flavonoid, suppresses lupus by inhibiting autoantigen presentation for expansion of autoreactive Th1 and Th17 cells. Arthritis Res Ther 2009;11:R59. [CrossRef]
63. Hougee S, Sanders A, Faber J, Graus YM, van den Berg WB, Garssen J, et al. Decreased pro-inflammatory cytokine production by LPS-stimulated PBMC upon in vitro incubation with the flavonoids apigenin, luteolin or chrysin, due to selective elimination of monocytes/macrophages. Biochem Pharmacol 2005;69:241-8. [CrossRef]
64. Shin EK, Kwon HS, Kim YH, Shin HK, Kim JK. Chrysin, a natural flavone, improves murine inflammatory bowel diseases. Biochem Biophys Res Commun 2009;381:502-7. [CrossRef]
65. Bae Y, Lee S, Kim SH. Chrysin suppresses mast cell-mediated allergic inflammation:Involvement of calcium, caspase-1 and nuclear factor-κB. Toxicol Appl Pharmacol 2011;254:56-64. [CrossRef]
66. Chandrashekar N, Selvamani A, Subramanian R, Pandi A, Thiruvengadam D. Baicalein inhibits pulmonary carcinogenesis-associated inflammation and interferes with COX-2, MMP-2 and MMP-9 expressions in-vivo. Toxicol Appl Pharmacol 2012;261:10-21. [CrossRef]
67. Fan GW, Zhang Y, Jiang X, Zhu Y, Wang B, Su L, et al. Anti-inflammatory activity of baicalein in LPS-stimulated RAW264.7 macrophages via estrogen receptor and NF-κB-dependent pathways. Inflammation 2013;36:1584-91. [CrossRef]
68. Lee W, Ku SK, Bae JS. Anti-inflammatory effects of Baicalin, Baicalein, and Wogonin in vitro and in vivo. Inflammation 2015;38:110-25. [CrossRef]
69. Li P, Yin YL, Li D, Kim SW, Wu G. Amino acids and immune function. Br J Nutr 2007;98:237-52. [CrossRef]
70. Ye M, Wang Q, Zhang W, Li Z, Wang Y, Hu R. Oroxylin A exerts anti-inflammatory activity on lipopolysaccharide-induced mouse macrophage via Nrf2/ARE activation. Biochem Cell Biol 2014;92:337-48. [CrossRef]
71. Shaik YB, Castellani ML, Perrella A, Conti F, Salini V, Tete S, et al. Role of quercetin (a natural herbal compound) in allergy and inflammation. J Biol Regul Homeost Agents 2006;20:47-52.
72. Kleemann R, Verschuren L, Morrison M, Zadelaar S, van Erk MJ, Wielinga PY, et al. Anti-inflammatory, anti-proliferative and anti-atherosclerotic effects of quercetin in human in vitro and in vivo models. Atherosclerosis 2011;218:44-52. [CrossRef]
73. Choi SJ, Tai BH, Cuong NM, Kim YH, Jang HD. Antioxidative and anti-inflammatory effect of quercetin and its glycosides isolated from mampat (Cratoxylum formosum). Food Sci Biotechnol 2012;21:587-95. [CrossRef]
74. Shehzad A, Rehman G, Lee YS. Curcumin in inflammatory diseases. Biofactors 2013;39:69-77. [CrossRef]
75. Peng Y, Ao M, Dong B, Jiang Y, Yu L, Chen Z, et al. Anti-inflammatory effects of curcumin in the inflammatory diseases:Status, limitations and countermeasures. Drug Des Devel Ther 2021;15:4503-25. [CrossRef]
76. Gao X, Kuo J, Jiang H, Deeb D, Liu Y, Divine G, et al. Immunomodulatory activity of curcumin:Suppression of lymphocyte proliferation, development of cell-mediated cytotoxicity, and cytokine production in vitro. Biochem Pharmacol 2004;68:51-61. [CrossRef]
77. Valles SL, Dolz-Gaiton P, Gambini J, Borras C, Lloret A, Pallardo FV, et al. Estradiol or genistein prevent Alzheimer's disease-associated inflammation correlating with an increase PPAR gamma expression in cultured astrocytes. Brain Res 2010;1312:138-44. [CrossRef]
78. Stefanska J, Pawliczak R. Apocynin:Molecular aptitudes. Mediators Inflamm 2008;2008:106507. [CrossRef]
79. Kim SY, Moon KA, Jo HY, Jeong S, Seon SH, Jung E, et al. Anti-inflammatory effects of apocynin, an inhibitor of NADPH oxidase, in airway inflammation. Immunol Cell Biol 2012;90:441-8. [CrossRef]
80. Kong L, Luo C, Li X, Zhou Y, He H. The anti-inflammatory effect of kaempferol on early atherosclerosis in high cholesterol fed rabbits. Lipids Health Dis 2013;12:115. [CrossRef]
81. Liu X, Mei Z, Qian J, Zeng Y, Wang M. Puerarin partly counteracts the inflammatory response after cerebral ischemia/reperfusion via activating the cholinergic anti-inflammatory pathway. Neural Regen Res 2013;8:3203-15.
