1. INTRODUCTION
Nitrogen (N) is a crucial primary nutrient for plant growth. It is a major constituent of amino acids, proteins, and nucleic acid precursors in plant regimes. It helps build the structural integrity of proteins and chlorophyll, enhancing plants’ leaf growth [1]. The atmosphere contains ~78% gaseous nitrogen. Atmospheric nitrogen (N2) is a nonreactive molecule. Thus, it cannot be assimilated in plant biomass following central metabolic cascades. However, plants access the soluble form of nitrogen, that is, nitrate (NO3-) and ammonia (NH4+), as sole nitrogen sources [2,3]. Naturally, continuous farming, leaching, and evaporation reduced plant-accessible nitrogen continuously in the soil [4]. In this context, inorganic agrochemicals and biological nitrogen-fixing (BNF) microbes can provide plant’s accessible nitrogen precursors [5,6]. Long-term application of inorganic agrochemicals at high concentrations has shown extensive detrimental impacts on soil fertility and plant productivity. It is also increasing environmental pollution and affecting human health. In addition, the reduction in soil fertility and the high expense of inorganic agrochemicals have gradually increased agricultural production costs [5,7]. BNF is a more economically and environmentally sustainable way of converting elemental nitrogen into plant-usable ammonium. Several bacteria are involved in the BNF molecular event. Nitrogen-fixing bacterial assemblages utilize the nitrogenase enzyme to fix atmospheric nitrogen in the soil to increase soil fertility [8,9]. Moreover, BNF annually provides 19,080 metric tonnes of the accessible form of nitrogen (i.e., ammonium salt) worldwide [10].
Several rhizospheric bacterial isolates, that is, Azotobacter sp., Azospirillum sp., Beijerinckia sp., Burkholderia sp., Clostridium sp., Enterobacter sp., Gluconacetobacter sp., Herbaspirillum sp., Ideonella sp., Klebsiella sp., Methanosarcina sp., Pseudomonas sp., and Paenibacillus sp. have been reported to show significant potential for N-fixing efficacy [11-14]. Therefore, the use of N-fixing bacterial regimes as crop inoculants could be a practical goal to overcome economic and commercial barriers in the agricultural sector [15,16]. Increasing soil salinity and many abiotic stresses have also been shown to be significant issues upon the use of N-fixing bacterial inoculants. The stress-associated microenvironment of soil (i.e., agricultural landscape) affects nitrogenase enzymatic activity, bacterial N-fixation, and proliferation [17-19]. Therefore, it is essential to carry out extensive research to identify the potential N-fixing bacteria that can be commercially viable and tolerant of abiotic stress factors in soil.
In this context, isolation of N-fixing bacteria needs to be performed from a microbially diversified region/ecosystem with salinity stress. The Sundarban mangrove soil ecosystem is suitable for evaluating the impact of salt stress along with N2 fixation of bacterial inoculants. Anoxic conditions nurture higher nitrogenase enzyme activity. Sundarban mangrove soil ecosystems provide permissive anoxic conditions to explore the potential nitrogen-fixing bacteria within the aforementioned soil habitat [20]. In addition, the commercialization of N-fixing bacteria (as growth-promoting biotic factors) depends on their viability in association with compatible carriers [16]. To this end, the current study used two potential carriers to check the metabolic viability of N-fixing bacterial inoculants in association with carriers, that is, lignite and cow dung compost (CDC) [21]. These two natural carriers are biocompatible, environmentally friendly, inexpensive, and highly available in the market, which can easily imply commercial sustainability [22,23]. Brownish-black lignite contains carbon (50–60%), nitrogen, hydrogen, oxygen, sulfur, and some other minerals [24,25]. In CDC contains carbon (30–40%), nitrogen (3%), phosphorus (2%), and potassium (1%), including essential minerals for bacterial and plant growth [26]. In addition, the pathogenicity and toxicity of N-fixing bacterial inoculants are other major burning issues at present. Pseudomonas aeruginosa, Klebsiella pneumoniae, and Clostridium botulinum are more effective N-fixing bacteria that can cause many acute and chronic diseases in the human body [27-29]. However, pathogenic N-fixing bacteria usually have negative impacts on soil ecology and the biogeochemical cycle. Thus, pathogenicity studies on N-fixing bacterial inoculants need to be considered before their commercialization and agricultural field application. Finally, we compared culture-dependent and culture-independent methodologies to assess the growth of the green chili plant (Capsicum frutescens L.) using the putative best newly isolated N-fixing bacterial inoculants, that is, Enterobacter sp. GG1 to accelerate its commercial feasibility and plant growth promotion. Hence, the current study focuses on isolating and characterizing most potential free-living N-fixing bacteria from mangrove ecosystems. Enterobacter sp. GG1 was found to have the highest nitrogen-fixing capacity. Moreover, pathogenicity, viability, carrier study, and plant growth-promoting efficacies of Enterobacter sp. GG1 have also been studied.
2. MATERIALS AND METHODS
2.1. Study Area for N-fixing Bacterial Isolation
In the winter season, soil samples were taken from Dobanki (Bidyadhari river beach: 21.988228oN, 88.753554oE), Pakhiralay (Datta river beach: 22.140086oN, 88.843076oE), and Mathurakhanda (Junction of Bidyadhari river and Datta river beach: 22.058277°N, 88.738887°E) in the Sundarban mangrove forests of West Bengal. Soil samples were collected from 4-inch depth of the rhizospheric soil surface (ground level). The adhered soil sample was transported within 24 h to the MEAB laboratory of JIS University in Kolkata, India, at a controlled 4°C temperature. However, the pH of the collected soil samples from the mangrove ecosystem was determined to be pH 7.3.
2.2. Screening and Isolation of N-fixing Bacteria
Collected samples were mixed in a sterile phosphate buffer solution (PBS) (pH7). After that, the supernatant was collected and serially diluted up to 10-8 using PBS under aseptic conditions. Then, N-fixing bacteria were screened and isolated using selective Nfb (nitrogen-free bromothymol blue) semi-solid medium (5 g DL-malic acid, 4 g KOH, 0.5 g K2HPO4, 0.1 g MgSO4.7H2O, 0.01 g MnSO4.H2O, 0.02 g NaCl, 0.01 g CaCl2, 0.05 g FeSO4.7H2O, 0.002 g Na2MoO4.2H2O, 1.75 g agar?agar, and 2 mL of 0.5% bromothymol blue, pH 7). After 3 days of incubation at 37°C, growing bacterial samples appeared green to blue color on selective Nfb media [30,31]. N-fixing bacteria were streaked on BMS media (potatoes 200 g, DL-malic acid 2.5 g, KOH 2.0 g, vitamin (biotin) 1 mL, agar?agar 18.0 g, distilled water 1000 mL) for single bacterial colony isolation to end up with pure axenic cultures for further studies [32].
First, 12 individual single-cell colonies were isolated randomly (4 colonies from each place) and grown in a different container in Nfb broth media. After that, 12 isolated bacterial cultures were subjected to primary screening by the well diffusion method on Nfb agar plates. For further study, the six best bacterial isolates were chosen based on colony diameter. Six isolates are nomenclature as N1, N2, N3, N4, N5, and N6. After isolation, six pure N-fixing bacterial cultures were qualitatively studied by the well diffusion method. In due course, 100 μL of broth culture from the exponential growth phase of the N-fixing bacterial samples was transferred aseptically into wells on Nfb agar medium [31]. Finally, blue zones were observed in the Nfb medium plate after72 h of incubation at 37°C.
2.3. Determination of the Isolated Bacterial Cellular Response to N-fixation
Broth culture from the exponential growth phase of the N-fixing bacterial sample was centrifuged at 10,000 rpm for 10 min. After that, the supernatant was discarded. Then, the intracellular response was investigated by crude extract of N-fixing bacterial cell pellet (sonicated in PBS buffer) transfer in Nfb agar plate media. Accordingly, the extracellular response was investigated by N-fixing bacterial cell pellet (without sonication) transfer in Nfb agar media with PBS buffer. After 72 h of incubation at 37°C, a blue zone was observed in the Nfb medium plate. In case, the enzymatic response of N-fixing bacteria is inside the cell; then, the intercellular reaction fixes the nitrogen and creates a blue color. In addition to the enzymatic response of N-fixing bacteria outside the cell, the extracellular reaction fixes nitrogen and creates a blue color.
2.4. Quantitative Analysis of Ammonium to Determine Potential N-fixing Bacterial Isolates Using the Phenate Method
Nitrogen-fixing bacteria fix atmospheric nitrogen into ammonium by nitrogenase enzymatic regulation. To compare the nitrogen fixation capabilities of the six new isolates, the analysis must quantify the final product, that is, ammonium using the phenate method. In this assay, stock solution 1 (sodium phenate solution) was prepared with the following composition: 25 g phenol and 20% NaOH were added to 6 mL acetone, and finally, the volume was 200 mL. Stock solution 2 (sodium nitroprusside): 0.15 g sodium nitroprusside was added to 1 L distilled water. Stock solution 3 (sodium hypochlorite) was also prepared using commercially available 5% sodium hypochlorite solution. According to the standard ammonium curve preparation, 0.3819 g anhydrous NH4Cl was dissolved (after drying at 100°C) in distilled water and diluted to 1 L (100 ppm). Then, 100 μL, 200 μL, 300 μL, 400 μL, 500 μL, and 600 μL of standard ammonium solution (100 ppm) were added into distilled water up to makeup 1 mL to prepare the working standards. After that, 500 μL of stock solution-1 (sodium phenate solution) was added to 100 μL of stock solution-2 (sodium nitroprusside) and 400 μL of stock solution-3 (hypochlorite solution) spontaneously and mixed gently at 20–30°C for 30 min. The final working standard concentrations of each solution were made upto 5 ppm, 10 ppm, 15 ppm, 20 ppm, 25 ppm, and 30 ppm spontaneously. Finally, the reaction mixture ended up with blue indophenol in basic solution, and absorbance was measured at 630 nm using a spectrophotometer (Shimadzu, Model: UV-1800) [33,34].
Isolated bacterial ammonium production comparative estimation for the initially equivalent quantity of biomass from exponential growth phase culture treated in Nfb broth media (without bromothymol blue). After 5 days of treatment, the bacterial culture was centrifuged for 10 min at 10000 rpm. Then, 1 mL of supernatant was collected, and following the typical standard curve preparation, the stock solution was mixed spontaneously. Finally, absorption was measured at 630 nm using a spectrophotometer [33,34]. In addition, the pH change was measured in treated culture broth media before (day 0) and after treatment (day 5).
2.5. Molecular Characterization of Potential N-fixing Bacteria N4 (Enterobacter sp. GG1)
The nitrogen-fixing N4 isolate (axenic pure culture) was harvested by centrifugation (10,000 rpm for 5 min), and genomic DNA of the nitrogen-fixing N4 isolate was extracted using a bacterial DNA isolation kit. Afterward, genomic DNA of the nitrogen-fixing N4 isolate was purified, followed by RNaseA treatment using a bacterial genomic DNA purification kit (Cat. No.: MB505-50PR). The purity and quantity of extracted genomic DNA of the nitrogen-fixing N4 isolate was determined using a Denovix DS-11 spectrophotometer. PCR was used to amplify a DNA fragment (amplicon) of 16S rRNA using the universal forward primer 10F (5′- AGTTTGATCATGGCTCAGATTG-3′) and the universal reverse primer 800R (5′- TACCAGGGTATCTAATCC-3′) [35,56]. These primers were used to amplify fragments of ~800 kb that included nearly complete 16S rDNA sequences. Afterwards, 1 μL (≈500 ng template) total DNA samples were used in a 50 μL PCR with 25 μL of EmeraldAmp GT PCR Master Mix (2X Premix); primers (forwards and reverse primer conc. 0.2 μM) 2.5 μL; depending on the volume of the sample DNA solution used, add an appropriate amount of dH2O to obtain a final volume of 50 μL (using Bacterial 16S rDNA PCR kit Fast (800), Cat. No. #RR310A). The PCR conditions were as follows: 98°C for 10 s; 60°C for 30 s; and 72°C for 1 min; for a total of 30 reaction cycles in a thermal cycler (Lablife) [37,38].