82. Li H, Gong X, Zhang L, Zhang Z, Luo F, Zhou Q, et al. Madecassoside attenuates inflammatory response on collagen-induced arthritis in DBA/1 mice. Phytomedicine 2009;16:538-46. [CrossRef]
83. Hartog A, Smit HF, van der Kraan PM, Hoijer MA, Garssen J. In vitro and in vivo modulation of cartilage degradation by a standardized Centella asiatica fraction. Exp Biol Med (Maywood) 2009;234:617-23. [CrossRef]
84. Yoo H, Ku SK, Baek YD, Bae JS. Anti-inflammatory effects of rutin on HMGB1-induced inflammatory responses in vitro and in vivo. Inflamm Res 2014;63:197-206. [CrossRef]
85. Zhao F, Nozawa H, Daikonnya A, Kondo K, Kitanaka S. Inhibitors of nitric oxide production from hops (Humulus lupulus L.). Biol Pharm Bull 2003;26:61-5. [CrossRef]
86. Liu L, Lu W, Ma Z, Li Z. Oxymatrine attenuates bleomycin-induced pulmonary fibrosis in mice via the inhibition of inducible nitric oxide synthase expression and the TGF-β/Smad signaling pathway. Int J Mol Med 2012;29:815-22. [CrossRef]
87. Seksaria S, Mehan S, Dutta BJ, Gupta GD, Ganti SS, Singh A. Oxymatrine and insulin resistance:Focusing on mechanistic intricacies involve in diabetes associated cardiomyopathy via SIRT1/AMPK and TGF-β signaling pathway. J Biochem Mol Toxicol 2023;37:e23330. [CrossRef]
88. Andújar I, Recio MC, Bacelli T, Giner RM, Ríos JL. Shikonin reduces oedema induced by phorbol ester by interfering with Ikappa Balpha degradation thus inhibiting translocation of NF-kappaB to the nucleus. Br J Pharmacol 2010;160:376-88. [CrossRef]
89. Yun KJ, Shin JS, Choi JH, Back NI, Chung HG, Lee KT. Quaternary alkaloid, pseudocoptisine isolated from tubers of Corydalis turtschaninovi inhibits LPS-induced nitric oxide, PGE(2), and pro-inflammatory cytokines production via the down-regulation of NF-kappaB in RAW 264.7 murine macrophage cells. Int Immunopharmacol 2009;9:1323-31. [CrossRef]
90. Chung JW, Choi RJ, Seo EK, Nam JW, Dong MS, Shin EM, et al. Anti-inflammatory effects of (Z)-ligustilide through suppression of mitogen-activated protein kinases and nuclear factor-κB activation pathways. Arch Pharm Res 2012;35:723-32. [CrossRef]
91. Liu HT, Du YG, He JL, Chen WJ, Li WM, Yang Z, et al. Tetramethylpyrazine inhibits production of nitric oxide and inducible nitric oxide synthase in lipopolysaccharide-induced N9 microglial cells through blockade of MAPK and PI3K/Akt signaling pathways, and suppression of intracellular reactive oxygen species. J Ethnopharmacol 2010;129:335-43. [CrossRef]
92. Hu JZ, Huang JH, Xiao ZM, Li JH, Li XM, Lu HB. Tetramethylpyrazine accelerates the function recovery of traumatic spinal cord in rat model by attenuating inflammation. J Neurol Sci 2013;324:94-9. [CrossRef]
93. Wu CJ, Wang YH, Lin CJ, Chen HH, Chen YJ. Tetrandrine down-regulates ERK/NF-κB signaling and inhibits activation of mesangial cells. Toxicol In Vitro 2011;25:1834-40. [CrossRef]
94. Chen DP, Wong CK, Leung PC, Fung KP, Lau CB, Lau CP, et al. Anti-inflammatory activities of Chinese herbal medicine sinomenine and Liang Miao San on tumor necrosis factor-α-activated human fibroblast-like synoviocytes in rheumatoid arthritis. J Ethnopharmacol 2011;137:457-68. [CrossRef]
95. Oh YC, Kang OH, Kim SB, Mun SH, Park CB, Kim YG, et al. Anti-inflammatory effect of sinomenine by inhibition of pro-inflammatory mediators in PMA plus A23187-stimulated HMC-1 Cells. Eur Rev Med Pharmacol Sci 2012;16:1184-91.