After PCR completion, analyze a part of the reaction mixture by agarose gel (2% gel) electrophoresis. The sample DNA solution and the positive control (Escherichia coli) each yielded amplification products of approximately 0.8 kb [Supplementary Figure S1]. After confirming the amplification products by electrophoresis, the PCR products were purified for sequence analysis. PCR yielded a single band of approximately 0.8 kb; the remaining reaction mixture was purified directly using a purification kit such as the GeneJET Extraction kit (Cat. No. #K0691). After purification of the target band from the agarose gel, the quantity of the purified PCR products was determined by measuring their absorbance at A260. Then, before sequencing, sequencing was performed with an ABI 3730 Genetic Analyser using the original PCR primers.
2.6. Pathogenicity Study of Enterobacter sp. GG1 by Hemolysis Assay
The pathogenicity test of Enterobacter sp. GG1 was inoculated in blood agar base media (containing 5% v/v defibrinated sheep blood) aseptically. Dispense of Enterobacter sp. GG1 was grown in two wells, one as an overnight pure culture and the other as a crude culture (by sonication) [39]. Accordingly, for the positive control, an ammonium-chloride-potassium (ACK buffer) lysis buffer solution (ammonium chloride 8.26 g, EDTA 0.037 g, potassium bicarbonate 1 g, in 1 L DW) was inoculated with the same volume in a sheep blood agar plate. After 48 h of treatment at 30°C, the result was observed [40].
In the broth media investigation, 5 mL of sheep blood was mixed well with 10 mL of PBS (pH 7.2). The supernatant was removed after 10 min of centrifugation at 2000 rpm at 4°C. This washing technique was repeated 3 times. Following the final washing, 2 mL of PBS (pH 7.2) was mixed with the pellet homogenously for erythrocyte suspension stock [41]. After that, 0.5 mL of erythrocyte suspension was mixed with 1.5 mL of hemolysis buffer (ACK) for the positive control. A positive control was made by adding 0.5 mL of stock erythrocyte suspensions to 1.5 mL of hemolysis buffer solution. Enterobacter sp. GG1 pathogenicity was investigated by adding 0.5 mL of erythrocyte suspension to 1.5 mL of overnight culture solution. Accordingly, intracellular pathogenicity was investigated in Enterobacter sp. GG1 by adding a crude extract of supernatant culture (sonicated in PBS buffer) in erythrocyte suspension stock. After preparation, the tested vials were incubated at 30°C for 2 h. After incubation, the samples were centrifuged at 8000 rpm for 8 min, the supernatant was carefully and aseptically collected from each vial, and the absorbance was measured at 412 nm [42].
2.7. Viability Study of Enterobacter sp. GG1 in Solid Carrier (CDC and Lignite)
This study was carried out to determine the viability of isolated bacteria in lignite and CDC (cow dung compost) carriers. This can occur while maintaining the physio-chemical parameters of carriers, such as pH and moisture conditions. Two individual carriers (CDC and lignite) were separately neutralized (pH-7) using CaCO3. Then, the two carriers individually maintain moisture at 25–30% by drying [43,44].
Following that, the dilution plate count technique was used to test the viability of the Enterobacter sp. GG1 bacterium in lignite and the CDC carrier. CDC was stored in two polypropylene bags with 20 g. Accordingly lignite was transferred into two polypropylene bags. Then, the four bags with the carrier were sterilized for 1 h at 121°C at 15 pounds per square inch pressure using an autoclave machine. After carrier sterilization Enterobacter sp. GG1 culture (1 × 1012 CFU/mL) was inoculated aseptically on laminar airflow at exponential phage [45]. These four inoculated bags were (i) CDC- control (without mix any isolates in CDC); (ii) CDC-GG1 (Enterobacter sp. GG1 had been inoculated in CDC carrier); (iii) Lignite-control (don’t mix any isolates in lignite); and (iv) Lignite-GG1 (inoculated Enterobacter sp. GG1 in lignite in the carrier). After inoculation, Enterobacter sp. GG1 in carriers was stored at room temperature (~ 25–30°C) for 180 days (i.e. 6 months).
Survival Enterobacter sp. GG1 population was estimated at monthly intervals for 6 months, such as days 0, 30, 60, 90, 120, 150, and 180. For the viability study, 1 g of treated carrier mix was added to 10 mL of sterile distilled water and serially diluted up to 10-7 in laminar. Then, 50 μL of diluted culture was spread on a nutrient agar media plate. After 1 day of incubation at 37°C in a B.O.D incubator, the bacterial cell colony grew. Then, the colony was counted manually, and the results were expressed as colony-forming units per gram (Cfu/g) using Equation 1 [44].
 |
2.8. Application of Enterobacter sp. GG1 Effects on Green Chili Plant Growth Characterization
The soil was collected from the Pataspur region (21.984060°N, 87.462378°E) for the plant growth study. It is one of the most important agricultural regions in the Purba Medinipur district of West Bengal. Clayey soils in nature dominate this region. This soil contains higher salt as salinity is too high [46]. In addition, clay soil nitrogen capacity has been deficient [47].
In this work, plants were harvested to use Enterobacter sp. GG1 N-fixing bacteria to analyze their above- and belowground growth features in the spring season [48,49]. The aboveground growth characteristics, such as plant shoot height and numbers of leaves, were measured after the 2-month change. Accordingly, belowground factors such as root length and the number of secondary root changes were measured after a 2-month interval.
3. RESULTS AND DISCUSSION
In the experiment, after 72 h of inoculation fixing the nitrogen to ammonium, Nfb semi-solid media’s green color changed to blue [Figure 1a and b, Supplementary Figure S2]. Isolate several efficient nitrogen-fixing bacteria from blue color Nfb semi-solid media. This several N-fixing bacteria were a streak in BMS media. After overnight growth in BMS medium, 12 essential N-fixing bacterial single-cell colony isolates were obtained from the BMS medium plate [Figure 1c and Supplementary Figure S3]. After that, six effective N-fixing bacterial isolates (nomenclatures such as N1, N2, N3, N4, N5, and N6) were selected from the preliminary well diffusion diameter [Supplementary Figure S4].
 | Figure 1: Screening and isolation of N-fixing bacteria; (a) control of Nfb semi-solid media (after 72 h incubation), (b) inoculated soil sample in Nfb semi-solid media (after 72 h incubation), (c) nitrogen-fixing bacteria streak in BMS agar media (after 24 h incubation). [Click here to view] |
3.1. Qualitative Primary Screening of N-fixing Bacterial Isolates
Six N-fixing bacterial isolates were extensively studied in Nfb selective medium to determine whether they could develop enough biomass under the same growth conditions to be used as inoculums in this comparative experiment. Inoculum development was carried out on Nfb selective media bacterial isolates in the exponential phase, showing N1 isolates at 10 h, N2 isolates at 10 h, N3 isolates at 16 h, N4 isolates at 10 h, N5 isolates at 12 h, and N6 isolates at 15 h [Supplementary Figure S5]. Then, inoculating the same number of N-fixing bacteria in exponential conditions showed an incredible amount of nitrogen-fixing ability in Nfb selective agar medium by precise blue zone formation compared to the control. In this experiment, N-fixing bacterial isolates were grown in the Nfb medium from the green to blue zone [Supplementary Figure S6]. This study’s potential results show the six isolates [Figure 2]. In addition, all types of bacteria showed extracellular activity for nitrogen fixation by blue zone creation in Nfb media [Supplementary Figure S7]. The extracellular response is quite effective for soil application because the extracellular response exposes the maximum reaction shown outside the cell.
 | Figure 2: Quality study of N-fixing bacterial isolates; (a) comparative study of N-fixing ability by blue zone diameter in Nfb agar media plate (After 72 h incubation) (n = 3), (b) One best plate shows of qualitative study (After 72 h of incubation). [Click here to view] |
3.2. Quantitative Identification of Potential N-fixing Bacteria
In the phenate method, the standard curve was a very accurate result, with an R2 value of 0.9971 [Figure 3a and b]. The method utilized to select the best potential isolate samples. N4 also demonstrates a great quantity of ammonium-generating capability in Nfb selective broth by indophenol blue color development compared to the control [Figure 3c-e]. During the 5-day experiment, a rise in pH was detected in N-fixing culture inoculates (pH 7–8) [Figure 4].
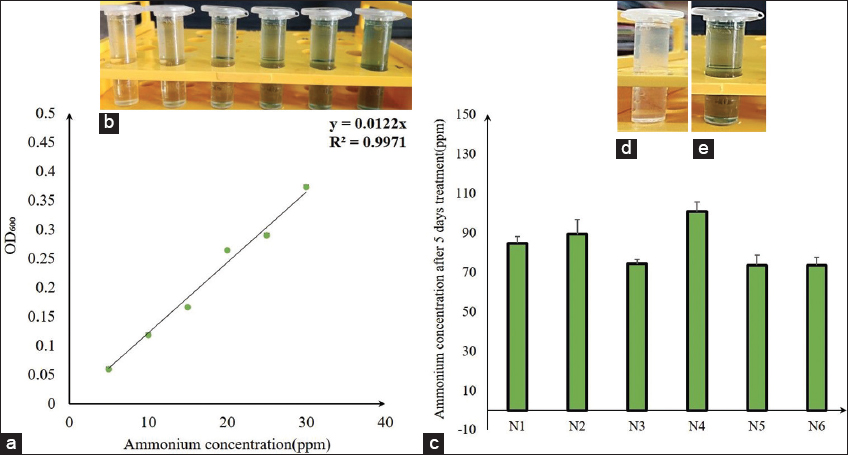 | Figure 3: Quantitative study of N-fixing bacterial isolates; (a) standard curve of ammonium (n = 3), (b) one set of ammonium standard curve measurements using the phenate method, (c) comparative quantification of ammonia at 5 days treatment in Nfb media (n = 3), (d) control of phenate test; (e) phenate test of one best result showing isolate (N4 isolate). [Click here to view] |
 | Figure 4: Measurement of pH change by isolated bacterial treatment at 5 days (n = 3). [Click here to view] |
3.3. Molecular Characterization and Phylogenetic Analysis of Potential N-fixing Bacterial Isolation N4 (Enterobacter sp. GG1)
The16S rRNA consensus sequence of the N4 isolate was identified by genetic analyzer-generated BLAST analysis for comparison with the sequences in the NCBI GenBank database. The N4 isolate was more similar (98.93%) to Enterobacter sp. [Supplementary Figure S8]. Finally, N4 isolate was abbreviated as Enterobacter sp. GG1. Furthermore, the 16S rRNA nucleotide sequences from Enterobacter sp. GG1 was compared with other most popular free-living nitrogen-fixing bacterial sequences in the databases using multiple sequence alignment [Supplementary Data files SD1 and SD2] to infer an integrated phylogenetic relationship. A phylogenetic tree representing 14 major groups of free-living nitrogen-fixing 16S rRNA sequences was generated and had been shown in Figure 5. Enterobacter sp. GG1 has a very close molecular character to the other prudential N-fixing bacterial strains Azospirillum lipoferum B7 and Methanosarcina sp. T40UC2.
 | Figure 5: Sequence alignments for molecular phylogenetic analysis were generated using SnapGene and optimized using MEGA11. Nearest-Neighbor-Interchange (NNI) method, Tamura-Nei model and maximum likelihood (ML) statistical models were used to infer the phylogenetic relationship among strains of closely associated N-fixing free living bacterial based on 16s rRNA gene sequences. A substitution model for phylogenetics of each nucleotide sequence data set was selected using the Tamura-Nei model with model selection in MEGA11. Representative phylogenetic trees were drawn using the NNI, ML methods [50,51]. [Click here to view] |
3.4. Pathogenicity Prediction
The blood agar test was used to evaluate germs for biosafety. Blood cell lysis or halo zone development on the blood agar medium was not observed compared to the positive control by Enterobacter sp. GG1. A positive control (ACK lysis buffer) was observed in beta-hemolysis (clear zones). The bacteria Enterobacter sp. GG1 exhibits gamma-hemolysis (no clear zones around colonies) [Figure 6a].