96. Lee IT, Lin CC, Lee CY, Hsieh PW, Yang CM. Protective effects of (-)-epigallocatechin-3-gallate against TNF-α-induced lung inflammation via ROS-dependent ICAM-1 inhibition. J Nutr Biochem 2013;24:124-36. [CrossRef]
97. Youn J, Lee JS, Na HK, Kundu JK, Surh YJ. Resveratrol and piceatannol inhibit iNOS expression and NF-kappaB activation in dextran sulfate sodium-induced mouse colitis. Nutr Cancer 2009;61:847-54. [CrossRef]
98. Vang O, Ahmad N, Baile CA, Baur JA, Brown K, Csiszar A, et al. What is new for an old molecule?Systematic review and recommendations on the use of resveratrol. PLoS One 2011;6:e19881. [CrossRef]
99. Vaillancourt F, Silva P, Shi Q, Fahmi H, Fernandes JC, Benderdour M. Elucidation of molecular mechanisms underlying the protective effects of thymoquinone against rheumatoid arthritis. J Cell Biochem 2011;112:107-17. [CrossRef]
100. Kannaiyan R, Shanmugam MK, Sethi G. Molecular targets of celastrol derived from Thunder of God Vine:Potential role in the treatment of inflammatory disorders and cancer. Cancer Lett 2011;303:9-20. [CrossRef]
101. Nie Y, Fu C, Zhang H, Zhang M, Xie H, Tong X, et al. Celastrol slows the progression of early diabetic nephropathy in rats via the PI3K/AKT pathway. BMC Complement Med Ther 2020;20:321. [CrossRef]
102. Kang SR, Park KI, Park HS, Lee DH, Kim JA, Nagappan A, et al. Anti-inflammatory effect of flavonoids isolated from Korea Citrus aurantium L. on lipopolysaccharide-induced mouse macrophage RAW 264.7 cells by blocking of nuclear factor-kappa B (NF-κB) and mitogen-activated protein kinase (MAPK) signalling pathways. Food Chem 2011;129:1721-8. [CrossRef]
103. Zhang Y, Wang S, Li Y, Xiao Z, Hu Z, Zhang J. Sophocarpine and matrine inhibit the production of TNF-alpha and IL-6 in murine macrophages and prevent cachexia-related symptoms induced by colon26 adenocarcinoma in mice. Int Immunopharmacol 2008;8:1767-72. [CrossRef]
104. Gao Y, Jiang W, Dong C, Li C, Fu X, Min L, et al. Anti-inflammatory effects of sophocarpine in LPS-induced RAW 264.7 cells via NF-kB and MAPKs signaling pathways. Toxicol In Vitro 2012;26:1-6. [CrossRef]
105. Kang J, Zhang Y, Cao X, Fan J, Li G, Wang Q, et al. Lycorine inhibits lipopolysaccharide-induced iNOS and COX-2 up-regulation in RAW264.7 cells through suppressing P38 and STATs activation and increases the survival rate of mice after LPS challenge. Int Immunopharmacol 2012;12:249-56. [CrossRef]