 | Figure 6: Enterobacter sp. GG1 bacterial hemolysis assay; (a) hemolysis assay in sheep blood agar plate, when Hb = ACK lysis buffer, C = Enterobacter sp. GG1 pure culture, and S = Enterobacter sp. GG1 crude culture; (b) comparative study of hemolysin activity in sheep blood broth media (n = 3), (c) hemolysin activity of ACK lysis buffer, (d) hemolysin activity of Enterobacter sp. GG1 pure culture, (e) hemolysin activity of Enterobacter sp. GG1 crude culture. [Click here to view] |
In liquid medium, ACK lysis buffer absorbance was assigned the value of 100% hemolysis. The construction was used to calculate the percentage hemolysis of the remaining absorptions. Homolysis activity was measured in pure culture and crude extract at 7.83% and 2.04%, respectively, [Figure 6b-e]. Enterobacter sp. GG1 hemolysin activity was shallow in the broth medium experiment. This indicates that the implanted bacteria may not be harmful to the environment.
3.5. Viable Potentiality of Enterobacter sp. GG1 in CDC and Lignite Carrier
The commercialization of Enterobacter sp. GG1 bacteria requires the study of the 6-month viability in a selected carrier’s lignite and CDC. This result showed that after 180 days (i.e. 6 months) of treatment at room temperature (25–30°C), the satisfying viable cells of Enterobacter sp. were sustained. Enterobacter sp. GG1 was the qualitatively highest number of viable cells shown in the lignite carrier at day 30 [Figure 7a]. After 180 days of storage treatment, potential viability was obtained in lignite carriers compared to CDC. After 6 months, compared to initial inoculations, nearly 40% of Enterobacter sp. GG1 cells survive in lignite carriers. In the CDC carrier satisfactory survival report, Enterobacter sp. GG1 at 180 days of treatment. Moreover, 21% of Enterobacter sp. GG1 survived as a CDC carrier for 6 months [Figure 7b and Supplementary Figure S9].
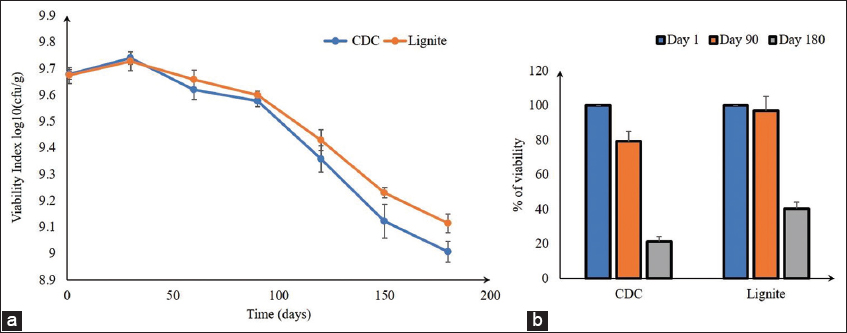 | Figure 7: Viability study of Enterobacter sp. GG1 in carrier (n = 3); (a) 180 days viability study in lignite and CDC carrier using best N-fixing bacterial isolates Enterobacter sp. GG1; (b) Comparative study percentage (%) of viability after 180 days treatment of Enterobacter sp. GG1 bacteria in CDC and lignite carriers. [Click here to view] |
3.6. Effect of Enterobacter sp. GG1 (N-fixing biofertilizer) Application on Green Chili Plant Growth
3.6.1. Effects of Enterobacter sp. GG1 on green chili plant shoot height and diameter
In the current study, nitrogen impacted chili plant morphology, including shoot growth [Figure 8a and b]. Table 1 had been showing the shoot height of green chili plants treated with Enterobacter sp. GG1 60 days after seedling. Enterobacter sp. GG1 treatment resulted in chili plant shoots that were 11.2 cm in height and 0.7 cm in diameter. The lowest chili plant shoot height and shoot diameter were recorded in the control (without biofertilizer) at 10.4 cm and 0.4 cm, respectively. Based on the practical results of Enterobacter sp. GG1, as a nitrogen biofertilizer, significantly affected chili plant shoot length (7.7% increases) and diameter (75% increases) compared to control growth.
 | Figure 8: Studies on green chili plant growth upon nitrogen biofertilizer (Enterobacter sp. GG1) treatment at 2 months; months; (a) N = Enterobacter sp. GG1 treated plant growth after 2 months, C = Untreated plant growth after 2 months; (b) N = Enterobacter sp. GG1 treated plant root and shoot growth after 2 months, C = Untreated plant root and shoot growth after 2 months; (c) N = Enterobacter sp. GG1 treated plant leaf growth after 2 months, C = Untreated leaf growth after 2 months. [Click here to view] |
Table 1: Studies on green chili plant growth upon nitrogen biofertilizer (Enterobacter sp. GG1) treatment at 2 months (n=3).
| Study sample character | Shoot height (cm) | Shoot diameter (cm) | Root length (cm) | No of branch root | No. of leaf | Leaf height (cm) | Leaf width (cm) |
|---|---|---|---|---|---|---|---|
| Control | 10.4 | 0.4 | 12.6 | 16 | 7 | 4.9 | 1.8 |
| N biofertilizer | 11.2 | 0.7 | 13.9 | 39 | 11 | 5.9 | 2.8 |
3.6.2. Effect of Enterobacter sp. GG1 on chili root length and number of branches
Green chili root morphology is significantly influenced by N-biofertilizer Enterobacter sp. GG1 treatments [Figure 8b]. The chili root length was significantly prolonged, 13.9 cm in the Enterobacter sp. GG1 treatment compared to the without fertilized treatment was 12.6 cm. In the case of the root branches (secondary root), the Enterobacter sp. GG1-treated plant yielded a total of 39 numbers, while the unfertilized plant yielded the lowest total of 16. In this report, it had clearly been shown that Enterobacter sp. GG1 had a significant effect on plant root morphology. Based on the effective results of Enterobacter sp. GG1 as an nitrogen biofertilizer on chili plants resulted in considerable increases in root length (10.9% increase) and the number of secondary roots (143.75% increase) compared to control growth, as shown in Table 1.
3.6.3. Effects of Enterobacter sp. GG1 on leaf number and morphology
Nitrogen biofertilization treatments significantly influenced the green chili plant leaf number [Figure 8a]. Treatment with Enterobacter sp. GG1 was found to have more than four leaves compared with the without fertilized treatment. It was thought that the absence of Enterobacter sp. GG1 could not produce a more significant number of leaves. This result from the present study revealed that the growth of the green chili plant was also significantly influenced (57.1%) by the treatments of Enterobacter sp. GG1 as an N biofertilizer.
The leaf width and length were significantly influenced by Enterobacter sp. GG1 treatments [Figure 8c]. Chili leaf width increased positively with Enterobacter sp. GG1 application. The minimum values of 1.8 cm were obtained from the plants grown in pots where no fertilizers were applied. The highest value of 2.8 cm of chili plant leaf width character was obtained from the Enterobacter sp. GG1 treatments. The Enterobacter sp. GG1, as an N biofertilizer, showed significant differences in leaf width (56.6% increases) compared with the untreated plant. The leaf length also increased (20.4% rise) when the chili plants were exposed to several Enterobacter sp. GG1 treatments. The Enterobacter sp. GG1 produced chili leaf lengths of 5.9 cm at 60 days to planting. The minimum leaf length was observed in the control treatment with a value of 4.9 cm, as shown in Table 1. Furthermore, leaf color did not significantly influence the impact of nitrogen fertilizer on Enterobacter sp. GG1 on the leaf. No higher differences were observed among the treatments.
4. CONCLUSION
A recent study indicated that nitrogen-fixing bacteria were concentrated and accumulated in mangrove ecosystem soil. Bacteria obtained in this investigation were shown to exhibit plant growth-stimulating properties such as ammonium generation. Enterobacter sp. GG1 was identified as the best isolate based on its biochemical features. After 5 days of treatment with Enterobacter sp. GG1 nitrogen fixation to ammonium was 100.68 ppm. However, current study’s results confirmed that Enterobacter sp. GG1 bacterial viability satisfactory in CDC and lignite carriers. Almost 40% and 21% of Enterobacter sp. GG1 isolate had been survived in lignite and CDC carrier spontaneously until 6 months, respectively. Furthermore, this strain is also nontoxic to humans, as confirmed by a hemolysis assay. Accordingly, a plant growth study demonstrated that the shoot height (7.7%), shoot diameter (75%), root length (10.87%), number of branch roots (143.75%), number of leaves (57.14%), leaf height (20.41%), and leaf width (56.60%) of green chili seedlings were significantly affected by Enterobacter sp. GG1 treatment in the soil. The present results conclude that optimal levels of Enterobacter sp. GG1 use in soil could be given essential nutrient elements to ensure the growth and health of green chili plants. Compared to more efficient N-fixing bacteria, ammonium production ability is slightly lower, but it has more potential for other effective significant factors, such as growth, viability, pathogenicity, and salinity soil tolerance [Supplementary Table 1]. Therefore, in future studies, the multi process parameter optimization of various independent variables and genetic modification need to be carried out to increase the nitrogenase enzyme catalytic efficacy and ammonia accumulation for more commercial potential [36]. Therefore, in the current study, the enzymatic activity and properties of Enterobacter sp. GG1 N-fixing bacteria develop microbial metabolic networks to improve soil fertility for the development of sustainable eco-friendly agriculture.
4.1. Supplementary Information
Supplementary Figure S1, Supplementary Figure S2; Supplementary Figure S3; Supplementary Figure S4; Supplementary Figure S5; Supplementary Figure S6; Supplementary Figure S7; Supplementary Figure S8;Supplementary Figure S9; Supplementary Datafile SD1; Supplementary Datafile SD2; Supplementary Table 1.
5. ACKNOWLEDGMENTS
We would like to thank JIS University Kolkata and JIS Group Educational Initiatives.
6. AUTHORS’ CONTRIBUTIONS
Ms. Palash Ghorai: All wet laboratory experimentation, data collection, data analysis, data set tabulation, data validation, investigation, wet laboratory experimental dataset visualization, original drafting, writing, and formatting. Dr. Dipankar Ghosh: Conceptualization, original drafting, writing, editing, reviewing, computation experimentation and computation dataset analysis, computational results visualization, and supervision.
7. FUNDING
There is no funding to report.
8. CONFLICT OF INTEREST
The authors declare that they do not have any conflict of interest.
9. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
10. DATA AVAILABILITY
All data supporting this study are available on request.
11. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Mager S, Ludewig U. Massive loss of DNA methylation in nitrogen-, but not in phosphorus-deficient Zea mays roots is poorly correlated with gene expression differences. Front Plant Sci 2018;9:497. [CrossRef]
2. Krouk G, Kiba T. Nitrogen and Phosphorus interactions in plants:From agronomic to physiological and molecular insights. Curr Opin Plant Biol 2020;57:104-9. [CrossRef]
3. Ghorai P, Ghosh D. Ameliorating the performance of NPK biofertilizers to attain sustainable agriculture with special emphasis on bioengineering. Bioresour Technol Rep 2022;19:101117. [CrossRef]
4. Dogbatse JA, Arthur A, Awudzi GK, Quaye AK, Konlan S, Amaning AA. Effects of organic and inorganic fertilizers on growth and nutrient uptake by young cacao (Theobroma cacao L.). Int J Agron 2021;2021:5516928. [CrossRef]
5. Aryal JP, Sapkota TB, Krupnik TJ, Rahut DB, Jat ML, Stirling CM. Factors affecting farmers'use of organic and inorganic fertilizers in South Asia. Environ Sci Pollut Res 2021;28:51480-96. [CrossRef]
6. Dal Cortivo C, Ferrari M, Visioli G, Lauro M, Fornasier F, Barion G, et al. Effects of seed-applied biofertilizers on rhizosphere biodiversity and growth of common wheat (Triticum aestivum L.) in the field. Front Plant Sci 2020;11:72. [CrossRef]
7. Reid TE, Kavamura VN, Abadie M, Torres-Ballesteros A, Pawlett M, Clark IM, et al. Inorganic chemical fertilizer application to wheat reduces the abundance of putative plant growth-promoting rhizobacteria. Front Microbiol 2021;12:642587. [CrossRef]
8. Ghosh D, Ghorai P, Debnath S, Indrama T, Kondi V, Tiwari ON. Algal biofertilizer towards green sustainable agriculture. In:Singh HB, Vaishnav A, editors. New and Future Developments in Microbial Biotechnology and Bioengineering. Netherlands:Elsevier;2022. 27-45. [CrossRef]
9. Zhang X, Tong J, Dong M, Akhtar K, He B. Isolation, identification and characterization of nitrogen fixing endophytic bacteria and their effects on cassava production. PeerJ 2022;10:e12677. [CrossRef]
10. Ikhwani, Novisrayani, Husna N, Yuniarti E. Formula application of N fixation and P solubilizing isolate from two soil type of paddy field on the growth and yield of rice (Oryza sativa Linn.). IOP Conf Ser Earth Environ Sci 2022;976:012040. [CrossRef]
11. Aasfar A, Bargaz A, Yaakoubi K, Hilali A, Bennis I, Zeroual Y, et al. Nitrogen fixing Azotobacter species as potential soil biological enhancers for crop nutrition and yield stability. Front Microbiol 2021;12:628379. [CrossRef]
12. Singh RK, Singh P, Sharma A, Guo DJ, Upadhyay SK, Song QQ, et al. Unraveling nitrogen fixing potential of endophytic diazotrophs of different Saccharum species for sustainable sugarcane growth. Int J Mol Sci 2022;23:6242. [CrossRef]
13. Wang M, Bian Z, Shi J, Wu Y, Yu X, Yang Y, et al. Effect of the nitrogen-fixing bacterium Pseudomonas protegens CHA0-DretS-nif on garlic growth under different field conditions. Ind Crops Prod 2020;145:111982. [CrossRef]
14. Ji C, Liu Z, Hao L, Song X, Wang C, Liu Y, et al. Effects of Enterobacter cloacae HG-1 on the nitrogen-fixing community structure of wheat rhizosphere soil and on salt tolerance. Front Plant Sci 2020;11:1094. [CrossRef]
15. Berninger T, López ÓG, Bejarano A, Preininger C, Sessitsch A. Maintenance and assessment of cell viability in formulation of non-sporulating bacterial inoculants. Microb Biotechnol 2018;11:277-301. [CrossRef]
16. Aloo BN, Mbega ER, Makumba BA, Tumuhairwe JB. Effects of carrier materials and storage temperatures on the viability and stability of three biofertilizer inoculants obtained from potato (Solanum tuberosum L.) rhizosphere. Agriculture 2022;12:140. [CrossRef]
17. Nandanwar S, Yele Y, Dixit A, Goss-Souza D, Singh R, Shanware A, et al. Effects of pesticides, temperature, light, and chemical constituents of soil on nitrogen fixation. In:Rigobelo E, Serra A, Blumenberg M, editors. Nitrogen Fixation. London:IntechOpen;2020. [CrossRef]
18. Kirova E, Kocheva K. Physiological effects of salinity on nitrogen fixation in legumes-a review. J Plant Nutr 2021;44:2653-62. [CrossRef]
19. Severin I, Confurius-Guns V, Stal LJ. Effect of salinity on nitrogenase activity and composition of the active diazotrophic community in intertidal microbial mats. Arch Microbiol 2012;194:483-91. [CrossRef]
20. Das S, De TK. Microbial assay of N2 fixation rate, a simple alternate for acetylene reduction assay. MethodsX 2018;5:909-14. [CrossRef]
21. Fasusi OA, Cruz C, Babalola OO. Agricultural sustainability:Microbial biofertilizers in rhizosphere management. Agriculture 2021;11:163. [CrossRef]
22. Li C, Xiong Y, Zou J, Dong L, Ren P, Huang G. Impact of biochar and lignite-based amendments on microbial communities and greenhouse gas emissions from agricultural soil. Vadose Zone J 2021;20:e20105. [CrossRef]
23. Gupta KK, Aneja KR, Rana D. Current status of cow dung as a bioresource for sustainable development. Bioresour Bioprocess 2016;3:28. [CrossRef]
24. Hu X, Wang T, Dong Z, Zhang H, Dong C. Research on the gas reburning in a circulating fluidized bed (CFB) system integrated with biomass gasification. Energies 2012;5:3167-77. [CrossRef]
25. Vivekanandhan P, Anand S, Thanikaselam A. Investigation on wear resistance of varying load of particulate lignite fly ash reinforced aluminium alloy 6063 composites. Int J Emerg Technol Adv Eng 2012;2:145-8.
26. Liu M, Sui X, Hu Y, Feng F. Microbial community structure and the relationship with soil carbon and nitrogen in an original Korean pine forest of Changbai Mountain, China. BMC Microbiol 2019;19:218. [CrossRef]
27. Li B, Zhao Y, Liu C, Chen Z, Zhou D. Molecular pathogenesis of Klebsiella pneumoniae. Future Microbiol 2014;9:1071-81. [CrossRef]
28. Reynolds D, Kollef M. The epidemiology and pathogenesis and treatment of Pseudomonas aeruginosa infections:An update. Drugs 2021;81:2117-31. [CrossRef]
29. Bhardwaj T, Haque S, Somvanshi P. In silico identification of molecular mimics involved in the pathogenesis of Clostridium botulinum ATCC 3502 strain. Microb Pathog 2018;121:238-44. [CrossRef]
30. Widawati S, Suliasih S. Role of indigenous nitrogen-fixing bacteria in promoting plant growth on post tin mining soil. Makara J Sci 2019;23:4. [CrossRef]
31. Arsita R, Karim H, Hala Y, Iriany N, Jumadi O. Isolation and identification of nitrogen-fixing bacteria in the corn rhizosphere (Zea mays l.) originating from Jeneponto Regency, South Sulawesi. IOP Conf Ser Earth Environ Sci 2020;484:012051. [CrossRef]
32. Raffi MM, Charyulu PB. Nitrogen fixation by the Native Azospirillum sp. Ssolated from Rhizosphere and Non-Rhizosphere of foxtail Millet. Asian J Biol Life Sci 2012;1:213-8.
33. Zhou L, Boyd CE. Comparison of Nessler, phenate, salicylate and ion selective electrode procedures for determination of total ammonia nitrogen in aquaculture. Aquaculture 2016;450:187-93. [CrossRef]
34. Park JE, Oh HN, Ahn S. Improvement of the ammonia analysis by the phenate method in water and wastewater. Bull Korean Chem Soc 2009;30:2032-8. [CrossRef]
35. Hirai J, Sakanashi D, Hagihara M, Haranaga S, Uechi K, Kato H, et al. Bacteremia due to Streptococcus tigurinus:A case report and literature review. J Infect Chemother 2016;22:762-6. [CrossRef]
36. Ghosh D, Sobro IF, Hallenbeck PC. Optimization of the hydrogen yield from single-stage photofermentation of glucose by Rhodobacter capsulatus JP91 using response surface methodology. Bioresour Technol 2012;123:199-206. [CrossRef]
37. Franco-Duarte R, ?ernákováL, Kadam S, Kaushik KS, Salehi B, Bevilacqua A, et al. Advances in chemical and biological methods to identify microorganisms-from past to present. Microorganisms 2019;7:130. [CrossRef]
38. Weisburg WG, Barns SM, Pelletier DA, Lane DJ. 16S ribosomal DNA amplification for phylogenetic study. J Bacteriol 1991;173:697-703. [CrossRef]
39. Denaya S, Yulianti R, Pambudi A, Effendi Y. Novel microbial consortium formulation as plant growth promoting bacteria (PGPB) agent. IOP Conf Ser Earth Environ Sci 2021;637:012030. [CrossRef]
40. Brown WE, Hu JC, Athanasiou KA. Ammonium-chloride-potassium lysing buffer treatment of fully differentiated cells increases cell purity and resulting neotissue functional properties. Tissue Eng Part C Methods 2016;22:895-903. [CrossRef]
41. Huang B, Liang Y, Pan H, Xie L, Jiang T, Jiang T. Hemolytic and cytotoxic activity from cultures of Aureococcus anophagefferens-a causative species of brown tides in the North-Western Bohai Sea, China. Chemosphere 2020;247:125819. [CrossRef]
42. Elleboudy N, Aboulwafa M, Hassouna N. Phospholipases C from Pseudomonas aeruginosa and Bacillus cereus isolates, chromosome-mediated enzymes with roles in virulence. Turk J Biol 2013;37:433-42. [CrossRef]
43. Bello A, Begianpuye A, Alfa A, Abdulraheem G, Adamu Y, Anthony D. Development of biofertilizer from composted cow-dung. Sci Forum (J Pure Appl Sci) 2019;16:1-7. [CrossRef]
44. Gade RM, Chaithanya BH, Khurade KC. A comparitive study of different carriers for shelflife of Pseudomonas fluorescens. Bioscan 2014;9:287-90.
45. Abd El-Fattah DA, Eweda WE, Zayed MS, Hassanein MK. Effect of carrier materials, sterilization method, and storage temperature on survival and biological activities of Azotobacter chroococcum inoculant. Ann Agric Sci 2013;58:111-8. [CrossRef]
46. Sahu A. Status of Soil in Purba Medinipur District, West Bengal-a review. Indian J Geogr Environ 2014;13:121-6.
47. Dempster DN, Jones DL, Murphy DV. Clay and biochar amendments decreased inorganic but not dissolved organic nitrogen leaching in soil. Soil Res 2012;50:216. [CrossRef]
48. Raza A, Ejaz S, Saleem MS, Hejnak V, Ahmad F, Ahmed MA, et al. Plant growth promoting rhizobacteria improve growth and yield related attributes of chili under low nitrogen availability. PLoS One 2021;16:e0261468. [CrossRef]
49. Bhutia LK, Vk K, Meetei NG, Bhutia DN. Effects of climate change on growth and development of Chilli. Agrotechnology 2018;7:180. [CrossRef]
50. Saitou N, Nei M. The neighbor-joining method:A new method for reconstructing phylogenetic trees. Mol Biol Evol 1987;4:406-25.
51. Felsenstein J. Confidence limits on phylogenies:An approach using the bootstrap. Evolution 1985;39:783-91. [CrossRef]
52. Sellstedt A, Richau KH. Aspects of nitrogen-fixing Actinobacteria, in particular free-living and symbiotic Frankia. FEMS Microbiol Lett 2013;342:179-86. [CrossRef]
53. Giri S, Pati BR. A comparative study on phyllosphere nitrogen fixation by newly isolated Corynebacterium sp. and Flavobacterium sp. and their potentialities as biofertilizer. Acta Microbiol Immunol Hung 2004;51:47-56. [CrossRef]
54. Sumbul A, Ansari RA, Rizvi R, Mahmood I. Azotobacter:A potential bio-fertilizer for soil and plant health management. Saudi J Biol Sci 2020;27:3634-40. [CrossRef]
55. Ramalho TP, Chopin G, Salman L, Baumgartner V, Heinicke C, Verseux C. On the growth dynamics of the cyanobacterium Anabaena sp. PCC 7938 in Martian regolith. NPJ Microgravity 2022;8:43. [CrossRef]
56. Sandström JP, Russell JA, White JP, Moran NA. Independent origins and horizontal transfer of bacterial symbionts of aphids. Mol Ecol 2001;10:217-28. [CrossRef]